Abstract
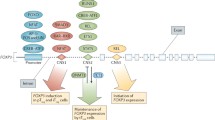
FOXP3 is a member of the forkhead transcription factor family. Unlike other members, it is mainly expressed in a subset ofCD4+ T-cells that play a suppressive role in the immune system. A function of FOXP3 is to suppress the function of NFAT and NFKB and this leads to suppression of expression of many genes including IL-2 and effector T-cell cytokines. FOXP3 acts also as a transcription activator for many genes including CD25, Cytotoxic T-Lymphocyte Antigen4 (CTLA4), glucocorticoid-induced TNF receptor family gene (GITR) and folate receptor 4. FOXP3+ T-cells are made in the thymus and periphery. The FOXP3+ T-cells made in the thymus migrate to secondary lymphoid tissues and suppress antigen priming of lymphocytes. Antigen priming of naïve FOXP3+ T-cells and naïve FOXP3− T-cells leads to generation of memory FOXP3+ T-cells which are efficient in migration to nonlymphoid tissues. Memory FOXP3+ T-cells are, therefore, effective in suppression of effector T-cell function, while naive FOXP3+ T-cells are adept at suppressing the early immune responses in lymphoid tissues. Both naïve and memory FOXP3+ T-cells are required for effective maintenance of tolerance and prevention of autoimmune diseases throughout the body. Many factors such as cytokines and noncytokine factors regulate the generation of FOXP3+ T-cells. For example, retinoic acid, produced by the dendritic cells and epithelial cells in the intestine, works together with TGF-β1 and promotes generation of small intestine-homing FOXP3+ T-cells by upregulating the expression of FOXP3 and gut homing receptors. FOXP3+ T-cells can be produced in vitro from autologous naïve T-cells and, therefore, have great therapeutic potentials in treating a number of inflammatory diseases and graft rejection.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
- Retinoic Acid
- Experimental Autoimmune Encephalomyelitis
- Foxp3 Expression
- Forkhead Transcription Factor
- FOXP3 Gene
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Bates GJ, Fox SB, Han C et al. Expression of the forkhead transcription factor FOXP1 is associated with that of estrogen receptor-beta in primary invasivebreast carcinomas. Breast Cancer Res Treat 2008; 111:453–459.
Campbell DJ, Ziegler SF. FOXP3 modifies the phenotypic and functional properties of regulatory T-cells. Nat Rev Immunol 2007; 7:305–310.
Shevach EM, DiPaolo RA, Andersson J et al. The lifestyle of naturally occurring CD4+ CD25+ Foxp3+ regulatory T-cells. Immunol Rev 2006; 212:60–73.
Ziegler SF. FOXP3: of mice and men. Annu Rev Immunol 2006; 24:209–226.
Kim CH. Molecular targets of FoxP3+ regulatory T-cells. Mini Rev Med Chern 2007; 7:1136–1143.
Sakaguchi S, Ono M, Setoguchi R et al. Foxp3+ CD25+ CD4+ natural regulatory T-cells in dominant self-tolerance and autoimmune disease. Immunol Rev 2006; 212:8–27.
Sakaguchi S. Naturally arising Foxp3-expressing CD25+CD4+ regulatory T-cells in immunological tolerance to self and nonself. Nat Immunol 2005; 6:345–352.
Beyer M, Schultze JL. Regulatory T-cells in cancer. Blood 2006; 108:804–811.
Chang X, Gao JX, Jiang Q et al. The Scurfy mutation of FoxP3 in the thymus stroma leads to defective thyrnopoiesis. J Exp Med 2005; 2002:1141–1151.
Zuo T, Wang L, Morrison C et al. FOXP3 is an X-linked breast cancer suppressor gene and an important repressor of the HER-2/ErbB2 oncogene. Cell 2007; 129:1275–1286.
Bennett CL, Yoshioka R, Kiyosawa H et al. X-Linked syndrome of polyendocrinopathy, immune dysfunction and diarrhea maps to Xp11.23–Xq13.3. Am J Hum Genet 2000; 66:461–468.
