Abstract
Background
The mainstay of treatment of well-differentiated thyroid cancer (WDTC) is surgery followed by adjuvant radioactive iodine therapy. Postoperative radiation therapy (PORT) is rarely used.
Objective
The aim of our study was to report our experience of patients with WDTC who were selected to receive PORT.
Materials and Methods
After Institutional Review Board approval, patients who received PORT were identified from a departmental database of 6259 patients with WDTC treated with primary surgery from 1986 to 2015. We carried out propensity matching to compare outcomes with a cohort of patients who did not receive PORT. The main outcome of interest was central neck recurrence-free probability (CNRFP), while secondary outcomes were lateral neck recurrence-free probability (LNRFP), disease-specific survival (DSS), and overall survival (OS).
Results
From 6259 patients, 32 (0.5%) patients with a median age of 65.2 years received PORT. Tall-cell variant papillary thyroid carcinoma was the most common pathology (45%). Patients who received PORT had no difference in CNRFP compared with patients treated without PORT (10-year CNRFP 88% vs. 73%; p = 0.18). Furthermore, patients who received PORT had superior LNRFP (10-year LNRFP 100% vs. 62%; p = 0.001) compared with the no-PORT cohort. Despite this, patients who received PORT had similar DSS (71% PORT vs. 75% no-PORT) and OS (65% PORT vs. 58% no-PORT group) as the no-PORT cohort.
Conclusions
Our data show that select patients who received PORT had improved locoregional recurrence-free probability; however, this did not translate into improved DSS and OS. At our institution, we recommend the use of PORT only in highly selected patients with locally advanced primary tumors who are deemed to have a high risk of central neck recurrence for which salvage surgery would result in unacceptable risk to the airway.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The incidence of differentiated thyroid cancer is increasing worldwide. Every year in the US alone more than 64,000 new cases are diagnosed and the increase is mostly attributable to papillary thyroid cancer.1 This dramatic increase in early-stage thyroid cancer is largely due to improved early diagnosis.2 However, the incidence of stage IV thyroid cancer is also increasing. Differentiated thyroid cancer is treated surgically by lobectomy or total thyroidectomy with or without neck dissection.3 Postoperative radioactive iodine (RAI) therapy is administered to select patients in the intermediate-risk group and nearly all patients in the high-risk group according to the recommendations of the American Thyriod Association (ATA) or other similar guidelines.4
External-beam radiotherapy (EBRT) is rarely utilized in the management of patients with well-differentiated thyroid cancer (WDTC), with publications typically from single institutions.5,6 National Comprehensive Cancer Network (NCCN) guidelines recommend that EBRT should be considered for unresectable disease or gross residual tumor after surgical resection.7 These indications apply mainly to patients with locally advanced primary thyroid tumors with invasion of surrounding structures such as the larynx, trachea, or esophagus. In these cases, positive margins, microscopic residual disease, or gross residual disease may be present. Often patients with positive margins and microscopic residual disease can be managed with RAI or observation. Patients with gross residual disease after surgery are relatively uncommon as the vast majority of patients with differentiated thyroid cancer present with surgically resectable disease. EBRT is therefore rarely indicated in the management of patients with WDTC. The decision to use EBRT must always be weighed against the potential adverse effects and long-term sequela of treatment, including swallowing and speech problems as a result of radiation to the central compartment neck structures. This is why EBRT is rarely administered in patients with microscopic positive margins, relying on RAI to achieve local control with the avoidance of toxicity from EBRT. Consequently, there are no prospective randomized trials comparing the use of EBRT with other options, such as surgical salvage, observation, or RAI treatment. Existing literature from retrospective data report conflicting results, with some reporting no improvement in outcome whereas others report EBRT prolongs recurrence-free survival and even disease-specific survival (DSS) of patients with advanced thyroid cancer.8 However, these studies contained a heterogeneous cohort of patients that included not only patients who had EBRT performed after surgery (PORT) but also patients who had EBRT as palliative treatment for inoperable recurrence. The radiation dose used in these studies also varied. In addition, these studies included patients with mixed histology and included poorly differentiated thyroid cancer as well as WDTC.
