Abstract
After 50 years of clinical experience with the aminoglycoside agents, there is continuing debate over the most appropriate administration regimen for these drugs. In recent years, once daily administration has been used increasingly, in the hope of both improving efficacy and reducing toxicity. At least 30 controlled clinical trials have compared once versus conventional multiple daily administration. Efficacy was assessed in some, but not all, studies using clinical and/or bacteriological cure. Toxicity was generally determined using rather nonsensitive end-points such as measurement of serum creatinine for nephrotoxicity and clinically detectable hearing loss for ototoxicity.
The results of individual clinical trials and subsequent meta-analyses have been variable. However, 5 of 9 meta-analyses found clinical efficacy to be significantly better with once daily administration, and in 3 of the 9 there were significantly less nephrotoxicity with once daily administration. The results were not significant for ototoxicity in any of the meta-analyses.
There is debate about how therapeutic drug monitoring should be performed, and whether it is still required with once daily administration. Previous experience with the aminoglycosides, especially in patients with impaired drug clearance caused by renal impairment, suggests that monitoring is still prudent. Results from the once daily administration trials appear to support this.
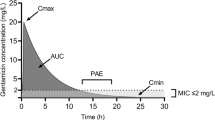
Various methods of monitoring and dose adjustment have been proposed. The most common is to measure a 24-hour trough concentration and to adjust the dose to maintain the trough concentration below a value of 2, 1 or 0.5 mg/L. However, this method allows for greater total aminoglycoside exposure than has been permitted with conventional dosages, increasing the likelihood of toxicity in patients with impaired aminoglycoside clearance.
Other methods measure drug concentrations at a time-point or points within the dose interval (when the concentration is still measurable), and adjust the dose according to concentration-time curve nomograms or to a target area under the concentration-time curve. This allows the use of higher doses in those with high drug clearance. Furthermore, in patients with impaired clearance, drug exposure is limited to the same extent as, or less than, that with conventional multiple daily administration. To date no controlled trials have compared methods of dose-individualisation.
In summary, in addition to a slight overall improvement in efficacy, once daily administration has resulted in a small reduction in nephrotoxicity. In the studies using more sensitive measures of toxicity, the differences in toxicity were greater, strengthening the case for once daily administration. Therapeutic drug monitoring is probably required with once daily administration. Methods which use mid-dosage interval concentrations to gauge drug exposure would seem to be preferable over trough concentration measurement.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Begg EJ, Barclay ML. Aminoglycosides: 50 years on. Br J Clin Pharmacol 1995; 39: 597–603.
Ali MZ, Goetz MB. A meta-analysis of the relative efficacy and toxicity of single daily dosing versus multiple daily dosing of aminoglycosides. Clin Infect Dis 1997; 24: 796–809.
Bailey TC, Little JR, Littenberg B, et al. A meta-analysis of extended-interval dosing versus mutiple daily dosing of aminoglycosides. Clin Infect Dis 1997; 24: 786–95.
Barza M, Ioannides JP, Capelleri JC, et al. Single or multiple daily doses of aminoglycosides: a meta-analysis. BMJ 1996; 312: 338–45.
Ferriol-Lisart R, Alós-Almiñana M. Effectiveness and safety of once-daily cimonglycosides: a meta-analysis. Am J Health Syst Pharm 1996; 53: 1141–50.
Freeman CD, Strayer AH. Mega-analysis of meta-analysis: an examination of meta-analysis with an emphasis on once-daily aminoglycoside comparative trials. Pharmacotherapy 1996; 16: 1093–102.
Galloe AM, Graudal N, Christensen HR, et al. Aminoglycosides: single or multiple daily dosing? Eur J Clin Pharmacol 1995; 48: 39–43.
Hatala R, Dinh TT, Cook DJ. Once-daily aminoglycoside dosing in immunocompetent adults: a meta-analysis. Ann Intern Med 1996; 124: 717–25.
Hatala R, Dinh TT, Cook DJ. Single daily dosing of aminoglycosides in immunocompromised adults: a systemic review. Clin Infect Dis 1997; 24: 810–5.
