Abstract
Background and Objectives: In a previous article, we showed that the pharmacokinetic set of remifentanil used for target-controlled infusion (TCI) might be biased in obese patients because it incorporates flawed equations for the calculation of lean body mass (LBM), which is a covariate of several pharmacokinetic parameters in this set. The objectives of this study were to determine the predictive performance of the original pharmacokinetic set, which incorporates the James equation for LBM calculation, and to determine the predictive performance of the pharmacokinetic set when a new method to calculate LBM was used (the Janmahasatian equations).
Methods: This was an observational study with intraoperative observations and no follow-up. Fifteen morbidly obese inpatients scheduled for bariatric surgery were included in the study. The intervention included manually controlled continuous infusion of remifentanil during the surgery and analysis of arterial blood samples to determine the arterial remifentanil concentration, to be compared with concentrations predicted by either the unadjusted or the adjusted pharmacokinetic set. The statistical analysis included parametric and non-parametric tests on continuous variables and determination of the median performance error (MDPE), median absolute performance error (MDAPE), divergence and wobble.
Results: The median values (interquartile ranges) of the MDPE, MDAPE, divergence and wobble for the James equations during maintenance were −53.4% (−58.7% to −49.2%), 53.4% (49.0–58.7%), 3.3% (2.9–4.7%) and 1.4% h−1 (1.1–2.5% h−1), respectively. The respective values for the Janmahasatian equations were −18.9% (−24.2% to −10.4%), 20.5% (13.3–24.8%), 2.6% (−0.7% to 4.5%) and 1.9% h−1 (1.4–3.0% h−1). The performance (in terms of the MDPE and MDAPE) of the corrected pharmacokinetic set was better than that of the uncorrected one.
Conclusion: The predictive performance of the original pharmacokinetic set is not clinically acceptable. Use of a corrected LBM value in morbidly obese patients corrects this pharmacokinetic set and allows its use in obese patients. The ‘fictitious height’ can be a valid alternative for use of TCI infusion of remifentanil in morbidly obese patients until commercially available infusion pumps and research software are updated and new LBM equations are implemented in their algorithms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Obesity is defined as an excess of fat tissue when compared with normal values for an individual of the same age and sex. The WHO classification of obesity is based upon the body mass index (BMI), calculated as total bodyweight (TBW) in kilograms divided by the square of the height in metres. Obesity is defined as a BMI ≥30 kg/m2, and morbid (or class III) obesity is defined as a BMI ≥40 kg/m2.[1] Obesity affects almost every body system, resulting in important changes in drug disposition,[2–5] and potentially has significant effects on use of drugs in anaesthesia.
Remifentanil is a synthetic μ opioid receptor agonist and an opioid analgesic with unique pharmacokinetics, which make it ideally suited for target-controlled infusion (TCI). The pharmacokinetic parameters incorporated into the commercially available TCI pumps and used for almost all research studies in anesthesia are those derived from a study published by Minto et al.[6] (table I). It is evident that lean body mass (LBM) is a covariate for the central volume of distribution (V1), rapid peripheral volume of distribution (V2) and metabolic clearance (CLm), and its value is critical for the calculation of these parameters.