The aim of our study was to report our experience of patients with WDTC managed with surgery followed by postoperative radiation therapy (PORT). The primary outcomes we were interested in were central neck recurrence-free probability (CNRFP) and impact on survival. We carried out propensity matching using a cohort of patients with similar clinical and pathological characteristics who did not receive PORT to oncological outcomes.
Materials and Methods
Patients included in this study were selected from our existing database of 6259 patients with WDTC primarily treated with surgery by our service between 1986 and 2015. The study was approved by the Institutional Review Board at our center.
Of the entire database, only 32 (0.5%) patients received PORT. Most patients in this group received PORT due to incomplete tumor resection or microscopic positive margins in high-risk areas of the central neck. Patients were pathologically staged according to the 8th Edition of the American Joint Committee on Cancer (AJCC).
Propensity Matching
Statistical analysis was performed using R version 3.6.2 (The R Foundation for Statistical Computing, Vienna, Austria). To select the control group of patients who did not receive PORT in order to compare with the patients treated with PORT, we specified age, sex, histology type, and TNM stage as our propensity score matching criteria. The R package ‘MatchIt’ and ‘optmatch’ were used at a 1:1 ratio, as the the most precise match pairs are achieved using 1:1 matching, not with 1:2 or 1:3 matching.9,10 Optimal matching finds the matched samples with the smallest average absolute distance across all matched pairs. The Pearson Chi-square test or Fisher’s exact test were used for the comparison of categorical covariates between the two groups, and Student’s t-test was used for comparison of the continuous variable age.
Outcomes Analysis
The main outcome of interest was CNRFP, which includes thyroid bed and level VI node recurrences. We analyzed secondary outcomes, including lateral neck recurrence-free probability (LNRFP), distant recurrence-free probability (DRFP), DSS, and overall survival (OS). Cumulative incidence function was used to compare CNRFP, LNRFP and DRFP, with death as a competing risk, and a Gray’s test was performed to compare the groups. The Kaplan–Meier method was used to compare the remaining outcomes and a log-rank test was performed to compare the two groups. All local and regional recurrences were structural biopsy-proven recurrences. Thyroglobulin data were not available for these cases and thus biochemical recurrence was not recorded. The follow-up interval was calculated in months from the date of initial surgery. A p-value of <0.05 was considered statistically significant.
Results
Clinical and Pathological Characteristics of the Postoperative Radiation Therapy (PORT) Group
The clinical and pathological characteristics of the selected patients are shown in Table 1. The median age of the patients receiving PORT was 65.2 years (range 34.7–83.7 years) and the majority of cases were male (n = 23, 72%). Aggressive types of WDTC were prevalent in this group of patients; 14 (44%) patients had tall cell variant papillary thyroid carcinoma (PTC) and 3 (9%) patients had Hurthle cell carcinoma. Twenty six patients (81%) had a primary tumor invading strap muscles, and 29 (91%) additionally invaded adjacent structures such as the trachea, larynx, recurrent laryngeal nerves, and esophagus. One case (3%) had gross residual disease, while the remaining patients had microscopic positive margins in high-risk areas of the central neck. Metastases to regional lymph nodes in the central and lateral neck were present in 26 patients (81%). The majority of cases (66%) had RAI therapy, with a median dose of 193 mCi (interquartile range 144–216) (electronic supplementary Table 1).
The median time from surgery to PORT was 3.2 months (range 0.9–8.4 months), and patients received PORT with a median dose of 62 Gy (range 50–70 Gy). The central and lateral necks were included in the target volume in 25 patients (76%), thyroid bed with central neck in four patients (12%), and in four cases (12%) the radiation fields were unknown (electronic supplementary Table 2).
