Abstract
The last common ancestor of cephalopods and vertebrates lived about 580 million years ago, yet coleoid cephalopods, comprising squid, cuttlefish and octopus, have evolved an extraordinary behavioural repertoire that includes learned behaviour and tool utilization. These animals also developed innovative advanced defence mechanisms such as camouflage and ink release. They have evolved unique life cycles and possess the largest invertebrate nervous systems. Thus, studying coleoid cephalopods provides a unique opportunity to gain insights into the evolution and development of large centralised nervous systems. As non-model species, molecular and genetic tools are still limited. However, significant insights have already been gained to deconvolve embryonic brain development. Even though coleoid cephalopods possess a typical molluscan circumesophageal bauplan for their central nervous system, aspects of its development are reminiscent of processes observed in vertebrates as well, such as long-distance neuronal migration. This review provides an overview of embryonic coleoid cephalopod research focusing on the cellular and molecular aspects of neurogenesis, migration and patterning. Additionally, we summarize recent work on neural cell type diversity in embryonic and hatchling cephalopod brains. We conclude by highlighting gaps in our knowledge and routes for future research.
Similar content being viewed by others
Background
Coleoid cephalopods, i.e. cuttlefish, octopuses, and squids, are protostome invertebrates with large nervous systems, a broad behavioural repertoire and camera-type eyes and belong to the Mollusca [1,2,3]. They have divergent life cycles in line with their habitats that can change over different phases of their lives, and have recently been described as holopelagic (whole life cycle pelagic), holobenthic (entire life cycle benthic), merobenthic (with pelagic paralarval (= direct developing larval) phase before becoming benthic) and meropelagic (with alternating pelagic and benthic phases but no paralarval phase) [4].
The cephalopod nervous system comprises a large centralised brain located between the eyes, a neural cord in each arm, and distributed ganglia in the mantle important for body shape control, and control of gastrointestinal function [2, 5]. The total nervous system of the studied adult coleoid species such as octopus is estimated to account for approximately five hundred million neurons, making it the largest of all invertebrates [5, 6]. This neuronal count exceeds that of the other protostomes and some deuterostomes, and is in the same range as that of a medium-sized mammalian brain such as a ferret brain (Fig. 1) [7]. Their extended behavioural repertoire reflects the complex cognitive abilities they have developed throughout their evolution. They can change their skin colour and texture in milliseconds due to the direct control of their chromatophores by the nervous system. They camouflage as a form of communication and defensive mimicry [8]. They use tools like rocks, shells, and other items to cover their bodies and create a mobile den [8]. They have developed novel morphological features compared to other mollusks, such as a eight sucker-lined arms and in addition to that, for decapods, 2 tentacles [9]. Their large and repetitive genome displays innovations such as significant genome reorganisations and specific gene family expansions, for example, within the protocadherin genes and zinc-finger transcription factors, which are thought to play crucial roles during neural development [10, 11]. In addition, they have evolved extensive RNA editing that endows them with immense neural plasticity and the capacity to adapt to changing environmental conditions[11, 12].
Neural cell count in the nervous system of Bilateria, Cnidaria and Porifera. The total number of neurons in the nervous system (depending on the size of the nervous system and availability of the data) of Bilateria, Cnidaria and Porifera. Bilateria are divided into Protostomia and Deuterostomia. Urbilateria is the last common ancestor between protostomes and deuterostomes. Cephalopods have the highest number of neurons in their nervous system among protostomes. Their neuronal count is even higher than that of some deuterostomes. The mammalian brain is in a neuronal count range similar to the cephalopod nervous system. Porifera has no nervous system. The neuronal count number representatives for the selected groups: Cephalopoda: [5, 6]; Gastropoda: [13]; Annelida: [14, 15]; Platyhelminthes*: [16, 17]; Nematoda: [18]; Arthropoda*: [19]; Mammalia*: [20, 21]; Teleostei*: [22]; Echinodermata: [23]; Cnidaria: [24]; Porifera: [25]. Asterisk (*) symbolises that the number range indicated is for either the brain or the CNS
Coleoid cephalopods have evolved and expanded their central nervous system independently from mammalian brains, which makes them ideal organisms to investigate the mechanisms of large centralised nervous system evolution from an unconventional perspective [26, 27]. Studying neurogenesis and neural patterning in coleoid cephalopods is a starting point to provide insights into this fundamental question.