Minto pharmacokinetic parameters estimated using nonlinear mixed-effects modelling (NONMEM)[6]
LBM, according to the James equations,[7] is calculated from TBW in kilograms and height (Ht) in centimetres, as follows (equation 1):
Unfortunately, the James equations suffer from an ‘odd’ property because they are inverted parabolas, which means that, beyond a certain bodyweight (which can be called the ‘critical weight’ [CW])[8] LBM values start to decrease, leading to underdosing in obese patients. Using dual-energy x-ray absorptiometry, Janmahasatian et al.[9] have recently derived equations yielding the adjusted fat-free mass (FFM; almost equal to LBM) for a wide range of bodyweights (40.7–216.5 kg) and BMIs (17.1–69.9 kg/m2) [equation 2]:
It should be noted that 71% of patients enrolled in this study were overweight or obese. The difference between the two ways to calculate LBM (in females) is depicted in figure 1. In theory, the prediction of a pharmacokinetic set is most accurate when it is used in a population similar to the one in which it was originally developed (e.g. non-obese patients), and using it in another set of population is basically wrong. Nevertheless, it is still possible to examine how the original pharmacokinetic set fits obese patients. Table II presents the effect of using the original pharmacokinetic set in a morbidly obese patient compared with a non-obese subject in terms of pharmacokinetic parameters and the total dose infused. The other two columns represent the same situation if a new method for calculating LBM is used by changing the patient’s true height into their ‘fictitious height’ (FH).[8] It is evident that the use of a different LBM value has at least two significant implications: (i) creation of a vastly different pharmacokinetic parameter set for the same patient; and (ii) different predictions of plasma concentration in the same patient.
Nomogram relating lean body mass (LBM), total bodyweight (TBW) and height in females (height range: 150–180 cm), adapted from the James[7] and Janmahasatian[9] equations. The circles show the critical weight (CW) and the divergence weight (DW; i.e. the weight beyond which LBM values calculated with these two methods start diverging) for each height.
Illustration of the different pharmacokinetic parameters calculated according to either the James or the Janmahasatian lean body mass (LBM) equation in two different female persons (each 1.7 m tall), one weighing 205 kg (body mass index [BMI] 70.9 kg/m2) and the other weighing 55 kg (BMI 19 kg/m2) after a 120-minute, target-controlled infusion of remifentanil 2 ng/mL
In Italy (similarly to other countries), there are three commercially available pumps that incorporate the Minto pharmacokinetic set: the Alaris® (previously Asena) Infusion System (CareFusion Corporation, San Diego, CA, USA), the Orchestra® Base Primea (Fresenius Kabi AG, Bad Homburg, Germany) and the new Injectomat® TIVA Agilia (also from Fresenius Kabi). Until 2007, all of them presented the aforementioned problem. However, in late 2007, following our first report,[10] these brands decided to update their software. While Fresenius Kabi opted for the introduction of an upper BMI value of 42 kg/m2 for males and 35 kg/m2 for females, Alaris introduced a maximum weight, dependent on the patient’s height and sex (this weight is almost equal to our CW[8]). In both cases, TCI is not available if the patient’s BMI or bodyweight is greater than the values set by the updated algorithms, thus it is now impossible to administer TCI of remifentanil to morbidly obese patients.
The aims of this prospective study were to determine the predictive performance of the original pharmacokinetic set of remifentanil (Minto) in obese patients, and to determine the predictive performance of the pharmacokinetic set of remifentanil when a new method to calculate LBM is used (through the introduction of the FH, in our case).[8]
Patients and Methods
After ethics committee approval was granted for this prospective study, written informed consent was obtained from 15 consecutive, obese, female patients (American Society of Anesthesiologists [ASA] physical status II–III, aged 20–64 years) undergoing elective Roux-en-Y bypass surgery. Patients with an ASA physical status >III, aged <18 or ≥65 years, or with a history of alcohol (ethanol) or drug abuse, were excluded a priori.
Patients fasted for 8 hours before surgery and received no premedication. After the patient arrived in the operating room, two 18-gauge intravenous cannulas were placed in the forearm and Ringer’s lactate solution (6 mL/kg) was infused. A radial artery catheter was inserted for arterial blood sampling and invasive arterial blood pressure monitoring. Standard monitoring was used throughout the study, including ECG, heart rate (lead II) and pulse oximetry. In all patients, the EEG was monitored using the bispectral index (BIS XP A-2000 monitor; Aspect Medical Systems Inc., Natick, MA, USA).
General anaesthesia was induced with fentanyl 1.5 µg/kg and propofol 1–2 mg/kg. If ventilation was possible, suxamethonium chloride 1 mg/kg was administered and the patients’ tracheas were intubated.
