Abstract
Various drug delivery systems are being rapidly developed for controlled drug release, improved efficacy, and reduced side effects with the goal of improving quality of life for patients and curing disease. Poly(lactic acid) (PLA) possesses numerous advantages compared with other polymers, including biocompatibility, biodegradability, low cost, environmental friendliness, and easily modified mechanical properties. These properties make PLA a promising polymer for biomedical applications. This review introduces the specific characteristics of PLA that enable its application for controlled drug delivery and describes different forms of PLA used for drug delivery, including nanoparticles, microspheres, hydrogels, electrospun fibers, and scaffolds. Previous work is summarized and future development is discussed.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
There are serious problems associated with the therapeutic use of small molecule drugs (SMDs), including insolubility, instability (e.g., rapid degradation in physiological environments), sequestration within the blood space by endothelial barriers, and poor uptake by tissues and cells [1,2,3]. A wide variety of drug delivery systems (DDSs) have been designed to overcome these challenges, which are of crucial importance in healthcare and clinical applications [3,4,5,6,7]. Drug delivery is a process, by which specific drugs are delivered to a target area in the organism (i.e., animals or humans) to achieve a therapeutic effect [8]. Controlled DDSs can be specially designed to increase the solubility of SMDs, improve stability by preventing SMD degradation under physiological conditions, reduce side effects by targeting the lesion regions without affecting healthy sites, and maintain sustained drug release at optimal doses [9, 10]. Moreover, the use of DDSs removes the need for frequent administration, which is the primary cause of various degrees of bodily injury. As a result, DDSs have shown significant efficacy in improving quality of life in patients. Packaging an existing clinically approved drug into an effective delivery system as an advanced formulation can also reduce the economic cost and time required for new drug development.
In controlled DDSs, the carriers are as important as the bioactive drugs. Successful delivery of drugs to target tissues requires that the carriers sustain good stability during administration. Additionally, these carriers should be biocompatible. A variety of polymers, including natural and synthetic polymers, can serve as drug carriers, but few can meet the requirements of acceptable biocompatibility, biodegradability, and absorbability [9, 11]. Recently, the field has begun to pay more attention to the use of biodegradable polymers as drug carriers because of their extraordinary performance [9, 12]. Many forms of carriers, including nanoparticles (NPs), microspheres, hydrogels, electrospun fibers, and scaffolds, have been investigated for the delivery of different types of drugs or for adaptation to different situations in in vivo microenvironments.
Among the numerous biodegradable polymers that have been used as drug carriers, poly(lactic acid) (PLA) is one of the most promising candidates (Fig. 1). PLA can be fabricated into various forms for controlled drug release, including NPs [13, 14], microspheres [15], hydrogels [16], electrospun fibers [17], and scaffolds [18]. Its various advantages include being environmentally friendly, existing in multiple forms, exhibiting good biocompatibility and biodegradability, sustaining long drug retention times, having easily modified mechanical properties, and being low cost [19, 20].
PLA-based carriers for controlled drug delivery. (a) TEM image of PLA nanoparticles [13]. (b–e) SEM images of (b) microspheres [15], (c) hydrogels [119], (d) electrospun fibers [17], and (e) scaffolds [18]. Reproduced from [13] with permission of Springer, from [15, 119] with permission of Elsevier, from [17] with permission of American Chemical Society, and from [18] with permission of John Wiley and Sons, respectively
PLA is derived from renewable sources, such as sugar, maize, potato, sugarcane, and beet [21]. It is prepared by different polymerizations of lactic acid, which is in turn typically produced by bacterial fermentation [22] or glycolysis [23], with little environmental pollution. Furthermore, lactic acid, the degradation product of PLA, can be removed completely in vivo. Therefore, PLA is a green biomaterial.
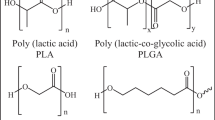
Lactic acid is a chiral molecule existing as l and d isomers (Fig. 2). PLA exists primarily in three forms: poly(l-lactic acid) (PLLA), poly(d-lactic acid) (PDLA), and poly(d,l-lactic acid) (PDLLA) [22, 24, 25]. Of these, PLLA has attracted the most attention in DDSs as a result of its favorable mechanical properties. However, because PLLA has a long degradation time and is therefore likely to cause inflammatory responses in vivo, it is often used in combination with polymerization of d,l-lactide monomers [22, 26].
Biocompatibility is a highly desirable trait in DDSs and has been the focus of much research. PLA is biodegradable with high biocompatibility and does not circulate in vivo for extended periods of time. Although PLA is hydrophobic, it can undergo scission, primarily by hydrolysis, to monomeric units of lactic acid in vivo even in the absence of enzymes [21]. This degradation process leaves no foreign or toxic substances because lactic acid is a natural intermediate of carbohydrate metabolism [27, 28]. As a result, PLA is highly biocompatible, biodegradable, and bioresorbable in vivo.
Another advantage of biodegradable PLA DDSs is their long retention time. Compared with other polymers, PLA possesses better biodegradability, which is relatively moderate and can be properly controlled. This biodegradability mainly relies on the crystallinity, morphology, and relative molecular mass of the polymer [20]. PLA tends to be crystalline when the PLLA content is higher than 90%, whereas the less optically pure form is amorphous [22]. Auras et al. [29] reported that the densities of solid PLLA, PDLLA, crystalline PLA, and amorphous PLA are 1.36, 1.33, 1.36, and 1.25 g cm−3, respectively. PLA of low molecular weight is preferred for use as a drug carrier because it has a shorter degradation time, giving better release properties [30]. For drug carriers composed of nondegradable polymers, the drug release rate slows gradually with a reduction in the amount of encapsulated drug. PLA systems avoid this problem because the structure of PLA gradually loosens with continued degradation in vivo. As a result, the resistance to drug diffusion out of the PLA carrier is reduced, and the drug release rate is upregulated. Because the increased drug release rate counteracts the reduced drug concentration, a long-term constant release of drug from the carrier can be achieved [31].
The chemical and physical properties of PLA, especially its biocompatibility and biodegradability, are easily influenced by adding different surfactants or changing the molecular weight, size, shape, temperature, and moisture [21, 22, 29]. This enables the creation of desired DDSs under specific conditions with different formulations [32,33,34,35,36,37,38]. It is also possible to control the distribution and release behavior of drugs in the PLA devices. As a typical example, Fernandez et al. [39] extracted proanthocyanidins from grapes and stabilized them with PDLA using an emulsion-evaporation method. They evaluated three factors in the formulation: sonication time for the emulsion process, loading of grape extracts, and concentration of stabilizing agent. They concluded that the extract load and stabilizer concentration were closely related to the properties of this drug model. Wei et al. [25] loaded oxaliplatin (OXA) into NP and compared the drug delivery characteristics of poly(ethylene glycol) (PEG)−PLA NP with that of PLA-only NP. They found that the OXA concentration in the tumor in the PEG−PLA NP group was higher than that in the PLA NP group. Furthermore, less OXA accumulated in the liver and lungs after PEG−PLA NP was administered. The results indicated that the PEG-modified platform possessed good drug retention ability and could deliver more drugs to the target sites. Many others have demonstrated that PEG can resist nonspecific absorption of proteins in the blood [40,41,42,43,44]. However, PEG has some limitations as a coating for PLA DDSs, particularly in achieving effective PEG surface densities [45, 46]. To combat this problem, Deng et al. [47] applied hyperbranched polyglycerol (HPG) as an alternative coating of PLA NP. They found that the antitumor agent camptothecin (CPT) had a longer blood circulation time, higher stability, less accumulation in the liver, and better therapeutic effectiveness against tumors in the HPG−PLA NP group than that in the PEG−PLA NP group. They concluded that HPG is a better surface coating for NPs than PEG for applications in drug delivery. In a study performed by Yamakawa et al. [48], neurotensin analogue-loaded PDLLA microspheres with different PLA molecular weights were prepared. The authors found that when the molecular weight changed, the rate of initial burst and the length of time, over which the drug was released, varied. Zeng et al. [49] examined the influence of surfactants on the diameter of electrospun PLLA fibers by adding cationic, anionic, and nonionic surfactants, that is, triethyl benzyl ammonium chloride, sodium dodecyl sulfate, and aliphatic PPO−PEO ether, respectively. Rifampin (RIF; a drug for tuberculosis) and paclitaxel (PTX; an anticancer drug) were used as model drugs and loaded into PLLA fibers. It was revealed that the addition of each of the three types of surfactant could reduce the diameter and narrow the distribution of electrospun fibers. Furthermore, RIF contained in these fibrous mats could be released constantly with no burst release behavior.
All the advantages of PLA make it a popular drug carrier matrix, and it has been approved by the Food and Drug Administration (FDA) for in vivo applications in humans [21]. The next section gives details on different forms of PLA for controlled drug delivery, including NPs, microspheres, hydrogels, electrospun fibers, and scaffolds.
2 PLA-Based Carriers for Drug Delivery
With the assistance of DDSs, it is possible to achieve a much greater therapeutic effect for many clinical applications, including a reduced pain burden and improved quality of life. It is also possible to limit side effects by controlling drug release rate and specifically transporting drugs to target sites [50]. A variety of advanced delivery systems have been developed to achieve high efficiency and safety in drug delivery and overcome the disadvantages of traditional formulations [24, 25]. The biomedical applications of PLA-based drug carriers discussed in this section are summarized in Table 1.
2.1 Nanoparticles
NPs are spherical skeletons composed of polymer matrix, with diameters ranging from 1 to 500 nm.
2.1.1 Properties
Of the various categories of drug carrier systems, NPs have attracted the most attention because of their unique properties [34, 51]. NPs have many advantages compared with other DDS formulations. First, NPs have a high drug retention rate, which can prevent inactivation in vivo; second, NPs allow well-controlled drug distribution by delivering drugs to disease sites with few side effects in other areas; and third, NPs allow long-term drug release [52, 53]. A variety of biocompatible and biodegradable biomaterials, especially PLA, have been used as raw materials for NPs, thereby increasing their clinical utility.