Munckhof WJ, Grayson ML, Turnidge JD. A meta-analysis of studies on the safety and efficacy of aminoglycosides given either once daily or as divided doses. J Antimicrob Chemother 1996; 37: 645–63.
Marra F, Partovi N, Jewesson P. Aminoglycoside administration as a single daily dose: an improvement to current practice or a repeat of previous errors? Drugs 1996; 52: 344–70.
Barclay ML, Begg EJ, Chambers ST. Adaptive resistance following single doses of gentamicin in a dynamic in vitro model of infection. Antimicrob Agents Chemother 1992; 36: 1951–7.
Just M, Erdmann G, Habermann E. The renal handling of polyphasic drugs: 1. Gentamicin and aprotonin in intact animals. Naunyn Schmeidebergs Arch Pharmacol 1977; 300: 57–66.
Silverblatt FJ, Kuehn C. Autoradiography of gentamicin uptake by the rat proximal tubule cell. Kidney Int 1979; 15: 335–45.
Wedeen RP, Batuman V, Cheeks C, et al. Transport of gentamicin in rat proximal tubule. Lab Invest 1983; 48: 212–23.
Giuliano RA, Verpooten GA, Verbist L, et al. In vivo uptake kinetics of aminoglycosides in the kidney cortex of rats. J Pharmacol Exp Ther 1986; 236: 470–5.
Nedden R, Fuchs T, Schroder K, et al. Die renale Ausscheidung von Gentamicin beim Menschern. Deutsch Med Wochenschr 1972; 97: 1496–8.
Kaloyanides GJ. Aminoglycoside nephrotoxicity. In: Schrier W, Gottschalk X, editors. Diseases of the kidney. 5th ed. Boston: Little Brown and Company, 1992: 1131–64.
Ibrahim S, Derde MP, Kaufman L, et al. Safety, pharmacokinetics and efficacy of once-a-day netilmicin and amikacin versus their conventional schedules in patients suffering from pelvic inflammatory disease. Ren Fail 1990; 12: 199–203.
Barclay ML, Begg EJ. Aminoglycoside toxicity and relation to dose regimen. Adverse Drug React Toxicol Rev 1994; 13: 207–34.
Beck PR, Thomson RB, Chaudhuri AKR. Aminoglycoside antibiotics and renal function: changes in urinary gamma-glutamyl transferase excretion. J Clin Pathol 1977; 30: 432–7.
Gibey R, Dupond J-L, Alber D, et al. Predictive value of urinary N-acetyl-beta-glucosaminidase (NAG), alanine-aminopeptidase (AAP), and beta-2-microglobulin (B2M) in evaluating nephrotoxicity of gentamicin. Clin Chem Acta 1981; 116: 25–34.
Schentag JJ, Sutfin TA, Plaut ME, et al. Early detection of aminoglycoside nephrotoxicity with urinary beta-2-microglobulin. J Med 1978; 9: 201–10.
Schentag JJ. Specificity of renal tubular damage criteria for aminoglycoside nephrotoxicity in critically ill patients. J Clin Pharmacol 1983; 23: 473–83.
Lietman PS, Smith CR. Aminoglycoside nephrotoxicity in humans. Rev Infect Dis 1983; 5: S284.
Houghton DC, Lee D, Gilbert DM, et al. Chronic gentamicin nephrotoxicity: continued tubular injury with preserved glomerular filtration function. Am J Pathol 1986; 123: 183–94.
Laurent G, Toubeau G, Heuson-Stiennon JA, et al. Kidney tissue repair after nephrotoxic injury: biochemical and morphological characterization. CRC Crit Rev Toxicol 1988; 19: 147–83.
Sens MA, Hazen-Martin DJ, Blackburn JG, et al. Growth characteristics of cultured human proximal tubule cells exposed to aminoglycoside antibiotics. Ann Clin Lab Sci 1989; 19: 266–79.
Fausti SA, Henry JA, Schaffer HI, et al. High-frequency audiometric monitoring for early detection of aminoglycoside ototoxicity. J Infect Dis 1992; 165: 1026–32.
Powell SH, Thompson WL, Luthe MA, et al. Once-daily vs continuous aminoglycoside dosing: efficacy and toxicity in animal and clinical studies of gentamicin, netilmicin and tobramycin. J Infect Dis 1983; 147: 918–32.
