Abstract
Neural mechanisms underlying alcohol use disorder remain elusive, and this lack of understanding has slowed the development of efficacious treatment strategies for reducing relapse rates and prolonging abstinence. While synaptic adaptations produced by chronic alcohol exposure have been extensively characterized in a variety of brain regions, changes in intrinsic excitability of critical projection neurons are understudied. Accumulating evidence suggests that prolonged alcohol drinking and alcohol dependence produce plasticity of intrinsic excitability as measured by changes in evoked action potential firing and after-hyperpolarization amplitude. In this chapter, we describe functional changes in cell firing of projection neurons after long-term alcohol exposure that occur across species and in multiple brain regions. Adaptations in calcium-activated (KCa2), voltage-dependent (KV7), and G protein-coupled inwardly rectifying (Kir3 or GIRK) potassium channels that regulate the evoked firing and after-hyperpolarization parallel functional changes in intrinsic excitability induced by chronic alcohol. Moreover, there are strong genetic links between alcohol-related behaviors and genes encoding KCa2, KV7, and GIRK channels, and pharmacologically targeting these channels reduces alcohol consumption and alcohol-related behaviors. Together, these studies demonstrate that chronic alcohol drinking produces adaptations in KCa2, KV7, and GIRK channels leading to impaired regulation of the after-hyperpolarization and aberrant cell firing. Correcting the deficit in the after-hyperpolarization with positive modulators of KCa2 and KV7 channels and altering the GIRK channel binding pocket to block the access of alcohol represent a potentially highly effective pharmacological approach that can restore changes in intrinsic excitability and reduce alcohol consumption in affected individuals.
The original version of this chapter was revised. A correction to this chapter is available at https://doi.org/10.1007/164_2018_192.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
The term addiction is commonly used to denote an unnatural or unbalanced amount of attention directed toward an object, substance, or activity. In clinical terms, the term addiction has largely been replaced by the phrase “use disorder” that reflects the understanding that these conditions are diseases or illnesses of the brain that result from some underlying alteration in normal physiology. Substance use disorders (SUDs) and more specifically those involving alcohol (ethanol) are operationally defined as the inability of the individual to control or reduce drug or alcohol intake even in the face of negative social, emotional, or health-related consequences (DSM-5 2013).
An emerging theme in the study of drug and alcohol addiction is that different neural systems and neurochemicals underlie various stages associated with drug and alcohol intake that contribute toward the progression of a drug-/alcohol- dependent state (Koob 2013). In one such model, a binge/intoxication stage is thought to involve midbrain dopamine, opioid, and endocannabinoid reward pathways that are activated by acute ingestion of drugs/alcohol and induce positive and pleasurable feelings. Metabolic clearance of the drug or alcohol then induces a stage of acute withdrawal that may engage brain stress systems involving hypothalamic and extrahypothalamic circuits leading to negative or unpleasant symptoms that drive additional binge/intoxication stages. The desire to retake a drug or alcohol following some period of abstinence represents the preoccupation/anticipation stage and is associated with feelings of craving characterized as a need to seek out and consume the substance. This phase is thought to reflect activity in higher cortical areas including areas of the prefrontal and orbitofrontal cortex that are involved in guiding behavior by assessing risk and reward associated with making choices between competing activities.
While the brain circuitry underlying these three phases of the cycle is evolutionarily adapted for pursuing natural rewards, repeated episodes of drug or alcohol consumption may lead to an imbalance between these systems with reductions in reward signaling and altered activity in brain stress and preoccupation/anticipation pathways that combine to produce escalations in drug/alcohol use and the development of an SUD. The yearly incidence for developing an alcohol use disorder (AUD) varies among different populations and is approximately 6% for US adults age 18 and older (Substance Abuse and Mental Health Administration (SAMHSA) 2015). Multiple factors enhance an individual’s susceptibility for developing an AUD including genetic and epigenetic mechanisms that influence the sensitivity of brain reward, stress, and cognitive systems to alcohol. Alcohol dependence disrupts systems designed to maintain and regulate excitability, and in this chapter, we focus on the role of three subtypes of K+-selective ion channels (KCa2, KV7, and Kir3 channels) in regulating intrinsic excitability and discuss how alcohol exposure alters the expression and function of these channels. We also highlight findings from recent studies showing how manipulating these channels with selective pharmacological agents can moderate alcohol consumption and facilitate extinction of cue-induced alcohol-seeking behaviors.
2 Alcohol and Intrinsic Excitability
Although several types of cells including those of skeletal and cardiac muscle are capable of generating action potentials, neurons have exploited this process to generate the complex set of electrical signals that underlie brain function. Action potentials reflect the opening of sodium permeable ion channels activated during periods of membrane depolarization elicited by excitatory synaptic inputs. The rapid entry of positively charged sodium ions drives the membrane potential toward the sodium equilibrium potential that is approximately +40 mV for most mammalian neurons. Voltage-dependent K+ ion channels are activated at these highly depolarized potentials and, combined with sodium channel inactivation, repolarize the neuronal membrane. Repolarization is often accompanied by an overshoot called the after-hyperpolarization (AHP) that briefly drives the membrane potential past its normal resting value. The AHP helps regulate the frequency of action potential generation and is mediated by a variety of ion channels including those regulated by changes in intracellular calcium (e.g., KCa1 and KCa2 channels). Neuronal firing is also regulated by activation of channels in the KV7 family whose activity is normally inhibited by G protein-coupled neurotransmitter receptors and by inwardly rectifying K+ channels (Kir3) that are activated by G proteins (also called GIRK channels).
Intrinsic excitability is a measure of a neuron’s ability to fire and is experimentally determined by measuring the number of action potentials generated spontaneously or during a series of current pulses delivered to the neuron using the current-clamp mode of whole-cell electrophysiology. Importantly, plasticity of intrinsic excitability is a critical mechanism underlying synaptic integration and learning processes (Sehgal et al. 2013), and alterations in cellular firing by abused substances may facilitate drug-induced pre- and postsynaptic adaptations (Kourrich et al. 2015). The following section briefly reviews studies that have examined the effects of acute and chronic alcohol exposure on the firing properties of neurons (for review, see Harrison et al. 2017). Later sections focus on how alcohol-induced changes in the expression or function of KCa2, KV7, and GIRK channels may underlie changes in the current-spike relationship, thus altering the intrinsic excitability of neurons.
2.1 Acute Alcohol and Spike Firing
Brain slice recordings have revealed that under basal conditions, neurons in ventral tegmental area (VTA), globus pallidus, cerebellum, and lateral habenula (LHb) are spontaneously active, while those from dorsal striatum, nucleus accumbens (NAc), hippocampus, and most cortical areas are silent although firing can be induced by direct current injection. Acute application of relatively high concentrations of alcohol (~40–80 mM) enhances firing of dopamine VTA neurons (Brodie et al. 1999), while a lower concentration (20 mM) was shown to increase firing of DA neurons located in the medial VTA (Mrejeru et al. 2015). Concentrations of alcohol as low as 1 mM increased spontaneous firing of glutamatergic neurons in the LHb, while 50 mM alcohol increased the frequency of action potential spiking in cerebellar Golgi neurons (Carta et al. 2004). In contrast, alcohol (10–80 mM) inhibits spontaneous firing of low-frequency (<30 Hz) globus pallidus neurons while having no effect on those that fire at higher frequencies (Abrahao et al. 2017). This effect was occluded by a blocker of KCa1 channels that are a known target for alcohol (Mulholland et al. 2009). Studies using current-evoked spiking report that alcohol also reduces firing of GABAergic neurons in the VTA (Gallegos et al. 1999), pyramidal neurons in the lateral orbitofrontal cortex (lOFC) (Badanich et al. 2013; Nimitvilai et al. 2016, 2017a), and serotonergic neurons of the dorsal raphe (Maguire et al. 2014). Alcohol inhibition of VTA GABAergic neuron firing may involve α6-containing nicotinic acetylcholine receptors (Schilaty et al. 2014), while the alcohol-induced reduction in the firing of lOFC and dorsal raphe neurons requires activation of strychnine-sensitive glycine receptors (Badanich et al. 2013; Maguire et al. 2014).
2.2 Chronic Alcohol and Spike Firing
Several studies have reported changes in intrinsic excitability following chronic exposure to alcohol (Table 1). Spike firing was increased in NAc medium spiny neurons (MSNs) recorded from rats following operant self-administration of alcohol (Hopf et al. 2010), and this was accompanied by reduced AHP amplitude and function of apamin-sensitive KCa2 channels. Similarly, repeated systemic alcohol treatment and long-term drinking in an intermittent access model increased spontaneous action potentials and reduced AHP amplitude in LHb neurons (Agrawal et al. 2012). In contrast, no changes in spike firing or KCa2 channel currents were reported for NAc MSNs from rats given repeated oral doses of alcohol (Marty and Spigelman 2012). Following repeated cycles of chronic intermittent ethanol (CIE) vapor exposure in mice, spike firing of NAc medium spiny neurons and lOFC pyramidal neurons was increased along with reduced AHP and loss of apamin-sensitive currents (Nimitvilai et al. 2016; Padula et al. 2015). Renteria and colleagues reported that the increased firing after CIE exposure was observed only in D1R-expressing MSNs in the NAc shell (Renteria et al. 2017). The increase in spike firing in lOFC neurons persisted for up to 10 days and was associated with a loss of monoamine (DA, NE, 5HT) and GIRK channel modulation of firing and a tolerance to the acute inhibitory actions of alcohol (Harrison et al. 2017; Nimitvilai et al. 2016, 2017b). A similar attenuation of alcohol’s inhibitory effect on spike firing was observed in lOFC neurons from long-term drinking (~9 months) monkeys, while current-evoked spiking was reduced in these animals (Nimitvilai et al. 2017a). In contrast, intrinsic excitability of putamen MSNs was increased in monkeys following prolonged drinking and repeated periods of abstinence (Cuzon Carlson et al. 2011). Region-specific changes in spike firing following CIE treatment of mice have also been reported with increases for neurons in the ventral BNST (Marcinkiewcz et al. 2015; Pleil et al. 2015) and dorsal raphe (Lowery-Gionta et al. 2015), decreases in medial central nucleus of the amygdala (Pleil et al. 2015), and no change in infralimbic cortex (Pleil et al. 2015).
One key factor to consider from these types of studies is the time of recording relative to the last alcohol exposure. For example, in the mouse lOFC studies, CIE-exposed animals showed elevated spike firing by the 3-day withdrawal period, and excitability was still elevated at the 10-day withdrawal time point (Nimitvilai et al. 2016), while no change in excitability was seen in animals undergoing acute (<6 h) withdrawal (Nimitvilai et al. 2017a). Studies examining chronic alcohol-induced changes in intrinsic excitability in prelimbic cortical pyramidal neurons are inconsistent, with two studies showing no alterations in evoked firing in layer V neurons and one study reporting an increase in excitability of layer II/III neurons (Pleil et al. 2015; Hu et al. 2015; Trantham-Davidson et al. 2014). Changes in NAc neuron excitability in alcohol-exposed mice and rats were observed during early withdrawal (3–7 days; Padula et al. 2015) as well as following extended abstinence (Hopf et al. 2010). The increase in dorsal raphe neuron spiking following CIE exposure in mice was present 24 h after the last treatment but was lost after 1 week of withdrawal (Lowery-Gionta et al. 2015). Overall, these findings highlight the dynamic nature of processes that regulate the intrinsic excitability of neurons across species and reveal that not all alcohol exposure models produce the same effect on cell firing, even within the same brain region.