2.1.2 Applications
PLA NPs have been the subject of much interest as DDSs to access the central nervous system (CNS) [54]. The blood–brain barrier (BBB) is a significant challenge for drug delivery to the CNS, because it is composed of special endothelial cells that form tight junctions, blocking drug transport into the CNS [34, 52]. Drugs are typically unable to pass through the BBB in free form [34]. However, by varying the molecular weight of PLA and using surfactants or surface modifications, the PLA NPs loaded with different drugs can be successfully delivered to the CNS. For example, Liu et al. [13] prepared the breviscapine (BVP)-loaded PDLLA NPs of different sizes and investigated the distribution of BVP in rats. The mean diameters of these NPs were 177 and 319 nm. The BVP-loaded PDLLA NPs could not only avoid capture by the reticuloendothelial system (RES), but also penetrated the BBB and enhanced accumulation of BVP in the brain. Additionally, a larger NP could deliver more BVP to the CNS. In another study, the PLLA NP loaded with ritonavir (a protease inhibitor) was attached to a trans-activating transcriptional activator (TAT) peptide [55]. The results indicated that TAT increased the transport of NPs across the BBB. In addition, the TAT-conjugated NPs were able to maintain a therapeutic drug level in the brain, which could be effective in controlling viral replication in the CNS. In a recent study, Sun et al. [56] demonstrated that the coating was necessary for drug delivery across the BBB. Compared with the modified PLA NP, the unmodified platform was able to deliver only a small amount of drug to the brain [34, 57,58,59]. However, in all cases, less than 1% of the administered dose reached the CNS, far less than enough to achieve a significant therapeutic effect.
By-passing the BBB is another efficient way of achieving drug delivery for CNS diseases. Intranasal delivery is one such method for circumventing the BBB and not only provides rapid access to the CNS, but also avoids first-pass hepatic clearance and tends to avoid systemic side effects [60]. Unfortunately, the brain concentration of drugs delivered intranasally is often still too low to achieve a desired therapeutic effect. NPs may help to solve this problem: loading drugs into NPs can protect them from degradation in the nasal enzymatic microenvironment. For example, Jain et al. [61] delivered methotrexate (MTX) by designing the thermosensitive PLA NP that exhibited enhanced residence time in the nasal cavity and by-passed the BBB. The results indicated that more NP was detected in the brain than free drug. Xia et al. [62] applied low molecular weight protamine (LMWP) to decorate the surface of methoxy poly(ethylene glycol) (mPEG)−PLA NP and determined the percentage of drug delivered to the brain after intranasal administration. Their results showed that the LMWP-modified mPEG−PLA NP could be more effectively delivered to the CNS than the unmodified one.
PLA NPs are also widely used for the diagnosis and treatment of cancers. Li et al. [63] first fabricated PLA NP encapsulating Endostar, then conjugated the near-infrared (NIR) dye IRDye 800CW and GX1 peptide onto NP (IGPNE). With NIR, fluorescence molecular imaging, and bioluminescence imaging, they were able to use this composite to attain a real-time image of U87MG tumor. Furthermore, IGPNE accumulated in the tumor site and had an antiangiogenic therapeutic effect. Miller et al. [64] incorporated dechloro-4-iodo-fenofibrate (IFF) into the core of PEGylated PDLLA (PEG−PDLLA) micelle and investigated its effect on tumor targeting, as shown in Fig. 3a. The results showed that the separation process of the drug from the carrier was extremely fast and that the drug accumulated more in the tumor than did the carrier (Fig. 3b, c). Mishra et al. [65] investigated the angiogenesis inhibition effect of mPEG-b-PDLLA micelle loaded with PTX and rapamycin (RAP). The results indicated that the PTX−RAP dual drug micelle had an enhanced antiangiogenesis effect that was promising for cancer chemotherapy. Other studies have also reported the anticancer effects of PLA DDSs loaded with chemotherapy drugs [66,67,68,69,70,71].
Micelle composition and metabolism in vivo [64]. (a) The core component consists of PDLLA, which hosts the radioactively labeled drug 131IFF or 125IFF. The particle shell is covered with PEG. The surfaces show a mixture of 111In-DOTA-HN-PEG and H3CO-PEG. (b) Biodistribution (%ID) of polymer carrier and (c) IFF drug payload (%ID). All statistical data are presented as mean ± standard deviation (SD; n = 3). Reproduced from [64] with permission of Elsevier
PLA NPs also play an important role in other areas, such as live cell imaging and treatment, and diagnosis of various other diseases. For instance, Contreras et al. [72] prepared the PLA NP based on difluoroboron dibenzoylmethane dye (BF2dbmPLA). They found that the BF2dbmPLA NP could be internalized into cultured HeLa cells by endocytosis and that the NP retained its fluorescence property, suggesting that the unique optical property of this complex could be harnessed for live cell imaging. In another study, the Fab′-bearing siRNA tumor necrosis factor α (TNFα)-loaded PEG−PLA NP was prepared and studied for use in inflammatory bowel disease [73]. The in vivo experiment indicated that colitis was inhibited efficiently as the TNFα-siRNA-loaded NP was released to and accumulated in the diseased area. Babak et al. [74] prepared PLA DDSs for long-term antibacterial applications. They designed the composite platforms of poly(ε-caprolactone) (PCL) with different concentrations of the triclosan (TC)-loaded PLA NPs and investigated their drug release properties and antibacterial effects. Because of the advantages of PLA, including its higher glass transition temperature (T g) and lower flexibility, this biocomposite showed reduced burst release of TC and an antibacterial effect that lasted approximately 2 years.
Various other studies have shown the therapeutic effect of drugs loaded into PLA NPs against many diseases, including hearing loss by cisplatin (CDDP) chemotherapy [75], periodontitis [44, 76], colitis [73], acute hepatic failure [73], bone fracture [77], dermatitis [78], autoimmune uveoretinitis [79], and Chagas disease [80].
2.2 Microspheres
Microspheres are another kind of fine particle dispersion system in which drug molecules are dispersed or adsorbed. The diameters of microspheres range from 1 to 250 μm, and the main difference between NPs and microspheres is their size [81]. Because microspheres are larger than NPs in size, they are unlikely to cross biological barriers. Furthermore, microspheres tend to stay in place if injected into certain tissues. Additionally, microspheres can be only taken up by phagocytosis, whereas NPs can be taken up by both phagocytosis and pinocytosis [81]. For these reasons, microspheres are most widely used in cancer treatment as DDSs. However, NPs can be applied in many medical areas, as discussed in Sect. 2.1.
2.2.1 Properties
The drug-loaded microspheres can disperse specifically to target tissues in vivo, improving local drug concentrations and reducing systemic side effects. As a result of their excellent biocompatibility and biodegradability, PLA and its copolymers are the most frequently used polymers for DDSs. However, discovering which factors affect the rate of microsphere degradation and achieving more appropriate degradation behaviors is extremely important. Previous studies have found that the degradation of microspheres is associated with molecular weight, polymer crystallinity, microsphere size, and the presence of drugs [82]. Li et al. [83] found that a copolymer with 50% lactic acid and 50% glycolic acid (50:50 PLGA) has a shorter half-life period than 75:25 PLGA, and degrades faster than PLA.
The PLA-based microspheres loaded with different drugs are primarily delivered by intravascular injection, subcutaneous injection, in situ injection, and oral administration. Various types of drugs have been loaded into microspheres for medical applications, including anticancer drugs, antibiotics, antituberculosis drugs, antiparasitic drugs, asthma drugs, and vaccines. Of these, anticancer therapeutics have been most studied.
The microspheres composed of biocompatible and biodegradable polymers like PLA can also increase the stability and bioavailability of drugs, reduce gastrointestinal irritation, prolong the duration of drug release, and deliver drugs to target sites [84, 85]. Guan et al. [86] formulated the PLA microspheres loaded with lovastatin (LVT) for oral administration, and evaluated the in vitro and in vivo characteristics of the microspheres. They concluded that PLA microspheres could significantly prolong the circulation time of LVT in vivo and also significantly increase the relative bioavailability of LVT. Ding et al. [15] evaluated the drug loading ability and drug release behavior of amorphous calcium phosphate (ACP) microspheres containing mPEG–PDLLA. The ACP porous hollow microspheres were found to have a high docetaxel (DTX) loading capacity, thus causing more damage to tumor cells. Lu et al. [87] prepared the RIF-loaded PLA microspheres using the electrospray technique and showed that drug release from the microspheres lasted for more than 60 h in vitro. In another study, Chen et al. [88] formulated the emodin (ED)-loaded PLA microspheres and studied their lung-targeting effect. Apart from determining the optimal parameters for formulation and sustained drug release, this research also indicated that ED was mainly delivered to lung tissue without causing toxicity to the liver and kidneys. Mirella et al. [89] compared PLLA microspheres with poly(lactide-co-glycolide) (PLGA) microspheres prepared by the same technique. They concluded that the PLLA microspheres had the best physical properties, the highest drug loading content, and the most efficient drug release behavior without any burst effect. Other studies have similarly shown that PLA microspheres increase drug release time, stability, and bioactivity, all of which increase their medical applicability [90,91,92,93,94,95,96].
