Abstract
Background
Soil-borne plant pathogenic fungi with a wide host range of crops cause a significant limitation on the global production of agronomic crops. Applications of synthetic pesticides are an important tool for managing plant diseases, but have deleterious influences on the environment as well as its incompatibility with organic agriculture. Recently, Streptomyces spp. became one of the best bio-control agents as a promising environmentally eco-friendly method for effective management of plant diseases.
Results
In a previous research, three species of Streptomyces spp., i.e., S. griseus (MT210913 “DG5”), S. rochei (MN700192 “DG4”) and S. sampsonii (MN700191 “DG1” strains) were identified, as exhibiting potent antifungal activities against plant pathogenic fungus, Sclerotinia sclerotiorum in vitro and greenhouse. GC–Mass analysis revealed the presence of 44, 47 and 54 substances of S. sampsonii DG1, S. griseus DG5 and S. rochei DG4, respectively. GC–MS revealed substances, with bio-control activity, were categorized as volatile organic compounds (VOCs), fatty acids and plant growth regulators, etc. GC–MS analysis exhibited the presence of 7, 13 and 20 volatile compounds produced by S. sampsonii, S. rochei and S. griseus, respectively. These substances exhibited potent antifungal activity against various plant pathogenic fungi, i.e., Botrytis cinerea, Macrophomina phaseolina, Rhizoctonia solani and S. sclerotiorum in vitro, by dual-culture assay. The three strains inhibited all the pathogenic fungi in dual-culture assay in the range of 30–73.67%. Also, the produced substances were applied in vivo (in the field) and supported their potential biocontrol agent against S. sclerotiorum as well as possessed significant biological properties for plant health and growth. Applying Streptomyces spp. culture broth in the field enhanced physiological responses of phenols, sugar, chlorophyll, protein contents and parameters as well as the yield of bean plants.
Conclusion
In field experiments, foliar application of Streptomyces spp. and their metabolites proved to be a great potential, as promising biocontrol agents, for controlling S. sclerotiorum and enhanced plant growth and yield. S. rochei and S. griseus proved to be strong antifungal, plant growth promoters and environmentally eco-friendly fungicides.
Similar content being viewed by others
Background
Plant diseases caused by fungi and bacteria accounted for 42 and 27%, respectively, of the global crop production loss (14%) which represents $200 billion a year, and this destruction can worsen under climate changes (Elad and Pertol 2014). Botrytis cinerea, Macrophomina phaseolina, Rhizoctonia solani and Sclerotinia sclerotiorum are the most prevalent and destructive fungal diseases affecting crops in all growing regions. However, these soil-borne pathogens are very difficult to control, because of their broad-host range and high survival rate of sclerotia under various environmental conditions. Excessive applications of fungicides lead to harmful effects, i.e., higher input costs, human health problems, environmental pollution, pathogens resistance and phytotoxicity (Le et al. 2022). Nowadays, researchers are focused on identifying and applying environmentally friendly alternatives for controlling plant diseases and improving crop productivity, which are recommended within an integrated crop management ecosystem (Boukhatem et al. 2022).
Last few decades, controlling plant diseases using biocontrol agent antagonists developed much attention. Recently, the most important bioagents used in biological control are the Streptomyces genus. Streptomyces spp. belonging to this genus are of great importance in the field of biotechnology, as producers of beneficial bioactive secondary metabolites with extensive industrial, medical and agricultural applications (Harir et al. 2018). The secondary metabolites produced by Streptomyces spp. are diversified in their biological activities and functions as antifungal, antibacterial, insecticidal, enzymes, antibiotics and activities (Katarzyna et al. 2018). Streptomyces spp. have the potential inhibitory effect on the growth of various plant pathogens, i.e., fungi (Le et al. 2022), bacteria (Guo et al. 2020) and viruses (Li et al. 2019). Moreover, Streptomyces spp. are known to improve the availability of nutrients and minerals, enhance the production of metabolites and promote plant growth regulators (Boukhatem et al. 2022). The objective of the present research is to: (a) evaluate three Streptomyces spp. in vitro on four plant pathogenic fungi (B. cinerea; M. phaseolina; R. solani and S. sclerotiorum), (b) identify their secondary metabolites by GC–MS analysis and (c) evaluate its ability in controlling S. sclerotiorum causative agent of bean white rot disease in vivo.
Methods
The experiments were carried out in two phases, firstly in vitro antagonism assay of microorganisms against various plant pathogenic agents and secondly in vivo biocontrol trials on susceptible bean plants cultivated in the producer's fields located in Dahshur Province, Giza Governorate, Egypt, during two successive seasons (2018–2019 and 2019–2020).