The patients’ lungs were mechanically ventilated using a 50% oxygen in air mixture and controlled using a Cato-Dräger anaesthesia workstation (Dräger AG, Lubeck, Germany) set to maintain an end-tidal partial pressure of carbon dioxide ranging between 32 and 35 mmHg. Cisatracurium besilate was used for neuromuscular blockade.
Anaesthesia was maintained with a continuous infusion (via a syringe pump) of propofol (no specific protocol; infusion speed at the discretion of the anaesthesiologist), administered according to the needs of each patient and targeted to maintain a stable bispectral index value ranging between 40 and 50.
Remifentanil was used to provide intraoperative analgesia and administered with a continuous infusion (via a syringe pump) without any strict protocol, with the only aim being to maintain haemodynamic stability (defined as heart rate and mean blood pressure values within ±10% of the basal values, i.e. the values recorded at the patient’s arrival in the operating theatre). The remifentanil infusion was started immediately after tracheal intubation, and no boluses or loading doses were administered.
The time course and speed of the remifentanil infusion were manually recorded, stored on a computer and used for subsequent analyses and simulations.
Arterial blood samples for determination of plasma remifentanil concentrations were collected before the start of the infusion and then every 10 minutes thereafter until the infusion was discontinued (usually at the time of the last skin stitch).
Arterial blood samples for determination of plasma remifentanil concentrations were collected in syringes and immediately transferred into test tubes containing citric acid and sodium fluoride, which were then immediately stored at −30°C. The plasma remifentanil concentration was determined by high-performance liquid chromatography with mass spectromety (HPLC-MS).[11] The mean coefficient of variation of the HPLC-MS method was 6.58%, and the lowest limit of detection was 0.09 ng/mL.
Based on the patients’ anthropometric characteristics and the recorded infusion speeds, it was possible to calculate the time course of the predicted plasma concentration (Cpred) values of remifentanil by means of commercially available research software (TIVAtrainer© version 5.1[12] and Rugloop© version 3.28[13]).
We performed two different simulations for each patient, one using the original parameter set (the James equation predictions, referred to here as ‘James’ for simplicity), which incorporates the James equations for LBM calculation, and the other using the same parameter set, incorporating the Janmahasatian equations for LBM calculation (the Janmahasatian equation predictions, referred to here as ‘Janmahasatian’). This has been possible (and will be possible for every anaesthesiologist until corrected pumps become available) by simply changing the real height of patients into their FH.[8] FH equations[8] are the result of a mathematical process which, if used to calculate the input height, can force the algorithm to use ‘true’ values of LBM[9] to calculate pharmacokinetic parameter values (table II). Using FH as the input height has the same effect on the pharmacokinetic set as if we had changed only LBM equations in the original set.
Statistical Analysis
The predictive performance of the pharmacokinetic set of remifentanil was evaluated by calculating the performance errors (PEs) for each blood sample, according to the methods described by Varvel et al.[14] Intrasubject bias (i.e. the direction and size of the deviation from the predicted concentration) and inaccuracy (i.e. the size of the typical miss) were assessed by determination of the median PE (MDPE) and the median absolute PE (MDAPE). Divergence, a measure of the expected systematic time-related changes in performance (that is, the tendency towards narrowing or widening over time of the gap between the observed and predicted concentrations in a given subject) was calculated as the slope obtained from linear regression of that individual’s ∣PE∣s against time. Wobble, a measure of the total intraindividual variability in PEs, which is directly related to the ability to achieve stable drug concentrations, was calculated as the median value of the absolute differences between the individual PEs at each sampling time and the MDPE for that patient.