2.2.2 Applications
PLA microspheres are primarily used for delivering anticancer drugs to target sites. In a study conducted by Chen et al. [97], the in vitro and in vivo antitumor efficacy of magnetic composite microspheres of the MTX-loaded Fe3O4-PLLA-PEG-PLLA (MMCMs) was investigated. The results from experiments at the cellular, molecular, and integrated level indicated that MMCMs with magnetic induction possess the ability to accumulate MTX in tumor tissue, leading to apoptosis of the tumor cells. Zhao et al. [98] investigated the antitumor efficacy of dextran/PLGA−PLA core/shell microspheres loaded with recombinant interleukin-2 (rIL-2), as depicted in Fig. 4. They injected a single dose of microspheres intratumorally in a subcutaneous colon carcinoma BALB/c mouse model and demonstrated that the antitumor effect of the microspheres was promising. Tumor growth was significantly suppressed in the rIL-2-loaded microsphere group (Fig. 4). Zhou et al. [99] explored the use of epirubicin (EPI)-loaded PDLLA microspheres for treating hepatocellular carcinoma (HCC) in mice. Compared with the blank microsphere group and the normal saline control group, the group treated with the EPI-loaded PDLLA microspheres had the longest survival time, which indicated that the PLA microspheres combined with EPI are highly effective in treating HCC in mice. In another study, the PLLA microspheres containing 5-fluorouracil (5-FU) were prepared [100]. The authors found that the microspheres were primarily located in the liver and were more efficient than free 5-FU in prolonging the survival time of rats with liver tumors. In a study performed by Kuang et al. [101], the PDLLA microspheres loaded with CDDP were injected into mammary tumors in rats. These microspheres had a similar antitumor effect as aqueous CDDP solution in that the tumor became significantly smaller or disappeared 16 days after treatment. Fascinatingly, the CDDP-loaded PDLLA microspheres showed less nephrotoxicity than the aqueous CDDP solution. Other studies have also revealed the anticancer effects of drug-loaded PLA microspheres and provided an experimental basis for further therapies [102,103,104,105,106].
Properties and antitumor efficacy of rIL-2-loaded dextran/PLGA−PLA core/shell microsphere [98]. (a) SEM image of drug-loaded microsphere. (b) In vitro cumulative rlL-2 release profile of loading microsphere in phosphate-buffered saline (PBS) of pH 7.4 at 37 °C. Error bars represent the SD (n = 3). (c–f) In vivo antitumor efficacy of rlL-2-loaded dextran/PLGA−PLA core/shell microsphere toward BALB/c mice bearing colon carcinoma. All mice were euthanized on day 22, and tumors were stripped, weighed, and photographed. (c) Representative photographs of tumors. (d) Representative photographs of BALB/c mice bearing tumors. (e) Tumor volumes in the different groups (blank microsphere, rIL-2 solution, and rIL-2-loaded microsphere) as a function of days post-treatment. Arrow represents the day that each formulation was administrated for the first time. (f) Tumor weights after euthanizing on day 22. Data are expressed as mean ± SD (n = 4). Reproduced from [98] with permission of Elsevier
In addition to anticancer drugs, other kinds of drugs, including antibiotics [107,108,109], anti-TB drugs [110], asthma drugs [111, 112], and vaccines [113,114,115,116,117,118] have been loaded into PLA microspheres. Consequently, PLA microspheres are another widely used DDS for medical applications.
2.3 Hydrogels
Hydrogels, a type of three-dimensional (3D) polymer network containing significant amounts of water [119, 120], have received increasing attention in the fields of drug delivery and tissue engineering [121, 122].
2.3.1 Properties
The environmentally sensitive hydrogels are widely studied because of their biocompatibility and resemblance to biological tissues [123]. As a result of its biocompatibility, PLA has gained favor for the construction of hydrogels. The most attractive feature of PLA hydrogels is their thermal sensitivity: the PLA copolymer is soluble at room temperature and changes into a gel at body temperature. In addition, PLA hydrogels have good controlled drug release properties and can maintain drug release for over a month. However, crystallization and subsequent precipitation in solution is a major challenge for PLA use in hydrogel fabrication. To solve this problem, PLGA−PEG systems are often chosen. The PLGA−PEG solution is liquid at room temperature and immediately forms a hydrogel at body temperature. Furthermore, its mechanical properties are superior to those of PLA-only hydrogels [124].
Compared with hydrophobic materials, hydrogels interact less strongly with immobilized biomolecules. Because of the biocompatibility of PLA and the high water content of hydrogels, the use of PLA hydrogels is ideal for sustained drug release. Various studies have demonstrated this idea: for example, Lai et al. [125] developed a thermosensitive methoxy poly(ethylene glycol)-co-poly(lactic acid-co-aromatic anhydride) (mPEG−PLCPHA) hydrogel for cefazolin (CEF) delivery that exhibited long-term antibacterial effects. Wang et al. [119] studied the safety of a pH-sensitive hydrogel consisting of mPEG, PLA, and itaconic acid, and concluded that it might be used as a safe method for drug delivery. Other studies have also provided convincing evidence that PLA hydrogels can serve as efficient DDSs [126,127,128].
2.3.2 Applications
PLA hydrogels have already shown great potential as DDSs for medical applications. In one study, researchers dispersed NPs of DTX and LL-37 peptide into a PLA−L64−PLA thermosensitive hydrogel and evaluated the intraperitoneal effect of this composite in a colorectal peritoneal carcinomatosis HCT116 model [129]. They found that the hydrogel showed significant antitumor efficacy both in vitro and in vivo. Manaka et al. [130] evaluated the bone formation effect of a PDLLA−p-dioxanone−PEG hydrogel carrier for siRNA delivery. The hydrogel was found to be safe and efficient for siRNA delivery, and could promote new bone formation (Fig. 5). Devin et al. [131] synthesized a degradable methacrylate PLA hydrogel loaded with bioactive basic fibroblast growth factor (bFGF) and insulin-like growth factor-1 (IGF-1). This hydrogel loaded with growth factors was injected into infarcts in Lewis rats and found to improve cardiac function and geometry compared with the saline control. The results indicated that PLA hydrogel can act as a carrier of growth factors to influence cardiac remodeling. In another study, a diblock hydrogel of mPEG−PLA was studied for adhesion prevention [122]. The results showed that the hydrogel system was equally effective compared with the commercial anti-adhesion product. Furthermore, this hydrogel system could be more promising for adhesion prevention if it were loaded with antifibrosis and anti-inflammatory drugs.
Characterization of ectopic bone formation [130]. (a) Soft X-ray examination of newly formed ectopic bone induced by hydrogel containing 2.5 mg rhBMP-2 without Noggin siRNA [BMP(+)siRNA(−)] or with Noggin [BMP(+)siRNA(+)] for 2 weeks. (b) Bone mineral contents of ossicles measured by dual-energy X-ray absorptiometry (DXA). *P < 0.05, compared with control group. (c) Von Kossa and van Gieson staining of sections of undercalcified BMP-induced ectopic bone without or with Noggin siRNA. Bone volume per tissue volume (BV/TV) is expressed as mean. *P < 0.05 compared with control group. Reproduced from [130] with permission of Elsevier
Even though PLA hydrogels are not as widely used as PLA NPs and microspheres, they still play an important role in controlled drug delivery.
2.4 Electrospun Fibers
Electrospinning is a facile and economic technique for producing nanoscale or microscale fibers from different polymers. The fibers can then be used for a variety of biomedical applications [132,133,134,135,136,137].
2.4.1 Properties
Electrospun fibers have attracted increasing attention as DDSs because of their specific advantages, including a high surface to area ratio, which can lead to high drug loading capacity, variable pore size, and mechanical flexibility [135, 136, 138]. Electrospinning is an efficient and simple method of rapidly and reproducibly manufacturing fiber networks incorporating different kinds of drugs [139]. However, hydrophilic water-soluble drugs cannot be directly mixed into solutions of PLA. Fortunately, the techniques of emulsion electrospinning and coaxial electrospinning can be used to encapsulate hydrophilic drugs in the core of the fibers to mitigate the initial burst release of drug [137]. Furthermore, the characteristics of electrospun fibers as DDSs can be modified by biological, chemical, optical, thermal, magnetic, and electric stimuli [139]. As a result, electrospun fibers can be designed to achieve the desired drug transport properties for medical application. PLA electrospun fibers play a significant role in this area.
2.4.2 Applications
Like other PLA DDSs, electrospun PLA fibers are widely used in cancer therapy. Zhang et al. [140] manufactured the PLLA electrospun fibers loaded with 5-FU and OXA for treatment of colorectal cancer. They found that the PLLA electrospun fiber loaded with chemotherapy drugs significantly suppressed tumor growth and prolonged mouse survival time. In another study (Fig. 6), researchers prepared the electrospun PLLA fibers loaded with multiwalled carbon nanotubes (MWCNTs) and doxorubicin (DOX) [141]. As demonstrated in Fig. 6, the combination of photothermal therapy using MWCNTs and chemotherapy induced with DOX greatly suppressed tumor growth, with less damage to nearby normal tissue.
Fabrication of DOX/MWCNT co-loaded electrospun PLA fibers for treatment of U14 cervical cancer in mice [141]. (A) SEM image of co-loaded fibers. (B) TEM of co-loaded fiber, and fluorescence image of (a) PLA fiber, (b) MWCNT-loaded PLA fiber, and (c) co-loaded PLA fiber. (C) Release profiles of DOX from co-loaded fiber in PBS at 37°C without or with NIR irradiation of 2 W/cm2. The column zone indicates when the NIR irradiation was applied. (D) Temperature of tumor region at different time points under NIR irradiation of 1.5 W/cm2. Control group: (a) tumor surface, (b) 3 mm inside tumor. Fiber dressing group: (c) tumor surface, (d) 3 mm inside tumor. (E) Evolution of U14 tumor volumes of KM mice as a function of time. (F) Relative body weight changes with time of U14 tumor-bearing mice. All the fibers were implanted only once in the beginning at an equivalent DOX dose of 0.1 mg and MWCNT dose of 0.1 mg. In groups of M/laser and DM/laser, tumor regions were exposed to NIR irradiation (1.5 W/cm2) for 10 min after fiber dressing for 24 h. Reproduced from [141] with permission of Elsevier
The electrospun PLA fibers loaded with various kinds of drugs are also used for adhesion prevention [137]. Tissue adhesion is one of the most common postoperative complications, in most cases requiring a second operation to remove the adhesions [142]. Electrospun PLA membranes not only act as a physical barrier, but can also be loaded with many kinds of drugs to prevent post-surgical adhesions. Because of the excellent biocompatibility and biodegradability of PLA, antibacterial drugs [142,143,144], anti-inflammatory drugs [145,146,147], drugs that facilitate healing [148], and synergistic combinations [149] have also been loaded into electrospun PLA fibers.