Isolation of microorganisms and in vitro antagonism assay
This experiment was conducted in the laboratory at the Central Lab. of Organic Agri., Agri. Research Center (ARC) and Plant Pathology Dept., Fac. of Agric., Cairo Univ., Giza, Egypt. Three Streptomyces species were previously characterized molecularly, using PCR technique, then sequenced and submitted to NCBI GenBank and gave accession numbers, i.e., S. griseus (MT210913"DG5"), S. rochei (MN700192"DG4") and S. sampsonii (MN700191"DG1") strains. Streptomyces spp. were cultured and maintained on starch casein (StC) medium as described in our previous report (Gebily et al. 2021).
Concerning the tested fungi, various plant pathogenic fungi, i.e., B. cinerea, M. phaseolina, R. solani and S. sclerotiorum were isolated from Strawberry, Sesame, Sweet pepper and Bean plants, respectively. These fungi were maintained and stored at the laboratory, using a potato dextrose agar (PDA) medium. Five-mm-diameter inoculum disks of the above phytopathogenic fungi were cut from the margin of the fresh colonies on PDA plates. Each disk was inoculated on the center of the PDA pour plate and incubated at 20 °C (S. sclerotiorum and B. cinerea) and 25 °C (R. solani and M. phaseolina) for 7 days. To evaluate bioagent activity, each of the three Streptomyces species was dually cultured with all above-mentioned phytopathogenic fungi. The growth inhibitory effects on the pathogenic fungi were represented as a percentage of pathogen growth inhibition (PIG) and calculated according to Zambrano et al. (2021) following equation:
where R1 = Growth of pathogenic fungus in the control plate. R2 = Growth of pathogenic fungus interacting with the antagonist.
Gas chromatography–mass (GC–Mass) analysis of Streptomyces spp. secondary metabolites
This experiment aimed to identify secondary metabolites produced by Streptomyces spp. which play an important role in their antagonistic activities against the phytopathogenic fungi in two steps as follows: Extraction of the secondary metabolites using ethyl acetate according to Parthasarathi et al. (2012) and gas chromatography–mass spectrometry (GC–Mass) analysis as described by El-Kareem et al. (2016). The obtained pellets of Streptomyces spp. (S. griseus, S. rochei and S. sampsonii) were categorized using GC–Mass and performed using Trace GC-ISQ mass spectrometer (Thermo Scientific, Austin, TX, USA) with a direct capillary column TG–5MS (30 m × 0.25 mm × 0.25-µm film thickness). The components were identified by comparison of their retention times and mass spectra with those of WILEY 09 and NIST14 mass spectral databases.
Application of Streptomyces spp. on the control bean white rot disease in vivo
Nowadays, due to the harmful effect on the environment caused by excessive usage of pesticides, the urge to use an alternative method became precisely important and motivated toward managing the diseases in sustainable agriculture. For this reason, a field experiment was conducted to (a) apply an eco-friendly manner such as microbial bioagents and/or their mycoparasitic activity toward pathogen sclerotia for decreasing their number in the soil and infection percent; (b) improve the quality and quantity of bean pods by controlling white rot disease in the field under natural infection; (c) check the ability of the selected three Streptomyces spp. in controlling white rot disease caused by S. sclerotiorum, bean plants were sprayed by Streptomyces spp. as individual or mixture treatments and (d) determine the most effective Streptomyces spp. and their concentrations to control white rot disease and enhancing plant parameters components (phenols, sugars, chlorophyll and protein contents).
Effect of applying Streptomyces spp. under field conditions on disease reduction and enhancing plant growth
In vivo experiment was carried out to evaluate the effect of different concentrations of Streptomyces spp. to control S. sclerotiorum infecting bean. Field experiments were conducted in light sandy soil, irrigated by a flood system and naturally infested due to our observation from the previous plantations. All experiments were designed in complete randomized blocks design. Three replicates were used for each treatment; each plot (2.8 × 5 m) was used as a replicate. Each replicate contained 100 bean seeds cv. Paulista. All plants received the same fertilizers and irrigation regime. Streptomyces spp. were applied individually or in combinations under field conditions. All the used Streptomyces spp. were grown into liquid starch casein medium for 7 days. All media components of the Streptomyces spp. were prepared as culture broth (containing cell-free extract, mycelia + spores) + 5% acacia gum (Arabic gum) and 0.5% potassium soap. Each suspension was adjusted using sterilized distilled water to be contained (15 × 106 c.f.u./ml). The application was carried out 4 times (15 days intervals) on the plants after 30-day post-sowing. Corporal Max was used as a positive control (commercial fungicide produced by the Central Lab. for Organic Agric.). Control treatment plants were sprayed with water.
Impact of applying different concentrations of Streptomyces culture broth
To determine the most efficient and economic concentration of each Streptomyces spp., three different concentrations (diluted by water) were applied for each treatment 1/50, 1/100, and 1/150 (v/v). Plants were sprayed by three isolates of Streptomyces spp. either individually or their combinations to evaluate disease reduction, plant parameters and components as the following:
Evaluation of disease reduction and plant parameters
The percentages of disease incidence and reduction in white rot disease were calculated after the development of natural disease symptoms on the control plants (60-day post-sowing). Healthy plants of three-month-old were collected from each treatment, and the plant's dry matter and pods were dried using an electric oven at 70 °C. The total yield and number of pods/plant in the different treatments were calculated per feddan.