Statistical analysis was performed using Systat® software (Systat Software Inc., San Jose, CA, USA) and Microsoft® Excel (Microsoft Corporation, Redmond, WA, USA). The aim of this analysis was to evaluate the clinical suitability of the unadjusted and adjusted tests according to Glass et al.[15] and Schuttler et al.[16]
Based on our previous (published and unpublished) data and simulations, and considering a 50% reduction in the calculated MDPE value of 50% ± 20% to be significant, a sample size of 12 patients per group was adequate to detect a difference with an α error of 0.05 and a power of 0.8.
The normality of the distribution was assessed by means of the Shapiro-Wilk test. The Student’s t-test for unpaired groups or the Mann-Whitney-Wilcoxon rank-sum test was also used to test the differences between groups. The significance of the difference between the slopes of the linear regression of predicted versus measured plasma remifentanil concentrations was tested using the t-test (starting from the slope value, the size of the sample and the standard error of the regression).
A p-value of <0.05 was considered significant. The results are presented as median [interquartile range].
Results
Fifteen consecutive, obese, female patients were enrolled in this study. The patients’ demographics and significant Minto pharmacokinetic parameters are shown in tables III and IV. A total of 155 arterial blood samples were drawn and analysed.
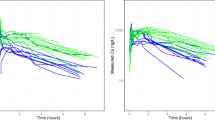
The plasma remifentanil concentrations ranged from 0.3 to 16.1 ng/mL. On visual inspection, the observed plasma concentration (Cobs) values were generally lower than the Cpred values, particularly in the James set (figure 2). Regression analysis of Cpred versus Cobs values of remifentanil yielded the following results: Cobs = 0.46Cpred + 0.27, R2 = 0.83 for the James pharmacokinetic parameter set; and Cobs = 0.83Cpred + 0.14, R2 = 0.91 for the Janmahasatian pharmacokinetic parameter set (p < 0.001).
For the James set, PEs were distributed within a range of −89.7% to 52.2%, with a median value of −53.4%. For the Janmahasatian set, the PEs were distributed within a range of −77.9% to 80.6%, with a median value of −18.9% (figure 3). In figure 4, the PEs are plotted against time. MDPE values, a measure of the intrasubject bias (i.e. the direction and size of the deviation from the targeted concentration), ranged from −64.0% to 28.2% in the James set, with a median value of −53.4% and an interquartile range of −58.7% to −49.2%; the negative values suggested a significant overprediction (table V). In the Janmahasatian set, MDPE values ranged from −32.2% to 45.7%, with a median value of −18.9% and an interquartile range of −24.2% to −10.4% (table V). In figure 5, the patients with the best and worst agreement between Cobs and Cpred values (i.e. the lowest and highest MDPE values, respectively) are depicted for both groups. MDAPE values, a measure of inaccuracy (i.e. the size of the typical miss), also differed significantly between the groups. In the James set, MDAPE values ranged from 28.2% to 64.0%, with a median value of 53.4% and an interquartile range of 49.0–58.7%; in the Janmahasatian set, MDAPE values ranged from 3.6% to 32.2%, with a median value of 20.5% and an interquartile range of 13.3–24.8% (table V). Even though the absolute values of the wobble (a measure of total intraindividual variability in PEs) were significantly different, this was not clinically important. In the James set, the median value of the wobble was 1.4% h−1, with an interquartile range of 1.1–2.5% h−1; in the Janmahasatian set, the median value of the wobble was 1.9% h−1, with an interquartile range of 1.4–3.0% h−1 (table V). Wobble can also be inferred from figure 6, in which the difference between the PEs and MDPEs for every patient in each group is plotted against time.
Plots of the remifentanil percentage performance errors (PEs) vs time for (a) the James set and (b) the Janmahasatian set. The thin solid lines indicate the median values of the pooled PEs and the dotted lines indicate the 25th and 75th percentile values. The thick solid lines indicate the regression analysis of PEs vs time (PE = −0.10Time − 54.05, R 2 = 0.02 for the James set; PE = −0.09Time − 18.62, R 2 = 0.01 for the Janmahasatian set).