3 Potassium Channels
The K+ channel family is the largest known group of ion channels with at least 79 mammalian K+ channel genes that encode the wide variety of channel subtypes. K+ channels are highly conserved and ubiquitously expressed in almost all species (Kuo et al. 2005). Currently, there are several distinct categories of K+ channels based on the mode of activation and gating, including calcium-activated (KCa), voltage-gated (KV), inward rectifier (Kir), and two-pore domain K+ channels (Table 2). Moreover, alternative splicing and co-assembly of varying K+ channel subunits add to the diversity in K+ channel subtypes and function (Zandany et al. 2015; King et al. 2016). Despite the large number of K+ channel subtypes, all share the common feature of a highly selective K+ ion-permeable transmembrane pore. Given the high intracellular K+ concentration compared to that in the extracellular environment, channel opening leads to rapid K+ ion efflux and hyperpolarization/repolarization of the cell membrane potential (Doyle et al. 1998). K+ channels thus act as an opposing force to cellular depolarization and excitability or as a modifying factor for the shaping of action potentials. Due to the immense number of K+ channel subtypes and the limited scope of this chapter, we will highlight only three of the K+ channel families, the KCa channels, the KV channels, and the G protein-activated inward rectifier (Kir3) channels (Fig. 1). The structure and function of other K+ channel families and subtypes not mentioned here have been extensively reviewed by others and are referenced for the reader’s consideration (Coetzee et al. 1999; Miller 2000; Gonzalez et al. 2012; Jenkinson 2006; Fonseca 2012; Vandenberg et al. 2015).
Activation of KCa2, KV7, and GIRK channels in neural membranes. KCa2 channels are activated by elevations in intracellular calcium via influx through voltage-gated calcium channels (VGCCs), NMDA receptors (NMDARs), or release from intracellular stores. KV7 channels are activated near the resting membrane potential and during membrane depolarization. Ligand binding of G protein-coupled receptors (GPCRs) releases G proteins that bind to and activate GIRK channels. Opening of these KCa2, KV7, and GIRK channels allows potassium ions to flow outside of the cell causing hyperpolarization and shunting of neuronal excitability. CaM calmodulin, RyR ryanodine receptors. Images were acquired with permission from www.servier.com and subsequently modified
There are 12 subgroups (KV1–KV12) in the family of voltage-gated K+ channels. KV channel subtypes are gated by varying voltage dependencies through a voltage-sensing domain formed by four transmembrane segments (S1–S4) in each channel subunit, while the channel pore is comprised of two other transmembrane segments that form a loop (S5–S6). KV1, KV4, and KV7 channels are activated at relatively low membrane potentials and are critical in regulating the number of action potentials during early phases of membrane depolarization (Johnston et al. 2010). The transient A-type current is generated by KV4 channels and has a unique feature in which hyperpolarization is necessary for the removal of steady-state inactivation before activation (Maffie and Rudy 2008). KV7 voltage-gated K+ channels produce the M-current, a slow-developing, low-voltage activated AHP current that is sensitive to muscarinic receptor activation (discussed in detail below). KV2 and KV3 currents are activated at high voltages, particularly during the firing of action potentials, and play an important role in membrane repolarization and regulation of firing in neurons with a fast-spiking phenotype. An important feature that differentiates KV2 and KV3 channels is the rate of activation with KV2 channels showing much slower activation, while KV3 channels open rapidly contributing to their prominent role in regulating firing of fast-spiking neurons. Interestingly, KV5, KV6, KV8, and KV9 are commonly named the silent electrical K+ channel subunits. These subunits do not form functional channels as homomers but readily form heteromers with KV2 channels with diverse functions within tissue types (Bocksteins 2016). The KV10–KV12 channels are referred to as ether-à-go-go (denoted KV10), ether-à-go-go-related (erg, KV11), and ether-à-go-go-like (elk, KV12) and are members of the KCNH gene family. Few studies exist that directly characterize the exact function of these channels, but generally, these channels become activated at subthreshold voltages and display highly variable gating kinetics (Vandenberg et al. 2015; Zhang et al. 2009).
KCa channels are unique in that they are activated by increasing levels of intracellular calcium. Each member of this K+ channel family is further distinguished by the level of single-channel conductance upon activation (Vergara et al. 1998). Large-conductance calcium-activated KCa1 channels that are also voltage-gated have a single-channel conductance of ∼100–300 pS, while intermediate (KCa3; ∼20–80 pS) and small (KCa2; ∼2–20 pS, Marty and Neher 1985) subtypes have more modest values. As discussed below, small-conductance calcium-activated KCa2 channels are of particular interest due to their critical role in regulating intrinsic excitability in relation to alcohol addiction. Alcohol effects on large-conductance calcium-activated KCa1 channels are discussed in the chapter by Dopico and colleagues in this volume (Dopico et al. 2017).
Inward-rectifying K+ channels consist of seven subfamilies (Kir1–Kir7) and are distributed in multiple cell types, including cardiomyocytes, endothelial cells, kidneys, and neurons (for review, see Hibino et al. 2010). Among these, G protein-coupled inwardly rectifying K+ (Kir3 or GIRK) channels are considered as important neuronal regulators and are associated with numerous neuropsychiatric diseases, including schizophrenia (Yamada et al. 2012), depression (Llamosas et al. 2015), epilepsy (Signorini et al. 1997), as well as drug and alcohol abuse disorders (Hill et al. 2003). Unlike KV and KCa, GIRK channels are activated by ligand-stimulated G protein-coupled receptors (GPCRs), including dopamine, serotonin, and GABA. Binding of an agonist to the GPCR triggers the dissociation of Gαi and Gβγ. Once released from its bound Gαi, the Gβγ dimeric protein can directly activate the GIRK channel (Logothetis et al. 1987; Reuveny et al. 1994) and increase its affinity for the membrane-bound phospholipid-phosphatidylinositol 4,5-bisphosphate (PIP2), which is required to stabilize the open state of the GIRK channel (Huang et al. 1998; Xiao et al. 2003). The activated GIRK channel is now permeable to K+ ions, leading to hyperpolarization of the cell membrane and inhibition of neuronal activity. Interestingly, concentrations of alcohol relevant to human consumption (18 mM alcohol or 0.08% blood alcohol level) directly activate GIRK channels independently from GPCR-dependent activation (Bodhinathan and Slesinger 2013; Kobayashi et al. 1999; Lewohl et al. 1999). As many therapeutic drugs that target GPCR-linked GIRK channels have been used to treat a number of neuropsychiatric diseases including alcoholism and alcohol itself directly activates GIRK channels, the role of these channels on neuronal activity and its involvement in alcohol addiction is also addressed in this chapter.
4 Intrinsic Excitability
K+ channel regulation of cellular excitability is among the most important functions related to proper neuronal activity. Excitability of neuronal activity is regulated in many ways including alteration of resting membrane potential (RMP) and limiting neuronal spike activity. The AHP that follows neuronal spiking is a manifestation of outward K+ current that regulates subsequent spiking episodes and, thus, establishes firing frequency. In addition, summation of the AHP after bursts of action potentials leads to spike-frequency adaptation that ultimately leads to the inhibition of spiking and is key for protecting neurons from over-excitation and epileptiform activity (Alger and Williamson 1988; Garduno et al. 2005; Schulz et al. 2012). The generation and regulation of action potentials by K+ channels are critical for neuronal encoding of input- and output-specific information and are mediated by the AHP. Intrinsic excitability is also crucial to processes such as spike timing-dependent plasticity in which changes in the strength of neuronal connectivity rely on the relative timing of synaptic events and back-propagating action potentials and is essential for certain types of learning (Debanne and Poo 2010). Therefore, K+ channel regulation of excitability has important implications in establishing and mediating learning and memory processes. KCa2, KV7, and GIRK channels play significant roles in controlling intrinsic excitability by regulating RMP, spike frequency, and the AHP. Their functional roles and importance in alcohol-related neuroadaptations and behaviors are discussed in more detail below.
5 KCa2, KV7, and GIRK Channel Regulation of Excitability
The small-conductance calcium-activated K+ (KCa2) channels play a key role in regulating intrinsic excitability and underlie the medium AHP (mAHP) that follows action potentials. KCNN1–3 genes encode the pore-forming α subunits of KCa2.1, KCa2.2, and KCa2.3 channels that are predominantly expressed in the central nervous system (Stocker 2004). These channels are structurally similar to the KV channels in that they form tetramers and each α subunit contains six transmembrane regions. KCa2 channels are potently inhibited by the bee venom toxin apamin that produces an increase in cellular excitability and reduction in the amplitude of the mAHP (Blatz and Magleby 1986). Subunits of KCa2 channels contain a calcium-sensitive calmodulin-binding domain formed by constitutively bound calmodulin (Schumacher et al. 2001; Xia et al. 1998) that senses changes in intracellular calcium concentration arising from activation of various calcium channel subtypes, NMDA receptors, and intracellular stores of the endoplasmic reticulum (Adelman et al. 2012). The increase in intracellular calcium concentration that occurs during membrane depolarization induces a conformational change in the calmodulin-binding domain of the KCa2 channel resulting in activation and K+ ion efflux (Xia et al. 1998). Thus, KCa2 channels are critical for monitoring and regulating intracellular calcium signaling and action potential generation.
