Abstract
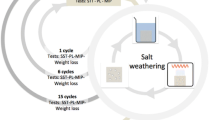
Salt crystals are able to exert stress on the pore walls in stone because there is a repulsive force between the salt and mineral surfaces, so that a film of supersaturated solution is in contact with the growing crystal. Damage from salt could be prevented if the repulsion were eliminated, so we have screened a variety of organic systems to find species that adsorb on carbonates and on salts. Several candidates were identified and tested for their influence on nucleation and growth of sodium sulfate; promising systems were applied to Indiana limestone, which was then subjected to cycles of soaking in sodium sulfate solution followed by drying. The treatment that offered the most protection was polyacrylic acid with very low molecular weight.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
REFERENCES
I.S. Evans, Salt crystallization and rock weathering, Rev. Géomorphologie dynamique XIX (4) 153–177 (1969-70)
Goudie and H. Viles, Salt Weathering Hazards (Wiley, Chichester, 1997)
G.W. Scherer, R. Flatt, and G. Wheeler, Materials Science Research for Conservation of Sculpture and Monuments, MRS Bulletin, Jan. 2001, pp. 44–50
G.W. Scherer, Crystallization in pores, Cement Concr. Res. 29(8) 1347–1358 (1999); Reply to discussion of Crystallization in pores, Cement and Concr. Res. 30(4), 673–675 (2000)
G.W. Scherer, Stress from crystallization of salt, Cement Concr. Res. 34, 1613–1624 (2004)
G.F. Becker and A.L. Day, Note on the linear force of growing crystals, J. Geology XXIV(4) 313–333 (1916)
S. Taber, The growth of crystals under external pressure, Am. J. Sci. 41, 532–556 (1916)
C.W. Correns, Growth and dissolution of crystals under linear pressure, Disc. Faraday Soc. 5, 267–271 (1949)
A.W. Neumann, S.N. Omenyi, and C.J. van Oss, Attraction and repulsion of solid particles by solidification fronts. 3. van der Waals interactions, J. Phys. Chem., 86, 1267– 1270 (1982)
Israelachvili, Intermolecular & Surface Forces, 2nd ed. (Academic, London, 1992)
R.J. Flatt, Salt damage in porous materials: how high supersaturations are generated, J. Cryst. Growth 242, 435–454 (2002)
J. Freundlich, Colloid & Capillary Chemistry (Methuen, London, 1926) pp. 154–157
G. Wulff, Zur Frage der Geschwindigkeit des Wachsthums und der Auflösung der Krystallfláchen (On the question of the growth and dissolution rates of crystalline faces), Z. Krist. 34, 449–530 (1901)
A.W. Adamson, Physical Chemistry of Surfaces, 5th ed., (Wiley, New York, 1990)
L.A. Rijniers, Salt crystallization In porous materials: an NMR study, Ph.D. thesis, Technische Universiteit Eindhoven, Nov. 2004
G.W. Scherer, Stress from crystallization of salt in pores, pp. 187–194 in Proc. 9th Int. Congress on Deterioration and Conservation of Stone, Vol. 1, ed. V. Fassina (Elsevier, Amsterdam, 2000)
S. Sexmith and E.Q. Petrey, The use of polyelectrolytes for scale control in sea water evaporators, Desalination 13, 87–90 (1973)
L. Gustafson, Polymer applications in stone: An attempt to decrease deterioration due to salt crystallization, Senior thesis, Dept. Chemical Eng., Princeton University, 1998
N. Alleyne, Art conservation: Testing interfacial energies, Senior thesis, Dept. Chemical Eng., Princeton University, 2001
L. Tangeman, The effects of polymer coatings in decreasing the interfacial energy between salt and stone, Senior thesis, Dept. Chemical Eng., Princeton University, 2002
M.M. Thomas, J.A. Couse, and J. Longo, Adsorption of organic compounds on carbonate minerals, Chemical Geology, 109, 201–213 (1993)
R.D. Cody, Organo-crystalline interactions in evaporite systems: The effects of crystallization inhibition, J. Sedimentary Petrology, 65 (5) 704–715 (1990)
S. Sarig, A. Glasner, and J. Epstein, Crystal habit modifiers, J. Crystal Growth, 28, 295– 299 (1975)
S. Sarig and F. Kahana, On the association between sparingly soluble carbonates and polyelectrolytes, J. Crystal Growth, 35, 145–152 (1976)
S. Sarig, F. Kahana, and R. Leshem, Selection of threshold agents for calcium sulfate scale control on the basis of chemical structure, Desalination, 17, 215–229 (1975)
N. Tsui, R.J. Flatt, G.W. Scherer, Crystallization damage by sodium sulfate, J. Cultural Heritage, 4, 109–115 (2003)
C.A. Price, The use of the sodium sulphate crystallisation test for determining the weathering resistance of untreated stone, in Proc. UNESCO-RILEM Symposium on the Deterioration and Protection of Stone Monuments, Vol. I. Sect. 3-6 (UNESCO/RILEM, Paris, 1978) pp. 1–24
G.W. Scherer, “Fundamentals of drying and shrinkage”, in Science of Whitewares, eds. V.E. Henkes, G.Y. Onoda, and W.M. Carty (Am. Ceram. Soc., Westerville, OH, 1996) pp. 199–211
E. Wendler, D.D. Klemm, and R. Snethlage, Consolidation and hydrohobic treatment of natural stone, in Proc. 5th Int. Conference on Durability of Building Materials and Components, eds. J.M. Baker, P.J. Nixon, A.J. Majumdar, and H. Davies (Chapman & Hall, London, 1991) pp. 203–212
K.L. Nagy, R.T. Cygan, C.S. Scotto, C.J. Brinker, and C.S. Ashley, Use of coupled passivants and consolidants on calcite mineral surfaces, in Materials Issues in Art and Archaeology V (Mater. Res. Soc., Pittsburgh, PA, 1997) pp. 301–306
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2006 Springer
About this chapter
Cite this chapter
Houck, J., Scherer, G.W. (2006). Weathering. In: KOURKOULIS, S.K. (eds) Fracture and Failure of Natural Building Stones. Springer, Dordrecht. https://doi.org/10.1007/978-1-4020-5077-0_19
Download citation
DOI: https://doi.org/10.1007/978-1-4020-5077-0_19
Publisher Name: Springer, Dordrecht
Print ISBN: 978-1-4020-5076-3
Online ISBN: 978-1-4020-5077-0
eBook Packages: EngineeringEngineering (R0)