Abstract
In this chapter, we provide an overview of the dynamical properties of spontaneous activity in the cortex, as represented by the subthreshold membrane potential fluctuations of the cortical neurons. First, we discuss the main findings from various intracellular recording studies performed in anesthetized animals as well as from a handful of studies in awake animals. Then, we focus on two specific questions pertaining to random and deterministic properties of cortical spontaneous activity. One of the questions is the relationship between excitation and inhibition, which is shown to posses a well-defined structure, owing to the spatio-temporal organization of the spontaneous activity in local cortical circuits at the millisecond scale. The other question regards the spontaneous activity at a scale of seconds and minutes. Here, examination of repeating patterns in subthreshold voltage fluctuations failed to reveal any evidence for deterministic structures.
Equal contribution.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
Introduction
Even in the absence of sensory stimuli, cortical activity is highly prominent. At the single-cell level, spontaneous activity in the cortex is observed using extracellular, intracellular, and calcium imaging recordings, whereas populations of cells can be seen using voltage sensitive dyes. At a larger scale, spontaneous activity can be observed in EEG, MEG, and fMRI recordings. In this chapter, we focus on the ongoing cortical activity as expressed by the dynamics of the subthreshold membrane potential of single neurons. Since synaptic inputs are the main cause of membrane potential fluctuations in cortical neurons [51], this technique is one of the most powerful tools to probe the network activity. The intracellular recording technique provides the most accurate data in terms of spatial and temporal precision, which comes at the expense of low yield of recorded cells and limited recording duration, because of the mechanical sensitivity of the technique. Nevertheless, an increasing number of studies have used this method to unveil the dynamics of spontaneous activity in the cortex.
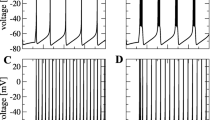
A particularly distinctive feature of the subthreshold dynamics in cortical neurons is the appearance of Up-Down states of membrane potential, originally described in anesthetized cats [52] and rats [17]. The Up-Down dynamics is characterized by large (10–20 mV) depolarizations relative to the baseline potential, lasting for several hundreds of milliseconds (the Up state), resulting in bimodal membrane potential distribution (Fig. 1a). This activity pattern was also observed in other species, including mice [42] and ferrets [23, 25]. Indirect EEG evidence for the presence of Up-Down states is also available for monkeys [39] and humans [4, 52]. In a series of studies in drug-free cats, it was found that Up-Down dynamics occurs during slow wave sleep (SWS) [53, 54]. Similar behavior during SWS and periods of drowsiness was observed in rats and mice as well [33, 42]. On the other end of scale, Up-Down dynamics was also reproduced in slices [49].
Examples of subthreshold spontaneous activity. (a) Up-Down dynamics in a neuron in the primary somatosensory cortex (S1) of ketamine-anesthetized rat, and the resulting membrane potential distribution. (b) Spontaneous activity in parietal association cortex of an awake cat, data from [9]. (c) Spontaneous activity in A1 of an awake rat, data from [27]. (d) Spontaneous activity in S1 of an awake rat, data from [40]. (e) Spontaneous activity in S1 of an awake mouse, data from [18] (To have a uniform scale, in all panels the data is reprinted in modified form from the original publication, and spikes are cut.)
While Up-Down dynamics is readily observed under some conditions of anesthesia (urethane, ketamine-xylazine), quite a different activity pattern, characterized by rather short (10–50 ms) depolarizations and membrane potential distribution that is not bimodal, emerges with other anesthetics (most distinctively the inhaled ones, such as isoflurane and halothane). This kind of activity appears to be a manifestation of lighter anesthesia when compared with the Up-Down dynamics, since the bimodal distribution of the membrane potential tends to appear when the concentration of the inhaled anesthetic is increased (unpublished results). Furthermore, under light gas anesthesia membrane dynamics is more similar to the activity observed in awake animals (see below).
Since it is plausible that the spontaneous dynamics in awake animals differs substantially from the anesthetized condition, intracellular recordings of cortical neurons in awake animals have been performed as well. Rather unfortunately these data are also most experimentally demanding to obtain, since intracellular recordings are extremely sensitive to mechanical instabilities, which are almost inevitable in awake, drug-free animals. At present time only a handful of such studies were performed, mostly in head fixed animals: monkeys [14, 35], cats [8, 53, 54], rats [12, 20, 22, 34, 40], mice [18, 43], and bats [16]. A methodology for whole-cell recording in behaving rodents is being developed as well [32].