Electrospun PLA fibers can be applied in many other medical fields, including tissue engineering [150,151,152], antibiotic therapy [144, 153,154,155], bone repair [156, 157], and wound healing [158, 159]. For example, Screerekha et al. [150] developed a fibrin-based electrospun composite scaffold that provided a natural environment for cell attachment, migration, and proliferation. The results indicated that the electrospun-based composite was promising for myocardial tissue engineering. In another study, Spasova et al. [155] prepared PLA stereocomplex fibers using an amphiphilic block copolymer and demonstrated good antibacterial properties in experiments on blood cells and pathogenic microorganisms. Ni et al. [156] developed an electrospun PEG/PLA fibrous scaffold to provide an interconnected porous environment for attachment of mesenchymal stem cells (MSCs). The results showed good cell response, excellent osteogenic ability, and outstanding biocompatibility of the electrospun PEG/PLA fibrous composite for bone repair. Kobsa et al. [158] developed a PLA-based electrospun scaffold integrating nucleic acid delivery and studied its effect in the treatment of full thickness wounds. They found that the scaffold could serve as a protective barrier in the early stages of wound healing, as well as induce cell migration and growth.
2.5 Scaffolds
Tissue engineering scaffolds, especially those prepared from biocompatible and biodegradable polymers, are increasingly widely used. The scaffolds loaded with different drugs are crucial for the regeneration of large defects.
2.5.1 Properties
PLA has also become a popular scaffold for tissue engineering, again due to its outstanding biocompatibility and biodegradability. PLA scaffolds can be designed to match the mechanical properties of native tissues [160]. Furthermore, because the concentration of degradation products is reduced with increased porosity, PLA is a favorable material for scaffold fabrication [160].
2.5.2 Applications
PLA is promising for tissue engineering applications, not only as a scaffold material, but also for its drug delivery properties [24, 25, 161]. PLA scaffolds can be implanted at injured sites to support injured tissues and enhance the repair process. By loading drugs into the scaffolds, it is also possible to generate multifunctional PLA scaffolds for various applications [162]. However, release properties are important when scaffolds are also harnessed as DDSs. Many parameters, such as loading method, scaffold properties, and choice of polymer, can all play an important role in the mechanism of drug release, which occurs primarily via desorption, degradation, and diffusion in the electrospun PLA scaffolds [160].
The PLA scaffolds loaded with various agents have been the subject of much research in bone, vascular, and other tissue engineering applications. As a typical example, Zhou et al. [163] exploited a calcium phosphate−PLA composite as a coating for a tantalum porous scaffold (Fig. 7). Vascular endothelial growth factor (VEGF) and transforming growth factor (TGF) were loaded into the scaffold and used for bone defect repair. Their results indicated that the scaffold provided growth factors, physical support, structural guidance, and interfaces for new bone growth, and was therefore useful to guide new bone regeneration. Other studies have also reported the superior effects of various PLA scaffolds for tissue engineering [158, 164,165,166,167,168,169,170,171,172,173]. For example, Hu et al. [166] fabricated a nanofibrous PLLA scaffold for blood vessel regeneration. The results showed that the scaffold preferentially supported the reconstruction of tissue-engineered vascular graft. In another study, Niu et al. [165] developed a microencapsulated chitosan (CM), nanohydroxyapatite/collagen (nHAC), and PLLA-based microsphere–scaffold delivery system. Bone morphogenetic protein-2 (BMP-2)-derived synthetic peptide was incorporated into the synthesized composite. The results showed that the CM/nHAC/PLLA composite can accelerate the regeneration of cancellous bone defect with controlled release of the incorporated peptide. Haddad et al. [168] developed a 3D PLA scaffold with polyallylamine to introduce amine groups, followed by grafting of epidermal growth factor (EGF) onto the scaffold. They found that neural stem-like cells were able to proliferate on the EGF-grafted substrates and might be promising for repair of the CNS. The PLA scaffolds loaded with drugs, such as ibuprofen IBU, alkannin ALK, and curcumin (CUR), are also used for promoting cutaneous wound healing [160]. However, a detailed discussion is beyond the scope of this review on DDSs.
Strategy for preparation and bone defect repair application of porous tantalum scaffold coated with a composite of calcium phosphate and PLA. Reproduced from [163] with permission of Elsevier
3 Conclusions and Perspectives
This review introduces the characteristics of PLA as a promising matrix for DDSs in its five most commonly used forms: NPs, microspheres, hydrogels, electrospun fibers, and scaffolds. The PLA DDSs can effectively deliver drugs to the target sites, reduce drug toxicity, and increase the therapeutic effect. In addition, PLA can be modified to achieve various desired properties in DDSs. As a result, PLA has great potential for DDS development.
The PLA DDSs loaded with different drugs can be used for the treatment of many diseases. For example, the PLA DDSs loaded with anticancer agents like PTX can directly deliver drugs to tumor sites, thereby increasing drug accumulation and retention time in the tumor while reducing systemic side effects. Meanwhile, the PLA DDSs loaded with different cytokines, such as BMP, can control drug release via degradation and play an important role in bone repair. The PLA DDSs loaded with other drugs, including anti-inflammatory agents [107, 108], antihypotensors [174], painkillers [158], and vaccines [113, 114, 118], can also be used for other medical applications.
With the development of biotechnology, peptide and protein drugs have become increasingly prevalent. However, the short retention time of these drugs limits their application, as continuous administration is often impractical. PLA DDSs may be able to improve the application of peptide- and protein-based therapeutics. Furthermore, PLA DDSs can be engineered to be intelligent drug delivery vehicles. For example, a smart PLA glucose monitor may be engineered to be inserted into body tissue and release the proper dose of insulin according to glucose fluctuations.
Although some aspects of PLA DDSs still need to be improved, further research will allow PLA to play an increasingly important role as a matrix promising material for DDSs and provide more efficacious treatment methods for many diseases.
References
Datta M, Via LE, Kamoun WS, Liu C, Chen W, Seano G, Weiner DM, Schimel D, England K, Martin JD, Gao X, Xu L, Barry CE, Jain RK (2015) Anti-vascular endothelial growth factor treatment normalizes tuberculosis granuloma vasculature and improves small molecule delivery. Proc Natl Acad Sci 112(6):1827–1832
Chen J, Ding J, Xiao C, Zhuang X, Chen X (2015) Emerging antitumor applications of extracellularly reengineered polymeric nanocarriers. Biomater Sci 3(7):988–1001
Gu X, Ding J, Zhang Z, Li Q, Zhuang X, Chen X (2015) Polymeric nanocarriers for drug delivery in osteosarcoma treatment. Curr Pharm Des 21(36):5187–5197
Pacardo DB, Ligler FS, Gu Z (2015) Programmable nanomedicine: synergistic and sequential drug delivery systems. Nanoscale 7(8):3381–3391
Ding J, Xiao C, Yan L, Tang Z, Zhuang X, Chen X, Jing X (2011) pH and dual redox responsive nanogel based on poly(L-glutamic acid) as potential intracellular drug carrier. J Control Release 152:E11–E13
Ding J, Chen J, Li D, Xiao C, Zhang J, He C, Zhuang X, Chen X (2013) Biocompatible reduction-responsive polypeptide micelles as nanocarriers for enhanced chemotherapy efficacy in vitro. J Mater Chem B 1(1):69–81
Xiao H, Qi R, Liu S, Hu X, Duan T, Zheng Y, Huang Y, Jing X (2011) Biodegradable polymer-cisplatin(IV) conjugate as a pro-drug of cisplatin(II). Biomaterials 32(30):7732–7739
Tiwari G, Tiwari R, Sriwastawa B, Bhati L, Pandey S, Pandey P, Bannerjee SK (2012) Drug delivery systems: an updated review. Int J Pharm Investig 2(1):2–11