Brummett RE, Fox KE. Aminoglycoside-induced hearing loss in humans. Antimicrob Agents Chemother 1989; 33: 797–800.
Dulon D, Zajic G, Aran J-M, Schacht J. Aminoglycoside antibiotics impair calcium entry but not viability and motility in isolated cochlear outer hair cells. J Neurosci Res 1989; 24: 338–46.
Nakagawa T, Kakehata S, Akaike N, et al. Effects of Ca2+ antagonists and aminoglycoside antibiotics on Ca2+ current in isolated outer hair cells of guinea pig cochlea. Brain Res 1992; 580: 345–7.
Black FO, Pesznecker RN. Vestibular toxicity: clinical considerations. Otolaryngol Clin North Am 1993; 5: 705–12.
Matz GJ. Aminoglycoside cochlear ototoxicity. Otolaryngol Clin North Am 1993; 5: 705–12.
Mattie H, Craig WA, Pechere PC. Determinants of efficacy and toxicity of aminoglycosides. J Antimicrob Chemother 1989; 24: 281–93.
Beaubien AR, Desjardins S, Ormsby E, et al. Incidence of amikacin ototoxicity: a sigmoid function of total drug exposure independent of plasma levels. Am J Otolaryngol 1989; 10: 234–43.
Barclay ML, Begg EJ, Hickling KG. What is the evidence for once daily aminoglycoside therapy? Clin Pharmacokinet 1994; 27: 32–48.
Giamarellou H, Yiallouros K, Petrikkos G, et al. Comparative kinetics and efficacy of amikacin administered once or twice daily in the treatment of systemic Gram-negative infections. J Antimicrob Chemother 1991; 27: 73–9.
Hansen M, Achen F, Carstensen C, et al. Once- versus thrice-daily dosing of netilmicin in febrile immunocompromised patients: a randomized, controlled study of efficacy and safety. J Drug Dev 1988; 1 Suppl. 3: 119–24.
Marik PE, Lipman J, Kobilski S, et al. A propspective randomized study comparing once- versus twice-daily amikacin dosing in critically ill adult and paediatric patients. J Antimicrob Chemother 1991; 28: 753–64.
Mauracher EH, Lau W-Y, Kartowisastro H, et al. Comparison of once-daily and thrice-daily netilmicin regimens in serious systemic infections: a multicenter study of six Asian countries. Clin Ther 1989; 11: 604–13.
Raz R, Adawi, M, Romano S. Intravenous administration of gentamicin once daily versus thrice daily in adults. Eur J Clin Microbiol Infect Dis 1995; 14: 88–91.
Prins JM, Buller HR, Kuijper EJ, et al. Once versus thrice daily gentamicin in patients with serious infections. Lancet 1993; 341: 335–9.
Tulkens PM. Pharmacokinetic and toxicological evaluation of a once-daily regimen versus conventional schedules of netilmicin and amikacin. J Antimicrob Chemother 1991; 27 Suppl. C: 49–61.
Bailey RR, Begg EJ, Smith AH, et al. Prospective, randomised, controlled study comparing two dosing regimens of gentamicin/oral ciprofloxacin switch therapy for acute pyelonephritis. Clin Nephrol 1996; 46: 183–6.
Tulkens PM, Clerkx-Braun F, Donnez J, et al. Safety and efficacy of aminoglycosides once-a-day: experimental data and randomized, controlled evaluation in patients suffering from pelvic inflammatory disease. J Drug Dev 1988; 1 Suppl. 3: 71–82.
Darrell JH, Waterworth PM. Dosage of gentamicin for pseudomonas infections. BMJ 1967; 2: 535–7.
Jackson GG, Riff LJ. Pseudomonas bacteremia: pharmacologic and other bases for failure of treatment with gentamicin. J Infect Dis 1971; 124 Suppl. 124: S185–91.
Moore RD, Smith CR, Lietman PS. The association of aminoglycoside plasma levels with mortality in patients with gramnegative bacteremia. J Infect Dis 1984; 149: 443–8.