Clinical and Pathological Characteristics of the Propensity Matched No-PORT Group
We achieved similar clinical and pathologic characteristics of the matched cohort (Table 1). The median age was 60.1 years (range 25.5–91.3 years), with males being the most common sex (69%). There were no significant differences in age, sex, histology, TNM stage, RAI and extrathyroidal extension (ETE) between the PORT and no-PORT groups. In addition, there was no significant difference in the positive margin rate or extent of initial surgery between groups. In the matched control group obtained using optimal matching, the absolute standardized difference ranged from 0.069 to 0.471.
Central Neck and Lateral Neck Recurrence-Free Probability
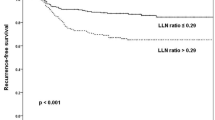
The median follow-up of the PORT group was 91.7 months (range 7–281 months), and 90.6 months (range 12–385 months) for the control no-PORT group. Patients treated with PORT had no difference in CNRFP compared with patients treated without PORT (10-year CNRFP 88% vs. 73%; Grays’s test p = 0.18) (Fig. 1a). The PORT group had three local recurrences and no central neck nodal recurrences, compared with the matched no-PORT group, where three local and four central neck nodal recurrences occurred.
Estimated cumulative incidence curves with death as a competing factor of A central local recurrence-free probability, B lateral neck recurrence-free probability, and D distant recurrence-free probability, and Kaplan–Meier curves of (C) disease-specific survival. CNRFP central neck recurrence-free probability, LNRFP lateral neck recurrence-free probability, DRFP distant recurrence-free probability, DSS disease-specific survival, PORT postoperative radiotherapy
Patients treated with PORT had superior LNRFP compared with patients treated without PORT (10-year LNRFP 100% vs. 62%; Grays’s test p = 0.001) (Fig. 1b). In the PORT group, no patients had a lateral neck recurrence, compared with nine in the no-PORT group. The majority of patients (76%) in the PORT group had the lateral neck included in the target field for RT. Univariate analysis showed that the PORT group had a hazard ratio (HR) of 0.227 (95% confidence interval 0.042–1.221; p = 0.0232) compared with the matched control group.
Disease-Specific and Overall Survival
To determine if the improved LNRFP translated into improved survival, we compared DSS and OS between the groups. Thirty deaths were recorded in both groups, 13 of which were disease-specific.
There was no difference in OS between the groups (OS: 5- and 10-year OS was 87.8% and 64.8% for the PORT group compared with 71.1% and 57.8% for the no-PORT group, respectively; p = 0.41) [electronic supplementary Fig. 1]. Furthermore, there was no difference in DSS between the groups (DSS: 5- and 10-year DSS was 89.7% and 71% for the PORT group compared with 80.6% and 75.2% for the no-PORT group, respectively; p = 0.88) (Fig. 1c).
Distant Recurrence-Free Probability
To determine if the lack of improved DSS in the PORT group was due to poorer DRFP, we compared the DRFP between the groups. There was no difference in DRFP (10-year DRFP 70% vs. 65%; Grays’s test p = 0.65) (Fig. 1d). In those patients who received PORT, eight developed distant metastasis during the follow-up period compared with six patients in the no-PORT group.
Salvage Treatment in Patients Treated Without PORT
We next analyzed the treatment of recurrence in patients who did not receive PORT in comparison with patients who did receive PORT (details are shown in Table 2). Of 11 recurrences, two (18%) were local, two (18%) were central with lateral neck node, and seven (64%) were lateral neck node recurrences. Of those patients who did not receive PORT, six (55%) with recurrence were able to be treated with surgery; two had a bilateral neck dissection, three had a lateral neck dissection, and one had a central neck dissection. Of the remaining five patients, three had observation only, due to distant metastasis or concurrent diseases preventing safe surgery, and two patients were treated with RAI. The detailed characteristics of each patient with recurrence are given in Table 3. The ability to salvage a significant portion of recurrences explains the similar OS and DSS in the PORT and no-PORT groups.