Bennett CL, Christie J, Ramsdell F et al. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat Genet 2001; 27:20–21.
Kaufmann E, Muller D, Knochel W. DNA recognition site analysis of Xenopus winged helix proteins. J Mol Biol 1995; 248:239–254.
Schubert LA, Jeffery E, Zhang Y et al. Scurfin (FOXP3) acts as a repressor of transcription and regulates T-cell activation. J Biol Chem 2001; 276:37672–37679.
Lopes JE, Torgerson TR, Schubert LA et al. Analysis of FOXP3 reveals multiple domains required for its function as a transcriptional repressor. J Immunol 2006; 177:3133–3142.
Bettelli E, Dastrange M, Oukka M. FOXP3 interacts with nuclear factor of activated T-cells and NF-kappa B to repress cytokine gene expression and effector functions of T helper cells. Proc Natl Acad Sci USA 2005; 102:5138–5143.
Kamine J, Elangovan B, Subramanian T et al. Identification of a cellular protein that specifically interacts with the essential cysteine region of the HIV-1 Tat transactivator. Virology 1996; 216:357–366.
Yang XJ. The diverse superfamily of lysine acetyltransferases and their roles in leukemia and other diseases. Nucleic Acids Res 2004; 32:959–976.
Bennett CL, Ochs HD. IPEX is a unique X-linked syndrome characterized by immune dysfunction, polyendocrinopathy, enteropathy and a variety of autoimmune phenomena. Curr Opin Pediatr 2001; 13:533–538.
Ochs HD, Gambineri E, Torgerson TR. IPEX, FOXP3 and regulatory T-cells: a model for autoimmunity. Immunol Res 2007; 38:112–121.
van der Vliet HJ, Nieuwenhuis EE. IPEX as a result of mutations in FOXP3. Clin Dev Immunol 2007; 2007:89017.
Chen, C, Rowell EA, Thomas RM et al. Transcriptional regulation by Foxp3 is associated with direct promoter occupancy and modulation of histone acetylation. J Biol Chem 2006; 281:36828–36834.
Zheng Y, Josefowicz SZ, Kas A et al. Genome-wide analysis of Foxp3 target genes in developing and mature regulatory T-cells. Nature 2007; 445:936–940.
Marson A, Kretschmer K, Frampton GM et al. Foxp3 occupancy and regulation of key target genes during T-cell stimulation. Nature 2007; 445:931–935.
Wu Y, Borde M, Heissmeyer V et al. FOXP3 controls regulatory T-cell function through cooperation with NFAT. Cell 2006; 126:375–387.
Kim CH. Migration and function of FoxP3+ regulatory T-cells in the hematolyrnphoid system. Exp Hematol 2006; 34:1033–1040.
Mantel PY, Ouaked N, Ruckert B et al. Molecular mechanisms underlying FOXP3 induction in human T-cells. J Immunol 2006; 176:3593–3602.
Bisikirska B, Colgan J, Luban J et al. TCR stimulation with modified anti-CD3 mAb expands CD8+ T-cell population and induces CD8+CD25+ Tregs. J Clin Invest 2005; 115:2904–2913.
Fontenot JD, Dooley JL, Farr AG et al. Developmental regulation of Foxp3 expression during ontogeny. J Exp Med 2005; 2002:901–906.
Kim CH. Trafficking of FoxP3+ regulatory T-cells: myths and facts. Arch Immunol Ther Exp (Warsz) 2007; 55:151–159.
Lee JH, Kang SG, Kim CH. FoxP3+ T-cells undergo conventional first switch to lymphoid tissue homing receptors in thymus but accelerated second switch to nonlymphoid tissue homing receptors in secondary lymphoid tissues. J Immunol 2007; 178:301–311.
Tuovinen H, Kekalainen E, Rossi LH et al. Cutting edge: human CD4-CD8-thyrnocytes express FOXP3 in the absence of a TCR. J Immunol 2008; 180:3651–3654.