Moore RD, Smith CR, Lietman PS. Association of aminoglycoside plasma levels with therapeutic outcome in gram-negative pneumonia. Am J Med 1984; 77: 657–62.
Noone P, Parsons TMC, Pattison JR, et al. Experience in monitoring gentamicin therapy during treatment of serious gram-negative sepsis. BMJ 1974; 1: 477–81.
Noone P, Rogers BT. Pneumonia caused by coliforms and Pseudomonas aeruginosa. J Clin Pathol 1976; 29: 652–6.
Dahlgren JG, Anderson ET, Hewitt WL. Gentamicin blood levels: a guide to nephrotoxicity. Antimicrob Agents Chemother 1975; 8: 58–62.
Goodman EL, Van Gelder J, Holmes R, et al. Prospective comparative study of variable dosage and variable frequency regimens for administration of gentamicin. Antimicrob Agents Chemother 1975; 8: 434–8.
Smale F. Commentary: single daily dose of aminoglycoside is the preferred mode of administration. BMJ; 312; 344–5.
Parker SE, Davey PG. Practicalities of once daily dosing. J Antimicrob Chemother 1993; 31: 4–8.
Prins JM, Weverling GJ, de Blok K, et al. Validation and nephrotoxicity of a simplified once-daily aminoglycoside dosing schedule and guidelines for monitoring therapy. Antimicrob Agents Chemother 1996; 40: 2494–9.
Appel GB. Aminoglycoside nephrotoxicity. Am J Med 1990; 88 Suppl. 3c: 16S–20S.
Bertino JSJ, Brooker LA, Franck PA, et al. Incidence of the significant risk factors for aminoglycoside-associated nephrotoxicity in patients dosed by using individualized pharmacokinetic monitoring. J Infect Dis 1993; 167: 173–9.
Moore RD, Smith CR, Lipsky JD, et al. Risk factors for nephrotoxicity in patients treated with aminoglycosides. Ann Intern Med 1984; 100: 352–7.
Barclay ML, Begg EJ, Duffull SB, et al. Experience of once-daily aminoglycoside dosing using a target area under the concentration-time curve. Aust NZ J Med 1995; 25: 230–5.
Verpooten GA, Giuliano RA, Pattyn VM, et al. Renal cortical uptake kinetics of gentamicin in rats with impaired renal function. Am J Kidney Dis 1986; 8: 304–7.
McCormack JP, Jewesson PJ. A critical reevaluation of the ‘therapeutic range’ of aminoglycosides. Clin Infect Dis 1992; 14: 320–39.
Singer C, Smith C, Krieff D. Once-daily aminoglycoside therapy: potential ototoxicity. Antimicrob Agents Chemother 1996; 40: 2209–11.
Nicolau DP, Freeman CD, Belliveau PP, et al. Experience with once-daily aminoglycoside program administered to 2,184 adult patients. Antimicrob Agents Chemother 1995; 39: 650–5.
Begg EJ, Barclay ML, Duffull SB. A suggested approach to once-daily aminoglycoside dosing. Br J Clin Pharmacol 1995; 39: 605–9.
Cronberg S. Antimicrobial practice: simplified monitoring of aminoglycosides. J Antimicrob Chemother 1994; 34: 819–27.
Duffull SB, Kirkpatrick CMJ, Begg EJ. Comparison of Bayesian approaches to dose-individualisation for once-daily aminoglycoside regimens. Br J Clin Pharmacol 1997; 43: 125–35.
Hickling KG, Begg EJ, Perry RE, et al. Serum aminoglycoside clearance is predicted as poorly by renal aminoglycoside clearance as by creatinine clearance in critically ill patients. Crit Care Med 1991; 19: 1041–7.
Kirkpatrick CMJ, Begg EJ. The dose response relationships of gentamicin-induced phospholipiduria and nephrotoxicity in Coopworth sheep. Proc Aust Soc Clin Exp Pharmacol Toxicol 1996; 3: 59.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Barclay, M.L., Kirkpatrick, C.M.J. & Begg, E.J. Once Daily Aminoglycoside Therapy. Clin Pharmacokinet 36, 89–98 (1999). https://doi.org/10.2165/00003088-199936020-00001
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-199936020-00001