Impact of Streptomyces spp. treatments on plant components
Analyses of plant contents were carried out at the Central Lab. of Organic Agric. Samples of the treated plants were collected from different tested treatments to assess their contents (phenols, sugars and protein in pods as well as leaves chlorophyll). Collected samples were washed with tap water and then, prepared to determine reducing and non-reducing sugars using the method developed by AOAC (2005). Total phenols, free and conjugated were also determined using a method described by Simons and Ross (1971). Protein and leaf chlorophyll contents were also determined using the methods described by Bradford (1976) and Askar and Treptow (1993), respectively.
Statistical analysis
Data were statistically analyzed according to the general linear models procedure of Statistical Package for Social Sciences (SPSS 2008) Version 17.0.0 Software. Least significant difference test (LSD) at ≤ 5% level of probability (P ≤ 0.05) was used. The presented data were calculated for two successive seasons.
Results
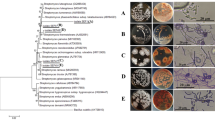
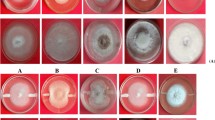
Streptomyces spp. exhibited broad-spectrum antifungal activity against four phytopathogenic fungi, i.e., B. cinerea, M. phaseolina, R. solani and S. sclerotiorum. In vitro dual-culture assay revealed that the volatile organic compounds (VOCs) produced by the three strains of Streptomyces spp. showed inhibitory activity against the mycelial growth of all the tested phytopathogenic fungi in the range of 30–73.67%. S. sampsonii revealed the highest antifungal activity (73.67%) against B. cinerea, followed by S. rochei (70.83%) and S. griseus (70.33%), while S. sampsonii showed the lowest antifungal activity (35.33%) against R. solani, followed by S. griseus (33.33%) and S. rochei (30%) (Table 1 and Fig. 1).
Identification of potent bioactive compounds by GC–MS
The GC–MS analysis profiles of the active fraction and its constituents from the selected strains were presented in (Figs. 2, 3). A total of 44, 47 and 54 compounds for S. sampsonii DG1; S. griseus DG5 and S. rochei DG4, respectively, were detected and compared with those entries in the NIST14 database, and the nearest compound resembling those peaks was identified, as well as its molecular weight (MW), retention time (RT), and other characteristics. It was noticed that the fraction contained various types of major VOCs compounds including groups of alkenes, sulfides, ketones, benzenoids, aromatic hydrocarbons, odors, hormones and alcohols. The most abundant volatile compounds produced by the three Streptomyces spp. were phenol-2-methoxy4,2 propenyl (Eugenol); 1,2 benzenedicarboxylic acid; phenols and heptanol, 6-methyl; 1-hexadecanol, acenaphthenol, behenic alcohol, 2,4-di-tert-butylphenol; benzene acetic acid-butyl ester; 2-acetoxy-1,1,10-trimethyl-6,9-epidioxydecalin; tetrahydroactinidiolide; 4-hydroxybenzaldehyde; benzene, 1,1-(1-butene-1,4-diyl)bis-(Z); N-(4-hydroxyphenethyl) acetamide, 7,9-Di-tertbutyl-1-oxaspiro decadienedione and 2-(3,4-dimethoxyphenyl)-2,3-dihydro-3-hydroxy-5,7-dimethoxy-3-phenyl-4H-1-benzopyranone. The results indicated the inhibitory activities of one or more volatile compounds produced by each Streptomyces strain on mycelial growth, suppressing spore germination or sclerotia formation of all pathogenic fungi. Also, the result of in vitro bioassay showed that the VOCs from the three Streptomyces spp. had a strong antifungal activity against B. cinerea, followed by M. phaseolina and S. sclerotiorum (Fig. 1).