Hori S, Nomura T, Sakaguchi S. Control of regulatory T-cell development by the transcription factor Foxp3. Science 2003; 299:1057–1061.
Khattri R, Cox T, Yasayko SA et al. An essential role for Scurfin in CD4+CD25+ T regulatory cells. Nat Immunol 2003; 4:337–342.
Allan SE, Passerini L, Bacchetta R et al. The role of 2 FOXP3 isoforms in the generation of human CD4 Tregs. J Clin Invest 2005; 115:3276–3284.
Sauer S, Bruno L, Hertweck A et al. T-cell receptor signaling controls Foxp3 expression via PI3K, Akt and mTOR. Proc Natl Acad Sci USA 2008; 105:7797–7802.
Zorn E, Nelson EA, Mohseni M et al. IL-2 regulates FOXP3 expression in human CD4+CD25+ regulatory T-cells through a STAT-dependent mechanism and induces the expansion of these cells in vivo. Blood 2006; 108:1571–1579.
Floess S, Freyer J, Siewert C et al. Epigenetic control of the foxp3 locus in regulatory T-cells. PLoS Biol 2007; 5:e38.
Kim HP, Leonard WJ. CREB/ATF-dependent T-cell receptor-induced FoxP3 gene expression: a role for DNA methylation. J Exp Med 2007; 204:1543–1551.
Chen W, Jin W, Hardegen N et al. Conversion of peripheral CD4+CD25− naive T-cells to CD4+CD25+ regulatory T-cells by TGF-beta induction oftranscription factor Foxp3. J Exp Med 2003; 198:1875–1886.
Fantini MC, Becker C, Monteleone G et al. Cutting edge: TGF-beta induces a regulatory phenotype in CD4+CD25− T-cells through Foxp3 induction and down-regulation of Smad7. J Immunol 2004; 172:5149–5153.
Tone Y, Furuuchi K, Kojima Y et al. Smad3 and NFAT cooperate to induce Foxp3 expression through its enhancer. Nat Immunol 2008; 9:194–202.
Baron U, Floess S, Wieczorek G et al. DNA demethylation in the human FOXP3 locus discriminates regulatory T-cells from activated FOXP3(+) conventional T-cells. Eur J Immunol 2007; 37:2378–2389.
Janson PC, Winerdal ME, Marits P et al. FOXP3 promoter demethylation reveals the committed Treg population in humans. PLoS ONE 2008; 3:e1612.
Polansky JK, Kretschmer K, Freyer J et al. DNA methylation controls Foxp3 gene expression. Eur J Immunol 2008; 38:1654–1663.
Yao Z, Kanno Y, Kerenyi M et al. Nonredundant roles for Stat5a/b in directly regulating Foxp3. Blood 2007; 109:4368–4375.
Takaki H, Ichiyama K, Koga K et al. STAT6 Inhibits TGF-betal-mediated Foxp3 induction through direct binding to the Foxp3 promoter, which is reverted by retinoic acid receptor. J Biol Chem 2008; 283:14955–14962.
Nishihara M, Ogura H, Ueda N et al. IL-6-gp130-STAT3 in T-cells directs the development of IL-17+ Th with a minimum effect on that of Treg in the steady state. Int Immunol 2007; 19:695–702.
Veldhoen M, Hocking RJ, Atkins CJ et al. TGFbeta in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17-producing T-cells. Immunity 2006; 24:179–189.
Samanta A, Li B, Song X et al. TGF-beta and IL-6 signals modulate chromatin binding and promoter occupancy by acetylated FOXP3. Proc Natl Acad Sci USA 2008; 105:14023–14027.
Michie SA, Streeter PR, Bolt PA et al. The human peripheral lymph node vascular addressin. An inducible endothelial antigen involved in lymphocyte homing. Am J Pathol 1993; 143:1688–1698.
Picker LJ, Michie SA, Rott LS et al. A unique phenotype of skin-associated lymphocytes in humans. Preferential expression of the HECA-452 epitope by benign and malignant T-cells at cutaneous sites. Am J Pathol 1990; 136:1053–1068.