Plot of plasma remifentanil concentrations of the patient with the lowest median performance error (MDPE) and the patient with the highest MDPE vs time for (a) the James set and (b) the Janmahasatian set. The solid lines represent measured plasma concentrations (Cobs) and the dashed lines represent predicted plasma concentrations (Cpred). The thick lines represent the patient with the highest MDPE value and the thin lines represent the patient with the lowest MDPE value. Although the y-axis displays remifentanil concentrations, the terms ‘lowest’ and ‘highest’ refer to MDPEs, not concentrations.
The divergence values (i.e. time-related changes in PEs) were similar (not significantly different) in both pharmacokinetic parameter sets. In the James equation prediction set, the median value of the divergence was 3.3 h−1 with an interquartile range of 2.9–4.7 h−1; in the Janmahasatian equation prediction set, the median value of the divergence was 2.6 h−1, with an interquartile range of −0.7 to 4.5 h−1. These values suggest that the overprediction increased slightly with time for most patients in each set.
Discussion
Over the years, the pharmacokinetics of remifentanil have been evaluated in different clinical settings and classes of patients (e.g. healthy subjects, abdominal surgery, hepatic and renal failure). While the pharmacokinetic/pharmacodynamic population analysis by Minto et al.[6] has not shown the best bias value compared with other remifentanil pharmacokinetic sets,[17] it has nearly always been chosen for TCI studies because it is generally agreed that it better suits a wider and more heterogeneous population of patients. Moreover, this pharmacokinetic set has been incorporated into commercially available TCI pumps in those countries where TCI of remifentanil is available.
Because LBM[7] is a covariate of several pharmacokinetic parameters in the Minto model (namely, the V1, V2 and CLm)[6] and is also flawed when applied to morbidly obese patients,[18] it is probable that remifentanil will be underdosed in such patients, leading to poor predictive performance of the Minto pharmacokinetic set (table II).[8] After our initial report,[10] both TCI pump manufacturers updated their software (see Background section), each setting a different limit on their pumps.
Egan et al.[19] compared the pharmacokinetics of remifentanil in 12 obese subjects (bodyweight 113 ± 17 kg, height 171 ± 9 cm, age 38 ± 8 years) and 12 matched lean subjects (bodyweight 64 ± 10 kg, height 170 ± 10 cm, age 38 ± 7 years) and concluded that obesity does not significantly affect the pharmacokinetics of remifentanil. In the light of our results, they were correct, but if they had tested morbidly obese patients, using the flawed equations to calculate LBM, their results would probably have been different. In fact, LBM would not have turned out to be a significant covariate, given the fact that ‘wrong’ LBM values do not correlate with volumes and clearance. This is why the obese patients enrolled in the study by Egan et al.[19] appeared to be around their value of ‘CW’ in the LBM-TBW relationship. Therefore, the difference between ‘true’ and calculated LBM was not so great as to result in significant differences in the analysis.
As stated in the Methods section, the method we have used[8] forces the model to use the ‘true’ LBM values for calculations, even though it incorporates the traditional, flawed LBM equations. This is possible by simply modifying the input height (FH). We consider the ‘true’ LBM value to be the value that would be found following dual-energy x-ray absorptiometry.[9] This value has recently been found to normalize renal function indices in obese patients, as well.[20]
It should be noted that in order to test the new LBM formula and its incorporation into the Minto model, we have probably not chosen the most scientifically correct and straightforward method. In fact, we could have simply forced (in our simulations) the original pharmacokinetic parameter set to incorporate the corrected LBM equations rather than leaving the pharmacokinetic set unchanged (i.e. the old LBM equations) and ‘fiddled’ it to get new LBM values calculated by means of the FH. While the final result is absolutely identical (and, therefore, our choice is still scientifically correct), the first method would not have helped readers in that they would have had to wait for new and updated infusion pumps to use TCI of remifentanil in obese patients. The choice to introduce the concept of the FH and to use it to calculate the correct LBM value enables each anaesthesiologist to use TCI from now on until new pumps become available.