Perhaps somewhat surprisingly, there exist large discrepancies between these studies. Two recent investigations reported diametrically opposing results: one group recorded neurons from the primary auditory cortex (A1) of rats [20, 27] and the other recorded from the parietal association cortex in cats [46]. According to Zador and his colleagues, the spontaneous subthreshold activity in the rat A1 is characterized by infrequent large positive excursions (“bumps”), resulting in membrane potential distribution with sharp peak and heavy tail at its positive side (average kurtosis of ∼ 15), quite distinct from the Gaussian distribution. On the contrary, in [46] the membrane potential exhibits activity resembling a continuous Up state, characterized by frequent, small fluctuations and membrane potential distribution which is close to normal. In particular, independent excitatory and inhibitory synaptic inputs that follow the Ornstein–Uhlenbeck stochastic differential equation are shown to provide a rather accurate approximation of the observed activity.
Intracellular recordings in awake animals were carried out in several additional works, but unlike the two papers above, in these studies the investigation of spontaneous dynamics was not the primary goal. Nevertheless, they provide an additional opportunity to examine the ongoing activity in the cortex. In most of these studies recordings were conducted in the rodent barrel cortex: [12, 18, 22, 34, 43]. When inspecting these recordings, as well as our own (Fig. 1d), the dynamics appears to be somewhere in between the two extremes of [20, 27] and [46]. On the one hand, the potential fluctuations do not seem to be produced by entirely uncorrelated synaptic inputs, as suggested in [46] while at the same time the bumps are smaller and more frequent than in [20]. In particular, in our recordings voltage distribution is approximately normal (kurtosis ∼ 0). However, we note that the presently available experimental data on the patterns of spontaneous activity in the barrel cortex of awake rodents are not fully consistent on their own, since recordings in mice [18, 43], see Fig. 1e, show a very bumpy activity. In these mice studies, bump amplitude appears to be several times larger than in rats and more importantly their durations are substantially longer than in the rat traces.
At the present stage we are not aware of any persuasive explanation for the discrepancies just described. Possible factors that might contribute to the observed differences are the animal species used, the cortical areas, layers, and specific neuron types from which the recordings were made, as well as the state of the animal (its level of stress, alertness, etc.). At the first sight the discrepancies between the handful of currently available datasets seem to be of a highly significant nature. Because ongoing activity can have substantial effect on the neural response to sensory stimuli, e.g., see [24, 42], cortical spontaneous activity may play a significant role in sensory processing. However, it is not clear whether the differences in spontaneous cortical dynamics are manifested during behavioral states, such as sensory processing, memory tasks, attention, and awareness. Though it is unlikely, it might be the case that these large differences from the point of view of the researcher are of no major importance for the processing of information in the cortex.
Synchrony in Spontaneous Activity
A significant difference in the amount of synchrony at the network level exists between the model of spontaneous activity proposed in [46] (Fig. 1b) and that of [20, 27] (Fig. 1c). In the first case, the dynamics is suggested to be asynchronous, with each presynaptic neuron firing independently of the others. However, the distinctive short bumps, as in Fig. 1c, indicate that firing of hundreds of presynaptic neurons is synchronized, since unitary synaptic potentials (uPSPs) are of the order of 1 mV or less. Owing to the enormous connectivity in the cortex, even if the presynaptic neurons do fire in synchrony, it is possible that nearby neurons receive inputs from independent pools of inputs. Simultaneous dual intracellular recordings, however, indicate that neurons in the local cortical network receive synaptic inputs with highly similar pattern and magnitude. In anesthetized animals, dual recordings reveal a very high correlation between the subthreshold activities in pairs of cells (Fig. 2a) [25, 31, 40, 56]. Since there is good evidence that the probability of nearby cells to receive inputs from the same presynaptic neuron is low, e.g., [50], and since a lag of several milliseconds may exist between their activities [31, 33, 40, 56], this synchrony indicates that most of the cortical spontaneous activity consists of waves, with large portions of local cortical circuits participating in their propagation [29, 33, 42].
Correlation of excitatory and inhibitory synaptic inputs during spontaneous activity. (a–d) Examples of simultaneous dual intracellular recordings, when both cells are hyperpolarized (a), depolarized (b) and intermixed (c, d). (e) Synaptic events in the second cell (Cell 2) of a pair are shown at the bottom, sorted by their amplitude (indicated by the color intensity). The corresponding events in Cell 1 are shown above with the same color. At first, both neurons were in the hyperpolarized mode. Then, the first cell was recorded in the depolarized mode. (f) Amplitudes of the events presented in (e). A significant correlation between the amplitudes is clearly visible in each case.
It was shown that synchrony in the cortex increases with the level of anesthesia [15]. For example, the Up-Down activity (Fig. 1a) is synchronous across large areas of the cortex (several mm apart) [56]. The high synchrony of large neuronal populations during Up-Down activity is further evidenced by high correlation between membrane potential and EEG, e.g., see [52]. Moreover, even the small synaptic events that appear within individual Up-states are synchronized across cells [25].