Similar to other voltage-gated K+ channels, KV7 channels are tetramers formed by assembly of six transmembrane KV7 protein subunits (KV7.1–7.5) that are encoded by the KCNQ family of genes (KCNQ1–5, respectively). KV7.1 subunits are found almost exclusively in cardiomyocytes and do not co-assemble with any of the other KV7 channel proteins and instead associate with KCNE proteins that act as ancillary modulatory subunits (Abbott and Goldstein 2001; McCrossan and Abbott 2004; Roura-Ferrer et al. 2010). The remaining four subunits are expressed throughout the nervous system and form functional homotetramers (KV7.2, KV7.4, and KV7.5) or heterotetramers with KV7.3 (Howard et al. 2007; Jentsch 2000). These channels generate the M-current (I M), a voltage-sensitive K+ current so named as it is suppressed by activation of muscarinic receptors (Wang et al. 1998; Shah et al. 2002). Subsequent research has demonstrated that the M-current can also be inhibited by a number of different neurotransmitters/hormones that act on Gq- and G11-coupled receptors, including, but not limited to, mGlu1/5, histamine H1, 5-HT2C, substance P, bradykinin, and angiotensin II (Brown and Passmore 2009; Zaika et al. 2006; Marrion 1997). Despite the abundance of KV7.2/7.3 channels and the physiological evidence that these heterotetramers contribute substantially to the native M-current, all KV7 channels produce M-like currents (Brown and Passmore 2009), with KV7.4-containing channels contributing to M-current in midbrain dopaminergic neurons (Hansen et al. 2008). Although KV7.5-containing channels show a less ubiquitous expression profile, they are nonetheless found in cortical, hippocampal, and striatal tissue (Shah et al. 2002; Schroeder et al. 2000). Regardless of composition, each M-channel subunit contains a voltage-sensor domain (transmembrane domains S1–S4), and the channels are slow-activating starting at subthreshold potentials of around −60 mV. Uniquely, M-channels do not inactivate, thus producing a sustained voltage-dependent outward current that stabilizes the membrane potential in the presence of depolarizing currents, thus regulating neuronal excitability by dampening repetitive or burst firing of action potentials (Jentsch 2000; Brown and Passmore 2009; Brown and Adams 1980). These channels also regulate interspike intervals and contribute substantially in determining whether neurons naturally fire tonically or phasically (Wang and McKinnon 1995; Lawrence et al. 2006). They also contribute to aspects of the AHP differentially depending on the brain region, channel composition, and presence/distribution of other K+ channels. In CA1 pyramidal neurons, M-channels almost exclusively mediate the mAHP (Gu et al. 2005), though not as much in other cell types (Mateos-Aparicio et al. 2014). In contrast, M-channels have been shown to mediate the fAHP and sAHP in VTA dopamine neurons (Koyama and Appel 2006). Suppression of the M-current by application of the KV7 channel blocker, XE-991, diminishes respective AHP components and increases cellular excitability and spike discharge (Brown and Passmore 2009; Gu et al. 2005; Koyama and Appel 2006). Thus, KV7 channels are an essential regulator of intrinsic excitability, the importance of which is underscored by the finding that a mere 25% reduction in function of KV7.2/7.3 channels induces seizures, as found in cases of benign familial neonatal convulsions caused by KCNQ2 and KCNQ3 mutations (Schroeder et al. 1998).
KCa2 and KV7 channels play complementary roles in regulating neuronal excitability and the AHP. KCa2 channels within dentate gyrus granule cells have been identified as the central contributor to the mAHP and spike-frequency adaptation, while KV7 channels contribute minimally to the isolated mAHP and spike-frequency adaptation but strongly regulate the action potential threshold by activating at subthreshold potentials (Mateos-Aparicio et al. 2014). This complementary function is critical because dentate granule cell activity can regulate the output of interneurons and pyramidal cells through synaptic activity within hippocampal mossy fibers (Henze et al. 2002), and these channels also function to support stable long-term potentiation through facilitating protein synthesis (Barnes et al. 2010). In contrast, in CA1 hippocampal pyramidal cells, KCa2 channels seem to contribute minimally to the mAHP following action potential bursts, whereas KV7 channels are the predominant regulators of the mAHP (Chen et al. 2014). Moreover, when KV7 channel function is inhibited or compromised in CA1 pyramidal cells, KCa2 channels usurp regulation of the AHP and intrinsic excitability and function as a “fail-safe” to limit spike discharge (Chen et al. 2014). This has been proposed to have functional implications for conditions in which KV7 channel function may be disrupted including hyposmolarity-induced epileptiform seizures (Kobayashi et al. 2008), benign familial neonatal convulsions (Singh et al. 2008), or chronic alcohol exposure (Kang et al. 2017). There is limited understanding of how KCa2 and KV7 channels may interact in other brain regions outside of the hippocampus. The diverse functional interactions within cellular subtypes of the hippocampus alone suggest that interplay between these channels may have varying influences on neuronal firing and AHP in other brain regions and/or cellular subtypes. Future studies are warranted to determine the full extent of KV7 and KCa2 channel interactions to regulate cell firing, especially as disruptions in KV7-KCa2 channel cross talk may influence plasticity of intrinsic excitability and alcohol-seeking behavior.
GIRK channels are comprised of four different subunits, GIRK1 to GIRK4. GIRK1–3 subunits (encoded by Kcnj3, Kcnj6, and Kcnj9, respectively, Table 2) are moderately or highly expressed throughout the brain, while expression of GIRK4 is low and is found only in a few brain regions such as the hypothalamus and cerebellum (Aguado et al. 2008; Karschin et al. 1996; Perry et al. 2008). Functional GIRK channels are heterotetramers of GIRK1/2, GIRK1/3, GIRK1/4 or GIRK2/3 subunits, or homotetramers of GIRK2 subunits (Luscher and Slesinger 2010). GIRK2 contains an endoplasmic reticulum (ER) export signal, enabling this subunit to form either homo- or heterotetramers. GIRK3 does not have an ER signal but contains a lysosomal targeting sequence that promotes degradation of GIRK channels. GIRK1 has neither an ER nor a lysosomal targeting signal so it must assemble with another GIRK subunit to express on the plasma membrane. GIRK currents are termed “inwardly rectifying” because current passes more easily in the inward direction than in the outward direction. Under physiological conditions where the RMP of neurons is positive to the equilibrium potential for K+, basal current or agonist-induced GIRK currents show large inward and small outward flow. The outward flow of GIRK-mediated K+ currents near the RMP hyperpolarizes the membrane potential and thus decreases the excitability of the neuron. Dysfunction of GIRK channels has been implicated in several diseases. For example, loss of GIRK function results in excessive excitability found in epilepsy (Signorini et al. 1997), while over-activation of GIRK can reduce neuronal activity and may trigger cell death as postulated in Down’s syndrome and Parkinson’s disease, respectively (Patil et al. 1995).
6 KCa2 Channels and Alcohol
6.1 Genetics
AUD is a complex brain disease that likely results from interactions among genetic, epigenetic, and environmental factors that combine to promote heavy alcohol drinking and dependence phenotypes. Meta-analyses and twin studies estimated that 23–79% of the variance in alcohol addiction is heritable (Agrawal et al. 2012; NIAAA 2016). The limited success of the current FDA-approved drugs for treating excessive drinking may reflect the heterogeneous population of those with AUD. As precision medicine approaches are showing efficacy across a number of diseases (i.e., cancer and cardiovascular disease), recent clinical and preclinical studies have searched for genetic mutations as possible pharmacogenetic targets to treat alcohol addiction (Kranzler et al. 2017; Rinker and Mulholland 2017). While not all pharmacogenetic studies to treat alcohol addiction are reproducible (Kranzler et al. 2017), some studies have shown that matching genetic variation to pharmacotherapies can improve relapse rates and heavy drinking days (Kranzler and McKay 2012; Heilig et al. 2011; Sturgess et al. 2011). Thus, advances in precision medicine approaches offer a promising strategy for treating alcohol addiction, and a number of novel targets have been identified from preclinical models of alcohol seeking and dependence (Padula et al. 2015; Rinker and Mulholland 2017; Rinker et al. 2017).
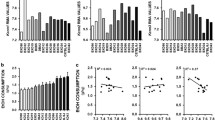
A recent preclinical study employing an integrative functional genomics approach revealed that KCNN genes are present in multiple quantitative trait loci (QTL) and gene sets related to alcohol intake and dependence (Padula et al. 2015), including an alcoholism susceptibility QTL on human chromosome 1. Each member of the family of Kcnn genes is found in QTLs for alcohol consumption and preference in rats and mice (Padula et al. 2015; Bachmanov et al. 2002; Carr et al. 2003; Foroud et al. 2000; Radcliffe et al. 2004). BXD recombinant inbred (RI) strains of mice are an excellent resource to study the genetic diversity of alcohol-related phenotypes (Philip et al. 2010). To gain a better understanding of Kcnn genes in controlling voluntary alcohol intake, the relationship between Kcnn and drinking was explored in BXD RI and their parental strains. Interestingly, Kcnn3 transcript levels in the NAc correlated negatively with voluntary drinking in a two-bottle choice limited-access and the drinking-in-the-dark model (Padula et al. 2015; Rinker et al. 2017). While there are some potential caveats of using BXD RI strains to study the genetic control of alcohol intake (such as the influence of taste and gene-environment interactions), Kcnn3 was also identified as a candidate signature gene that associated with alcohol preference in low- and high-alcohol-drinking rat lines (Lo et al. 2016). In alcohol-dependent BXD strains, the negative relationship between Kcnn3 and alcohol intake became more robust, suggesting that alcohol dependence alters Kcnn expression and that high levels of Kcnn expression protect against escalation of drinking in dependent mice (Padula et al. 2015). Thus, high expression levels of Kcnn3 may be a protective factor against developing alcohol addiction and dependence. Although not identified in the NAc, reductions in KCNN2 expression were reported in the frontal cortex and amygdala of alcohol-dependent individuals (Ponomarev et al. 2012). As described below in detail, studies consistently demonstrate that chronic alcohol exposure reduces KCa2 channel function across multiple preclinical models and in divergent cell types, further demonstrating that KCNN is a mediator of voluntary and heavy alcohol drinking. This finding is supported by evidence showing that KCa2 channel positive modulators reduce drinking in rodent models (see below). Lastly, KCNN genes are present in QTLs related to all abused substances and are altered in brains of addicts (Padula et al. 2015), suggesting that the KCNN family of genes is a common mechanism underlying addiction that spans multiple abused substances (Mulholland et al. 2016).
6.2 Alcohol and KCa2 Channel Function
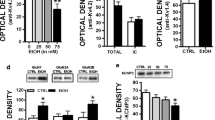
Alcohol has distinct effects on function and membrane trafficking of KCa2 channels that may underlie some of the neuropathology associated with alcohol addiction (Fig. 2). Early studies in rats demonstrated that 20 weeks of chronic alcohol exposure significantly reduced inhibitory postsynaptic potentials and AHP currents in CA1 pyramidal neurons and dentate gyrus granule cells (Durand and Carlen 1984). Similar effects on KCa2-mediated I AHP were later replicated in CA1 pyramidal neurons from organotypic hippocampal slices continuously exposed to alcohol in vitro (Mulholland et al. 2011). Interestingly, synaptic KCa2 channels are part of a negative feedback loop with NMDA receptors within dendritic spine head nanodomains (Mulholland et al. 2011; Ngo-Anh et al. 2005). Mulholland and colleagues (2011) reported a loss of KCa2 channel control of synaptic NMDA receptor activity after chronic alcohol that paralleled increases in excitotoxicity. This effect was attributed to reduced KCa2.2 subunit expression at synaptic sites, and the hyperexcitability was rescued by treatment with 1-EBIO, a KCa2 channel positive modulator. Together, these data demonstrate a relationship between prolonged alcohol exposure and deficits in hippocampal function that may underlie alcohol-induced cognitive impairments observed in clinical settings (Bartels et al. 2007).