Zhang Y, Chan HF, Leong KW (2013) Advanced materials and processing for drug delivery: the past and the future. Adv Drug Deliv Rev 65(1):104–120
Ikada Y, Tsuji H (2000) Biodegradable polyesters for medical and ecological applications. Macromol Rapid Commun 21(3):117–132
Couvreur P (2013) Nanoparticles in drug delivery: past, present and future. Adv Drug Deliv Rev 65(1):21–23
Doppalapudi S, Jain A, Domb AJ, Khan W (2016) Biodegradable polymers for targeted delivery of anti-cancer drugs. Expert Opin Drug Deliv 13(6):891–909
Liu M, Li H, Luo G, Liu Q, Wang Y (2008) Pharmacokinetics and biodistribution of surface modification polymeric nanoparticles. Arch Pharm Res 31(4):547–554
Feng C, Piao M, Li D (2016) Stereocomplex-reinforced PEGylated polylactide micelle for optimized drug delivery. Polymers 8(4):165
Ding GJ, Zhu YJ, Qi C, Lu BQ, Wu J, Chen F (2015) Porous microspheres of amorphous calcium phosphate: block copolymer templated microwave-assisted hydrothermal synthesis and application in drug delivery. J Colloid Interface Sci 443:72–79
Li J, Darabi M, Gu J, Shi J, Xue J, Huang L, Liu Y, Zhang L, Liu N, Zhong W, Zhang L, Xing M, Zhang L (2016) A drug delivery hydrogel system based on activin B for Parkinson’s disease. Biomaterials 102:72–86
Cheng M, Wang H, Zhang Z, Li N, Fang X, Xu S (2014) Gold nanorod-embedded electrospun fibrous membrane as a photothermal therapy platform. ACS Appl Mater Interfaces 6(3):1569–1575
Duan S, Yang X, Mei F, Tang Y, Li X, Shi Y, Mao J, Zhang H, Cai Q (2015) Enhanced osteogenic differentiation of mesenchymal stem cells on poly(L-lactide) nanofibrous scaffolds containing carbon nanomaterials. J Biomed Mater Res A 103(4):1424–1435
Pang XA, Zhuang XL, Tang ZH, Chen XS (2010) Polylactic acid (PLA): research, development and industrialization. Biotechnol J 5(11):1125–1136
Chang FY, Teng PT, Tsai TH (2013) Property measurement and processing parameter optimization for polylactide micro structure fabrication by thermal imprint. Jpn J Appl Phys 52(6S):06GJ09
Arrnentano I, Bitinis N, Fortunati E, Mattioli S, Rescignano N, Verdejo R, Lopez-Manchado MA, Kenny JM (2013) Multifunctional nanostructured PLA materials for packaging and tissue engineering. Prog Polym Sci 38(10–11):1720–1747
Lasprilla AJ, Martinez GA, Lunelli BH, Jardini AL, Filho RM (2012) Poly-lactic acid synthesis for application in biomedical devices – A review. Biotechnol Adv 30(1):321–328
Manome A, Okada S, Uchimura T, Komagata K (1998) The ratio of L-form to D-form of lactic acid as a criteria for the identification of lactic acid bacteria. J Gen Appl Microbiol 44(6):371–374
Wang J, Xu W, Ding J, Lu S, Wang X, Wang C, Chen X (2015) Cholesterol-enhanced polylactide-based stereocomplex micelle for effective delivery of doxorubicin. Materials 8(1):216–230
Wang J, Shen K, Xu W, Ding J, Wang X, Liu T, Wang C, Chen X (2015) Stereocomplex micelle from nonlinear enantiomeric copolymers efficiently transports antineoplastic drug. Nanoscale Res Lett 10(1):206
Ajiro H, Kuroda A, Kan K, Akashi M (2015) Stereocomplex film using triblock copolymers of polylactide and poly(ethylene glycol) retain paxlitaxel on substrates by an aqueous inkjet system. Langmuir 31(38):10583–10589
Chen C, Lv G, Pan C, Song M, Wu C, Guo D, Wang X, Chen B, Gu Z (2007) Poly(lactic acid) (PLA) based nanocomposites – A novel way of drug-releasing. Biomed Mater 2(4):L1–L4
Lasprilla AJR, Martinez GAR, Lunelli BH, Jardini AL, Maciel R (2010) Biomaterials for application in bone tissue engineering. J Biotechnol 150:S455–S455
Auras R, Harte B, Selke S (2004) An overview of polylactides as packaging materials. Macromol Biosci 4(9):835–864
Gupta AP, Kumar V (2007) New emerging trends in synthetic biodegradable polymers – polylactide: a critique. Eur Polym J 43(10):4053–4074
Townshend A (1985) Anal Chim Acta 177:304
Di Martino A, Sedlarik V (2014) Amphiphilic chitosan-grafted-functionalized polylactic acid based nanoparticles as a delivery system for doxorubicin and temozolomide co-therapy. Int J Pharm 474(1–2):134–145
Hou Z, Li L, Zhan C, Zhu P, Chang D, Jiang Q, Ye S, Yang X, Li Y, Xie L, Zhang Q (2012) Preparation and in vitro evaluation of an ultrasound-triggered drug delivery system: 10-Hydroxycamptothecin loaded PLA microbubbles. Ultrasonics 52(7):836–841
Li J, Sabliov C (2013) PLA/PLGA nanoparticles for delivery of drugs across the blood-brain barrier. Nanotechnol Rev 2(3):241–257
Sun D, Ding J, Xiao C, Chen J, Zhuang X, Chen X (2015) Drug delivery: pH-responsive reversible PEGylation improves performance of antineoplastic agent (Adv. Healthcare Mater. 6/2015). Adv Healthc Mater 4(6):786–786
Wu Y-L, Chen X, Wang W, Loh XJ (2016) Engineering bioresponsive hydrogels toward healthcare applications. Macromol Chem Phys 217(2):175–188
Zhang J, Wang X, Liu T, Liu S, Jing X (2016) Antitumor activity of electrospun polylactide nanofibers loaded with 5-fluorouracil and oxaliplatin against colorectal cancer. Drug Deliv 23(3):794–800
Li Z, Zhang FL, Pan LL, Zhu XL, Zhang ZZ (2015) Preparation and characterization of injectable mitoxantrone poly(lactic acid)/fullerene implants for in vivo chemo-photodynamic therapy. J Photochem Photobiol B Biol 149:51–57
Fernandez K, Aburto J, von Plessing C, Rockel M, Aspe E (2016) Factorial design optimization and characterization of poly-lactic acid (PLA) nanoparticle formation for the delivery of grape extracts. Food Chem 207:75–85
Feng X, Gao X, Kang T, Jiang D, Yao J, Jing Y, Song Q, Jiang X, Liang J, Chen J (2015) Mammary-derived growth inhibitor targeting peptide-modified PEG-PLA nanoparticles for enhanced targeted glioblastoma therapy. Bioconjug Chem 26(8):1850–1861
Dou S, Yang XZ, Xiong MH, Sun CY, Yao YD, Zhu YH, Wang J (2014) ScFv-decorated PEG-PLA-based nanoparticles for enhanced siRNA delivery to Her2(+) breast cancer. Adv Healthc Mater 3(11):1792–1803
Yao L, Song Q, Bai W, Zhang J, Miao D, Jiang M, Wang Y, Shen Z, Hu Q, Gu X, Huang M, Zheng G, Gao X, Hu B, Chen J, Chen H (2014) Facilitated brain delivery of poly(ethylene glycol)-poly(lactic acid) nanoparticles by microbubble-enhanced unfocused ultrasound. Biomaterials 35(10):3384–3395
Liu B, Han SM, Tang XY, Han L, Li CZ (2014) Cervical cancer gene therapy by gene loaded PEG-PLA nanomedicine. Asian Pac J Cancer Prev 15(12):4915–4918
Yao W, Xu P, Pang Z, Zhao J, Chai Z, Li X, Li H, Jiang M, Cheng H, Zhang B, Cheng N (2014) Local delivery of minocycline-loaded PEG-PLA nanoparticles for the enhanced treatment of periodontitis in dogs. Int J Nanomedicine 9:3963–3970
Amoozgar Z, Yeo Y (2012) Recent advances in stealth coating of nanoparticle drug delivery systems. Wiley Interdiscip Rev Nanomed Nanobiotechnol 4(2):219–233
Yeh P-YJ, Kainthan RK, Zou Y, Chiao M, Kizhakkedathu JN (2008) Self-assembled monothiol-terminated hyperbranched polyglycerols on a gold surface: a comparative study on the structure, morphology, and protein adsorption characteristics with linear poly(ethylene glycol)s. Langmuir 24(9):4907–4916
Deng Y, Saucier-Sawyer JK, Hoimes CJ, Zhang JW, Seo YE, Andrejecsk JW, Saltzman WM (2014) The effect of hyperbranched polyglycerol coatings on drug delivery using degradable polymer nanoparticles. Biomaterials 35(24):6595–6602
Yamakawa I, Tsushima Y, Machida R, Watanabe S (1992) Invitro and invivo release of poly(DL-lactic acid) microspheres containing neurotensin analog prepared by novel oil-in-water solvent evaporation method. J Pharm Sci 81(8):808–811
Jing Z, Xu XY, Chen XS, Liang QZ, Bian XC, Yang LX, Jing XB (2003) Biodegradable electrospun fibers for drug delivery. J Control Release 92(3):227–231
Maestrelli F, Bragagni M, Mura P (2016) Advanced formulations for improving therapies with anti-inflammatory or anaesthetic drugs: a review. J Drug Delivery Sci Technol 32:192–205
Zhu Z (2013) Effects of amphiphilic diblock copolymer on drug nanoparticle formation and stability. Biomaterials 34(38):10238–10248
Patel T, Zhou JB, Piepmeier JM, Saltzman WM (2012) Polymeric nanoparticles for drug delivery to the central nervous system. Adv Drug Deliv Rev 64(7):701–705
Ishihara T, Takahashi M, Higaki M, Mizushima Y (2009) Efficient encapsulation of a water-soluble corticosteroid in biodegradable nanoparticles. Int J Pharm 365(1–2):200–205
Choonara YE, Kumar P, Modi G, Pillay V (2016) Improving drug delivery technology for treating neurodegenerative diseases. Expert Opin Drug Deliv 13(7):1029–1043