Under lighter anesthesia conditions that do not exhibit Up-Down dynamics, coherent subthreshold activity in pairs of nearby neurons is still observed even though global brain synchrony is very low, which is evident from the low correlation of EEG and membrane potential [31, 40]. Possibly this kind of local synchrony is similar to the synchrony existing within single Up states. The degree of synchrony in an awake animal is probably even lower than in the lightly anesthetized one; however, recent imaging studies and dual intracellular recordings in awake animals [43] show that it does not disappear altogether.
Excitation and Inhibition During Spontaneous Activity
The interplay between the excitatory and inhibitory synaptic inputs is a long studied topic in the computational neuroscience community. A careful examination of the spiking statistics of single cortical neurons has suggested that they are constantly bombarded by excitatory and inhibitory inputs that on average balance each other [47, 48]. Furthermore, theoretical studies showed that networks which exhibit sustained activity with an approximate balance between the excitatory and inhibitory neurons indeed exist, e.g., see the review in [55].
Intracellular recordings allow direct measurement of the excitatory and the inhibitory synaptic inputs associated with some reproducible stereotypical event (such as a sensory stimulus), by a method introduced in [11] (for an up-to-date review see [38]). Using this methodology, a balanced excitatory and inhibitory activity was indeed discovered during Up states in the ferret cortex, both in vitro [49] and in vivo [23]. Specifically, it was found that in the beginning of an Up state, both synaptic conductances are high and they tend to progressively decrease, but their ratio remains constant and approximately equal to 1. It should be noted, however, that questions relating to the balance between excitation and inhibition during the Up state are still not settled. A study of Up-Down activity in association cortex of awake cats [46] reports inhibitory conductance that is several times higher than the excitatory one. On the contrary, it was also argued that during Up states in the rat somatosensory cortex the inhibitory conductance is only about 10% of the excitatory conductance and that the duration of an Up state is at least partially determined by intrinsic mechanisms [58].
It is important to observe that a particular membrane potential value can be produced by different combinations of excitatory and inhibitory synaptic inputs. For example, a positive change in membrane potential can be caused both by an increased excitation without any significant change in inhibition, and by withdrawal of inhibitory input (disinhibition). Hence, the single-cell conductance measurement methodology, whether in voltage or current clamp mode, can only provide the mean relation between these inputs, calculated from the average event recorded at different holding potentials. Because of this important limitation, the relationship between the excitatory and inhibitory inputs during spontaneous activity which does not exhibit stereotypic Up states remained unknown.
As we already discussed at length in the Introduction, different studies report very distinct dynamics of ongoing activity. However, the presence of synchrony in inputs of nearby neurons appears to be common to all types of activity, since it was observed in all studies involving dual intracellular recordings or single-electrode intracellular with nearby imaging or LFP recordings [19, 31, 40, 42, 56], though to a different degree. This synchrony provides a method for measuring at the same time both the excitatory and the inhibitory synaptic inputs to the local circuit, by means of simultaneous dual intracellular recording [40]. In each pair of neurons, one cell is recorded near the reversal potential of inhibition so that positive excursions of its membrane potential reflect excitatory currents, at the same time a positive current is injected into the other cell to reveal inhibitory potentials. In fact, this is the only presently available experimental methodology that provides an adequate single-trial picture of the magnitude and timing of both excitatory and inhibitory inputs and that is suitable for elucidating the excitatory–inhibitory dynamics during ongoing activity and evoked responses. An example of such a recording is presented in Fig. 2. The two cells receive synchronized excitatory (Fig. 2a) and inhibitory (Fig. 2b) inputs. Furthermore, when one cell was depolarized (by positive current injection) to reveal inhibitory potentials, while the second cell was recorded near its resting level (Fig. 2c and vice versa in Fig. 2d), a high synchrony between the excitatory and inhibitory potentials was revealed.
The shape and amplitude of the synaptic events are highly variable when recorded at the neuron’s resting potential (e.g., as in Fig. 2a). A priori, the variability in the amplitude of these events could reflect considerable changes in the contribution of the excitatory and inhibitory synaptic inputs, ranging between the following two diametrically opposing possibilities. It might be the case that large bumps occur when the inhibitory activity, which can shunt the excitation, is weak, whereas small bumps reflect shunted excitatory inputs. Alternatively, both types of synaptic inputs might reflect the overall level of activity in the local network and go hand in hand. We have used the same experimental approach to resolve this issue. We found that the amplitudes of spontaneous events were significantly correlated, both for the depolarizing potentials, when the two cells were held near their resting potential, and when one cell was depolarized to reveal inhibitory potentials (Fig. 2e–f). Hence, the latter alternative is correct; that is, the larger the excitatory drive in the local circuit, the larger the inhibitory one. In addition, we used one of the cells as a reference to measure the relative timing of excitatory and inhibitory bumps in the other cell. We found that during spontaneous activity the onset of inhibition lags by few milliseconds behind the excitatory input (Fig. 3).