Molecular structures of the representative compounds. Mass spectrum of the compounds obtained by GC–MS analysis of three Streptomyces spp. were compared with the WILEY 09 and NIST14 mass spectral databases. S. rochei (a1–8): (a1) 2,4-Di-Tert-butylphenol, (a2) Phenyl acetic acid, (a3) Gibberellic acid (GA), (a4) Palmitic acid, (a5) Ethanol, 2-(Octadecyloxy), (a6) Cholestanol, (a7) 2-methylene, & (a8) Hexadecenoic acid (Erucic acid). S. griseus (b1–8): (b1) Indole, (b2) Naphthalene, 2-ethenyl, (b3) Methanone, Bis-4-phenoxyphenyl, (b4) 1,2-benzenedicarboxylic acid (Phthalate), b5) Oleic acid, (b6) Thiocarbamic acid, (b7) O-Methylated Flavone, (Flavonoid or Genkwanin & (b8) 9-Octadecenoic acid (Oleic Acid). and S. sampsonii (c1–8): (c1) 1,2-Benzenedicarboxylic acid (Phthalic acid), (c2) Naphthalene, 1,6-Dimethyl-4-(1-Methylethyl), (b3) 3-Methyl-butyric acid (b4) Eugenol phenol,2-Methoxy-4-(2-Propenyl). (c5) 2,6-Ditert-butylbenzo-1,4-Quinone, (c6) 2 h-Naphthol, Octahydro-dimethyl, (c7) Heptanol and (c8) Hexane-1-carboxylic acid
Concerning the GC–MS chromatogram peak area percent, molecular weight and molecular formulae of three Streptomyces spp., the consequent compound were identified and shown in Fig. 2. Peak areas (%) were directly proportional to the degree of the bioactive compounds existing in the culture broth. GC–MS analysis revealed the presence of 7, 13 and 20 VOCs produced by S. sampsonii, S. rochei and S. griseus, respectively. Also, GC–MS analysis showed the existence of peak area percentages (0.87, 0.55 and 1.64%) of phthalate derivatives such 1,2- benzenedicarboxylic acid were produced by S. sampsonii, S. rochei and S. griseus, respectively. Further, GC–MS analysis indicated the presence of the highest peak area percentages of fatty acids for S. sampsonii strain. Peak area percentages were 6.63, 5.84, 3.74, 3.05 and 2.60% for oleic acid, palmitic acid, palmitoleic acid and hexadecanoic acid, respectively. Regarding S. rochei strain, data indicated that it was recognized by the highest peak area percentages of aromatic hydrocarbon derivatives, i.e., acenaphthene (10.25%), 1-acenaphthenol (2.59%), benzene acetic acid-butyl ester, (1.97%), 7,9-di-tert-butyl-1-oxaspirodeca-6,9-diene-2,8-dione, (1.81%). On the other hand, GC–MS chromatogram of S. griseus strain exhibited the highest peak area percentages of aromatic compound derivatives such as 1,4-benzenediol, 2-(1,1-dimethylethyl) 5, 2-propenyl (5.48%), 1-Acenaphthenol (3.83%), benzene,1,1-(2-butene-1,4-diyl) bis (2.25%), 1-benzoxirenol,2,2,5a-trimethyl-1a[2-(2-methyl)-1,3-dioxolanyl)1-ethenyl] hydro (1.82%), N(4-Hydroxyphenethyl) acetamide (0.83%), thiocarbamic acid, N,N-dimethyl, S-1,3-diphenyl-2-butenyl ester (1.90%) and methanone, bis (4-phenoxyphenyl) (1.25%). The aforementioned results indicated that these derivatives and components played an important role individually or combined with others as biologically active and effective against the tested phytopathogenic fungi. Additionally, GC–MS showed that S. rochei (DG4) and S. griseus (DG5) were able to produce secondary metabolites as indolic compounds, i.e., phenylacetic acid (PAA), 1H-Indole and gibberellic acid (GA) at a very low area percent (1.87, 0.59 and 0.28%, respectively). Thus, the result suggested that the application of Streptomyces spp. represented a promising sustainable solution to improve plant growth, productivity and disease reduction as well as alternative pesticides.
Impact of culture broth concentrations
Streptomyces spp. culture broth was used at various concentrations to prove the most effective one in reducing bean white rot disease, enhancing plant vigor and components in the treated plants as follows:
Streptomyces spp. efficacy on bean white rot disease reducing
To determine the most effective concentration of different Streptomyces spp. on disease control, different concentrations were tested as presented in Table 2. The various concentrations of each Streptomyces sp. led to a significant reduction in white rot disease compared with the control. In all cases, a clear positive correlation was noticed between the concentration of the antagonist and its efficacy. The highest reduction (61.33%) was obtained when used S. rochei (DG4) at the concentration (1:50). The highest reductions with the same concentration in the mixtures were noticed when S. griseus (DG5) mixed with S. sampsonii (DG1) or S. rochei (DG4), recording (86.67 and 84%) in disease reduction, respectively. Results also indicated that using a mixture of the three Streptomyces spp. together was the most effective treatment for disease control and resulted in a (92%) reduction when used at a concentration of 1:50 (v/v) compared with the control (0.00%) and Copral Max (74.67%).
Streptomyces spp. efficacy on enhancing green bean plant vigor
The used concentrations of each Streptomyces sp. culture broth exhibited significant differences in the percentages of plant and pods dry matter of the treated plants than the control. Results in Table 3 indicated that the most effective concentration in the individual treatment was S. rochei, since recorded the highest number of pods (22 pods/plant) and yield (3.9 ton/feddan). Also, it increased the dry matter of plants and pods to (67.33 and 18.67%), respectively. Also, the results revealed that the mixtures in all cases significantly enhanced plant vigor when compared with an individual treatment at the same concentration. Further, application of the triple mixture of S. rochei (DG4), S. sampsonii (DG1) and S. griseus (DG5) resulted in the highest control and increased all parameters related to plant vigor when using concentrations of (1:50 and 1:100), recording the highest number of pods (29 pods/plant) and yield (5.37 ton/feddan).