Rao KS, Reddy MK, Horning JL, Labhasetwar V (2008) TAT-conjugated nanoparticles for the CNS delivery of anti-HIV drugs. Biomaterials 29(33):4429–4438
Sun WQ, Xie CS, Wang HF, Hu Y (2004) Specific role of polysorbate 80 coating on the targeting of nanoparticles to the brain. Biomaterials 25(15):3065–3071
Kulkarni SA, Feng SS (2011) Effects of surface modification on delivery efficiency of biodegradable nanoparticles across the blood-brain barrier. Nanomedicine 6(2):377–394
Lu W, Wan J, She ZJ, Jiang XG (2007) Brain delivery property and accelerated blood clearance of cationic albumin conjugated PEGylated nanoparticle. J Control Release 118(1):38–53
Gan CW, Feng SS (2010) Transferrin-conjugated nanoparticles of poly(lactide)-D-alpha-tocopheryl polyethylene glycol succinate diblock copolymer for targeted drug delivery across the blood-brain barrier. Biomaterials 31(30):7748–7757
Hashizume R, Gupta N (2010) Telomerase inhibitors for the treatment of brain tumors and the potential of intranasal delivery. Curr Opin Mol Ther 12(2):168–175
Jain DS, Bajaj AN, Athawale RB, Shikhande SS, Pandey A, Goel PN, Gude RP, Patil S, Raut P (2016) Thermosensitive PLA based nanodispersion for targeting brain tumor via intranasal route. Mater Sci Eng C 63:411–421
Xia HM, Gao XL, Gu GZ, Liu ZY, Zeng N, Hu QY, Song QX, Yao L, Pang ZQ, Jiang XG, Chen J, Chen HZ (2011) Low molecular weight protamine-functionalized nanoparticles for drug delivery to the brain after intranasal administration. Biomaterials 32(36):9888–9898
Li Y, Du Y, Liu X, Zhang Q, Jing L, Liang X, Chi C, Dai Z, Tian J (2015) Monitoring tumor targeting and treatment effects of IRDye 800CW and GX1-conjugated polylactic acid nanoparticles encapsulating endostar on glioma by optical molecular imaging. Mol Imaging 14:356–365
Miller T, Breyer S, van Colen G, Mier W, Haberkorn U, Geissler S, Voss S, Weigandt M, Goepferich A (2013) Premature drug release of polymeric micelles and its effects on tumor targeting. Int J Pharm 445(1–2):117–124
Mishra GP, Nguyen D, Alani AW (2013) Inhibitory effect of paclitaxel and rapamycin individual and dual drug-loaded polymeric micelles in the angiogenic cascade. Mol Pharm 10(5):2071–2078
Xu W, Ding J, Li L, Xiao C, Zhuang X, Chen X (2015) Acid-labile boronate-bridged dextran-bortezomib conjugate with up-regulated hypoxic tumor suppression. Chem Commun 51(31):6812–6815
Liu Y, Wang X, Sun CY, Wang J (2015) Delivery of mitogen-activated protein kinase inhibitor for hepatocellular carcinoma stem cell therapy. ACS Appl Mater Interfaces 7(1):1012–1020
Zhang X, Yang Y, Liang X, Zeng X, Liu Z, Tao W, Xiao X, Chen H, Huang L, Mei L (2014) Enhancing therapeutic effects of docetaxel-loaded dendritic copolymer nanoparticles by co-treatment with autophagy inhibitor on breast cancer. Theranostics 4(11):1085–1095
Yang A, Liu Z, Yan B, Zhou M, Xiong X (2016) Preparation of camptothecin-loaded targeting nanoparticles and their antitumor effects on hepatocellular carcinoma cell line H22. Drug Deliv 23(5):1699–1706
Liu Y, Zhu YH, Mao CQ, Dou S, Shen S, Tan ZB, Wang J (2014) Triple negative breast cancer therapy with CDK1 siRNA delivered by cationic lipid assisted PEG-PLA nanoparticles. J Control Release 192:114–121
Yang XZ, Dou S, Sun TM, Mao CQ, Wang HX, Wang J (2011) Systemic delivery of siRNA with cationic lipid assisted PEG-PLA nanoparticles for cancer therapy. J Control Release 156(2):203–211
Contreras J, Xie J, Chen YJ, Pei H, Zhang G, Fraser CL, Hamm-Alvarez SF (2010) Intracellular uptake and trafficking of difluoroboron dibenzoylmethane-polylactide nanoparticles in HeLa cells. ACS Nano 4(5):2735–2747
Laroui H, Viennois E, Xiao B, Canup BSB, Geem D, Denning TL, Merlin D (2014) Fab’-bearing siRNA TNF alpha-loaded nanoparticles targeted to colonic macrophages offer an effective therapy for experimental colitis. J Control Release 186:41–53
Kaffashi B, Davoodi S, Oliaei E (2016) Poly(epsilon-caprolactone)/triclosan loaded polylactic acid nanoparticles composite: a long-term antibacterial bionanocomposite with sustained release. Int J Pharm 508(1–2):10–21
Sun C, Wang X, Chen D, Lin X, Yu D, Wu H (2016) Dexamethasone loaded nanoparticles exert protective effects against cisplatin-induced hearing loss by systemic administration. Neurosci Lett 619:142–148
Pinon-Segundo E, Ganem-Quintanar A, Alonso-Perez V, Quintanar-Guerrero D (2005) Preparation and characterization of triclosan nanoparticles for periodontal treatment. Int J Pharm 294(1–2):217–232
Chen L, Liu L, Li C, Tan Y, Zhang G (2011) A new growth factor controlled drug release system to promote healing of bone fractures: nanospheres of recombinant human bone morphogenetic-2 and polylactic acid. J Nanosci Nanotechnol 11(4):3107–3114
Rancan F, Papakostas D, Hadam S, Hackbarth S, Delair T, Primard C, Verrier B, Sterry W, Blume-Peytavi U, Vogt A (2009) Investigation of polylactic acid (PLA) nanoparticles as drug delivery systems for local dermatotherapy. Pharm Res 26(8):2027–2036
Sakai T, Kohno H, Ishihara T, Higaki M, Saito S, Matsushima M, Mizushima Y, Kitahara K (2006) Treatment of experimental autoimmune uveoretinitis with poly(lactic acid) nanoparticles encapsulating betamethasone phosphate. Exp Eye Res 82(4):657–663
Molina J, Urbina J, Gref R, Brener Z, Rodrigues Junior JM (2001) Cure of experimental Chagas’ disease by the bis-triazole DO870 incorporated into ‘stealth’ polyethyleneglycol-polylactide nanospheres. J Antimicrob Chemother 47(1):101–104
Kohane DS (2007) Microparticles and nanoparticles for drug delivery. Biotechnol Bioeng 96(2):203–209
Freiberg S, Zhu XX (2004) Polymer microspheres for controlled drug release. Int J Pharm 282(1–2):1–18
Li S (1999) Hydrolytic degradation characteristics of aliphatic polyesters derived from lactic and glycolic acids. J Biomed Mater Res 48(3):342–353
Floyd JA, Galperin A, Ratner BD (2015) Drug encapsulated polymeric microspheres for intracranial tumor therapy: a review of the literature. Adv Drug Deliv Rev 91:23–37
Edlund U, Albertsson AC (2002) Degradable polymer microspheres for controlled drug delivery. In: Degradable aliphatic polyesters, Advances in polymer science, vol 157. Springer, Heidelberg, pp 67–112
Guan Q, Chen W, Hu X (2015) Development of lovastatin-loaded poly(lactic acid) microspheres for sustained oral delivery: in vitro and ex vivo evaluation. Drug Des Devel Ther 9:791–798
Lu J, Hou R, Yang Z, Tang Z (2015) Development and characterization of drug-loaded biodegradable PLA microcarriers prepared by the electrospraying technique. Int J Mol Med 36(1):249–254
Chen X, Yang Z, Sun R, Mo Z, Jin G, Wei F, Hu J, Guan W, Zhong N (2014) Preparation of lung-targeting, emodin-loaded polylactic acid microspheres and their properties. Int J Mol Sci 15(4):6241–6251
Falconi M, Focaroli S, Teti G, Salvatore V, Durante S, Nicolini B, Orienti I (2014) Novel PLA microspheres with hydrophilic and bioadhesive surfaces for the controlled delivery of fenretinide. J Microencapsul 31(1):41–48
Pinto E, Zhang B, Song S, Bodor N, Buchwald P, Hochhaus G (2010) Feasibility of localized immunosuppression: 2. PLA microspheres for the sustained local delivery of a soft immunosuppressant. Pharmazie 65(6):429–435
Umeki N, Sato T, Harada M, Takeda J, Saito S, Iwao Y, Itai S (2010) Preparation and evaluation of biodegradable microspheres containing a new potent osteogenic compound and new synthetic polymers for sustained release. Int J Pharm 392(1–2):42–50
Rafat M, Cleroux CA, Fong WG, Baker AN, Leonard BC, O’Connor MD, Tsilfidis C (2010) PEG-PLA microparticles for encapsulation and delivery of Tat-EGFP to retinal cells. Biomaterials 31(12):3414–3421
Sheshala R, Peh KK, Darwis Y (2009) Preparation, characterization, and in vivo evaluation of insulin-loaded PLA-PEG microspheres for controlled parenteral drug delivery. Drug Dev Ind Pharm 35(11):1364–1374
Ding LY, Xia PF, Yang CQ, Lin YL, Wang J (2007) Preparation and evaluation of sustained-release microsphere of Sanguis Draconis in vitro. Zhongguo Zhong Yao Za Zhi 32(5):388–390
Ren J, Yu X, Ren T, Hong H (2007) Preparation and characterization of fenofibrate-loaded PLA-PEG microspheres. J Mater Sci Mater Med 18(8):1481–1487
Matsumoto A, Matsukawa Y, Suzuki T, Yoshino H (2005) Drug release characteristics of multi-reservoir type microspheres with poly(DL-lactide-co-glycolide) and poly(DL-lactide). J Control Release 106(1–2):172–180