Streeter PR, Rouse BT, Butcher EC. Immunohistologic and functional characterization of a vascular addressin involved in lymphocyte homing into peripheral lymph nodes. J Cell Biol 1988; 107:1853–1862.
Forster R, Schubel A, Breitfeld D et al. CCR7 coordinates the primary immune response by establishing functional microenvironments in secondary lymphoid organs. Cell 1999; 99:23–33.
Stein JV, Rot A, Luo Y et al. The CC chemokine thymus-derived chemotactic agent 4 (TCA-4, secondary lymphoid tissue chemokine, 6Ckine, exodus-2) triggers lymphocyte function-associated antigen 1-mediated arrest of rolling T-Iymphocytes in peripheral lymph node high endothelial venules. J Exp Med 2000; 191:61–76.
Warnock RA, Campbell JJ, Dorf ME et al. The role of chemokines in the microenvironmental control of T versus B-cell arrest in Peyer’s patch high endothelial venules. J Exp Med 2000; 191:77–88.
Wurbel MA, Malissen M, Guy-Grand D et al. Impaired accumulation of antigen-specific CD8 lymphocytes in chemokine CCL25-deficient intestinal epithelium and lamina propria. J Immunol 2007; 178:7598–7606.
Wendland M, Czeloth N, Mach N et al. CCR9 is a homing receptor for plasmacytoid dendritic cells to the small intestine. Proc Natl Acad Sci USA 2007; 104:6347–6352.
Stenstad H, Ericsson A, Johansson-Lindborn B et al. Gut-associated lymphoid tissue-primed CD4+ T-cells display CCR9-dependent and-independent homing to the small intestine. Blood 2006; 107:3447–3454.
Papadakis KA, Prehn J, Nelson V et al. The role of thymus-expressed chemokine and its receptor CCR9 on lymphocytes in the regional specialization of the mucosal immune system. J Immunol 2000; 165:5069–5076.
Kunkel EJ, Campbell JJ, Haraldsen G et al. Lymphocyte CC chemokine receptor 9 and epithelial thymus-expressed chemokine (TECK) expression distinguish the small intestinal immune compartment: Epithelial expression of tissue-specific chemokines as an organizing principle in regional immunity. J Exp Med 2000; 192:761–768.
Hamann A, Andrew DP, Jablonski-Westrich D et al. Role of alpha 4-integrins in lymphocyte homing to mucosal tissues in vivo. J Immunol 1994; 152:3282–3293.
Berlin C, Berg EL, Briskin MJ et al. Alpha 4 beta 7 integrin mediates lymphocyte binding to the mucosal vascular addressin MAdCAM-1. Cell 1993; 74:185–185.
Lim HW, Hillsamer P, Kim CH. Regulatory T-cells can migrate to follicles upon T-cell activation and suppress GC-Th cells and GC-Th cell-driven B-cell responses. J Clin Invest 2004; 114:1640–1649.
Huehn J, Siegmund K, Lehmann JC et al. Developmental Stage, Phenotype and Migration Distinguish Naive-and Effector/Memory-like CD4+ Regulatory T-Cells. J Exp Med 2004; 199:303–313.
Lee I, Wang L, Wells AD et al. Recruitment of Foxp3+ T regulatory cells mediating allograft tolerance depends on the CCR4 chemokine receptor. J Exp Med 2005; 201:1037–1044.
Sather BD, Treuting P, Perdue N et al. Altering the distribution of Foxp3(+) regulatory T-cells results in tissue-specific inflammatory disease. J Exp Med 2007; 204:1335–1347.
Schneider MA, Meingassner JG, Lipp M et al. CCR7 is required for the in vivo function of CD4+ CD25+ regulatory T-cells. J Exp Med 2007; 204:735–745.
Mucida D, Park Y, Kim G et al. Reciprocal TH17 and regulatory T-cell differentiation mediated by retinoic acid. Science 2007; 317:256–260.