The same experimental method was used more recently in an in vitro study of gamma oscillations in the hippocampus. This study also found a tight amplitude correlation between the excitatory and inhibitory inputs when analyzed on a cycle by cycle basis [6]. The coordinated activity of excitation and inhibition across two neurons strongly suggests that variations in excitation and inhibition reflect mostly changes in the local network activity rather than “private” variability of the inputs of individual cells.
Repeating Patterns in the Spontaneous Subthreshold Membrane PotentialFluctuations of Cortical Neurons
The notion that distinct and large depolarizing excursions of membrane potential reflect synchronized activity in the network was used in a recent study which reported on the surprising finding that patterns of spontaneous synaptic activity can repeat with high precision [28] (see also Chapter 3 in this volume). In this work, long and continuous records of spontaneous subthreshold membrane potential fluctuations obtained both in vivo and in brain slices were analyzed, and it was found that specific patterns of activity can reappear seconds to minutes apart (Fig. 4). These repeats of membrane potential fluctuations, also termed “motifs” [28, 37], typically span 1–2 s and include several large bumpy synaptic potentials separated by quiescent periods. What cortical mechanism could generate such precisely timed and long activity patterns? In [28], motifs were suggested to provide a strong supporting evidence for the existence of special cortical mechanisms, such as the synfire chain operation mode of the cortical network, which generate exact firing patterns with a millisecond precision.
Examples of repeating motifs from two different cells. The similarity between motif repetitions was quantified by the High Resolution Index (HRI, for further details see [28, 37] and Chapter 3 in this volume). Each row presents examples from the same cell, first the two motifs with the highest HRI values, then a motif which has the 1,000th highest HRI rank (still showing a marked similarity). Such highly similar motifs were found in all the recorded cells in spite of the differences in the statistical properties of their subthreshold activities.
According to the synfire chain model, cortical cells are organized into pools of cells [1]. Each pool is connected to the next by a set of diverging and converging connections, forming together a chain of pools. Despite the low transmission reliability between pairs of cells, a secure transmission of information is suggested to be accomplished by synchronous firing within each pool and its propagation from a pool of neurons to the following one. That is, after one pool of neurons was synchronously activated, a synchronous activation of the next pool of neurons follows with a typical synaptic delay. Processing of information may include multiple feedbacks, so that a single neuron might be a member in several pools along the chain. For such a neuron, activation of the chain would result in a sequence of large synaptic potentials, where each one is generated by a different group of presynaptic neurons. Because of the high reliability of the chain, such sequence of synaptic potentials is expected to repeat with high temporal precision once the first pool of neurons in the chain is reactivated, generating a repeated firing and synaptic pattern (motif).
The synfire chain model proposes an efficient mechanism for propagating a signal with a low number of spikes, in addition to a compact way of information encoding. The amount of information that might be encoded by precise temporal structures is far larger when compared with encoding by spike rate or spike count alone [36]. If motifs do not result from stimulation locking [41], then they may support higher level processes such as binding together different features of an object [1, 21, 57]. Few synaptic connections between reverberating synfire chains may facilitate the binding of several smaller chains, representing specific features, into a larger super-assembly [2, 3, 10] which could be a substrate of higher level perceptual processes. For example, it has been recently suggested that large-scale spatio-temporal activation patterns spreading over superior-temporal and inferior-frontal cortices, observed during processing of speech stimuli, are best explained by interarea synfire chain propagation [45].
The synfire chain model was supported by an analysis of spike sequences recorded simultaneously from several neurons of the frontal cortex of behaving monkeys [3, 44], which showed that the number of spike motifs exceeded what could be expected by chance in surrogate data [3]. While the propagation of the signal might be purely feedforward, so that a motif includes only one spike per neuron, most of the motifs that were described in the above studies were composed of patterns of spikes originating from a single unit. Therefore, the authors concluded that synfire chains typically contain feedback connections [3, 44].
Subsequent studies questioned the significance of temporal patterns by comparing the number of repeating patterns of spikes in the recordings and in surrogate data generated by shuffling the original spike times using different stochastic models [7, 41]. Their analysis of spike times recorded from the lateral geniculate nucleus (LGN) and primary visual cortex of behaving monkeys showed that adding more constraints to the surrogate data brings the number of repeating patterns closer to the recorded data. Hence, choosing the right stochastic model has critical consequences regarding the conclusions. For example, in surrogates that preserved the number of spikes per trial and the firing probability distribution, the number of motifs was much closer to the original than in surrogates generated with more simple assumptions such as a Poisson process. Baker and Lemon and Oram et al. [7, 41] therefore suggested that the appearance of spike motifs reflects the coarse dynamics of firing rate modulations rather than the existence of special network mechanisms for their generation. A lack of evidence for the occurrence of precise spike patterns beyond what is expected by chance was also reported in [7], where spike patterns recorded from multiple units of the primary motor cortex (M1) and the supplementary motor area (SMA) in monkeys were analyzed.