Toxicity of Radiation Treatment
Adverse effects from PORT are shown in Table 4. Data on toxicity were available for 31 patients. The majority of patients experienced grade 1–2 mucositis, xerostomia, and dermatitis. Grade 3–4 toxicities were rare—one case each of dermatitis and mucositis and three cases of dysphagia. Three patients (10%) required placement of the feeding tube during RT and one patient (3%) had a tracheostomy due to severe laryngeal congestion. Two patients (6%) required feeding tube placement in a late post-RT period.
Discussion
The use of EBRT in patients with WDTC is a very controversial topic with scarce published research and no prospective randomized clinical data. All existing papers have only a small number of patients, with a mixture of well and poorly differentiated pathology and comprising patients managed by surgery with PORT and also those treated with palliative RT without surgery. For example, a recently published paper by Samhouri et al.5 included a group of patients with diverse pathology, including medullary and anaplastic thyroid cancers. The study included EBRT administered both as PORT and also for metastatic cancer sites. Some earlier published studies excluded anaplastic thyroid cancers, focusing on differentiated thyroid cancer, and they also included medullary thyroid cancers.11 Medullary thyroid cancers are a specific pathological entity that should be studied in separate trials. Another paper published by Romesser et al. excluded both anaplastic and medullary thyroid cancers, concentrating on PTC and follicular thyroid carcinoma (FTC), but at least half of the patients were classified as having poorly differentiated thyroid cancers.12 That study also included patients with gross residual disease after surgery and also patients who did not have surgery because of unresectable disease. Conventional radiation techniques as well as intensity-modulated radiation therapy (IMRT) were reported. Beckham et al. reported on a more homogenous group of patients treated only with IMRT and only patients with PTC and FTC;13 however, that study still included unresectable disease at the time of presentation and those with unresectable recurrence in the cohort. Therefore, the current literature that includes this heterogeneity in histology and radiation indications makes it difficult to interpret the results of these studies and make any meaningful recommendations for PORT treatment in patients with WDTC.
PORT is rarely administered in patients with WDTC because surgery is considered to be safe and effective as a salvage procedure. This is especially true for salvage of lateral neck recurrence, which is usually achievable with a neck dissection with limited morbidity. Recurrence in the central neck, either due to local thyroid bed recurrence or paratracheal lymph node recurrence, is much more challenging. Surgical salvage carries increased risk in the central neck with possible injury to the recurrent laryngeal nerves and of permanent hypoparathyroidism. Surgical treatment of central neck recurrence may also require tracheal resection, laryngectomy, or even pharyngectomy and esophagectomy, resulting in diminished quality of life.14 Therefore, the site of possible recurrence will dramatically influence the surgeons’ decision to recommend PORT. Early series reported recurrence in the central neck as the most common reason of death in the management of DTC.15,16 However, due to improvements in surgery and the appropriate use of PORT, failure in the central neck with subsequent death is now quite rare. Nowadays, most deaths occur due to distant metastatic disease.17
Papers investigating the role of PORT usually use local and/or locoregional recurrence probability as the main outcome of interest;5,18,19 however, using only local recurrence-free probability underestimates failure in the central neck due to central neck node recurrences. In addition, locoregional recurrence-free probability includes both lateral and central neck recurrence.13 Since control of the central neck is the most important aspect in patients with locally advanced thyroid cancer, we used CNRFP as our primary endpoint, as this includes both the local thyroid bed and also level VI node recurrences.
The decision to use PORT also results in local toxicity, with detrimental effects on speech and especially on swallowing.20 Schuck et al. reported an increased rate of skin, mucosal, and salivary toxicity, and many patients experienced difficulty with swallowing. Although the toxicity markedly decreased 100 days after EBRT, a substantial number of patients still had salivary gland and swallowing issues. Similar results were reported by Shugard et al., with around one-third of patients receiving PORT reporting grade 2 dermatitis, mucositis, and xerostomia. Moreover, at 90 days post RT, 10–17% of cases still had symptoms of dysphagia and xerostomia.21
In our study, we report that patients selected to receive PORT had improved LNRFP compared with a propensity-matched no-PORT control group. At the same time, there was no difference in CNRFP. This may be due to the small number of cases and limited number of central neck recurrences. A larger dataset would be necessary to show the potential difference. Despite improved locoregional-free recurrence probability, this did not translate into improved OS and DSS compared with the propensity matched cohort of patients who did not receive PORT. We postulated that one possible explanation for the similar survival outcomes could be poorer DRFP in the PORT group. For this reason, we compared DRFP in each group. In fact, there was no significant difference between groups, which suggests that despite poorer locoregional recurrence-free probability, patients treated with surgery without PORT were able to be salvaged. In the propensity matched group, 11 patients had recurrence, of whom 55% were salvaged surgically and 27% of cases were managed with observation alone. This successful salvage treatment explains the similar survival outcomes in the two groups.