Benson MJ, Pino-Lagos K, Rosemblatt M et al. All-trans retinoic acid mediates enhanced T reg cell growth, differentiation and gut homing in the face of high levels of costimulation. J Exp Med 2007; 204:1765–1774.
Sun CM, Hall JA, Blank RB et al. Small intestine lamina propria dendritic cells promote de novo generation of Foxp3 T reg cells via retinoic acid. J Exp Med 2007; 204: 1775–1785.
Coombes JL, Siddiqui KR, Arancibia-Carcamo CV et al. A functionally specialized population of mucosal CDI03+ DCs induces Foxp3+ regulatory T-cells via a TGF-beta and retinoic acid-dependent mechanism. J Exp Med 2007; 204:1757–1764.
Kang SG, Lim HW, Andrisani OM et al. Vitamin A Metabolites Induce Gut-Homing FoxP3+ Regulatory T-Cells. J Immunol 2007; 179:3724–3733.
Schambach F, Schupp M, Lazar MA et al. Activation of retinoic acid receptor-alpha favours regulatory T-cell induction at the expense of IL-17-secreting T helper cell differentiation. Eur J Immunol 2007; 37:2396–2399.
Elias KM, Laurence A, Davidson TS et al. Retinoic acid inhibits Th17 polarization and enhances FoxP3 expression through a Stat-3/ Stat-5 independent signaling pathway. Blood 2008; 111:1013–1020.
Kim CH. Regulation of FoxP3 regulatory T-cells and Th17 cells by retinoids. Clin Dev Immunol 2008; 2008:416910.
Kang SG, Piniecki RJ, Hogenesch H et al. Identification of a chemokine network that recruits FoxP3(+) regulatory T-cells into chronically inAamed intestine. Gastroenterology 2007; 132:966–981.
Iwata M, Hirakiyama A, Eshima Y et al. Retinoic acid imprints gut-homing specificity on T-cells. Immunity 2004; 21:527–538.
Baumgart DC, Carding SR. In Aammatory bowel disease: cause and immunobiology. Lancet 2007; 369:1627–1640.
Hanauer SB. Inflammatory bowel disease: epidemiology, pathogenesis and therapeutic opportunities. In Aamm Bowel Dis 2006; (12 Suppl 1):S3–9.
Xiao S, Jin H, Korn T et al. Retinoic acid increases Foxp3+ regulatory T-cells and inhibits development of Th17 cells by enhancing TGF-beta-driven Smad3 signaling and inhibiting IL-6 and IL-23 receptor expression. J Immunol 2008; 181:2277–2284.
Suffia I, Reclding SK, Salay G et al. A role for CD 103 in the retention of CD4+CD25+ Treg and control of Leishmania major infection. J Immunol 2005; 174:5444–5455.
Annacker O, Coombes JL, Malmstrom V et al. Essential role for CDI03 in the T-cell-mediated regulation of experimental colitis. J Exp Med 2005; 202:1051–1061.
Ermann J, Hoffmann P, Edinger M et al. Only the CD62L+ subpopulation ofCD4+CD25+ regulatory T-cells protects from lethal acute GVHD. Blood 2005; 105:2220–2226.
Taylor PA, Panoskalrsis-Mortari A, Swedin JM et al. L-Selectin(hi) but not the L-selectin(lo) CD4+25+ T-regulatory cells are potent inhibitors of GVHD and BM graft rejection. Blood 2004; 104:3804–3812.
Nakamura K, Kitani A, Strober W Cell contact-dependent immuno suppression by CD4(+)CD25(+) regulatory T-cells is mediated by cell surface-bound transforming growth factor beta. J Exp Med 2001; 194:629–644.
Piccirillo CA, Letterio JJ, Thornton AM et al. CD4(+)CD25(+) regulatory T-cells can mediate suppressor function in the absence of transforming growth factor beta I production and responsiveness. J Exp Med 2002; 196:237–246.