Chronic alcohol reduces KCa2, KV7, and GIRK channel expression and signaling in the cortex and striatum. KCa2 and KV7 potassium channels are expressed in dendrites, axons, and along the soma of neurons where they function to reduce the action potential threshold, increase the after-hyperpolarization (AHP) amplitude, and hyperpolarize the resting membrane potential. GIRK channels localize to synaptic and perisynaptic regions of glutamatergic neurons where they function to dampen neuronal excitation. Chronic alcohol exposure and alcohol dependence reduce expression and function of KCa2 and KV7 potassium channels in neurons leading to increased intrinsic excitability and reduced AMP amplitude. GIRK channel signaling in the cortex is disrupted in alcohol-dependent mice. Images were acquired with permission from www.servier.com and subsequently modified
Alcohol exposure is also linked to reduced KCa2 channel function within the VTA, a brain region that is the origin of dopamine cell bodies and that plays a significant role in regulating motivation and the rewarding properties of alcohol (Gonzales et al. 2004; Gorelova et al. 2012; Cook et al. 2014). Indeed, studies have shown that bath application of alcohol reduces KCa2 channel function and increases excitability of dopaminergic neurons through modulation of the AHP (Brodie et al. 1999). It was later demonstrated that withdrawal from alcohol reduced AHP and KCa2-mediated I AHP amplitude in VTA neurons (Hopf et al. 2007). Similar effects have also been observed in the NAc and lOFC after induction of alcohol dependence (Nimitvilai et al. 2016; Padula et al. 2015) and voluntary consumption (Hopf et al. 2010). Decreases in NAc KCa2 subunit expression and trafficking also accompany reductions in the AHP amplitude and are associated with increased excitability of the NAc. Interestingly, there was a complete loss of apamin-sensitive I AHP current in NAc and lOFC neurons following chronic alcohol exposure (Nimitvilai et al. 2016; Padula et al. 2015), suggesting that alcohol may affect expression of apamin-sensitive and apamin-insensitive isoforms of KCa2.3 channels (Wittekindt et al. 2004). These data suggest that adaptations in KCa2 channels underlie plasticity of intrinsic excitability and the motivation for alcohol-seeking behavior, particularly during a period of abstinence.
6.3 KCa2 Channel Ligands and Drinking
Several preclinical studies have shown that pharmacological modulation of KCa2 channel activity alters alcohol consumption and seeking behavior. For example, chlorzoxazone is an FDA-approved KCa2 channel activator (Syme et al. 2000; Cao et al. 2001) used as a muscle relaxant that dose-dependently reduced alcohol consumption in rats using the chronic intermittent access drinking protocol (Hopf et al. 2011). The chlorzoxazone-induced reduction in alcohol consumption was associated with the rescue of diminished KCa2 channel activity in the NAc core as examined by ex vivo slice electrophysiology. In addition, infusion of 1-EBIO, a similar KCa2 positive modulator, in the NAc core selectively reduced alcohol, but not sucrose operant self-administration after a period of forced abstinence (Hopf et al. 2010). Using a 24-h intermittent access procedure, investigators were able to show that CyPPA, another KCa2 positive modulator, significantly reduced alcohol consumption in C57BL/6J mice (Padula et al. 2013). Taken together, these preclinical data indicate that treatment with KCa2 positive modulators is a potential promising pharmacotherapeutic approach to reduce alcohol consumption in individuals with alcohol use disorder (Mulholland 2012). Other studies have demonstrated that microinjection of apamin into the NAc significantly increased alcohol consumption in nondependent mice and also induced spontaneous seizure activity in dependent mice at high doses (Padula et al. 2015). These data indicate that there is a bidirectional role for KCa2 channels to modulate alcohol consumption and further emphasize the role of reduced KCa2 channel function in mediating excessive consumption.
In addition to its role in alcohol consumption, KCa2 channels are also involved in regulating responses to alcohol-associated cues. The prefrontal cortex is a key brain region that responds to the presentation of alcohol- and drug-associated cues (Otis et al. 2017), and KCa2 channels within this region have been linked with associative learning processes (Criado-Marrero et al. 2014). Recent evidence has shown that both systemic and local inhibition of infralimbic but not prelimbic cortex KCa2 channels with apamin facilitates extinction of alcohol-seeking behavior during cue extinction sessions (Cannady et al. 2017). This effect is hypothesized to be due to facilitation of newly formed extinction memories to compete with prior cue-associated memories that had once motivated alcohol-seeking behavior. These data demonstrate that KCa2 inhibition could act as a potential therapeutic target to facilitate cue-exposure therapy in problem drinkers.
7 KV7 Channels and Alcohol
7.1 Genetics
Similar to the relationship between KCNN genes and alcohol, integrative functional genomic analysis revealed that Kcnq2 and Kcnq3 lie within multiple alcohol-related QTLs in rodents, including those for alcohol consumption and preference (McGuier et al. 2016). Likewise, the remaining members of the Kcnq family of genes are present in alcohol preference-related QTLs in rodents (Table 3). In Drosophila, low doses of alcohol blocked KCNQ currents, and a loss-of-function mutation in KCNQ produced an increase in alcohol tolerance and sensitivity to its sedating effects (Cavaliere et al. 2012). Kcnq2 was identified as a positional candidate within the cis-eQTL for alcohol consumption and withdrawal (Metten et al. 2014), and there are two single nucleotide polymorphisms (SNPs) in Kcnq2 that could account for the differences in drinking phenotypes across multiple mouse crosses (McGuier et al. 2016). In BXD RI strains, Kcnq transcript levels in the NAc and prefrontal cortex negatively correlated with voluntary alcohol drinking in a limited-access model (Rinker et al. 2017). Transcript expression levels of genes encoding KV7.2, KV7.3, and KV7.5 channel subunits are altered in key brain regions within the addiction circuitry in postmortem brain tissue from alcoholics and rodent models of chronic alcohol exposure or intake (Rinker and Mulholland 2017; Rinker et al. 2017; McGuier et al. 2016). In the NAc of BXD RI strains, adaptations in Kcnq1 and Kcnq5 transcript levels correlated with the change in voluntary drinking in alcohol-dependent mice (Rinker et al. 2017). Moreover, a recent whole genome sequencing study identified Kcnq5 as a gene associated with alcohol preference in rats (Lo et al. 2016). These preclinical findings are intriguing given that SNPs in KCNQ1 and KCNQ5 are associated with early-onset alcoholism and symptoms of alcohol dependence (Kendler et al. 2011; Edenberg et al. 2010). Together, genetic findings linking alcohol action to the family of KCNQ genes across species as diverse as fruit flies and humans provide strong evidence that genetic diversity in KCNQ influences heavy alcohol intake and contributes to risk factors for developing an AUD.
7.2 Alcohol and KV7 Channel Function
There is accumulating evidence demonstrating that acute alcohol has direct effects on KV7 channel activity leading to alterations in the AHP and intrinsic excitability of neurons. In one of the first reports of this kind, Moore and colleagues showed that in rat hippocampal pyramidal cells, alcohol significantly reduced the M-current and blocked typical somatostatin-induced augmentation of the M-current (Moore et al. 1990). They also demonstrated that this effect of alcohol was mediated through mechanisms distinct from the muscarinic receptor, as bath application of atropine did not alter the alcohol-induced suppression of M-channel activity (Moore et al. 1990). More recent data in dissociated rat VTA dopaminergic neurons confirms that alcohol reduces M-current by acting directly on KV7 channels in a voltage-independent manner, suggesting that the site of action of alcohol is distinct from the voltage-sensing regions of KV7 channel subunits (Koyama et al. 2007). Additionally, this effect is conserved across species, as the M-channel ortholog in Drosophila, dKCNQ, is a direct target of alcohol (Cavaliere et al. 2012, 2013).
While the majority of studies in this area have focused on the acute effects of alcohol on M-channel function, Kang and colleagues have recently examined the effects of more chronic alcohol administration on M-channel function. They demonstrate that repeated alcohol exposure results in increased excitability of neurons in the LHb, as evidenced by increased evoked spike firing, as well as a reduction in the mAHP and decreased ability of XE-991 to increase LHb cell firing (Kang et al. 2017). The authors argued that these effects of alcohol on neuron physiology are attributable to a loss of M-channel activity because of an overall reduction of both KV7.2 and KV7.3 subunit expression in LHb neurons (Kang et al. 2017; Shah et al. 2017). Similarly, KV7.2 subunits in the NAc are differentially trafficked between detergent soluble and insoluble membrane fractions after chronic alcohol consumption, possibly due to changes in KV7.2 SUMOylation that could alter M-channel function (McGuier et al. 2016). These changes in cellular expression profiles and function of M-channels following alcohol exposure (Fig. 2) provide support for using KV7 channel positive modulators to reduce alcohol consumption associated with AUD.
7.3 Retigabine and Drinking
A number of preclinical studies demonstrate that increasing M-channel activity can alter alcohol-associated behaviors and, importantly, may represent a promising pharmacological target for treating AUD. Retigabine, a KV7 channel opener for treating epilepsy, has shown great promise in preclinical models as one such potential treatment. Initial demonstrations show that systemic retigabine administered acutely reduces alcohol consumption in rats in both a limited-access model of alcohol consumption (Knapp et al. 2014) and a more chronic alcohol-drinking model (McGuier et al. 2016). Interestingly, McGuier and colleagues demonstrated that retigabine was more effective in “high-drinking” rats than “low-drinking” rats and that positive modulation of KV7 in the NAc was similarly effective in reducing alcohol consumption in “high-drinking” rats (McGuier et al. 2016). In a mouse model of chronic alcohol consumption, retigabine was highly effective at reducing consumption in mice showing a high-drinking phenotype (Rinker et al. 2017). Additionally, repeated, prophylactic administration of retigabine decreased hippocampal sensitivity to an acute intravenous injection of alcohol, i.e., retigabine decreased alcohol-induced changes in hippocampal EEG activity in rabbits (Zwierzynska et al. 2015). In a subsequent study, Zwierzynska and colleagues demonstrated that chronic retigabine administration blocked alcohol-induced changes in EEG activity both during forced alcohol administration and during abstinence, highlighting the potential of retigabine to prevent alcohol dependence-related functional changes in activity of the frontal cortex, hippocampus, and midbrain (Zwierzynska et al. 2016). As well, LHb M-channels are sensitive to repeated alcohol exposure, and Kang and colleagues determined that microinfusion of retigabine into the LHb, but not the nearby paraventricular nucleus of the thalamus or the mediodorsal thalamic nuclei, significantly reduced alcohol consumption and alcohol withdrawal-induced anxiety (Kang et al. 2017). Taken together, these preclinical studies demonstrate that retigabine, or other KV7 channel openers, holds great therapeutic potential for treating AUD. Despite these promising results, only one study to date has examined the effects of retigabine in a clinical population of moderate social drinkers to determine interactive effects of alcohol on retigabine pharmacodynamics and pharmacokinetics (Crean and Tompson 2013). Thus, future studies are essential to determine the efficacy of KV7 channel positive modulators in treating AUD.