Neurogenesis is the process of generating neurons from neural stem cells. This process occurs during embryonic development to develop a nervous system, but in many species, continues later in life as adult neurogenesis [28, 29]. Neurogenesis is regulated by cell-intrinsic factors, such as transcription factors, as well as cell-extrinsic factors, such as secreted signalling molecules, that regulate the process in a precise temporospatial manner. First, neural fate is induced in specific ectodermal epithelial areas of the embryo, followed by the proliferation of neural stem cells. In species with larger-sized nervous systems, neuroepithelial stem cells initially proliferate symmetrically to expand the progenitor pool [30]. According to the canonical view, at the onset of neurogenesis, they switch to divide asymmetrically in order to generate neurons or secondary populations of proliferating progenitors [31]. The specification steps that neural progenitor cells follow varies depending on species and neurogenic region [32,33,34,35]. Current evidence from Drosophila neuroblast lineages and retinal progenitors in Xenopus indicate that the fate of the postmitotic daughter cells gets determined at the moment of the last cell division of the progenitors [36,37,38,39,40]. Different neuronal subtypes are consecutively generated from the same multipotent stem cell by a sequence of temporally regulated intrinsic changes in combination with extrinsic factors. In the Drosophila optic lobe, for instance, fate is regulated by the expression of temporally restricted transcription factors, ensuring the timely appearance and organisation of different neuronal subtypes [41, 42]. In the murine cerebral cortex, extrinsic factors from the cerebrospinal fluid as well as feedback factors from the postmitotic neurons regulate sequential generation of different cell types [43, 44]. Timing is also important for the coordination of neurogenesis in the mammalian cortex, where different neuronal layers are formed at specific time windows during corticogenesis. This process is orchestrated by the intrinsic capacity of a gradually depleting multipotent progenitor pool guided by extrinsic cues to generate 8-9 neurons that occupy different laminar positions [45, 46]. Besides time, the spatial location of neurogenic stem cells is an important factor in fate determination. In mammals, pallial and subpallial stem cells give rise to excitatory and inhibitory neurons of the neocortex, respectively [47]. In Drosophila, the ventral nerve cord neuroectoderm is patterned along the anterior-posterior axis in neuromeres that generate different neuronal subtypes [48, 49]. After neurogenesis and fate specification, newborn neurons further differentiate, mature and connect with other neurons. In vertebrate brains, newborn neurons migrate away from the neurogenic niche to other brain areas. During this process, they are guided by axons or by cues that are often also used by growing axons to connect more distant brain regions [50, 51]. In addition, during Drosophila neurogenesis newborn neurons have been observed to migrate into the optic lobe medulla during the pupal phase [52, 53]. Our current knowledge about the process of neurogenesis in species with larger, centralised nervous systems predominantly comes from model species, such as zebrafish, chicken and mouse, and Drosophila [54]. Recent advancements in technological tools, such as next-generation sequencing technologies, molecular biology and biotechnology techniques, and genome editing tools, have led to the investigation of neurogenesis and neural patterning in more species, including coleoid cephalopods [55,56,57]. Here, we summarise recent findings on neurogenesis, the molecular mechanisms responsible for its regulation and neural cell types in the embryonic coleoid cephalopod central nervous system.
Anatomical organisation of the coleoid cephalopod central nervous system
The coleoid cephalopod central nervous system (CNS) makes up one-third of the adult nervous system and comprises more than 30 differentiated lobes and around 12 nerve tracts [5, 58]. Each lobe has a central neuropil which contains mainly neurites and glial cells, and is surrounded by a perikaryal layer of neuronal cell bodies [27, 59]. Anatomically, the cephalopod CNS is located in between the eyes and consists of a circumesophageal central brain comprising supraesophageal (SEM) and subesophageal (SUB) masses that are surrounded by cartilaginous tissue and two periesophageal masses (PEM), which are located on each lateral side of the central brain (example of a 3D cephalopod brain atlas in Montague et al., 2023 [58]).