At our institution, the decision to treat patients with PORT is highly selective and involves a multidisciplinary team (MDT) decision. In general, PORT is only administered if the surgeon believes that there is a high risk of central neck recurrence for which salvage surgery is associated with a significant risk of RLN sacrifice, tracheal resection, or laryngectomy. In the majority of patients, surgical salvage is favored if possible. The rationale to favor surgical salvage is mainly due to the presumed toxicity of radiation. In the past, parallel opposed field radiation, and later three-dimensional (3D) conformal radiation encompassing the central and lateral neck from the hyoid bone to the mediastinum, were traditionally used. Such a wide field resulted in severe toxicity with regard to swallowing and speech dysfunction;11,22 however, nowadays it is possible to deliver more accurate and focused fields using IMRT. It is possible to spare the lateral neck, deliver less radiation to the pharyngeal constrictor muscles, and spare the submandibular gland function focusing only on the areas of high-risk of recurrence.11 This results in a better adverse effect profile.11,19,23,24 Therefore, one may argue that with present-day technology, a greater number of patients at risk for central neck recurrence may benefit from PORT and spare them future surgical salvage procedures. Another important factor is the patients’ preferences, which should be considered with a clear explanation of the different possible outcomes and the possible adverse effects of each treatment option. The possible indolent course of the disease should also be weighed against the possible toxicity of PORT. Lastly, other factors such as advanced age, extensive previous surgery, and the presence of distant disease also play an important role in the decision to administer PORT.
We recognize that our study has limitations due to its retrospective nature and that the data were from a single institution. We were limited by the fraction of patients who received PORT from this large cohort of patients with differentiated thyroid cancer. Moreover, our cohort received several different types of PORT techniques, including two-dimensional (2D; 2 cases), 3D conformal, and IMRT, over a span of several decades. Additionally, 11 patients did not receive RAI. These factors could impact outcomes, although in our cohort the difference between the PORT and no-PORT groups, based on some of the mentioned parameters, did not show significant difference. The strength of the study is that participants were selected from a large cohort of well-documented patients with differentiated thyroid cancer treated within the same comprehensive cancer center with a uniform institutional philosophy. Using our large database has allowed us to identify only patients with WDTC who received PORT, and allowed us to exclude patients with poorly differentiated or anaplastic thyroid cancer and also patients with unresectable disease treated without surgery. Given the lack of any prospective randomized trial, we believe the use of propensity matching allows more meaningful conclusions to be achieved for the use of PORT in these patients.
Conclusion
The decision to treat patients with PORT is highly selective and requires a comprehensive MDT discussion, taking into account patient preferences. Our data show that select patients who received PORT had improved locoregional recurrence-free probability with an acceptable adverse effect profile from radiation. At our institution, we recommend the use of PORT in highly selected patients who undergo surgery for locally advanced primary tumors (T4a) and/or significant central nodal disease and who have a high risk of central neck recurrence in whom surgical salvage is deemed to be unattractive due to unacceptable risks to the airway.
References
Howlader N, Noone AM, Krapcho M, Miller D, Brest A, Yu M, et al. (eds). SEER Cancer Statistics Review, 1975-2017, National Cancer Institute. Bethesda, MD, https://seer.cancer.gov/csr/1975_2017/, based on November 2019 SEER data submission, posted to the SEER web site, April 2020.