8 GIRK Channels and Alcohol
8.1 Genetics
A genome-wide QTL mapping study identified Kcnj9 (a gene encoding the GIRK3 subunit) as one of the genetic determinants of alcoholism in mice (Buck et al. 2012). This gene was also associated with withdrawal from alcohol and other sedative hypnotics (Herman et al. 2015; Kozell et al. 2009), alcohol drinking (Tarantino et al. 1998), alcohol-conditioned aversion (Risinger and Cunningham 1998), and acute sensitivity to alcohol (Crabbe et al. 1994; Demarest et al. 1999). In BXD RI strains, Kcnj3 (a gene encoding the GIRK1 subunit) transcript levels in the NAc positively correlated with voluntary alcohol drinking in nondependent mice, while Kcnj6 (a gene encoding the GIRK2 subunit) in the PFC negatively correlated with drinking in nondependent animals (Rinker et al. 2017). Following CIE exposure, significant adaptations in Kcnj3 and Kcnj6 transcript levels in the NAc have been detected (Rinker et al. 2017). In addition, there was a positive correlation between Kcnj9 and the change in voluntary drinking induced by alcohol dependence (Rinker et al. 2017). The genetic evidence in mice was correlated with behavioral studies in GIRK-knockout models. For example, GIRK2−/− mice showed reduced conditioned taste aversion and failed to develop a conditioned place preference for alcohol (Hill et al. 2003). Additionally, mice lacking GIRK3 exhibited excessive alcohol drinking (Herman et al. 2015) and demonstrated less severe withdrawal symptoms compared to their wild-type littermates (Kozell et al. 2009).
The involvement of KCNJ genes in the development of alcohol use disorders has also been identified in humans. One SNP, rs2836016 in KCNJ6, was found to be associated with alcohol dependence in adults. In addition, this KCNJ6 SNP was significantly associated with hazardous drinking, as defined by the Alcohol Use Disorders Identification Test (AUDIT), in adolescents but only in those exposed to early life stress (Clarke et al. 2011). Another study reported several SNPs in the promoter region of KCNJ6 in AUD subjects and in offspring at high risk to develop an AUD (Kang et al. 2012). In this study, electroencephalogram was used to record the theta event-related oscillations (EROs) that reflect processes underlying frontal inhibitory control, conscious awareness, and memory and processes that are often impaired in individuals with drug and alcohol use disorders. The results demonstrated a correlation between KCNJ6 SNPs and theta oscillations across the scalp, with the strongest associations for the frontal phenotype (Kang et al. 2012). A significant association of KCNJ6 SNP for nicotine dependence has also been reported (Saccone et al. 2007). Together, behavioral and genetic evidence in rodents and humans suggest the involvement of KCNJ family of genes with alcohol consumption and behaviors related to heavy drinking.
8.2 Alcohol and GIRK Channel Function
It is largely accepted that alcohol affects multiple neurotransmitters in the brain, many of which (i.e., dopamine and GABA) are linked to GIRK channel activation. For example, alcohol enhances GABAB-mediated inhibitory postsynaptic transmission on rat VTA neurons by facilitating GIRK currents (Federici et al. 2009). Withdrawal from repeated in vivo alcohol exposure produced a profound decrease in D2-/GIRK-mediated inhibition in VTA neurons of mouse brain slice while having no effect on GABAB-/GIRK-mediated inhibition (Perra et al. 2011). Recent findings from our group demonstrated that monoamines, including dopamine, serotonin, and norepinephrine, decreased evoked firing of lOFC neurons in C57BL/6J mice via the activation of Gi-coupled D2, 5HT1A, and α2-adrenergic receptors, respectively (Nimitvilai et al. 2017b). This effect was GIRK-dependent as blocking GIRK channel with barium attenuated monoamine inhibition, and the GIRK1-subunit selective activator, ML297, by itself reduced evoked spiking. Following CIE exposure, however, the inhibitory effects of each monoamine or ML297 were blunted (Nimitvilai et al. 2017b), indicating the importance of monoamine/GIRK system as a modulator of lOFC excitability and suggesting that disruption of this process could contribute to various deficits associated with alcohol use disorder. As mentioned earlier, alcohol at intoxicating concentrations in humans can directly activate GIRK channels independently from GPCR signaling pathway. In Xenopus oocytes co-injected with mRNAs encoding GIRK1/GIRK2 subunits or GIRK1/GIRK4 subunits, alcohol (100–200 mM) induced strong K+ currents, and this effect was blocked by barium, suggesting a direct action of alcohol on GIRK channels (Kobayashi et al. 1999). A similar finding was reported for GIRK-mediated currents in cerebellar granule cells (Lewohl et al. 1999). Recently, a high-resolution crystal structure of a GIRK channel (Aryal et al. 2009) combined with an alcohol-tagging approach (Bodhinathan and Slesinger 2013) revealed that GIRK channels contain an alcohol binding pocket located at the interface between two adjoining subunits within the cytoplasmic domains. The presence of alcohol in the pocket induces conformational changes and increases the affinity of PIP2 that helps stabilize the open state of the GIRK channel. Mutation of a leucine (L257) within the alcohol binding pocket significantly decreases alcohol-induced GIRK currents (Aryal et al. 2009). Therefore, changes in physical and chemical nature of the alcohol binding pocket could interfere with alcohol action on GIRK channels.
8.3 GIRK Channel Ligands and Drinking
Imbalance or dysfunction of GIRK channels has been implicated in altering neuronal excitability and is linked to many neuropsychiatric diseases and SUDs. As mentioned above, a number of studies demonstrate that GIRK channels can be activated directly by acute alcohol (Bodhinathan and Slesinger 2013; Kobayashi et al. 1999; Lewohl et al. 1999) and that GIRK function is blunted following chronic alcohol exposure (Nimitvilai et al. 2017b). In addition, the action of many Gi-coupled neurotransmitters, including dopamine, serotonin, and GABA, that are linked to GIRK channel activation is modulated by alcohol (Nimitvilai et al. 2017b; Federici et al. 2009; Perra et al. 2011). Genetic studies also depict the association of Kcnj genes encoding GIRK channels in alcohol use disorder and dependence (Rinker and Mulholland 2017; Rinker et al. 2017; Buck et al. 2012; Clarke et al. 2011). Therefore, selective manipulation of GIRK channels may represent a promising target for treating excessive drinking and relapse.
In 2012, baclofen, a GABAB agonist that is linked to GIRK channel activation, was approved as a treatment for alcohol addiction in France and is currently under clinical trials in the USA. It shows promising effects in managing alcohol withdrawal symptoms, reducing alcohol craving and consumption, and promoting alcohol abstinence in alcoholic animal models and humans, without serious or severe side effects (Addolorato et al. 2012; Maccioni and Colombo 2009; Morley et al. 2014). An increase in sedation on the BAES scale was reported in human subjects; however, no clinically significant sedative side effects, including sedation, tiredness, and sleepiness, were reported during the treatment session, confirming the safety of baclofen in alcohol-dependent patients (Evans and Bisaga 2009; Leggio et al. 2012). Despite these positive findings, other studies reported a lack of efficacy of baclofen in some alcohol-dependent individuals (Garbutt et al. 2010; Leggio et al. 2010) and a severe sedative effect of the drug (Garbutt et al. 2010). One clinical trial study examined the possible role of DRD4, a genetic modulator associated with risk of alcohol dependence (AD), on the effect of baclofen (Leggio et al. 2013). There is evidence of a robust relationship between urge for alcohol and the DRD4 or 5-HTTLPR polymorphisms, i.e., the presence and absence of the DRD4 allele 7-repeat or the short and long allele in the 5-HTTLPR promoter region (Kenna et al. 2012; McGeary 2009). When baclofen was given to AD participants, less drinking was observed in subjects with ≥7 DRD4 repeats, while the opposite was observed in AD patients with <7 DRD4 repeats. Baclofen caused a reduction in alcohol consumption regardless of 5HTTLPR form; however, individuals with a homozygous LL genotype drank significantly less than those with SS/SL genotype (Leggio et al. 2013). Therefore, the ability of baclofen to reduce alcohol consumption might be limited to specific endophenotypes of AD individuals. At present, there is no treatment for alcohol dependence that directly targets GIRK channels. One compound, ML297, has been recently designed to directly activate GIRK1-containing GIRK channels (Kaufmann et al. 2013) and has been shown to decrease anxiety-related behavior and exhibit antiepileptic properties without affecting locomotor activity or conditioned place preference (Kaufmann et al. 2013; Wydeven et al. 2014). It will be interesting to examine whether ML297 can also be used to alleviate symptoms related to alcohol withdrawal that are thought to contribute to the risk of relapse. Although both ML297 and alcohol enhance GIRK channel activity, ML297 may help restore normal GIRK channel tone that is lost following chronic alcohol exposure and reduce craving for alcohol.
9 Conclusions and Future Directions
In this chapter, we have summarized the effects of chronic alcohol exposure on intrinsic excitability and adaptations in expression and function of KCa, Kv7, and GIRK channels across different alcohol exposure models and species (Drosophila, rodent, monkey, and humans). In general, transcript levels of genes in these K+ channel families negatively correlate with higher levels of alcohol intake. Evidence presented here suggests that downregulation of KCa2 and KV7 channel function underpins the reduction in AHP amplitude and increased evoked cell firing after chronic alcohol exposure. In many cases, positive modulators of these channels restore or prevent aberrant physiology and behaviors that result from prolonged alcohol exposure, including alcohol-seeking and withdrawal- and anxiety-related behaviors (Table 4). Indeed, KCa2 and KV7 channel positive modulators reduce alcohol intake across many preclinical models of drinking and seeking, and both channels play a critical role in heavy alcohol drinking in dependent and nondependent rodents (Padula et al. 2013, 2015; Rinker et al. 2017; McGuier et al. 2016). Together, these findings reveal genetic variations and adaptations in KCa2 and KV7 channels that are important for the plasticity of intrinsic excitability and a heavy drinking phenotype.
The goal of preclinical studies is to identify neural mechanisms that underpin aberrant drug-seeking and relapse-like behaviors. As presented in this chapter, KCa2, KV7, and GIRK channels emerged from these preclinical studies as promising “translational” therapeutic targets for treating alcohol use disorder. The next step in the progression to FDA approval is to determine the efficacy of compounds that target KCa2, KV7, and GIRK channels in clinical trials of treatment-seeking individuals diagnosed with alcohol use disorder. Because activators of these channels have anticonvulsive properties, sedation and cognitive impairments may be an unwanted side effect. A small-scale trial in social and moderate drinkers (ClinicalTrials.gov record number: NCT01342341) was recently completed with chlorzoxazone, an FDA-approved drug prescribed as a skeletal muscle relaxant (Chou et al. 2004) that acts as a KCa2 channel positive modulator (Cao et al. 2001). Although chlorzoxazone was well tolerated in this moderate social drinking population, the dosing schedule used in this trial did not reduce the number of alcohol drinks across 2 weeks of treatment. These findings are in stark contrast to the preclinical studies showing that KCa2 channel positive modulators decrease consumption. The reasons for the discrepancy between the preclinical studies and this clinical trial are unclear but may relate to the short half-life, low EC50 for KCa2 channels, off-target actions of chlorzoxazone, conservative dosing approach, or population of drinkers that were recruited for the trial.