Research aimed to find in the subthreshold traces the synaptic inputs that create the precise spike patterns observed in some of the above described studies was reported by Ikegaya et al. in [28]. In contrast to the above studies, which searched for precise spike patterns in awake behaving monkeys, the repeating patterns reported in [28] were found in subthreshold membrane potential fluctuations recorded in-vivo in anesthetized cats and were generated spontaneously, in the absence of external sensory stimulation. Recently, we have reexamined the statistical significance of such spontaneously repeating patterns in intracellular recordings from the rat barrel cortex (S1) and the cat primary visual cortex (V1) of anesthetized animals [37]. In most of the recordings, the dynamics of spontaneous activity was similar to those reported in [28]. Using a search algorithm similar to the one described by Ikegaya and his colleagues, we found a large number of motifs. To test their statistical significance, we used three different methods to generate surrogate data, each corresponding to a different model of randomness. In the first method, the surrogate data were constructed by a time domain shuffling of the original trace (Fig. 5a). In the second method, the data were randomized in the frequency domain. In the third method, the parameters of Poisson distributed synaptic inputs of a simulated passive cell were optimized to elicit an activity with dynamics similar to that of the recorded cell. Perhaps surprisingly, a large number of motifs were found in all types of surrogate data.
Generation of surrogate data by time domain interval shuffling. (a) Time domain interval shuffling: Using two levels of potentials (determined from 1/3 and 2/3 of the density distribution), the data were fragmented into short segments. Each segment starts and ends at one of the two levels and its duration was the longest possible below 500 ms. Five different segments are marked. The fragments were then randomly assembled to generate a new continuous voltage trace. (b) Membrane potential distribution and (c) power spectrum of the recorded data and its time domain shuffled surrogate.
The close resemblance between the distributions of similarity scores of motifs found in physiological spontaneous activity and in the different types of surrogate data (Fig. 6a) suggests that the motifs in physiological data emerge by chance. Of the three methods for generating surrogate data, the time domain interval shuffling preserved both the power spectrum and the voltage distribution of recorded data most accurately (Fig. 5b–c). Surrogates produced with this method also had motif statistics that were closest to the original. These results suggest that physiological motifs could simply arise as a result of the coarse constraints on the subthreshold fluctuations dynamics, imposed by a wide range of properties of the cortical neuronal networks. An important issue of concern with any method that produces surrogate data from the original recordings is its effect on genuine motifs, if they do exist. To test this, we have implanted several highly similar repeats into a long continuous recording. Such artificial motif is easily detected by comparing the similarity scores distribution of the synthetic data to its shuffled versions (Fig. 6b). These results further support the conclusion that physiological records did not contain repeating motifs above what is expected at chance level.
(a) Example of HRI scores distributions in one of the cells analyzed in [37], sorted in decreasing order. Recorded data and the three types of surrogate data have very similar distributions of HRI scores. The interval shuffled curve is the average of 40 independent shuffles of the original recording, and the dashed green curves are its confidence intervals p = 0. 99. (b) Rank ordered HRI scores of all motifs found in surrogate data of 20 min in duration with 20 implanted motif repeats, and its 40 interval shuffled surrogates. To test whether motifs of high similarity that occur beyond chance level could be detected, we implanted into a surrogate data of 20 min in duration a single motif of 1-s duration that repeated every minute on average. The surrogate was produced from physiological recordings by shuffling it in the time domain. It is evident that the HRI scores of the top ranked motifs that were found in data with implanted motifs are much higher than those of all 40 shuffles. These results demonstrate that even a single motif that repeats several times with high similarity is identified by our methods.
Additional statistic that compared between physiological and surrogate data sets was the number of times motifs reappear. This particular comparison is of a particular interest since a theoretical analysis of synfire chains [26] demonstrates that typical cortical columns are not large enough to support many synfire chains of a duration as long as 1 s. Therefore, if the recorded 1 s repeats are generated at the column level, one would expect a small number of motifs that repeat numerous times rather than a large number of different motifs repeating small number of times. The analysis performed in [37] found that the statistics of the number of motif repetitions in the original traces and the surrogates was the same.