Olson E, Wintheiser G, Wolfe KM, Droessler J, Silberstein PT. Epidemiology of thyroid cancer: a review of the National Cancer Database, 2000–2013. Review Cureus. 2019;11(2):e4127. https://doi.org/10.7759/cureus.4127.
Harries V, Wang LY, McGill M, et al. Should multifocality be an indication for completion thyroidectomy in papillary thyroid carcinoma? Surgery. 2020;167(1):10–7. https://doi.org/10.1016/j.surg.2019.03.031.
Alexander EK, Pearce EN, Brent GA, et al. 2017 Guidelines of the American Thyroid Association for the Diagnosis and Management of thyroid disease during pregnancy and the postpartum. Thyroid. 2017;27(3):315–89. https://doi.org/10.1089/thy.2016.0457.
Samhouri L, Kriz J, Elsayad K, et al. The role of radiotherapy for patients with thyroid cancer in the modern era. Anticancer Res. 2020;40(6):3379–86. https://doi.org/10.21873/anticanres.14321.
Kiess AP, Agrawal N, Brierley JD, et al. External-beam radiotherapy for differentiated thyroid cancer locoregional control: a statement of the American Head and Neck Society. Head Neck. 2016;38(4):493–8. https://doi.org/10.1002/hed.24357.
Haddad RI, Nasr C, Bischoff L, et al. NCCN guidelines (R) insights thyroid carcinoma, Version 2.2018 featured updates to the NCCN guidelines. J Natl Comprehensive Cancer Network. 2018;16(12):1429–40. https://doi.org/10.6004/jnccn.2018.0089.
Fussey JM, Crunkhorn R, Tedla M, Weickert MO, Mehanna H. External beam radiotherapy in differentiated thyroid carcinoma: a systematic review. Review Head Neck. 2016;38:E2297–305. https://doi.org/10.1002/hed.24218.
Ho DE, Imai K, King G, Stuart EA. MatchIt: nonparametric preprocessing for parametric causal inference. J Stat Software. 2011;42(8):28.
Allan V, Ramagopalan SV, Mardekian J, et al. Propensity score matching and inverse probability of treatment weighting to address confounding by indication in comparative effectiveness research of oral anticoagulants. J Comp Eff Res. 2020;9(9):603–14. https://doi.org/10.2217/cer-2020-0013.
Rosenbluth BD, Serrano V, Happersett L, et al. Intensity-modulated radiation therapy for the treatment of nonanaplastic thyroid cancer. Int J Radiat Oncol Biol Phys. 2005;63(5):1419–26. https://doi.org/10.1016/j.ijrobp.2005.05.043.
Romesser PB, Sherman EJ, Shaha AR, et al. External beam radiotherapy with or without concurrent chemotherapy in advanced or recurrent non-anaplastic non-medullary thyroid cancer. J Surg Oncol. 2014;110(4):375–82. https://doi.org/10.1002/jso.23656.
Beckham TH, Romesser PB, Groen AH, et al. Intensity-modulated radiation therapy with or without concurrent chemotherapy in nonanaplastic thyroid cancer with unresectable or gross residual disease. Thyroid. 2018;28(9):1180–9. https://doi.org/10.1089/thy.2018.0214.
Nixon IJ, Simo R, Newbold K, et al. Management of invasive differentiated thyroid cancer. Review Thyroid. 2016;26(9):1156–66. https://doi.org/10.1089/thy.2016.0064.
Smith SA, Hay ID, Goellner JR, Ryan JJ, McConahey WM. Mortality from papillary thyroid-carcinoma: a case-control study of 56 lethal cases. Cancer. 1988;62(7):1381–8. https://doi.org/10.1002/1097-0142(19881001)62:7%3c1381::aid-cncr2820620724%3e3.0.co;2-r.
Tollefsen HR, Hutter RVP, Decosse JJ. Papillary carcinoma of thyroid: clinical + pathological study of 70 fatal cases. Cancer. 1964;17(8):1035–44. https://doi.org/10.1002/1097-0142(196408)17:8%3c1035::aid-cncr2820170810%3e3.0.co;2-w.