Mamura M, Lee W, Sullivan TJ et al. CD28 disruption exacerbates in Aammation in Tgf-betal-/-mice: in vivo suppression by CD4+CD25+ regulatory T-cells independent of autocrine TGF-beta1. Blood 2004; 103:4594–4601.
Huber S, Schramm C, Lehr HA et al. Cutting edge: TGF-beta signaling is required for the in vivo expansion and immuno suppressive capacity of regulatory CD4+CD25+ T-cells. J Immunol 2004; 173:6526–6531.
Nakamura K, Kitani A, Fuss I et al. TGF-beta 1 plays an important role in the mechanism of CD4+CD25+ regulatory T-cell activity in both humans and mice. J Immunol 2004; 172:834–842.
Kullberg MC, Hay V, Cheever AW et al. TGF-beta1 production by CD4+ CD25+ regulatory T-cells is not essential for suppression of intestinal inflammation. Eur J Immunol 2005; 35:2886–2895.
Annunziato F, Cosmi L, Liotta F et al. Phenotype, localization and mechanism of suppressionof CD4(+) CD25(+) human thymocytes. J Exp Med 2002; 196:379–387.
Manzotti CN, Tipping H, Perry LC et al. Inhibition of human T-cell proliferation by CTLA-4 utilizes CD80 and requires CD25+ regulatory T-cells. Eur J Immunol 2002; 32:2888–2896.
Grohmann U, Orabona C, Fallarino F et al. CTLA-4-lg regulates tryptophan catabolism in vivo. Nat Immunol 2002; 3:1097–1101.
Grossman WJ, Verbsky W, Barchet W et al. Human T regulatory cells can use the perforin pathway to cause autologous target cell death. Immunity 2004; 21:589–601.
Gondek DC, Lu LF, Quezada SA et al. Cutting edge: contact-mediated suppression by CD4+CD25+ regulatory cells involves a granzyme B-dependent, perforin-independent mechanism. J Immunol 2005; 174:1783–1786.
Zhao DM, Thornton AM, DiPaolo RJ et al. Activated CD4+CD25+ T-cells selectively kill B-Iymphocytes. Blood 2006; 107:3925–3932.
Choi BM, Pae HO, Jeong YR et al. Critical role of heme oxygenase-l in Foxp3-mediated immune suppression. Biochem Biophys Res Commun 2005; 327:1066–1071.
Otterbein LE, Soares MP, Yamashita K et al. Heme oxygenase-l:unleashing the protective properties of heme. Trends Immunol 2003; 24:449–455.
Godfrey VL, Wilkinson JE, Rinchik EM et al. Fatallymphoreticular disease in the scurfy (sf) mouse requires T-cells that mature in a sf thymic environment: potential model for thymic education. Proc Natl Acad Sci USA 1991; 88:5528–5532.
Godfrey VL, Wilkinson JE, Russell LB. X-linked lymphoreticular disease in the scurfy (sf) mutant mouse. AmJ Pathol 1991; 138:1379–1387.
Fontenot JD, Gavin MA, Rudensky AY. Foxp3 programs the development and function of CD4+CD25+ regulatory T-cells. Nat Immunol 2003; 4:330–336.
Huter EN, Punkosdy GA, Glass DD et al. TGF-beta-induced Foxp3+ regulatory T-cells rescue scurfy mice. Eur J Immunol 2008; 38:1814–1821.
Thornton AM, Shevach EM. CD4+CD25+ immunoregulatory T-cells suppress polyclonal T-cell activation in vitro by inhibiting interleukin 2 production. J Exp Med 1998; 188:287–296.
Piccirillo CA, Shevach EM. Cutting edge: control of CD8+ T-cell activation by CD4+CD25+ immunoregulatory cells. J Immunol 2001; 167:1137–1140.
Azuma T, Takahashi T, Kunisato A et al. Human CD4+ CD25+ regulatory T-cells suppress NKT cell functions. Cancer Res 2003; 63:4516–4520.
Taams LS, van Amelsfort JM, Tiemessen MM et al. Modulation of monocyte/macrophage function by human CD4+CD25+ regulatory T-cells. Hum Immunol 2005; 66:222–230.