The tight continuous synchrony of spontaneous membrane potential fluctuations of cortical neurons that is observed across extended cortical regions [5, 56] also stands in contrast to the idea that the observed long repeating patterns reflect a propagation of synfire chain within a single cortical column. This discrepancy is further demonstrated by our experiment in which intracellular voltages were recorded simultaneously from pairs of neurons (in the barrel cortex of an anesthetized rat). Some of the synchronized cells were laterally separated by about ∼ 500 μm, thus typically belonged to two distinct columns and to different layers. The same measure that was used to quantify the degree of similarity between repeats of a motif may also be used to measure similarity of simultaneously (or almost simultaneouslyFootnote 1) recorded epochs in a pair of cells. The inter-neuron similarity of simultaneous intervals was very high, much higher than the similarity between repeats within the recording of individual neuron (Fig. 7). This indicates that the vast majority of reoccurring temporal patterns in the spontaneous activity of the cortex do not reflect a column specific processing, rather they are a consequence of waves propagating across wider cortical regions. The dual recordings also provide an answer to an issue of concern not fully addressed in [37], regarding the possibility that the stochastic nature of motifs is due to intrinsic noise, unrelated to network activity. The results in Fig. 7 indicate that this is not the case. Finally, we note that [28] described in vitro motifs of firing sequences within a population of neurons whose size is of the order of a single cortical column, an indication that the mechanisms that are involved in their generation are different than those that may support the generation of motifs in the much larger scales of in vivo cortical activity.
Comparison between the similarity of motif repeats that were found within a continuous recording of a single cell, and the similarity of the first repeat to the corresponding epoch in another simultaneously recorded cell. Most points lie above the diagonal line. This implies that nearby cells have similar patterns of synaptic inputs and that these patterns do not reoccur with a comparable similarity at different times.
The above results provide no evidence for the existence of cortical mechanisms to support precisely timed, long-lasting spike patterns that propagate recurrently in the local cortical network. These results are consistent with other electrophysiological studies in vivo [30, 33, 48] that do not support a millisecond precision in cortical function. Moreover, [13] showed that the jitter of firing (measured by the standard deviation of the latency across multiple trials following local stimulation) is directly proportional to the latency of the propagating wave that activates them, suggesting that precise propagation of spike patterns cannot be maintained for long durations. However, we cannot exclude the existence of statistically significant motifs of much shorter duration. A study of Luczak et al. [33] has shown that different cortical cells exhibit unique patterns of modulation in firing rate that are associated with the onset of Up states and last around 100 ms. The typical patterns emerge regardless of the direction from which the propagating wave arrives, and therefore it has been hypothesized that they reflect the local functional organization of the network [33]. In summary, our data provide indications that long motifs of spontaneous activity are generated stochastically as a result of the coarse dynamics of cortical activity.
Conclusions
Spontaneous activity in the cortex was extensively investigated in the recent years, from the level of individual cells all the way to activity in whole cortical areas. The intracellular recording technique provides a powerful tool for probing cortical dynamics and synaptic inputs during spontaneous and evoked activities. In particular, our own works examined different and even opposing views regarding the organization of neuronal activity in the local cortical network, ranging from approximately independent firing to highly structured spike patterns, generated by synfire chains. On one hand, using in-vivo dual intracellular recordings of nearby cortical cells, we have shown that a highly coordinated activity in the local network exists at the millisecond time scale. This coordinated activity, in which both excitatory and inhibitory inputs participate, is manifested as brief bursts of network activity, interleaved with lower activity levels. On the contrary, on the time scale of seconds, spontaneous network activity appears to be governed by stochastic mechanisms, with no strong evidence for temporal patterns repeating above chance levels.
Notes
- 1.
In most of the cases, the temporal pattern of synaptic inputs recorded in one cell appeared in the other cell after a short delay of a few milliseconds, an indication of a traveling wave in the cortex.
References
Abeles M (1991) Corticonics. Cambridge: Cambridge University press.
Abeles M (2004) Neuroscience. Time is precious. Science 304:523–524.
Abeles M, Bergman H, Margalit E, Vaadia E (1993) Spatiotemporal firing patterns in the frontal cortex of behaving monkeys. J Neurophysiol 70:1629–1638.
Achermann P, Borbely AA (1997) Low-frequency ( < 1 Hz) oscillations in the human sleep electroencephalogram. Neuroscience 81:213–222.
Arieli A, Shoham D, Hildesheim R, Grinvald A (1995) Coherent spatiotemporal patterns of ongoing activity revealed by real-time optical imaging coupled with single-unit recording in the cat visual cortex. J Neurophysiol 73:2072–2093.
Atallah BV, Scanziani M (2008) Proportional excitatory and inhibitory conductances are maintained during gamma oscillations. In: COSYNE. Salt Lake City, UT.
Baker SN, Lemon RN (2000) Precise spatiotemporal repeating patterns in monkey primary and supplementary motor areas occur at chance levels. J Neurophysiol 84:1770–1780.
Baranyi A, Szente MB, Woody CD (1993) Electrophysiological characterization of different types of neurons recorded in vivo in the motor cortex of the cat. I. Patterns of firing activity and synaptic responses. J Neurophysiol 69:1850–1864.
Bazhenov M, Timofeev I, Steriade M, Sejnowski TJ (2002) Model of thalamocortical slow-wave sleep oscillations and transitions to activated states. J Neurosci 22:8691–8704.
Bienenstock E (1995) A model of neocortex. Network-Computation in Neural Systems 6:179–224.