Similar to KCa2 channels, an FDA-approved drug, retigabine, acts as a positive modulator of KV7 channels and is used to treat partial-onset seizures, and acute doses are well tolerated in moderate social drinkers (Crean and Tompson 2013). However, extended retigabine use produces pigment changes in the retina and skin due to accumulation of retigabine dimers with low solubility. Although the pigment changes are reversible, GlaxoSmithKline withdrew retigabine from the market in 2017. While these findings are somewhat discouraging, there are analogs of retigabine with selectivity for brain-specific KV7 channel subtypes with chemical scaffolds that appear not to form insoluble dimers. Thus, despite some setbacks, there is continued enthusiasm for developing additional KCa2 and KV7 channel positive modulators as pharmacotherapeutics for treating alcohol use disorder, especially in light of the vast preclinical genetic, functional, and pharmacological evidence supporting a role for these channels in alcohol-seeking behaviors. Finally, based on the compelling preclinical and human genetic evidence, future clinical studies are necessary to validate KCNQ SNPs as pharmacogenetic targets for a precision medicine approach for treating alcohol addiction.
In addition, although this chapter was focused on the role of discrete K+ channel subtypes in alcohol addiction, emerging evidence reveals that plasticity of intrinsic excitability contributes to addiction of all abused substances (Kourrich et al. 2015; Kourrich and Thomas 2009) and neuropsychiatric disorders (Beck and Yaari 2008). For example, protracted withdrawal from chronic morphine exposure increased the intrinsic excitability of NAc shell medium spiny neurons in rats (Wu et al. 2013), and this was accompanied by an attenuation of the apamin-sensitive AHP current in the morphine-withdrawn rats. Interestingly, members of the KCNN family of genes have links with opioid and alcohol addiction (Padula et al. 2015), and multiple studies have implicated KCNN3 mutations in schizophrenia (Bowen et al. 2001; Cardno et al. 1999; Chandy et al. 1998). A rare frameshift mutation in KCNN3 that generates a dominant-negative form of this channel was reported in a patient with schizophrenia (Bowen et al. 2001), and longer polyglutamine repeat alleles in KCa2.3 channels associate with negative symptoms and cognitive performance in schizophrenics (Cardno et al. 1999; Grube et al. 2011). Importantly, the rare frameshift mutation and longer polyglutamine repeats in KCNN3 suppress KCa2 channel surface expression and function (Grube et al. 2011; Miller et al. 2001). These findings are consistent with the overlap of some genes with abused substances and psychiatric diseases (Agrawal et al. 2012) and suggest that neural mechanisms that underlie adaptations in intrinsic excitability are critical factors that drive the risk for and emergence of neuropsychiatric disorders.
Change history
23 January 2019
In section 8.1 on the 10th line in first paragraph the reference citation Mateos-Aparicio et al. 2014 is incorrect.
References
Abbott GW, Goldstein SA (2001) Potassium channel subunits encoded by the KCNE gene family: physiology and pathophysiology of the MinK-related peptides (MiRPs). Mol Interv 1(2):95–107
Abrahao KP et al (2017) Ethanol-sensitive pacemaker neurons in the mouse external globus pallidus. Neuropsychopharmacology 42(5):1070–1081
Addolorato G et al (2012) Novel therapeutic strategies for alcohol and drug addiction: focus on GABA, ion channels and transcranial magnetic stimulation. Neuropsychopharmacology 37(1):163–177
Adelman JP, Maylie J, Sah P (2012) Small-conductance Ca2+-activated K+ channels: form and function. Annu Rev Physiol 74:245–269
Agrawal A et al (2012) The genetics of addiction – a translational perspective. Transl Psychiatry 2:e140
Aguado C et al (2008) Cell type-specific subunit composition of G protein-gated potassium channels in the cerebellum. J Neurochem 105(2):497–511
Alger BE, Williamson A (1988) A transient calcium-dependent potassium component of the epileptiform burst after-hyperpolarization in rat hippocampus. J Physiol 399:191–205
Aryal P et al (2009) A discrete alcohol pocket involved in GIRK channel activation. Nat Neurosci 12(8):988–995
Bachmanov AA et al (2002) Voluntary ethanol consumption by mice: genome-wide analysis of quantitative trait loci and their interactions in a C57BL/6ByJ × 129P3/J F2 intercross. Genome Res 12(8):1257–1268
Badanich KA et al (2013) Ethanol reduces neuronal excitability of lateral orbitofrontal cortex neurons via a glycine receptor dependent mechanism. Neuropsychopharmacology 38(7):1176–1188
Barnes SJ et al (2010) Stable mossy fiber long-term potentiation requires calcium influx at the granule cell soma, protein synthesis, and microtubule-dependent axonal transport. J Neurosci 30(39):12996–13004
Bartels C et al (2007) Recovery of hippocampus-related functions in chronic alcoholics during monitored long-term abstinence. Alcohol Alcohol 42(2):92–102
Beck H, Yaari Y (2008) Plasticity of intrinsic neuronal properties in CNS disorders. Nat Rev Neurosci 9(5):357–369
Blatz AL, Magleby KL (1986) Single apamin-blocked Ca-activated K+ channels of small conductance in cultured rat skeletal muscle. Nature 323(6090):718–720
Bocksteins E (2016) Kv5, Kv6, Kv8, and Kv9 subunits: no simple silent bystanders. J Gen Physiol 147(2):105–125
Bodhinathan K, Slesinger PA (2013) Molecular mechanism underlying ethanol activation of G-protein-gated inwardly rectifying potassium channels. Proc Natl Acad Sci U S A 110(45):18309–18314
Bowen T et al (2001) Mutation screening of the KCNN3 gene reveals a rare frameshift mutation. Mol Psychiatry 6(3):259–260
Brodie MS et al (1999) Pharmacological reduction of small conductance calcium-activated potassium current (SK) potentiates the excitatory effect of ethanol on ventral tegmental area dopamine neurons. J Pharmacol Exp Ther 290(1):325–333
Brown DA, Adams PR (1980) Muscarinic suppression of a novel voltage-sensitive K+ current in a vertebrate neurone. Nature 283(5748):673–676
Brown DA, Passmore GM (2009) Neural KCNQ (Kv7) channels. Br J Pharmacol 156(8):1185–1195
Buck KJ et al (2012) Discovering genes involved in alcohol dependence and other alcohol responses: role of animal models. Alcohol Res 34(3):367–374
Cannady R et al (2017) Prefrontal cortex KCa2 channels regulate mGlu5-dependent plasticity and extinction of alcohol-seeking behavior. J Neurosci 37(16):4359–4369
Cao Y et al (2001) Modulation of recombinant small-conductance Ca(2+)-activated K(+) channels by the muscle relaxant chlorzoxazone and structurally related compounds. J Pharmacol Exp Ther 296(3):683–689
Cardno AG et al (1999) CAG repeat length in the hKCa3 gene and symptom dimensions in schizophrenia. Biol Psychiatry 45(12):1592–1596
Carr LG et al (2003) Analyses of quantitative trait loci contributing to alcohol preference in HAD1/LAD1 and HAD2/LAD2 rats. Alcohol Clin Exp Res 27(11):1710–1717
Carta M, Mameli M, Valenzuela CF (2004) Alcohol enhances GABAergic transmission to cerebellar granule cells via an increase in Golgi cell excitability. J Neurosci 24(15):3746–3751
Cavaliere S, Gillespie JM, Hodge JJ (2012) KCNQ channels show conserved ethanol block and function in ethanol behaviour. PLoS One 7(11):e50279
Cavaliere S, Malik BR, Hodge JJ (2013) KCNQ channels regulate age-related memory impairment. PLoS One 8(4):e62445
Chandy KG et al (1998) Isolation of a novel potassium channel gene hSKCa3 containing a polymorphic CAG repeat: a candidate for schizophrenia and bipolar disorder? Mol Psychiatry 3(1):32–37
Chen S, Benninger F, Yaari Y (2014) Role of small conductance Ca(2)(+)-activated K(+) channels in controlling CA1 pyramidal cell excitability. J Neurosci 34(24):8219–8230
Chou R, Peterson K, Helfand M (2004) Comparative efficacy and safety of skeletal muscle relaxants for spasticity and musculoskeletal conditions: a systematic review. J Pain Symptom Manag 28(2):140–175
Clarke TK et al (2011) KCNJ6 is associated with adult alcohol dependence and involved in gene x early life stress interactions in adolescent alcohol drinking. Neuropsychopharmacology 36(6):1142–1148
Coetzee WA et al (1999) Molecular diversity of K+ channels. Ann N Y Acad Sci 868:233–285
Cook JB et al (2014) Overexpression of the steroidogenic enzyme cytochrome P450 side chain cleavage in the ventral tegmental area increases 3alpha,5alpha-THP and reduces long-term operant ethanol self-administration. J Neurosci 34(17):5824–5834
Crabbe JC et al (1994) Quantitative trait loci mapping of genes that influence the sensitivity and tolerance to ethanol-induced hypothermia in BXD recombinant inbred mice. J Pharmacol Exp Ther 269(1):184–192
Crean CS, Tompson DJ (2013) The effects of ethanol on the pharmacokinetics, pharmacodynamics, safety, and tolerability of ezogabine (retigabine). Clin Ther 35(1):87–93
Criado-Marrero M, Santini E, Porter JT (2014) Modulating fear extinction memory by manipulating SK potassium channels in the infralimbic cortex. Front Behav Neurosci 8:96
Cuzon Carlson VC et al (2011) Synaptic and morphological neuroadaptations in the putamen associated with long-term, relapsing alcohol drinking in primates. Neuropsychopharmacology 36(12):2513–2528
Debanne D, Poo MM (2010) Spike-timing dependent plasticity beyond synapse – pre- and post-synaptic plasticity of intrinsic neuronal excitability. Front Synaptic Neurosci 2:21
Demarest K et al (1999) Identification of an acute ethanol response quantitative trait locus on mouse chromosome 2. J Neurosci 19(2):549–561
Dopico AM, Bukiya AN, Bettinger JC (2017) Voltage-sensitive potassium channels of the BK type and their coding genes are alcohol targets in neurons. Handb Exp Pharmacol. https://doi.org/10.1007/164_2017_78
Doyle DA et al (1998) The structure of the potassium channel: molecular basis of K+ conduction and selectivity. Science 280(5360):69–77
DSM-5 (2013) Diagnostic and statistical manual of mental disorders, 5th edn. American Psychiatric Association, Arlington
Durand D, Carlen PL (1984) Decreased neuronal inhibition in vitro after long-term administration of ethanol. Science 224(4655):1359–1361
Edenberg HJ et al (2010) Genome-wide association study of alcohol dependence implicates a region on chromosome 11. Alcohol Clin Exp Res 34(5):840–852
Evans SM, Bisaga A (2009) Acute interaction of baclofen in combination with alcohol in heavy social drinkers. Alcohol Clin Exp Res 33(1):19–30