The SEM, located anteriorly to the esophagus, functions as the higher cognitive and motor center and consists of around 12 lobes (variation among species) organised in two major lobe complexes: vertical lobe complex and basal lobe complex. The vertical lobe complex is responsible for learning and memory and is comparable to the mammalian limbic system [60,61,62,63]. The basal lobe complex is the brain region mediating the higher motor functions involved in the movement of the body parts, and control of chromatophores and papillae [64, 65].
The SUB, located posteriorly to the esophagus, has intermediate and lower motor functions. It controls breathing, feeding and movements involved in defence mechanisms such as inking and camouflage [66]. The SUB comprises 4 lobe complexes made up of 17 lobes: brachial lobe complex, magnocellular lobe complex, palliovisceral lobe complex and pedal lobe complex. The brachial lobe complex is a motor control center for the arms and feeding. The magnocellular lobe complex is responsible for breathing. The palliovisceral lobe complex controls the locomotion. The pedal lobe complex is responsible for the movement of the body parts and is in control of chromatophores and papillae on the mantle and arms [58, 64].
Each PEM is formed from the optic tract complex, which comprises an optic lobe, a peduncle lobe, a dorsolateral lobe, an olfactory lobe and an optic gland. The optic tract complexes are in charge of anything relevant to visual processing, including visual learning and memory, and visio-motor integration [58, 67,68,69,70].
Neurogenesis and embryonic brain development
Embryonic brain growth
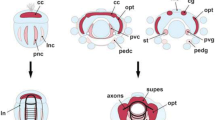
Molluscan nervous systems follow either a ganglionic or cordal pattern of embryonic neurogenesis. Conchiferan molluscs, such as bivalves, gastropods and scaphopods develop their nervous systems based on ganglia: dense, well-characterised clusters of neuronal cell bodies surrounding a centralised neuropil [13, 71,72,73]. In contrast, aculiferan molluscs, such as polyplacophorans, develop a nervous system organised as cords distinguished with a layer of neuronal cell bodies covering longitudinal neuropil [71, 74]. In the 20th and early 21st centuries, researchers hypothesised that coleoid cephalopods also followed a ganglionic pattern of neurogenesis [75,76,77,78]. Contrary to this, recent studies have shown that the coleoid cephalopod brain develops from cords [59, 79]. These studies shown that during early embryogenesis, development of extensive, rope-like neurogenic territories is observed during early allocation of the neurons which is common in cordal origin of neurogenesis, instead of nodular clusters which is typical for a ganglionic origin of neurogenesis [59, 79] (Fig. 2). This cordal patterning of neurogenesis is also observed in nautiloid embryos, indicating that multiple cord-based neurogenesis is a conserved origin across Cephalopoda [59, 80].
Overview of the expression of neurogenic genes in the cephalopod brain throughout the embryogenesis. A., B., D.-F. Schematic depiction of the expression domains of Ov-ngn (purple), Ov-ascl1 (green), Ov-neuroD (red) and Ov-elav (pink) from early organogenesis to late pre-hatching phases on horizontal cephalopod (O. vulgaris) brain sections. The larger solid color domains indicate high expression level of the specific gene in the majority of the cells located in that region. Points indicate either lower expression in the general region or high expression levels in a few cells. The orange arrows during late organogenesis indicate one of the migratory routes taken by the neural progenitor cells generated in the lateral lips, migrating through the posterior transition zone and entering the brain (based on the lineage tracing experiments). As the brain develops, the lateral lips diminish in size. C. Schematic illustration of the brain cords in a cephalopod (O. vulgaris) sagittal transection (inside view) during early organogenesis (stage XI). Schematic illustration of the embryo based on the developmental stage (O. vulgaris) for each brain section is provided on the top right. The approximate location and position of the section are delineated with a dashed line on the embryo. Abbreviations: A, anterior; ar, arm; ATZ, anterior transition zone; CC, cerebral cord; es, esophagus; ey, eye; fu, funnel; LL, lateral lips; mo, mouth; OC, optic cord; OL, optic lobe; P, posterior; PC, pedal cord; PTZ, posterior transition zone; PVC, palliovisceral cord; si, sinus ophthalmicus; SEM, supraesophageal mass; SUB, subesophageal mass. The embryonic schemes are based on Deryckere et al., 2020 [81]. Temporospatial patterning of the neurogenic genes and scheme illustrating interconnected nature of central brain cords in panel C are reproduced from Deryckere et al., 2021 [79]