Chen A, Dang T, Wang S, Tang N, Liu Y, Wu W (2014) The in vitro and in vivo anti-tumor effects of MTX-Fe3O4-PLLA-PEG-PLLA microspheres prepared by suspension-enhanced dispersion by supercritical CO2. Sci China Life Sci 57(7):698–709
Zhao H, Wu F, Cai Y, Chen Y, Wei L, Liu Z, Yuan W (2013) Local antitumor effects of intratumoral delivery of rlL-2 loaded sustained-release dextran/PLGA-PLA core/shell microspheres. Int J Pharm 450(1–2):235–240
Zhou JY, Wang XM, Zhang QQ, Ye SF (2009) Efficacy of intraperitoneally injected epirubicin-loaded poly (d, l)-lactic acid microspheres alone or combined with free epirubicin in treating hepatocellular carcinoma in mice. Zhongguo Yi Xue Ke Xue Yuan Xue Bao 31(5):603–606
Ciftci K, Hincal AA, Kas HS, Ercan TM, Sungur A, Guven O, Ruacan S (1997) Solid tumor chemotherapy and in vivo distribution of fluorouracil following administration in poly(L-lactic acid) microspheres. Pharm Dev Technol 2(2):151–160
Kuang LR, Yang DJ, Inoue T, Liu WC, Wallace S, Wright KC (1996) Percutaneous intratumoral injection of cisplatin microspheres in tumor-bearing rats to diminish acute nephrotoxicity. Anti-Cancer Drugs 7(2):220–227
Xia D, Yao H, Liu Q, Xu L (2012) Preparation of microspheres encapsulating a recombinant TIMP-1 adenovirus and their inhibition of proliferation of hepatocellular carcinoma cells. Asian Pac J Cancer Prev 13(12):6363–6368
Lu J, Jackson JK, Gleave ME, Burt HM (2008) The preparation and characterization of anti-VEGFR2 conjugated, paclitaxel-loaded PLLA or PLGA microspheres for the systemic targeting of human prostate tumors. Cancer Chemother Pharmacol 61(6):997–1005
Lu Y, Lin P, Lu B, Wang J, Zhang J, Huang X (2000) Studies on release characteristics and cytotoxicity of 5-fluorouracil loaded polylactide microspheres on lung cancer cell lines. Zhongguo Fei Ai Za Zhi 3(6):432–434
Burt HM, Jackson JK, Bains SK, Liggins RT, Oktaba AM, Arsenault AL, Hunter WL (1995) Controlled delivery of taxol from microspheres composed of a blend of ethylene-vinyl acetate copolymer and poly (D,L-lactic acid). Cancer Lett 88(1):73–79
Chandy T, Das GS, Rao GHR (2000) 5-Fluorouracil-loaded chitosan coated polylactic acid microspheres as biodegradable drug carriers for cerebral tumours. J Microencapsul 17(5):625–638
Macha IJ, Cazalbou S, Ben-Nissan B, Harvey KL, Milthorpe B (2015) Marine structure derived calcium phosphate-polymer biocomposites for local antibiotic delivery. Mar Drugs 13(1):666–680
Jain JP, Kumar N (2010) Development of amphotericin B loaded polymersomes based on (PEG)(3)-PLA co-polymers: factors affecting size and in vitro evaluation. Eur J Pharm Sci 40(5):456–465
Huang YY, Chung TW (2001) Microencapsulation of gentamicin in biodegradable PLA and/or PLA/PEG copolymer. J Microencapsul 18(4):457–465
Sharma R, Muttil P, Yadav AB, Rath SK, Bajpai VK, Mani U, Misra A (2007) Uptake of inhalable microparticles affects defence responses of macrophages infected with mycobacterium tuberculosis H37Ra. J Antimicrob Chemother 59(3):499–506
Selek H, Sahin S, Ercan MT, Sargon M, Hincal AA, Kas HS (2003) Formulation and in vitro/in vivo evaluation of terbutaline sulphate incorporated in PLGA (25/75) and L-PLA microspheres. J Microencapsul 20(2):261–271
Guiziou B, Armstrong DJ, Elliott PNC, Ford JL, Rostron C (1996) Investigation of in-vitro release characteristics of NSAID-loaded polylactic acid microspheres. J Microencapsul 13(6):701–708
Zha J, Chi XW, Yu XL, Liu XM, Liu DQ, Zhu J, Ji H, Liu RT (2016) Interleukin-1 beta-targeted vaccine improves glucose control and β-cell function in a diabetic KK-A(y) mouse model. PLoS One 11(5):16
Anugraha G, Madhumathi J, Prita PJJ, Kaliraj P (2015) Biodegradable poly-L-lactide based microparticles as controlled release delivery system for filarial vaccine candidate antigens. Eur J Pharmacol 747:174–180
Pavot V, Berthet M, Resseguier J, Legaz S, Handke N, Gilbert SC, Paul S, Verrier B (2014) Poly(lactic acid) and poly(lactic-co-glycolic acid) particles as versatile carrier platforms for vaccine delivery. Nanomedicine 9(17):2703–2718
Qiu SH, Wei Q, Liang ZL, Ma GH, Wang LY, An WQ, Ma XW, Fang X, He P, Li HM, Hu ZY (2014) Biodegradable polylactide microspheres enhance specific immune response induced by hepatitis B surface antigen. Hum Vaccin Immunother 10(8):2350–2356
Pandit S, Cevher E, Zariwala MG, Somavarapu S, Alpar HO (2007) Enhancement of immune response of HBsAg loaded poly(L-lactic acid) microspheres against hepatitis B through incorporation of alum and chitosan. J Microencapsul 24(6):539–552
Zhou SB, Liao XY, Li XH, Deng XM, Li H (2003) Poly-D,L-lactide-co-poly(ethylene glycol) microspheres as potential vaccine delivery systems. J Control Release 86(2–3):195–205
Wang K, Li WF, Xing JF, Dong K, Gao Y (2012) Preliminary assessment of the safety evaluation of novel pH-sensitive hydrogel. Eur J Pharm Biopharm 82(2):332–339
Markland P, Zhang Y, Amidon GL, Yang VC (1999) A pH- and ionic strength-responsive polypeptide hydrogel: synthesis, characterization, and preliminary protein release studies. J Biomed Mater Res 47(4):595–602
Gong C, Shi S, Dong P, Kan B, Gou M, Wang X, Li X, Luo F, Zhao X, Wei Y, Qian Z (2009) Synthesis and characterization of PEG-PCL-PEG thermosensitive hydrogel. Int J Pharm 365(1–2):89–99
Fu SZ, Li Z, Fan JM, Meng XH, Shi K, Qu Y, Yang LL, Wu JB, Fan J, Luot F, Qian ZY (2014) Biodegradable and thermosensitive monomethoxy poly(ethylene glycol)-poly(lactic acid) hydrogel as a barrier for prevention of post-operative abdominal adhesion. J Biomed Nanotechnol 10(3):427–435
Fan R, Deng X, Zhou L, Gao X, Fan M, Wang Y, Guo G (2014) Injectable thermosensitive hydrogel composite with surface-functionalized calcium phosphate as raw materials. Int J Nanomedicine 9:615–626
Basu A, Kunduru KR, Doppalapudi S, Domb AJ, Khan W (2016) Poly(lactic acid) based hydrogels. Adv Drug Deliv Rev 107:192–205
Lai PL, Hong DW, Ku KL, Lai ZT, Chu IM (2014) Novel thermosensitive hydrogels based on methoxy polyethylene glycol-co-poly(lactic acid-co-aromatic anhydride) for cefazolin delivery. Nanomedicine 10(3):553–560
Molina I, Li S, Martinez MB, Vert M (2001) Protein release from physically crosslinked hydrogels of the PLA/PEO/PLA triblock copolymer-type. Biomaterials 22(4):363–369
He X, Ma J, Jabbari E (2010) Migration of marrow stromal cells in response to sustained release of stromal-derived factor-1alpha from poly(lactide ethylene oxide fumarate) hydrogels. Int J Pharm 390(2):107–116
Shen W, Luan J, Cao L, Sun J, Yu L, Ding J (2015) Thermogelling polymer-platinum(IV) conjugates for long-term delivery of cisplatin. Biomacromolecules 16(1):105–115
Fan R, Tong A, Li X, Gao X, Mei L, Zhou L, Zhang X, You C, Guo G (2015) Enhanced antitumor effects by docetaxel/LL37-loaded thermosensitive hydrogel nanoparticles in peritoneal carcinomatosis of colorectal cancer. Int J Nanomedicine 10:7291–7305
Manaka T, Suzuki A, Takayama K, Imai Y, Nakamura H, Takaoka K (2011) Local delivery of siRNA using a biodegradable polymer application to enhance BMP-induced bone formation. Biomaterials 32(36):9642–9648
Nelson DM, Hashizume R, Yoshizumi T, Blakney AK, Ma Z, Wagner WR (2014) Intramyocardial injection of a synthetic hydrogel with delivery of bFGF and IGF1 in a rat model of ischemic cardiomyopathy. Biomacromolecules 15(1):1–11
Song Z, Shi B, Ding J, Zhuang X, Zhang X, Fu C, Chen X (2015) A comparative study of preventing postoperative tendon adhesion using electrospun polyester membranes with different degradation kinetics. Sci China Chem 58(7):1159–1168
Zhang ZZ, Jiang D, Wang SJ, Qi YS, Ding JX, Yu JK, Chen XS (2015) Scaffolds drive meniscus tissue engineering. RSC Adv 5(95):77851–77859
Wang X, Shan H, Wang J, Hou Y, Ding J, Chen Q, Guan J, Wang C, Chen X (2015) Characterization of nanostructured ureteral stent with gradient degradation in a porcine model. Int J Nanomedicine 10:3055–3064
Zhang J, Liu H, Ding JX, Zhuang XL, Chen XS, Li ZM (2015) Annealing regulates the performance of an electrospun poly(ε-caprolactone) membrane to accommodate tissue engineering. RSC Adv 5(41):32604–32608
Zhang J, Liu H, Xu H, Ding JX, Zhuang XL, Chen XS, Chang F, Xu JZ, Li ZM (2014) Molecular weight-modulated electrospun poly(ε-caprolactone) membranes for postoperative adhesion prevention. RSC Adv 4(79):41696–41704
Shi B, Ding J, Wei J, Fu C, Zhuang X, Chen X (2015) Drug-incorporated electrospun fibers efficiently prevent postoperative adhesion. Curr Pharm Des 21(15):1960–1966