Borg-Graham LJ, Monier C, Fregnac Y (1998) Visual input evokes transient and strong shunting inhibition in visual cortical neurons. Nature 393:369–373.
Bruno RM, Sakmann B (2006) Cortex is driven by weak but synchronously active thalamocortical synapses. Science 312:1622–1627.
Buonomano DV (2003) Timing of neural responses in cortical organotypic slices. Proc Natl Acad Sci U S A 100:4897–4902.
Chen D, Fetz EE (2005) Characteristic membrane potential trajectories in primate sensorimotor cortex neurons recorded in vivo. J Neurophysiol 94:2713–2725.
Contreras D, Steriade M (1997) State-dependent fluctuations of low-frequency rhythms in corticothalamic networks. Neuroscience 76:25–38.
Covey E, Kauer JA, Casseday JH (1996) Whole-cell patch-clamp recording reveals subthreshold sound-evoked postsynaptic currents in the inferior colliculus of awake bats. J Neurosci 16:3009–3018.
Cowan RL, Wilson CJ (1994) Spontaneous firing patterns and axonal projections of single corticostriatal neurons in the rat medial agranular cortex. J Neurophysiol 71:17–32.
Crochet S, Petersen CC (2006) Correlating whisker behavior with membrane potential in barrel cortex of awake mice. Nat Neurosci 9:608–610.
DeWeese MR, Zador AM (2004) Shared and private variability in the auditory cortex. J Neurophysiol 92:1840–1855.
DeWeese MR, Zador AM (2006) Non-Gaussian membrane potential dynamics imply sparse, synchronous activity in auditory cortex. J Neurosci 26:12206–12218.
Engel AK, Konig P, Kreiter AK, Schillen TB, Singer W (1992) Temporal coding in the visual cortex: new vistas on integration in the nervous system. Trends Neurosci 15:218–226.
Fee MS (2000) Active stabilization of electrodes for intracellular recording in awake behaving animals. Neuron 27:461–468.
Haider B, Duque A, Hasenstaub AR, McCormick DA (2006) Neocortical network activity in vivo is generated through a dynamic balance of excitation and inhibition. J Neurosci 26:4535–4545.
Hasenstaub A, Sachdev RN, McCormick DA (2007) State changes rapidly modulate cortical neuronal responsiveness. J Neurosci 27:9607–9622.
Hasenstaub A, Shu Y, Haider B, Kraushaar U, Duque A, McCormick DA (2005) Inhibitory postsynaptic potentials carry synchronized frequency information in active cortical networks. Neuron 47:423–435.
Herrmann M, Hertz JA, Prugelbennett A (1995) Analysis of synfire chains. Network: Comput Neural Syst 6:403–414.
Hromádka T (2007) Representation of Sounds in Auditory Cortex of Awake Rats, PhD thesis.
Ikegaya Y, Aaron G, Cossart R, Aronov D, Lampl I, Ferster D, Yuste R (2004) Synfire chains and cortical songs: temporal modules of cortical activity. Science 304:559–564.
Kenet T, Bibitchkov D, Tsodyks M, Grinvald A, Arieli A (2003) Spontaneously emerging cortical representations of visual attributes. Nature 425:954–956.
Kerr JN, Greenberg D, Helmchen F (2005) Imaging input and output of neocortical networks in vivo. Proc Natl Acad Sci U S A 102:14063–14068.
Lampl I, Reichova I, Ferster D (1999) Synchronous membrane potential fluctuations in neurons of the cat visual cortex. Neuron 22:361–374.
Lee AK, Manns ID, Sakmann B, Brecht M (2006) Whole-cell recordings in freely moving rats. Neuron 51:399–407.
Luczak A, Bartho P, Marguet SL, Buzsaki G, Harris KD (2007) Sequential structure of neocortical spontaneous activity in vivo. Proc Natl Acad Sci U S A 104:347–352.
Margrie TW, Brecht M, Sakmann B (2002) In vivo, low-resistance, whole-cell recordings from neurons in the anaesthetized and awake mammalian brain. Pflugers Arch 444:491–498.
Matsumura M, Chen D, Sawaguchi T, Kubota K, Fetz EE (1996) Synaptic interactions between primate precentral cortex neurons revealed by spike-triggered averaging of intracellular membrane potentials in vivo. J Neurosci 16:7757–7767.
McClurkin JW, Optican LM, Richmond BJ, Gawne TJ (1991) Concurrent processing and complexity of temporally encoded neuronal messages in visual perception. Science 253:675–677.
Mokeichev A, Okun M, Barak O, Katz Y, Ben-Shahar O, Lampl I (2007) Stochastic emergence of repeating cortical motifs in spontaneous membrane potential fluctuations in vivo. Neuron 53:413–425.