Streptomyces spp. efficacy on the treated plant components
Field experiments were conducted to determine the physiological responses of bean plants by applying an individual or mixture of Streptomyces spp. at different concentrations. Results of plant analyses included phenol, sugar, chlorophyll and protein contents are illustrated in Table 4. The presented data suggested that increased concentration of the tested Streptomyces spp. either individual or mixtures treatment increased its effectiveness which was reflected in the plants growth and changes in their components. Protein amount reached the highest content (9.18) when applying a triple mixture of Streptomyces spp., followed by the dual mixture of S. rochei (DG4) and S. griseus (DG5) (6.55), then S. rochei and S. sampsonii (DG1) (6.30) and S. sampsonii and S. griseus (5.80). Regarding phenols, all the treatments either individual or combination of Streptomyces spp. at a concentration of 1:50 increased the amount of total and free phenols than the control. On the contrary, the increase in conjugated phenols was correlated with the low concentration (1:150) in the different treatments. The mixture of three Streptomyces spp. resulted in the highest percentage of disease control and the highest amount of phenol content. A mixture of S. sampsonii (DG1), S. rochei (DG4) and S. griseus (DG5) reached the highest levels of free and total phenols (14.2 and 16.35, respectively) and 5.8 reduced sugars. Sugar contents were determined to clear positive correlation between the increase in the concentration of Streptomyces spp. and the number of reduced sugars in all cases.
Discussion
Until now, white rot disease of bean caused by S. sclerotiorum remains a significant challenge to recognize and control its infection mechanisms. To develop effective biocontrol agents to control the pathogen, three strains of Streptomyces spp., i.e., S. sampsonii (MN700191 “DG1”), S. rochei (MN700192 “DG4”) and S. griseus (MT210913 “DG5”), were isolated, tested in vitro and molecularly characterized in the previous research (Gebily et al. 2021). Streptomyces sp. is well-known for producing strong VOCs, odors, fatty acids and plant growth regulators. In the dual-culture assay, Streptomyces spp. induced inhibition of all the tested phytopathogenic fungi, which indicated the production of antifungal compounds that may be involved in the inhibition of mycelial growth, as there was no direct contact between the phytopathogenic fungi and Streptomyces spp. The result revealed that volatile substances of Streptomyces spp. had a potential for usage as a bio-fumigant to control plant fungal diseases. Such results are consistent with those described by other investigators working with Streptomyces species that often produce a wide diversity of secondary metabolites inhibiting the growth of phytopathogenic fungi and bacteria (Le et al. 2022).
Thus, the progress and exploitation of biological agents, such as antagonistic Streptomyces spp. and their secondary metabolites, became a promising alternative way to control plant diseases. Nowadays, GC–MS developed a novel technique to identify the secondary metabolites produced by Streptomyces species, and its analysis is very reliable to identify the compound in the complex biochemical products. The resultant GC–MS peak area, molecular weight, formula and the derived compounds were identified in the presented results. The peak area was directly relational to the level of the bioactive compounds prevailing in the composites. GC–MS analysis showed the presence of (44, 47 and 54) compounds for S. sampsonii DG1; S. griseus DG5 and S. rochei DG4, respectively. Obtained results of the antifungal effect of Streptomyces spp. culture broth on mycelial growth of different pathogens revealed that all bioagent strains tested produced several substances, i.e., alcohols, alkenes, aromatic hydrocarbons, benzenoids, fatty acids and alcohols, ketones, sulfides and steroids with varying efficiencies. GC–MS analysis showed various types of VOCs compounds produced by Streptomyces spp. and play an important role as one of the biocontrol mechanisms against plant diseases. Further, GC–MS analysis of three Streptomyces spp. revealed similarities in their producing many volatiles and other substances as mentioned before.
Results indicated significant differences among the number and components of the VOCs (7, 13 and 20) produced by S. sampsonii “MN700191 DG1,” S. rochei “MN700192 DG4” and S. griseus “MT210913 DG5,” respectively. The presented results of the three Streptomyces spp. were recorded as major aromatic compounds, i.e., phenol, acenaphthene, 1,4-benzenediol and phthalate derivatives (PAHs such benzenedicarboxylic acid or phthalic acid) with the highest number and area percentage, which supported their role in bioactivities against the tested pathogens. This result is resembling those obtained by Danaei et al. (2014) who mentioned that the antifungal activity of S. griseus cultures can be attributed to carvacrol, benzaldehyde, naphthalene and phenol as well as other compounds. Also, the results were in harmony with Ahsan et al. (2017) who reported that Streptomyces strain KX852460 produced major aromatic compounds with the highest peak number and had a good antifungal activity as well as potential biocontrol agent against R. solani. Furthermore, Memic et al. (2017) tested various polycyclic aromatic hydrocarbons (PAHs) against two ligninolytic fungi Hypoxylon fragiforme (wood white rot) and Coniophora puteana (wood brown rot). They found that the antifungal activity of PAHs depended on the number of aromatic or benzene rings and area percentage of the compound as well as molecular structure. Nevertheless, the VOCs components were identified as antimicrobial, antibacterial and antifungal to control bacterial and fungal diseases in vitro by destroying, deformation, and degrading cell membrane integrity and mycelial ultrastructure of the pathogens (Le et al. 2022); and in vivo (fields) and postharvest as alternative pesticides as well as environmentally friendly options (Kaari et al. 2022).