Nixon IJ, Ganly I, Palmer FL, et al. Disease-related death in patients who were considered free of macroscopic disease after initial treatment of well-differentiated thyroid carcinoma. Thyroid. 2011;21(5):501–4. https://doi.org/10.1089/thy.2010.0451.
Hamilton SN, Tran E, Berthelet E, Wu J. The role of external beam radiation therapy in well-differentiated thyroid cancer. Review Expert Rev Anticancer Therapy. 2017;17(10):905–10. https://doi.org/10.1080/14737140.2017.1361324.
Tam S, Amit M, Boonsripitayanon M, et al. Adjuvant external beam radiotherapy in locally advanced differentiated thyroid cancer. JAMA Otolaryngol-Head Neck Surg. 2017;143(12):1244–51. https://doi.org/10.1001/jamaoto.2017.2077.
Schuck A, Biermann M, Pixberg MK, et al. Acute toxicity of adjuvant radiotherapy in locally advanced differentiated thyroid carcinoma: first results of the multicenter study differentiated thyroid carcinoma (MSDS). Strahlentherapie Und Onkologie. 2003;179(12):832–9. https://doi.org/10.1007/s00066-003-1158-1.
Shugard E, Chen J, Quivey JM, et al. Does radiation dose matter in thyroid cancer? Patterns of local-regional failure in recurrent and metastatic well-differentiated thyroid cancers treated with dose-painted intensity-modulated radiation therapy. J Med Imaging Radiat Oncol. 2016;60(4):560–7. https://doi.org/10.1111/1754-9485.12452.
Nutting CM, Convery DJ, Cosgrove VP, et al. Improvements in target coverage and reduced spinal cord irradiation using intensity-modulated radiotherapy (IMRT) in patients with carcinoma of the thyroid gland. Radiotherapy Oncol. 2001;60(2):173–80. https://doi.org/10.1016/s0167-8140(01)00382-6.
Lee N, Puri DR, Blanco AI, Chao KSC. Intensity-modulated radiation therapy in head and neck cancers: an update. Headeck. 2007;29(4):387–400. https://doi.org/10.1002/hed.20332.
Yu N, Khurana A, Foote R, et al. Proton beam therapy for differentiated thyroid cancer: initial report of the Mayo Clinic Experience. Int J Radiat Oncol Biol Phys. 2020;108(2):E47–E47.
Funding
This research was funded in part through the NIH/NCI Cancer Center Support Grant P30 CA008748.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
Dauren Adilbay, Avery Yuan, Paul B. Romesser, Richard J. Wong, Jatin P. Shah, Ashok R. Shaha, Michael R. Tuttle, Snehal Patel, Nancy Y. Lee, and Ian Ganly declare no conflicts of interest pertinent to this work. Snehal G. Patel has a patent pending (PCT/US2016/026717: Methods of Cancer Detection Using PARPi-FL), holds equity in Summit Biomedical Imaging and ColdSteel Laser Inc., and has the following patents: US10,016,238B2: Apparatus, system and method for providing laser steering and focusing for incision, excision, and ablation of tissue in minimally invasive surgery; PCT/US2014/073053: Systems, methods, and apparatus for multichannel imaging of fluorescent sources in real time; PCT/US2015/065816: Cyclic peptides with enhanced nerve-binding selectivity, nanoparticles bound with said cyclic peptides, and use of same for real-time in vivo nerve tissue imaging; and PCT/US2016/066969: Imaging systems and methods for tissue differentiation, e.g., for intraoperative visualization. Nancy Lee reports advisory board participation for Merck, Merck EMD, Elsie, Mirati, and Roche.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Adilbay, D., Yuan, A., Romesser, P.B. et al. Well-Differentiated Thyroid Cancer: Who Should Get Postoperative Radiation?. Ann Surg Oncol 29, 5582–5590 (2022). https://doi.org/10.1245/s10434-022-11898-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-022-11898-2