Lim HW, Hillsamer P, Banham AH et al. Cutting Edge: Direct Suppression ofB-Cells by CD4+CD25+ Regulatory T-Cells. J Immunol 2005; 175:4180–4183.
Cederbom L, Hall H, Ivars F. CD4+CD25+ regulatory T-cells down-regulate costimulatory molecules on antigen-presenting cells. Eur J Immunol 2000; 30:1538–1543.
Smyth MJ, Teng MW, Swann J et al. CD4+CD25+ T regulatory cells suppress NK cell-mediated immunotherapy of cancer. J Immunol 2006; 176:1582–1587.
Salomon B, Lenschow DJ, Rhee L et al. B7/CD28 costimulation is essential for the homeostasis of the CD4+CD25+ immunoregulatory T-cells that control autoimmune diabetes. Immunity 2000; 12:431–440.
Kohm AP, Carpentier PA, Anger HA et al. Cutting edge: CD4+CD25+ regulatory T-cells suppress antigen-specific autoreactive immune responses and central nervous system inflammation during active experimental autoimmune encephalomyelitis. J Immunol 2002; 169:4712–4716.
Furtado GC, Olivares-Villagome ZD, Curotto de Lafaille MA et al. Regulatory T-cells in spontaneous autoimmune encephalomyelitis. Immunol Rev 2001; 182:122–134.
Sakaguchi S, Sakaguchi N, Asano M et al. Immunologic self-tolerance maintained by activated T-cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerancecauses various autoimmune diseases. J Immunol 1995; 155:1151–1164.
Singh B, Read S, Asseman C et al. Control of intestinal inflammation by regulatory T-cells. Immunol Rev 2001; 182:190–200.
Lee JH, Wang LC, Lin YT et al. Inverse correlation between CD4+ regulatory T-cell population and autoantibody levels in paediatric patients with systemic lupus erythematosus. Immunology 2006; 117:280–286.
Morgan ME, Sutmuller RP, Witteveen HJ et al. CD25+ cell depletion hastens the onset of severe disease in collagen-induced arthritis. Arthritis Rheum 2003; 48: 1452–1460.
Suri-Payer E, Amar AZ, Thornton AM et al. CD4+CD2S+ T-cells inhibit both the induction and effector function of autoreactive T-cells and represent a unique lineage of immunoregulatory cells. J Immunol 1998; 160:1212–1218.
Chen J, Huoam C, Plain K et al. CD4(+), CD2S(+) T-cells as regulators of alloimmune responses. Transplant Proc 2001; 33:163–164.
Wood KJ, Sakaguchi S. Regulatory T-cells in transplantation tolerance. Nat Rev Immunol 2003; 3:199–210.
Oluwole SF, Oluwole OO, DePaz HA et al. CD4+CD2S+ regulatory T-cells mediate acquired transplant tolerance. Transpl Immunol 2003; 11:287–293.
Belkaid Y, Piccirillo CA, Mendez S et al. CD4+CD2S+ regulatory T-cells control Leishmania major persistence and immunity. Nature 2002; 420:502–507.
Hori S, Carvalho TL, Demengeot J. CD2S+CD4+ regulatory T-cells suppress CD4+ T-cell-mediated pulmonary hyperinAammation driven by Pneumocystis carinii in immunodeficient mice. Eur J Immunol 2002; 32:1282–1291.
Rouse BT, Sarangi PP, Suvas S. Regulatory T-cells in virus infections. Immunol Rev 2006; 212:272–286.
Kullberg MC, Jankovic D, Gorelick PL et al. Bacteria-triggered CD4(+) T regulatory cells suppress Helicobacter hepaticus-induced colitis. J Exp Med 2002; 196:505–515.
Kawaida H, Kono K, Takahashi A et al. Distribution of CD4+CD2Shigh regulatory T-cells in tumor-draining lymph nodes in patients with gastric cancer. J Surg Res 2005; 124:151–157.