Also, the results of the present study indicated that the secondary metabolites such as fatty acids might contribute to VOC's inhibitory antagonistic effect on the mycelial growth and sclerotia formation of the tested phytopathogenic fungi. This result had already been described by Liu et al. (2008) on S. griseus, who evaluated in vitro inhibition of nine fatty acids against four phytopathogenic fungi. They detected that all fatty acids suppressed the mycelial growth of one or more tested fungi excluding oleic acid. Similarly, many researchers discussed that various Streptomyces spp. produced secondary metabolites as antibacterial, antifungal and antimicrobial bioactivities (Sholkamy et al. 2020).
Concerning the production of plant growth regulators, S. rochei “MN700192 DG4” followed by S. griseus “MT210913 DG5” proved to be the greatest effective producers with an area percentage of (1.87, 0.59 and 0.28%), respectively. Also, the result indicated that S. rochei was able to promote plant growth due to its secretion of some secondary metabolites as plant growth regulators such as auxin and gibberellin. Thus, they should be a great biological agent to control phytopathogens and promote plant growth. Such results were confirmed by several authors who applied Streptomyces fradiae NKZ-259 (Myo et al. 2019), Streptomyces tricolor HM10 (Rehan et al. 2021) and Streptomyces lydicus M01 (Wang et al. 2020). Similarly, Vurukonda et al. (2018) recorded that the S. rochei was able to promote plant growth due to its secretion of some secondary metabolites, i.e., auxin, gibberellin and cytokinin.
In the present research, the application of Streptomyces spp. to control bean white rot disease explained that the use of a mixture containing the three biological agents was the most efficient treatment due to their multiple weapons and mechanisms such as hyper-parasitism and secretion of secondary metabolites, which plays a role as potential biocontrol agents for controlling S. sclerotiorum pathogen in the field. The result agreed with Muiru et al. (2007) who tested secondary metabolites from three Streptomyces spp. isolates coded CS35, 28P, 14P against Pythium spp. The highest degree of activity was obtained from isolate 14P, it could be attributed to the presence of higher amounts and concentrations of antibiotics compared to metabolites from the other Streptomyces isolates.
Conferring Streptomyces spp. application to enhance plant vigor, the triple mixture of S. Streptomyces spp. proved to be the most efficient in increasing parameters (number of pods per plant, dry matter of plants (g.) and pods (g.)/plant as well as the weight of total yield [Ton/fed]) when concentrations of 1:50 and 1:100 were used. Similarly, Gopalakrishnan et al. (2015) tested six strains of Streptomyces spp. on the growth promotion and grain yield in chickpea (Cicer arietinum L.). Of six Streptomyces strains, CAI-85, CAI-93 and KAI-180 were found superior to CAI-155, CAI-140 and CAI-13, in terms of their effects on root and shoot development, nodule formation and crop productivity. These strains significantly enhanced the pod number and weight, leaf area and weight as well as stem weight and crop maturity, grain yield, total dry matter, pod weight seed number and seed weight compared with the untreated plants. Nevertheless, Mun et al. (2020) evaluated the ability of Streptomyces sp. LH4, a plant growth-promoting rhizobacteria (PGPR), when applied to colonize cucumber plant roots and enhance resistance against S. sclerotiorum. The results indicated that LH4 successfully colonized the root area promoting plant growth and proliferations. Applying Streptomyces spp. in the present paper resulted in changes including physiological responses of phenol, sugar, chlorophyll and protein contents of bean plants, indicating that their metabolites may have an excessive role as plant-growth-promoting, involvement in plant development and health. These results were consistent with those quoted by Nithya et al. (2020) who mentioned that Streptomyces applied plants had increased parameters such as carotenoid, sugars, nitrogen, amino acids, chlorophyll and protein contents. In the same respect, Cherif et al. (2007) mentioned that the percentage of chickpea blight disease (Ascochyta rabiei) reduction was correlated with the increase in free and total phenols. Further, Le et al. (2022) proved that Streptomyces sp. JCK-6131 revealed antagonistic activity in vitro against apple fire blight, tomato bacterial wilt cucumber, Fusarium wilt diseases caused by Erwinia amylovora, Ralstonia solanacearum and Fusarium oxysporum f. sp. cucumerinum, respectively. In vivo, JCK-6131 had suppressed the development of tomato bacterial wilt, cucumber Fusarium wilt and apple fire blight (on the detached leaf). Foliar application of tomato had induced pathogenesis-related (PR) genes and increased levels of salicylate (SA) as well as jasmonate (JA) in the treated plants compared with the untreated ones. They suggested that JCK-6131 played an efficient dual mechanism of antibiosis and induced resistance.