Jin HJ, Fridrikh SV, Rutledge GC, Kaplan DL (2002) Electrospinning Bombyx mori silk with poly(ethylene oxide). Biomacromolecules 3(6):1233–1239
Chen M, Li YF, Besenbacher F (2014) Electrospun nanofibers-mediated on-demand drug release. Adv Healthc Mater 3(11):1721–1732
Zhang J, Wang X, Liu T, Liu S, Jing X (2016) Antitumor activity of electrospun polylactide nanofibers loaded with 5-fluorouracil and oxaliplatin against colorectal cancer. Drug Deliv 23(3):784–790
Zhang Z, Liu S, Xiong H, Jing X, Xie Z, Chen X, Huang Y (2015) Electrospun PLA/MWCNTs composite nanofibers for combined chemo- and photothermal therapy. Acta Biomater 26:115–123
Zong X, Li S, Chen E, Garlick B, Kim KS, Fang D, Chiu J, Zimmerman T, Brathwaite C, Hsiao BS, Chu B (2004) Prevention of postsurgery-induced abdominal adhesions by electrospun bioabsorbable nanofibrous poly(lactide-co-glycolide)-based membranes. Ann Surg 240(5):910–915
Chen CH, Chen SH, Shalumon KT, Chen JP (2015) Dual functional core-sheath electrospun hyaluronic acid/polycaprolactone nanofibrous membranes embedded with silver nanoparticles for prevention of peritendinous adhesion. Acta Biomater 26:225–235
Liu S, Zhao J, Ruan H, Wang W, Wu T, Cui W, Fan C (2013) Antibacterial and anti-adhesion effects of the silver nanoparticles-loaded poly(L-lactide) fibrous membrane. Mater Sci Eng C 33(3):1176–1182
Hu C, Liu S, Zhang Y, Li B, Yang H, Fan C, Cui W (2013) Long-term drug release from electrospun fibers for in vivo inflammation prevention in the prevention of peritendinous adhesions. Acta Biomater 9(7):7381–7388
Jiang S, Zhao X, Chen S, Pan G, Song J, He N, Li F, Cui W, Fan C (2014) Down-regulating ERK1/2 and SMAD2/3 phosphorylation by physical barrier of celecoxib-loaded electrospun fibrous membranes prevents tendon adhesions. Biomaterials 35(37):9920–9929
Liu S, Hu C, Li F, Li XJ, Cui W, Fan C (2013) Prevention of peritendinous adhesions with electrospun ibuprofen-loaded poly(L-lactic acid)-polyethylene glycol fibrous membranes. Tissue Eng Part A 19(3–4):529–537
Liu S, Qin M, Hu C, Wu F, Cui W, Jin T, Fan C (2013) Tendon healing and anti-adhesion properties of electrospun fibrous membranes containing bFGF loaded nanoparticles. Biomaterials 34(19):4690–4701
Wang H, Li M, Hu J, Wang C, Xu S, Han CC (2013) Multiple targeted drugs carrying biodegradable membrane barrier: anti-adhesion, hemostasis, and anti-infection. Biomacromolecules 14(4):954–961
Sreerekha PR, Menon D, Nair SV, Chennazhi KP (2013) Fabrication of electrospun poly(lactide-co-glycolide)-fibrin multiscale scaffold for myocardial regeneration in vitro. Tissue Eng Part A 19(7–8):849–859
Jia L, Prabhakaran MP, Qin X, Ramakrishna S (2013) Stem cell differentiation on electrospun nanofibrous substrates for vascular tissue engineering. Mater Sci Eng C Mater Biol Appl 33(8):4640–4650
Vadala G, Mozetic P, Rainer A, Centola M, Loppini M, Trombetta M, Denaro V (2012) Bioactive electrospun scaffold for annulus fibrosus repair and regeneration. Eur Spine J 21(Suppl 1):S20–S26
Ahire JJ, Neppalli R, Heunis TD, van Reenen AJ, Dicks LM (2014) 2,3-Dihydroxybenzoic acid electrospun into poly(D,L-lactide) (PDLLA)/poly(ethylene oxide) (PEO) nanofibers inhibited the growth of gram-positive and gram-negative bacteria. Curr Microbiol 69(5):587–593
Llorens E, Calderon S, del Valle LJ, Puiggali J (2015) Polybiguanide (PHMB) loaded in PLA scaffolds displaying high hydrophobic, biocompatibility and antibacterial properties. Mater Sci Eng C Mater Biol Appl 50:74–84
Spasova M, Manolova N, Paneva D, Mincheva R, Dubois P, Rashkov I, Maximova V, Danchev D (2010) Polylactide stereocomplex-based electrospun materials possessing surface with antibacterial and hemostatic properties. Biomacromolecules 11(1):151–159
Ni P, Fu S, Fan M, Guo G, Shi S, Peng J, Luo F, Qian Z (2011) Preparation of poly(ethylene glycol)/polylactide hybrid fibrous scaffolds for bone tissue engineering. Int J Nanomedicine 6:3065–3075
Seyedjafari E, Soleimani M, Ghaemi N, Shabani I (2010) Nanohydroxyapatite-coated electrospun poly(L-lactide) nanofibers enhance osteogenic differentiation of stem cells and induce ectopic bone formation. Biomacromolecules 11(11):3118–3125
Kobsa S, Kristofik NJ, Sawyer AJ, Bothwell AL, Kyriakides TR, Saltzman WM (2013) An electrospun scaffold integrating nucleic acid delivery for treatment of full-thickness wounds. Biomaterials 34(15):3891–3901
Canton I, McKean R, Charnley M, Blackwood KA, Fiorica C, Ryan AJ, MacNeil S (2010) Development of an ibuprofen-releasing biodegradable PLA/PGA electrospun scaffold for tissue regeneration. Biotechnol Bioeng 105(2):396–408
Santoro M, Shah SR, Walker JL, Mikos AG (2016) Poly(lactic acid) nanofibrous scaffolds for tissue engineering. Adv Drug Deliv Rev 107:206–212
Liu DH, Ding JX, Xu WG, Song XF, Zhuang XL, Chen XS (2014) Stereocomplex micelles based on 4-armed poly ( ethylene glycol )-polylactide enantiomeric copolymers for drug delivery. Acta Polym Sin 9:1265–1273
Saffer EM, Tew GN, Bhatia SR (2011) Poly(lactic acid)-poly(ethylene oxide) block copolymers: new directions in self-assembly and biomedical applications. Curr Med Chem 18(36):5676–5686
Zhou R, Xu W, Chen F, Qi C, Lu BQ, Zhang H, Wu J, Qian QR, Zhu YJ (2014) Amorphous calcium phosphate nanospheres/polylactide composite coated tantalum scaffold: facile preparation, fast biomineralization and subchondral bone defect repair application. Colloids Surf B Biointerfaces 123:236–245
Pelto J, Bjorninen M, Palli A, Talvitie E, Hyttinen J, Mannerstrom B, Suuronen Seppanen R, Kellomaki M, Miettinen S, Haimi S (2013) Novel polypyrrole-coated polylactide scaffolds enhance adipose stem cell proliferation and early osteogenic differentiation. Tissue Eng Part A 19(7–8):882–892
Niu X, Fan Y, Liu X, Li X, Li P, Wang J, Sha Z, Feng Q (2011) Repair of bone defect in femoral condyle using microencapsulated chitosan, nanohydroxyapatite/collagen and poly(L-lactide)-based microsphere-scaffold delivery system. Artif Organs 35(7):E119–E128
Hu J, Sun X, Ma H, Xie C, Chen YE, Ma PX (2010) Porous nanofibrous PLLA scaffolds for vascular tissue engineering. Biomaterials 31(31):7971–7977
Tsuji T, Tamai H, Igaki K, Kyo E, Kosuga K, Hata T, Nakamura T, Fujita S, Takeda S, Motohara S, Uehata H (2003) Biodegradable stents as a platform to drug loading. Int J Cardiovasc Interv 5(1):13–16
Haddad T, Noel S, Liberelle B, El Ayoubi R, Ajji A, De Crescenzo G (2016) Fabrication and surface modification of poly lactic acid (PLA) scaffolds with epidermal growth factor for neural tissue engineering. Biomatter 6(1):e1231276
Zhao J, Han W, Tu M, Huan S, Zeng R, Wu H, Cha Z, Zhou C (2012) Preparation and properties of biomimetic porous nanofibrous poly(L-lactide) scaffold with chitosan nanofiber network by a dual thermally induced phase separation technique. Mater Sci Eng C 32(6):1496–1502
Lou T, Wang X, Song G, Gu Z, Yang Z (2014) Fabrication of PLLA/β-TCP nanocomposite scaffolds with hierarchical porosity for bone tissue engineering. Int J Biol Macromol 69:464–470
Binan L, Tendey C, De Crescenzo G, El Ayoubi R, Ajji A, Jolicoeur M (2014) Differentiation of neuronal stem cells into motor neurons using electrospun poly-L-lactic acid/gelatin scaffold. Biomaterials 35(2):664–674
Kontogiannopoulos KN, Assimopoulou AN, Tsivintzelis I, Panayiotou C, Papageorgiou VP (2011) Electrospun fiber mats containing shikonin and derivatives with potential biomedical applications. Int J Pharm 409(1–2):216–228
Nguyen TTT, Ghosh C, Hwang SG, Tran LD, Park JS (2013) Characteristics of curcumin-loaded poly(lactic acid) nanofibers for wound healing. J Mater Sci 48(20):7125–7133
Leroueil-Le Verger M, Fluckiger L, Kim YI, Hoffman M, Maincent P (1998) Preparation and characterization of nanoparticles containing an antihypertensive agent. Eur J Pharm Biopharm 46(2):137–143
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Li, J., Ding, J., Liu, T., Liu, J.F., Yan, L., Chen, X. (2017). Poly(lactic acid) Controlled Drug Delivery. In: Di Lorenzo, M., Androsch, R. (eds) Industrial Applications of Poly(lactic acid). Advances in Polymer Science, vol 282. Springer, Cham. https://doi.org/10.1007/12_2017_11
Download citation
DOI: https://doi.org/10.1007/12_2017_11
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-75458-1
Online ISBN: 978-3-319-75459-8
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)