Woo EY, Chu CS, Goletz TJ et al. Regulatory CD4(+)CD2S(+) T-cells in tumors from patients with early-stage nonsmall cell lung cancer and late-stage ovarian cancer. Cancer Res 2001; 61:4766–4772.
Somasundaram R, Jacob L, Swoboda R et al. Inhibition of cytolytic T-Iymphocyte proliferation by autologous CD4+/CD2S+ regulatory T-cells in a colorectal carcinoma patient is mediated by transforming growth factor-beta. Cancer Res 2002; 62:5267–5272.
Liyanage UK, Moore TT, Joo HG et al. Prevalence of regulatory T-cells is increased in peripheral blood and tumor microenvironment of patients with pancreas or breast adenocarcinoma. J Immunol 2002; 169:2756–2761.
Ghiringhelli F, Larmonier N, Schmitt E et al. CD4+CD2S+ regulatory T-cells suppress tumor immunity but are sensitive to cyclophosphamide which allows immunotherapy of established tumors to be curative. Eur J Immunol 2004; 34:336–344.
Fattorossi A, Battaglia A, Ferrandina G et al. Lymphocyte composition of tumor draining lymph nodes from cervical and endometrial cancer patients. Gynecol Oncol 2004; 92:106–115.
Sasada T, Kimura M, Yoshida Y et al. CD4+CD2S+ regulatory T-cells in patients with gastrointestinal malignancies: possible involvement of regulatory T-cells in disease progression. Cancer 2003; 98:1089–1099.
Schaefer C, Kim GG, Albers A et al. Characteristics of CD4+CD2S+ regulatory T-cells in the peripheral circulation of patients with head and neck cancer. Br J Cancer 2005; 92:913–920.
Unitt E, Rushbrook SM, Marshall A et al. Compromised lymphocytes infiltrate hepatocellular carcinoma: the role of T-regulatory cells. Hepatology 2005; 41:722–730.
Berger CL, Tigelaar R, Cohen J et al. Cutaneous T-cell lymphoma: malignant proliferation of T-regulatory cells. Blood 2005; 105:1640–1647.
Marshall NA, Christie LE, Munro LR et al. Immunosuppressive regulatory T-cells are abundant in the reactive lymphocytes of Hodgkin lymphoma. Blood 2004; 103:1755–1762.
Beyer M, Kochanek M, Darabi K et al. Reduced frequencies and suppressive function of CD4+CD25hi regulatory T-cells in patients with chronic lymphocytic leukemia after therapy with Audarabine. Blood 2005; 106:2018–2025.
Chen GY, Chen C, Wang L et al. Cutting edge: Broad expression of the FoxP3 locus in epithelial cells: a caution against early interpretation of fatal inAammatory diseases following in vivo depletion of FoxP3-expressing cells. J Immunol 2008; 180:5163–5166.
Zuo T, Liu R, Zhang H et al. FOXP3 is a novel transcriptional repressor for the breast cancer oncogene SKP2. J Clin Invest 2007; 117:3765–3773.
Ebert LM, Tan BS, Browning J et al. The regulatory T-cell-associated transcription factor FoxP3 is expressed by tumor cells. Cancer Res 2008; 68:3001–3009.
Karanikas V, Speletas M, Zamanakou M et al. Foxp3 expression in human cancer cells. J Transl Med 2008; 6:19.
Yamaguchi T, Hirota K, Nagahama K et al. Control of immune responses by antigen-specific regulatory T-cells expressing the folate receptor. Immunity 2007; 27:145–159.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Landes Bioscience and Springer+Business Media
About this chapter
Cite this chapter
Kim, C.H. (2009). FOXP3 and Its Role in the Immune System. In: Maiese, K. (eds) Forkhead Transcription Factors. Advances in Experimental Medicine and Biology, vol 665. Springer, New York, NY. https://doi.org/10.1007/978-1-4419-1599-3_2
Download citation
DOI: https://doi.org/10.1007/978-1-4419-1599-3_2
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4419-1598-6
Online ISBN: 978-1-4419-1599-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)