Abstract
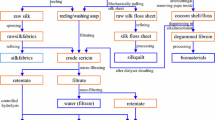
Mammalian cell cultures generally require supplementation with fetal bovine serum (FBS), or its replacement, into the culture media. Sera contain various unidentified and unknown factors and the risk of infections, including bovine spongiform encephalopathy (BSE), is of serious concern. Therefore, the supplementation of sera into culture media is a major obstacle for purification to recover cell products and this limits pharmaceutical acceptance of products. In this study, we examined whether the sericin protein, derived from the silkworm cocoon, can be effectively used as a substitute for FBS in mammalian cell culture. Together with fibroin, sericin is a major component of raw-silk and is removed from raw-silk by a treatment called degumming to make the silk lustrous and semitransparent. In order to investigate the effect of sericin on the proliferation and the productivity of mammalian cells, sericin was added to cultures of various mammalian cell lines, such as murine hybridoma 2E3-O. Sericin successfully accelerated the proliferation of the cells. Moreover, the production of MoAb by the hybridoma cells was also improved in the presence of sericin. Although heat easily denatures and inactivates most proteins, sericin maintained mitogenic activity after conventional autoclaving (20 minutes) and longer (60 minutes) autoclaving.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
- Bovine Spongiform Encephalopathy
- Mammalian Cell Line
- Mitogenic Activity
- Mammalian Cell Culture
- Viable Cell Number
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Terada, S., T. Nishimura, M. Sasaki, H. Yamada, M. Miki, 2002, Sercin, a protein derived from silkworms, accelerates the proliferation of several mammalian cell lines including a hybridoma, Cytotech. 40: 3–12
Kato, N., S. Sato, A. Yamanaka, H. Yamada, N. Fuwa and M. Nomura, 1998, Silk protein, sericin, inhibits lipid peroxidation and tyrosinase activity, Biosci. Biotechnol. Biochem. 62: 145–147
Sasaki, M., N. Kato, H. Watanabe and H. Yamada, 2000a, Silk protein, sericin, suppresses colon carcinogenesis induced by 1,2-dimethylhydrazine in mice, Oncol. Rep. 7: 1049–1052
Sasaki, M., H. Yamada and N. Kato, 2000b, Consumption of silk protein, sericin elevates intestinal absorption of zinc, iron, magnesium and calcium in rats, Nutr. Res. 20: 1505–1511
Takahashi, M., K. Tsujimoto, H. Yamada, H. Takagi and S. Nakamori, 2003, The silk protein, sericin, protects against cell death caused by acute serum deprivation in insect cell culture, Biotech. Lett. 25: 1805–1809
Tsujimoto, K., H. Takagi, M. Takahashi, H. Yamada and S. Nakamori, 2001, Cryoprotective effect of the serine-rich repetitive sequence in silk protein sericin, J. Biochem. 129: 979–986
Sasaki, M., Y. Kato, H. Yamada and S. Terada, Development of a novel serum-free freezing medium for mammalian cells using silk protein sericin, Biotechnology and Applied Biochemistry, in press
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2007 Springer
About this paper
Cite this paper
Terada, S., Takada, N., Itoh, K., Saitoh, T., Sasaki, M., Yamada, H. (2007). Silk Protein Sericin Improves Mammalian Cell Culture. In: Smith, R. (eds) Cell Technology for Cell Products., vol 3. Springer, Dordrecht. https://doi.org/10.1007/978-1-4020-5476-1_66
Download citation
DOI: https://doi.org/10.1007/978-1-4020-5476-1_66
Publisher Name: Springer, Dordrecht
Print ISBN: 978-1-4020-5475-4
Online ISBN: 978-1-4020-5476-1
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)