The choice to combine an opioid analgesic with propofol, although appropriate from a clinical point of view, could be considered a drawback of our study, as Mertens et al.[21] suggested that there is interdependence in the kinetics of alfentanil and propofol. This interrelationship is possibly due to the haemodynamic changes associated with the use of both drugs. The vasodilatory and possible negative inotropic effects of propofol, in particular, could have a major influence on systemic arterial blood pressure, the heart rate and thus cardiac output. These changes, in turn, could affect the delivery and redistribution of drugs to tissues. In this study, careful titration of remifentanil target concentrations to match patients’ requirements and to maintain systemic arterial blood pressures within ±10% of baseline values may have prevented the shifts in cardiac output that are possibly the basis of the interdependence in the kinetics of alfentanil and propofol observed by Mertens et al.[21]
The choice to use patients as their own controls might, at first, be considered another possible shortcoming of this study. However, not only is this method established,[17,22] but it is probably more correct (and ethical) than randomization of patients to different groups in this type of study and use of an incorrect pharmacokinetic parameter set in one group.
Our results indicate that during maintenance, use of the new formula to calculate LBM corrects the pharmacokinetic set of remifentanil. In fact, not only do the MDPE and MDAPE values differ significantly between the two sets, but the values obtained in the Janmahasatian equation prediction set are similar to those obtained in non-obese patients during propofol-remifentanil anaesthesia (the MDPE and MDAPE were −15% and −20%, respectively).[17] In addition, it has been suggested that the performance of a TCI system is clinically acceptable if the bias (MDPE) is not greater than 20%[16] and the inaccuracy (MDAPE) falls between 20% and 30%.[15,16] Considering these limits, the performance of the original pharmacokinetic set is not acceptable. On the other hand, the performance of the Janmahasatian set would be judged clinically acceptable, even though the 95% CI for MDPE (−28.4, −9.4) does not include zero.
Although the values obtained for divergence are similar to those obtained in similar studies, our values for the wobble are surprisingly low, suggesting lower total intraindividual variability in our group of morbidly obese female patients.
Conclusion
Although more studies are needed to confirm our results on a larger scale and also in morbidly obese male patients, our preliminary results and their implications for TCI are remarkable. They indicate that the predictive performance of the original remifentanil pharmacokinetic set is not acceptable if we use the current TCI pumps and research software, and therefore research software cannot be used in morbidly obese patients (taking this into consideration, the manufacturers of commercially available TCI pumps have already updated their software). The predictive performance of this set is preserved when ‘true’ values of LBM (the Janmahasatian equation prediction) are used to calculate the V1, V2 and CLm, indicating that it is possible to use this pharmacokinetic set even in morbidly obese patients. Until commercially available infusion pumps and research software are updated and new LBM equations are implemented in their algorithms, the FH can be a valid alternative to use in TCI infusion of remifentanil in morbidly obese patients when using both commercially available pumps and research software, since the FH forces the original pharmacokinetic parameter set to use the new formula to calculate LBM.