Conclusions
Obtained results confirmed that the foliar application of S. griseus (MT210913 “DG5”), S. rochei (MN700192 “DG4”) and S. sampsonii (MN700191 “DG1”) revealed a strong antifungal activity and inhibited the mycelial growth against various plant pathogenic fungi in vitro. It should be a valuable tool for controlling various plant pathogens, enhancing plant growth and yield of bean crop as well as reducing heavy usage of chemical fertilizers and fungicides. S. rochei and S. griseus proved to have a strong antifungal activity and plant growth promotion.
Availability of data and materials
All data are available in the manuscript.
Abbreviations
- DG1:
-
S. sampsonii
- DG4:
-
S. rochei
- DG5:
-
S. griseus
- GA:
-
Gibberellic acid
- PAA:
-
Phenylacetic acid
- PAHs:
-
Phthalate derivatives
- VOCs:
-
Volatile organic compounds
References
Ahsan T, Chen J, Zhao X, Irfan M, Wu Y (2017) Extraction and identification of bioactive compounds (eicosane and dibutyl phthalate) produced by Streptomyces strain KX852460 for the biological control of Rhizoctonia solani AG-3 strain KX852461 to control target spot disease in tobacco leaf. AMB Expr 7:1–10. https://doi.org/10.1186/s13568-017-0351-z
AOAC (2005) Official methods of association of official analytical chemists, 18th edn. AOAC International, Gaithersburg
Askar A, Treptow H (1993) Analytical methods. In: Askar A, Treptow H (eds) Quality assurance in tropical fruit processing. Springer, New York, pp 27–28
Boukhatem ZF, Merabet C, Tsaki H (2022) Plant growth promoting actinobacteria, the most promising candidates as bioinoculants? Front Agron 4:849911. https://doi.org/10.3389/fagro.2022.849911
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cherif M, Arafaoui A, Rhaiem A (2007) Phenolic compounds and their role in bio-control and resistance of chickpea to fungal pathogenic attacks. Tunis J Plant Prot 2:7–21
Danaei M, Baghizadeh A, Pourseyedi S, Amini J, Yaghoob M (2014) Biological control of plant fungal diseases using volatile substances of Streptomyces griseus. Eur J Exp Biol 4(1):334–339
El-Kareem MS, Rabbih MA, Selim ET, Elsherbiny EE, El-Khateeb AY (2016) Application of GC/EIMS in combination with semi-empirical calculations for identification and investigation of some volatile components in basil essential oil. Int J Anal Mass Spectrom Chromatogr 4:14–25. https://doi.org/10.4236/ijamsc.2016.41002
Elad Y, Pertot I (2014) Climate change impacts on plant pathogens and plant diseases. J Crop Improv 28:99–139
Gebily DAS, Ghanem GAM, Ragab MM, Ali AM, Soliman NK, Tawfik HM (2021) Characterization and potential antifungal activities of three Streptomyces spp. as biocontrol agents against Sclerotinia sclerotiorum (Lib.) de Bary infecting green bean. Egypt J Biol Pest Control 31:33. https://doi.org/10.1186/s41938-021-00373-x
Gopalakrishnan S, Srinivas V, Alekhya G, Prakash B (2015) Effect of plant growth-promoting Streptomyces sp. on growth promotion and grain yield in chickpea (Cicer arietinum L.). 3 Biotech. https://doi.org/10.1007/s13205-015-0283-8
Guo Q, Shi M, Chen L, Zhou J, Zhang L, Li Y, Xue Q, Lai H (2020) The biocontrol agent Streptomyces pactum increases Pseudomonas koreensis populations in the rhizosphere by enhancing chemotaxis and biofilm formation. Soil Biol Biochem 144:107755. https://doi.org/10.1016/j.soilbio.2020.107755
Harir M, Bendif H, Bellahcene M, Zohra-Fortas M, Pogni R (2018) Streptomyces secondary metabolites. In: Enany S (ed) Basic biology and applications of actinobacteria. IntechOpen, London, pp 99–122. https://doi.org/10.5772/intechopen.79890
Kaari M, Joseph J, Manikkam R, Ayswarya S, Gopikrishnan V (2022) Biological control of Streptomyces sp. UT4A49 to suppress tomato bacterial wilt disease and its metabolite profiling. J King Saud Univ Sci 34:101688
Katarzyna JK, Aleksandra RM, Adam G, Joannam Z, Jolanta S (2018) Secondary metabolites of actinomycetes and their antibacterial, antifungal and antiviral properties. Pol J Microbiol 67(3):259–272
Le KD, Yu NH, Park AR, Park DJ, Kim CJ, Kim JC (2022) Streptomyces sp. AN090126 as a biocontrol agent against bacterial and fungal plant diseases. Microorganisms 10:791. https://doi.org/10.3390/microorganisms10040791