Monier C, Fournier J, Fregnac Y (2008) In vitro and in vivo measures of evoked excitatory and inhibitory conductance dynamics in sensory cortices. J Neurosci Methods 169:323–365.
Okamoto H, Isomura Y, Takada M, Fukai T (2006) Combined Modeling and Extracellular Recording Studies of Up and Down Transitions of Neurons in Awake or Behaving Monkeys. In: The Basal Ganglia VIII, pp. 555–562.
Okun M, Lampl I (2008) Instantaneous correlation of excitation and inhibition during ongoing and sensory-evoked activities. Nat Neurosci 11:535–537.
Oram MW, Wiener MC, Lestienne R, Richmond BJ (1999) Stochastic nature of precisely timed spike patterns in visual system neuronal responses. J Neurophysiol 81:3021–3033.
Petersen CC, Hahn TT, Mehta M, Grinvald A, Sakmann B (2003) Interaction of sensory responses with spontaneous depolarization in layer 2/3 barrel cortex. Proc Natl Acad Sci U S A 100:13638–13643.
Poulet JF, Petersen CC (2008) Internal brain state regulates membrane potential synchrony in barrel cortex of behaving mice. Nature 454:881–885.
Prut Y, Vaadia E, Bergman H, Haalman I, Slovin H, Abeles M (1998) Spatiotemporal structure of cortical activity: properties and behavioral relevance. J Neurophysiol 79:2857–2874.
Pulvermuller F, Shtyrov Y (2009) Spatiotemporal signatures of large-scale synfire chains for speech processing as revealed by MEG. Cereb Cortex 19:79–88.
Rudolph M, Pospischil M, Timofeev I, Destexhe A (2007) Inhibition determines membrane potential dynamics and controls action potential generation in awake and sleeping cat cortex. J Neurosci 27:5280–5290.
Shadlen MN, Newsome WT (1994) Noise, neural codes and cortical organization. Curr Opin Neurobiol 4:569–579.
Shadlen MN, Newsome WT (1998) The variable discharge of cortical neurons: implications for connectivity, computation, and information coding. J Neurosci 18:3870–3896.
Shu Y, Hasenstaub A, McCormick DA (2003) Turning on and off recurrent balanced cortical activity. Nature 423:288–293.
Song S, Sjostrom PJ, Reigl M, Nelson S, Chklovskii DB (2005) Highly nonrandom features of synaptic connectivity in local cortical circuits. PLoS Biol 3:e68.
Steriade M (2001) Impact of network activities on neuronal properties in corticothalamic systems. J Neurophysiol 86:1–39.
Steriade M, Nunez A, Amzica F (1993) A novel slow ( < 1 Hz) oscillation of neocortical neurons in vivo: depolarizing and hyperpolarizing components. J Neurosci 13:3252–3265.
Steriade M, Timofeev I, Grenier F (2001) Natural waking and sleep states: a view from inside neocortical neurons. J Neurophysiol 85:1969–1985.
Timofeev I, Grenier F, Steriade M (2001) Disfacilitation and active inhibition in the neocortex during the natural sleep-wake cycle: an intracellular study. Proc Natl Acad Sci U S A 98:1924–1929.
Vogels TP, Rajan K, Abbott LF (2005) Neural network dynamics. Annu Rev Neurosci 28:357–376.
Volgushev M, Chauvette S, Mukovski M, Timofeev I (2006) Precise long-range synchronization of activity and silence in neocortical neurons during slow-wave oscillations. J Neurosci 26:5665–5672.
von der Malsburg C (1995) Binding in models of perception and brain function. Curr Opin Neurobiol 5:520–526.
Waters J, Helmchen F (2006) Background synaptic activity is sparse in neocortex. J Neurosci 26:8267–8277.
Acknowledgments
We would like to thank Profs. Carl Petersen, Igor Timofeev, and Tony Zador for providing examples of intracellular recordings in awake animals from experiments conducted in their laboratories (Fig. 1). We thank all the members of Lampl lab for their contribution to this work. This work was supported by grants from the Israel Science Foundation (1037/03, 326/07), the National Institute for Psychobiology in Israel, by the Henry S. and Anne Reich Research Fund for Mental Health, the Asher and Jeanette Alhadeff Research Award, and Sir Charles Clore fellowship.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Springer Science+Business Media, LLC
About this chapter
Cite this chapter
Okun, M., Mokeichev, A., Katz, Y., Lampl, I. (2009). On the Dynamics of Synaptic Inputs During Ongoing Activity in the Cortex. In: Josic, K., Rubin, J., Matias, M., Romo, R. (eds) Coherent Behavior in Neuronal Networks. Springer Series in Computational Neuroscience, vol 3. Springer, New York, NY. https://doi.org/10.1007/978-1-4419-0389-1_1
Download citation
DOI: https://doi.org/10.1007/978-1-4419-0389-1_1
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4419-0388-4
Online ISBN: 978-1-4419-0389-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)