References
Obesity: preventing and managing the global epidemic: report of a WHO consultation. World Health Organ Tech Rep Ser 2000; 894: i–xii, 1-253
Adams JP, Murphy PG. Obesity in anaesthesia and intensive care. Br J Anaesth 2000; 85: 91–108
Agarwal N, Shibutani K, SanFilippo JA, et al. Hemodynamic and respiratory changes in surgery of the morbidly obese. Surgery 1982; 92: 226–34
Alexander JK. Obesity and circulation. Mod Concepts Cardiovasc Dis 1963; 32: 799–803
Henegar JR, Bigler SA, Henegar LK, et al. Functional and structural changes in the kidney in the early stages of obesity. J Am Soc Nephrol 2001; 12: 1211–7
Minto CF, Schnider TW, Egan TD, et al. Influence of age and gender on the pharmacokinetics and pharmacodynamics of remifentanil: I. Model development. Anesthesiology 1997; 86: 10–23
James WPT, Waterlow JC. UK Department of Health and Social Security/Medical Research Council Group on Obesity Research. Research on obesity: a report of the DHSS/MRC Group. London: Her Majesty’s Stationery Office, 1976
La Colla L, Albertin A, La Colla G. Pharmacokinetic model-driven remifentanil administration in the morbidly obese: the ‘critical weight’ and the ‘fictitious height’, a possible solution to an unsolved problem? Clin Pharmacokinet 2009; 48(6): 397–8
Janmahasatian S, Duffull SB, Ash S, et al. Quantification of lean bodyweight. Clin Pharmacokinet 2005; 44: 1051–65
La Colla L, Albertin A, La Colla G, et al. Target controlled infusion of remifentanil: beware when using it in morbidly obese patients. First World Congress of Total Intravenous Anesthesia — TCI; 2007 Sep 27–29; Venice
Bossu E, Montinaro A, Lecce R, et al. LC-MS determination of remifentanil in maternal and neonatal plasma. J Pharm Biomed Anal 2006; 42: 367–71
TIVAtrainer© version 5.1 [computer program; online]. Available from URL: http://www.eurosiva.org/TivaTrainer/tivatrainer_main.htm [Accessed 2009 Nov6]
Rugloop© version 3.28 [computer program; online]. Available from URL: http://www.demed.be/rugloop.htm [Accessed 2009 Nov 6]
Varvel JR, Donoho DL, Shafer SL. Measuring the predictive performance of computer-controlled infusion pumps. J Pharmacokinet Biopharm 1992; 20: 63–94
Miller RD, editor. Anesthesia. 6th ed. New York: Churchill Livingstone, 2006
Schuttler J, Kloos S, Schwilden H, et al. Total intravenous anaesthesia with propofol and alfentanil by computer-assisted infusion. Anaesthesia 1988; 43 Suppl.: 2–7
Mertens MJ, Engbers FH, Burm AG, et al. Predictive performance of computer-controlled infusion of remifentanil during propofol/remifentanil anaesthesia. Br J Anaesth 2003; 90: 132–41
Green B, Duffull S. Caution when lean body weight is used as a size descriptor for obese subjects. Clin Pharmacol Ther 2002; 72: 743–4
Egan TD, Huizinga B, Gupta SK, et al. Remifentanil pharmacokinetics in obese versus lean patients. Anesthesiology 1998; 89: 562–73
Janmahasatian S, Duffull SB, Chagnac A, et al. Lean body mass normalizes the effect of obesity on renal function. Br J Clin Pharmacol 2008; 65: 964–5
Mertens MJ, Olofsen E, Burm AG, et al. Mixed-effects modeling of the influence of alfentanil on propofol pharmacokinetics. Anesthesiology 2004; 100: 795–805
Vuyk J, Engbers FH, Burm AG, et al. Performance of computer-controlled infusion of propofol: an evaluation of five pharmacokinetic parameter sets. Anesth Analg 1995; 81: 1275–82
Acknowledgements
These data were presented in part at the 2008 Congress of the Società Italiana di Anestesia Analgesia Rianimazione e Terapia Intensiva (Palermo, Italy; 14–17 October 2008). Support for this work was provided solely from institutional/departmental sources. The authors want to thank Daniele Pinna, MSc, from the Department of Physics at New York University (New York, NY, USA) for his help during the revision process. The authors have no conflicts of interest that are relevant to the contents of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
La Colla, L., Albertin, A., La Colla, G. et al. Predictive Performance of the ‘Minto’ Remifentanil Pharmacokinetic Parameter Set in Morbidly Obese Patients Ensuing from a New Method for Calculating Lean Body Mass. Clin Pharmacokinet 49, 131–139 (2010). https://doi.org/10.2165/11317690-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11317690-000000000-00000