Li Y, Guo Q, Li Y, Sun Y, Xue Q, Lai H (2019) Streptomyces pactum Act12 controls Tomato yellow leaf curl virus disease and alters rhizosphere microbial communities. Biol Fertil Soils 55:149–169. https://doi.org/10.1007/s00374-019-01339-w.Issue1
Liu S, Ruan W, Li J, Xu H, Wang J, Gao Y, Jingguo W (2008) Biological control of phytopathogenic fungi by fatty acids. Mycopathologia. https://doi.org/10.1007/s11046-008-9124-1
Memic M, Vrtacnik M, Boh M, Pohlevenc F, Mahmutovi O (2017) Biodegradation of PAHs by ligninolytic fungi Hypoxylon fragiforme and Coniophora puteana. Polycycl Aromat Compd. https://doi.org/10.1080/10406638.2017.1392326
Muiru WM, Mutitu EW, Mukunya DM (2007) Characterization of antibiotic metabolites from actinomycete isolates. In: African crop science conference proceedings, vol 8, pp 2103–2107
Mun BG, Lee WH, Kang SM, Lee S, Lee SM, Lee DY, Shahid M, Yun BW, Lee IJ (2020) Streptomyces sp. LH 4 promotes plant growth and resistance against Sclerotinia sclerotiorum in cucumber via modulation of enzymatic and defense pathways. Plant Soil 448:87–103. https://doi.org/10.1007/s11104-019-04411-4
Myo EM, Ge B, Ma J, Cui H, Liu B, Shi L, Jiang M, Zhang K (2019) Indole-3-acetic acid production by Streptomyces fradiae NKZ-259 and its formulation to enhance plant growth. BMC Microbiol 19(1):155. https://doi.org/10.1186/s12866-019-1528-1
Nithya M, Ponmurugan P, Gnanamangai BM, Robinson JP, Narayanasamy M, Senthilkumar J (2020) Evaluation of different native Streptomyces spp. for effective management of rhizome rot of turmeric. J Appl Bot Food Qual 93:225–233. https://doi.org/10.5073/JABFQ.2020.093.027
Parthasarathi S, Sathya S, Sathya G, Bupesh R, Durai-Samy M, Ram-Mohan G, Selva Kumar M, Manikandan C, Kim J, Balakrishnan K (2012) Isolation and characterization of antimicrobial compound from marine Streptomyces hygroscopicus BDUS 49. World J Fish Mar Sci 4(3):268–277. https://doi.org/10.5829/idosi.wjfms.2012.04.03.5658
Rehan M, Alsohim A, Abidou H, Rasheed Z, Abdulmonem W (2021) Isolation, identification, biocontrol activity, and plant growth-promoting capability of a superior Streptomyces tricolor Strain HM10. Pol J Microbiol 70:245–256. https://doi.org/10.33073/pjm-023
Sholkamy EN, Muthukrishnan P, Abdel-Raouf N, Nandhini X, Ibraheem BM, Ibraheem D, Mostafa AA (2020) Antimicrobial and antinematicidal metabolites from Streptomyces cuspidosporus strain SA4 against selected pathogenic bacteria, fungi and nematode. Saudi J Biol Sci 27:3208–3220
Simons TJ, Ross AF (1971) Changes in metabolism associated with enclosed systemic resistance to tobacco. Phytopathology 61:1261–1265
Spss (2008) Statistical package for social science. Version 17.0.0. Spss Corporation, Chicago
Vurukonda SKP, Giovanardi D, Stefani E (2018) Plant growth-promoting and biocontrol activity of Streptomyces spp. as endophytes. Int J Mol Sci 19:952. https://doi.org/10.3390/ijms19040952
Wang M, Xue J, Ma J, Feng X, Ying H, Xu H (2020) Streptomyces lydicus M01 regulates soil microbial community and alleviates foliar disease caused by Alternaria alternata on cucumbers. Front Microbiol 11:942. https://doi.org/10.3389/fmicb.2020.00942
Zambrano EC, Amanda AS, Ortiz AMM (2021) Biocontrol of rice sheath blight with microorganisms obtained in rice cultivated soils. Bragantia 80:e0921. https://doi.org/10.1590/1678-4499.20200356
Acknowledgements
Not applicable.
Funding
The authors have no financial or proprietary interests in any material discussed in this article.
Author information
Authors and Affiliations
Contributions
DG carried out laboratory works and analyses; GG supervisor, conceived, designed, led the study design, followed-up laboratory works and edited the manuscript. AA followed on some laboratory experiments. MR supervised some of the laboratory work. Two authors (NS and TM) are late/passed away. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Consent for publication
All authors consent to participate in publication of these data.
Competing interests
The author declares that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ghanem, G.A.M., Gebily, D.A.S., Ragab, M.M. et al. Efficacy of antifungal substances of three Streptomyces spp. against different plant pathogenic fungi. Egypt J Biol Pest Control 32, 112 (2022). https://doi.org/10.1186/s41938-022-00612-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41938-022-00612-9