Federici M et al (2009) Ethanol enhances GABAB-mediated inhibitory postsynaptic transmission on rat midbrain dopaminergic neurons by facilitating GIRK currents. Eur J Neurosci 29(7):1369–1377
Fonseca DS (2012) Potassium channels: types, structure, and blockers. In: Cell biology research progress. Nova Biomedical Books, New York, p xi, 145
Foroud T et al (2000) Identification of quantitative trait loci influencing alcohol consumption in the high alcohol drinking and low alcohol drinking rat lines. Behav Genet 30(2):131–140
Gallegos RA et al (1999) Adaptive responses of gamma-aminobutyric acid neurons in the ventral tegmental area to chronic ethanol. J Pharmacol Exp Ther 291(3):1045–1053
Garbutt JC et al (2010) Efficacy and safety of baclofen for alcohol dependence: a randomized, double-blind, placebo-controlled trial. Alcohol Clin Exp Res 34(11):1849–1857
Garduno J et al (2005) 1-Ethyl-2-benzimidazolinone (EBIO) suppresses epileptiform activity in in vitro hippocampus. Neuropharmacology 49(3):376–388
Gonzales RA, Job MO, Doyon WM (2004) The role of mesolimbic dopamine in the development and maintenance of ethanol reinforcement. Pharmacol Ther 103(2):121–146
Gonzalez C et al (2012) K(+) channels: function-structural overview. Compr Physiol 2(3):2087–2149
Gorelova N et al (2012) The glutamatergic component of the mesocortical pathway emanating from different subregions of the ventral midbrain. Cereb Cortex 22(2):327–336
Grube S et al (2011) A CAG repeat polymorphism of KCNN3 predicts SK3 channel function and cognitive performance in schizophrenia. EMBO Mol Med 3(6):309–319
Gu N et al (2005) Kv7/KCNQ/M and HCN/h, but not KCa2/SK channels, contribute to the somatic medium after-hyperpolarization and excitability control in CA1 hippocampal pyramidal cells. J Physiol 566(Pt 3):689–715
Hansen HH et al (2008) Kv7 channels: interaction with dopaminergic and serotonergic neurotransmission in the CNS. J Physiol 586(7):1823–1832
Harrison NL et al (2017) Effects of acute alcohol on excitability in the CNS. Neuropharmacology 122:36–45
Heilig M et al (2011) Pharmacogenetic approaches to the treatment of alcohol addiction. Nat Rev Neurosci 12(11):670–684
Henze DA, Wittner L, Buzsaki G (2002) Single granule cells reliably discharge targets in the hippocampal CA3 network in vivo. Nat Neurosci 5(8):790–795
Herman MA et al (2015) GIRK3 gates activation of the mesolimbic dopaminergic pathway by ethanol. Proc Natl Acad Sci U S A 112(22):7091–7096
Hibino H et al (2010) Inwardly rectifying potassium channels: their structure, function, and physiological roles. Physiol Rev 90(1):291–366
Hill KG et al (2003) Reduced ethanol-induced conditioned taste aversion and conditioned place preference in GIRK2 null mutant mice. Psychopharmacology 169(1):108–114
Hopf FW et al (2007) Withdrawal from intermittent ethanol exposure increases probability of burst firing in VTA neurons in vitro. J Neurophysiol 98(4):2297–2310
Hopf FW et al (2010) Reduced nucleus accumbens SK channel activity enhances alcohol seeking during abstinence. Neuron 65(5):682–694
Hopf FW et al (2011) Chlorzoxazone, an SK-type potassium channel activator used in humans, reduces excessive alcohol intake in rats. Biol Psychiatry 69(7):618–624
Howard RJ et al (2007) Structural insight into KCNQ (Kv7) channel assembly and channelopathy. Neuron 53(5):663–675
Hu W et al (2015) Effects of acamprosate on attentional set-shifting and cellular function in the prefrontal cortex of chronic alcohol-exposed mice. Alcohol Clin Exp Res 39(6):953–961
Huang CL, Feng S, Hilgemann DW (1998) Direct activation of inward rectifier potassium channels by PIP2 and its stabilization by Gbetagamma. Nature 391(6669):803–806
Jenkinson DH (2006) Potassium channels – multiplicity and challenges. Br J Pharmacol 147(Suppl 1):S63–S71
Jentsch TJ (2000) Neuronal KCNQ potassium channels: physiology and role in disease. Nat Rev Neurosci 1(1):21–30
Johnston J, Forsythe ID, Kopp-Scheinpflug C (2010) Going native: voltage-gated potassium channels controlling neuronal excitability. J Physiol 588(Pt 17):3187–3200
Kang SJ et al (2012) Family-based genome-wide association study of frontal theta oscillations identifies potassium channel gene KCNJ6. Genes Brain Behav 11(6):712–719
Kang S et al (2017) Ethanol withdrawal drives anxiety-related behaviors by reducing M-type potassium channel activity in the lateral habenula. Neuropsychopharmacology 42(9):1813–1824
Karschin C et al (1996) IRK(1-3) and GIRK(1-4) inwardly rectifying K+ channel mRNAs are differentially expressed in the adult rat brain. J Neurosci 16(11):3559–3570
Kaufmann K et al (2013) ML297 (VU0456810), the first potent and selective activator of the GIRK potassium channel, displays antiepileptic properties in mice. ACS Chem Neurosci 4(9):1278–1286
Kendler KS et al (2011) Genomewide association analysis of symptoms of alcohol dependence in the molecular genetics of schizophrenia (MGS2) control sample. Alcohol Clin Exp Res 35(5):963–975
Kenna GA et al (2012) Association of the 5-HTT gene-linked promoter region (5-HTTLPR) polymorphism with psychiatric disorders: review of psychopathology and pharmacotherapy. Pharmgenomics Pers Med 5:19–35
King BL et al (2016) Calcium activated K(+) channels in the electroreceptor of the skate confirmed by cloning. Details of subunits and splicing. Gene 578(1):63–73
Knapp CM et al (2014) The Kv7 potassium channel activator retigabine decreases alcohol consumption in rats. Am J Drug Alcohol Abuse 40(3):244–250
Kobayashi T et al (1999) Ethanol opens G-protein-activated inwardly rectifying K+ channels. Nat Neurosci 2(12):1091–1097
Kobayashi K et al (2008) K(+)-channel openers suppress epileptiform activities induced by 4-aminopyridine in cultured rat hippocampal neurons. J Pharmacol Sci 108(4):517–528
Koob GF (2013) Addiction is a reward deficit and stress surfeit disorder. Front Psych 4:72
Kourrich S, Thomas MJ (2009) Similar neurons, opposite adaptations: psychostimulant experience differentially alters firing properties in accumbens core versus shell. J Neurosci 29(39):12275–12283
Kourrich S, Calu DJ, Bonci A (2015) Intrinsic plasticity: an emerging player in addiction. Nat Rev Neurosci 16(3):173–184
Koyama S, Appel SB (2006) Characterization of M-current in ventral tegmental area dopamine neurons. J Neurophysiol 96(2):535–543
Koyama S, Brodie MS, Appel SB (2007) Ethanol inhibition of m-current and ethanol-induced direct excitation of ventral tegmental area dopamine neurons. J Neurophysiol 97(3):1977–1985
Kozell LB et al (2009) Mapping a barbiturate withdrawal locus to a 0.44 Mb interval and analysis of a novel null mutant identify a role for Kcnj9 (GIRK3) in withdrawal from pentobarbital, zolpidem, and ethanol. J Neurosci 29(37):11662–11673
Kranzler HR, McKay JR (2012) Personalized treatment of alcohol dependence. Curr Psychiatry Rep 14(5):486–493
Kranzler HR et al (2017) Precision medicine and pharmacogenetics: what does oncology have that addiction medicine does not? Addiction 112(12):2086–2094. https://doi.org/10.1111/add.13818
Kuo MM et al (2005) Prokaryotic K(+) channels: from crystal structures to diversity. FEMS Microbiol Rev 29(5):961–985
Lawrence JJ et al (2006) Somatodendritic Kv7/KCNQ/M channels control interspike interval in hippocampal interneurons. J Neurosci 26(47):12325–12338
Leggio L, Garbutt JC, Addolorato G (2010) Effectiveness and safety of baclofen in the treatment of alcohol dependent patients. CNS Neurol Disord Drug Targets 9(1):33–44
Leggio L et al (2012) Baclofen promotes alcohol abstinence in alcohol dependent cirrhotic patients with hepatitis C virus (HCV) infection. Addict Behav 37(4):561–564
Leggio L et al (2013) A human laboratory pilot study with baclofen in alcoholic individuals. Pharmacol Biochem Behav 103(4):784–791
Lewohl JM et al (1999) G-protein-coupled inwardly rectifying potassium channels are targets of alcohol action. Nat Neurosci 2(12):1084–1090
Llamosas N et al (2015) Deletion of GIRK2 subunit of GIRK channels alters the 5-HT1A receptor-mediated signaling and results in a depression-resistant behavior. Int J Neuropsychopharmacol 18(11):pyv051
Lo CL et al (2016) High resolution genomic scans reveal genetic architecture controlling alcohol preference in bidirectionally selected rat model. PLoS Genet 12(8):e1006178
Logothetis DE et al (1987) The beta gamma subunits of GTP-binding proteins activate the muscarinic K+ channel in heart. Nature 325(6102):321–326
Lowery-Gionta EG, Marcinkiewcz CA, Kash TL (2015) Functional alterations in the dorsal raphe nucleus following acute and chronic ethanol exposure. Neuropsychopharmacology 40(3):590–600
Luscher C, Slesinger PA (2010) Emerging roles for G protein-gated inwardly rectifying potassium (GIRK) channels in health and disease. Nat Rev Neurosci 11(5):301–315
Maccioni P, Colombo G (2009) Role of the GABA(B) receptor in alcohol-seeking and drinking behavior. Alcohol 43(7):555–558
Maffie J, Rudy B (2008) Weighing the evidence for a ternary protein complex mediating A-type K+ currents in neurons. J Physiol 586(23):5609–5623
Maguire EP et al (2014) Extrasynaptic glycine receptors of rodent dorsal raphe serotonergic neurons: a sensitive target for ethanol. Neuropsychopharmacology 39(5):1232–1244
Marcinkiewcz CA et al (2015) Ethanol induced adaptations in 5-HT2c receptor signaling in the bed nucleus of the stria terminalis: implications for anxiety during ethanol withdrawal. Neuropharmacology 89:157–167
Marrion NV (1997) Control of M-current. Annu Rev Physiol 59:483–504
Marty A, Neher E (1985) Potassium channels in cultured bovine adrenal chromaffin cells. J Physiol 367:117–141
Marty VN, Spigelman I (2012) Long-lasting alterations in membrane properties, k(+) currents, and glutamatergic synaptic currents of nucleus accumbens medium spiny neurons in a rat model of alcohol dependence. Front Neurosci 6:86
Mateos-Aparicio P, Murphy R, Storm JF (2014) Complementary functions of SK and Kv7/M potassium channels in excitability control and synaptic integration in rat hippocampal dentate granule cells. J Physiol 592(4):669–693
McCrossan ZA, Abbott GW (2004) The MinK-related peptides. Neuropharmacology 47(6):787–821
McGeary J (2009) The DRD4 exon 3 VNTR polymorphism and addiction-related phenotypes: a review. Pharmacol Biochem Behav 93(3):222–229
McGuier NS et al (2016) Kv7 channels in the nucleus accumbens are altered by chronic drinking and are targets for reducing alcohol consumption. Addict Biol 21(6):1097–1112
Metten P et al (2014) Dual-trait selection for ethanol consumption and withdrawal: genetic and transcriptional network effects. Alcohol Clin Exp Res 38(12):2915–2924
Miller C (2000) An overview of the potassium channel family. Genome Biol 1(4):REVIEWS0004
Miller MJ et al (2001) Nuclear localization and dominant-negative suppression by a mutant SKCa3 N-terminal channel fragment identified in a patient with schizophrenia. J Biol Chem 276(30):27753–27756
Moore SD, Madamba SG, Siggins GR (1990) Ethanol diminishes a voltage-dependent K+ current, the M-current, in CA1 hippocampal pyramidal neurons in vitro. Brain Res 516(2):222–228
Morley KC et al (2014) Baclofen for the treatment of alcohol dependence and possible role of comorbid anxiety. Alcohol Alcohol 49(6):654–660
Mrejeru A et al (2015) A subset of ventral tegmental area dopamine neurons responds to acute ethanol. Neuroscience 290:649–658
Mulholland PJ (2012) K(Ca)2 channels: novel therapeutic targets for treating alcohol withdrawal and escalation of alcohol consumption. Alcohol 46(4):309–315
Mulholland PJ et al (2009) Sizing up ethanol-induced plasticity: the role of small and large conductance calcium-activated potassium channels. Alcohol Clin Exp Res 33(7):1125–1135
Mulholland PJ et al (2011) Small conductance calcium-activated potassium type 2 channels regulate alcohol-associated plasticity of glutamatergic synapses. Biol Psychiatry 69(7):625–632
Mulholland PJ, Chandler LJ, Kalivas PW (2016) Signals from the fourth dimension regulate drug relapse. Trends Neurosci 39(7):472–485
Ngo-Anh TJ et al (2005) SK channels and NMDA receptors form a Ca2+-mediated feedback loop in dendritic spines. Nat Neurosci 8(5):642–649
NIAAA (2016) Alcohol facts and statistics. www.niaaa.nih.gov/alcohol-health/
Nimitvilai S et al (2016) Chronic intermittent ethanol exposure enhances the excitability and synaptic plasticity of lateral orbitofrontal cortex neurons and induces a tolerance to the acute inhibitory actions of ethanol. Neuropsychopharmacology 41(4):1112–1127
Nimitvilai S et al (2017a) Orbitofrontal neuroadaptations and cross-species synaptic biomarkers in heavy-drinking macaques. J Neurosci 37(13):3646–3660
Nimitvilai S et al (2017b) Ethanol dependence abolishes monoamine and GIRK (Kir3) channel inhibition of orbitofrontal cortex excitability. Neuropsychopharmacology 42(9):1800–1812
Otis JM et al (2017) Prefrontal cortex output circuits guide reward seeking through divergent cue encoding. Nature 543(7643):103–107
Padula AE et al (2013) Novel anticonvulsants for reducing alcohol consumption: a review of evidence from preclinical rodent drinking models. OA Alcohol 1(1):2
Padula AE et al (2015) KCNN genes that encode small-conductance Ca2+-activated K+ channels influence alcohol and drug addiction. Neuropsychopharmacology 40(8):1928–1939
Patil N et al (1995) A potassium channel mutation in weaver mice implicates membrane excitability in granule cell differentiation. Nat Genet 11(2):126–129
Perra S et al (2011) In vivo ethanol experience increases D(2) autoinhibition in the ventral tegmental area. Neuropsychopharmacology 36(5):993–1002
Perry CA et al (2008) Predisposition to late-onset obesity in GIRK4 knockout mice. Proc Natl Acad Sci U S A 105(23):8148–8153
Philip VM et al (2010) High-throughput behavioral phenotyping in the expanded panel of BXD recombinant inbred strains. Genes Brain Behav 9(2):129–159
Pleil KE et al (2015) Effects of chronic ethanol exposure on neuronal function in the prefrontal cortex and extended amygdala. Neuropharmacology 99:735–749
Ponomarev I et al (2012) Gene coexpression networks in human brain identify epigenetic modifications in alcohol dependence. J Neurosci 32(5):1884–1897
Radcliffe RA et al (2004) Quantitative trait loci mapping for ethanol sensitivity and neurotensin receptor density in an F2 intercross derived from inbred high and low alcohol sensitivity selectively bred rat lines. Alcohol Clin Exp Res 28(12):1796–1804
Renteria R, Buske TR, Morrisett RA (2017) Long-term subregion-specific encoding of enhanced ethanol intake by D1DR medium spiny neurons of the nucleus accumbens. Addict Biol. https://doi.org/10.1111/adb.12526
Reuveny E et al (1994) Activation of the cloned muscarinic potassium channel by G protein beta gamma subunits. Nature 370(6485):143–146
Rinker JA, Mulholland PJ (2017) Promising pharmacogenetic targets for treating alcohol use disorder: evidence from preclinical models. Pharmacogenomics 18(6):555–570
Rinker JA et al (2017) Differential potassium channel gene regulation in BXD mice reveals novel targets for pharmacogenetic therapies to reduce heavy alcohol drinking. Alcohol 58:33–45
Risinger FO, Cunningham CL (1998) Ethanol-induced conditioned taste aversion in BXD recombinant inbred mice. Alcohol Clin Exp Res 22(6):1234–1244
Roura-Ferrer M et al (2010) Impact of KCNE subunits on KCNQ1 (Kv7.1) channel membrane surface targeting. J Cell Physiol 225(3):692–700
Saccone SF et al (2007) Cholinergic nicotinic receptor genes implicated in a nicotine dependence association study targeting 348 candidate genes with 3713 SNPs. Hum Mol Genet 16(1):36–49
Schilaty ND et al (2014) Acute ethanol inhibits dopamine release in the nucleus accumbens via alpha6 nicotinic acetylcholine receptors. J Pharmacol Exp Ther 349(3):559–567
Schroeder BC et al (1998) Moderate loss of function of cyclic-AMP-modulated KCNQ2/KCNQ3 K+ channels causes epilepsy. Nature 396(6712):687–690
Schroeder BC et al (2000) KCNQ5, a novel potassium channel broadly expressed in brain, mediates M-type currents. J Biol Chem 275(31):24089–24095
Schulz R et al (2012) Network excitability in a model of chronic temporal lobe epilepsy critically depends on SK channel-mediated AHP currents. Neurobiol Dis 45(1):337–347
Schumacher MA et al (2001) Structure of the gating domain of a Ca2+-activated K+ channel complexed with Ca2+/calmodulin. Nature 410(6832):1120–1124
Sehgal M et al (2013) Learning to learn – intrinsic plasticity as a metaplasticity mechanism for memory formation. Neurobiol Learn Mem 105:186–199
Shah M et al (2002) Molecular correlates of the M-current in cultured rat hippocampal neurons. J Physiol 544(Pt 1):29–37
Shah A et al (2017) The lateral habenula and alcohol: role of glutamate and M-type potassium channels. Pharmacol Biochem Behav 162:94–102
Signorini S et al (1997) Normal cerebellar development but susceptibility to seizures in mice lacking G protein-coupled, inwardly rectifying K+ channel GIRK2. Proc Natl Acad Sci U S A 94(3):923–927
Singh NA et al (2008) Mouse models of human KCNQ2 and KCNQ3 mutations for benign familial neonatal convulsions show seizures and neuronal plasticity without synaptic reorganization. J Physiol 586(14):3405–3423
Stocker M (2004) Ca(2+)-activated K+ channels: molecular determinants and function of the SK family. Nat Rev Neurosci 5(10):758–770
Sturgess JE et al (2011) Pharmacogenetics of alcohol, nicotine and drug addiction treatments. Addict Biol 16(3):357–376
Substance Abuse and Mental Health Administration (SAMHSA) (2015) National Survey on Drug Use and Health (NSDUH) 2015
Syme CA et al (2000) Pharmacological activation of cloned intermediate- and small-conductance Ca(2+)-activated K(+) channels. Am J Physiol Cell Physiol 278(3):C570–C581
Tarantino LM et al (1998) Confirmation of quantitative trait loci for alcohol preference in mice. Alcohol Clin Exp Res 22(5):1099–1105
Trantham-Davidson H et al (2014) Chronic alcohol disrupts dopamine receptor activity and the cognitive function of the medial prefrontal cortex. J Neurosci 34(10):3706–3718
Vandenberg JN, Mann C, Perry S (2015) Reference module in biomedical sciences. Choice: Curr Rev Acad Libraries 52(10):1633–1634
Vergara C et al (1998) Calcium-activated potassium channels. Curr Opin Neurobiol 8(3):321–329
Wang HS, McKinnon D (1995) Potassium currents in rat prevertebral and paravertebral sympathetic neurones: control of firing properties. J Physiol 485(Pt 2):319–335
Wang HS et al (1998) KCNQ2 and KCNQ3 potassium channel subunits: molecular correlates of the M-channel. Science 282(5395):1890–1893
Wittekindt OH et al (2004) An apamin- and scyllatoxin-insensitive isoform of the human SK3 channel. Mol Pharmacol 65(3):788–801
Wu X et al (2013) Effects of morphine withdrawal on the membrane properties of medium spiny neurons in the nucleus accumbens shell. Brain Res Bull 90:92–99
Wydeven N et al (2014) Mechanisms underlying the activation of G-protein-gated inwardly rectifying K+ (GIRK) channels by the novel anxiolytic drug, ML297. Proc Natl Acad Sci U S A 111(29):10755–10760
Xia XM et al (1998) Mechanism of calcium gating in small-conductance calcium-activated potassium channels. Nature 395(6701):503–507
Xiao J, Zhen XG, Yang J (2003) Localization of PIP2 activation gate in inward rectifier K+ channels. Nat Neurosci 6(8):811–818
Yamada K et al (2012) Association study of the KCNJ3 gene as a susceptibility candidate for schizophrenia in the Chinese population. Hum Genet 131(3):443–451
Zaika O et al (2006) Angiotensin II regulates neuronal excitability via phosphatidylinositol 4,5-bisphosphate-dependent modulation of Kv7 (M-type) K+ channels. J Physiol 575(Pt 1):49–67
Zandany N et al (2015) Alternative splicing modulates Kv channel clustering through a molecular ball and chain mechanism. Nat Commun 6:6488
Zhang X et al (2009) Divalent cations slow activation of EAG family K+ channels through direct binding to S4. Biophys J 97(1):110–120
Zwierzynska E, Krupa A, Pietrzak B (2015) A pharmaco-EEG study of the interaction between ethanol and retigabine in rabbits. Am J Drug Alcohol Abuse 41(2):153–160
Zwierzynska E, Andrzejczak D, Pietrzak B (2016) Does retigabine affect the development of alcohol dependence? – a pharmaco-EEG study. Neurosci Lett 611:6–13
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG
About this chapter
Cite this chapter
Cannady, R., Rinker, J.A., Nimitvilai, S., Woodward, J.J., Mulholland, P.J. (2018). Chronic Alcohol, Intrinsic Excitability, and Potassium Channels: Neuroadaptations and Drinking Behavior. In: Grant, K., Lovinger, D. (eds) The Neuropharmacology of Alcohol . Handbook of Experimental Pharmacology, vol 248. Springer, Cham. https://doi.org/10.1007/164_2017_90
Download citation
DOI: https://doi.org/10.1007/164_2017_90
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-96522-2
Online ISBN: 978-3-319-96523-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)