Early histological studies on the embryonic brain development of squids Loligo vulgaris, Sepioteuthis lessoniana, Todarodes pacificus, Idiosepius paradoxus, and the octopod Octopus vulgaris have shown that morphogenesis of coleoid brains is highly similar [75,76,77,78, 82, 83]. The development of the cephalopod brain starts with the emergence of the neurogenic placodes during the epiboly stage of the embryo. The neurogenic cerebral, optic, palliovisceral and pedal placodes arise as ectodermal thickenings in the equatorial plane of the embryo [78]. The cerebral placodes emerge laterally to the mouth, the optic placodes arise anteriorly to the eye primordia, and the pedal and palliovisceral placodes are located anteriorly and posteriorly of the statocyst primordia respectively [78]. In the early stages of organogenesis, these ectodermal placodes are internalised, interconnected and longitudinally elongated to form the cordal anlagen of the circumesophageal cephalopod brain [59, 79]. As the brain develops, the earlier generated neurons mature and form the first neuropil. The formation of commissures in the developing cords leads to the formation of lobe complexes within the circumesophageal masses. The supraesophageal mass is formed from the anteriorly positioned cerebral cords, whereas posteriorly positioned palliovisceral and pedal cords join to develop into the subesophageal mass, and the bilaterally located optic cords grow into optic lobes [79].
Neurogenesis and the brain neurogenic niche
During early organogenesis, after the first cordal structures have been established, the growing embryonic cephalopod brain is predominantly populated by postmitotic, newborn neurons that express the pan-bilaterian neuronal marker embryonic lethal, abnormal vision (elav) and low to no synaptotagmin (a synaptic vesicle protein present in differentiated, mature neurons) [79]. After the primary cords have been established, the cephalopod brain seems to lack dividing neural progenitor cells within the cords. Proliferation, marked by proliferating cell nuclear antigen (pcna) and phosphohistone H3, is observed more in the paraocular area, previously described as the "Kopflappen" [75] then "Anterior Chamber Organ" [78, 84, 85] or and recently renamed to "Lateral lips" [79]. These neural progenitor cell populations have been identified using evolutionarily conserved proneuronal regulatory markers, such as basic helix-loop-helix (bHLH) transcription factors, achaete-scute (asc) and neurogenin (neurog) (Fig. 2) [59, 79]. SoxB1 (sox2), a common marker of early neural progenitors in many species, seems to be expressed beyond the neural progenitor zones, and also in postmitotic neuronal subpopulations, making it a less suitable neural progenitor marker in cephalopods [79, 86]. As neuronal progenitor cells differentiate, they start expressing another conserved bHLH transcription factor known as neuron differentiation marker neuroD [59, 79]. The sequential expression of bHLH transcription factors is highly conserved for neurogenesis throughout Bilateria [79]. neuroD is predominantly expressed in the areas which interconnect the lateral lips (LL) and brain cords anteriorly and posteriorly in a bow-shaped 3D structure, thus called anterior and posterior transition zones, respectively [87]. During embryonic development, the neural progenitor population at the lateral lips progressively gets consumed, and at hatching, only a small remnant is left (Fig. 2) [79]. Since the adult octopus brain is estimated to have a thousand times more neurons than the brain of the hatchling (200 million to 200,000), extensive neurogenesis must occur post-hatching [27]. The post-hatching source of neural progenitor cells and the process of post-hatching neurogenesis, which is responsible for the neural cell count increase and brain lobe development and maturation is still a mystery to be solved.
In contrast to the cephalopod retina [84] and the vertebrate neural tube [88], the lateral lip neurogenic zones are not organised as a pseudostratified neuroepithelium (a single epithelial layer which appears stratified because cell nuclei occupy different apicobasal positions) [89]. The primary, self-renewing progenitors (solely expressing ascl1+) appear more laterally, whereas differentiating postmitotic cells (neuroD+) are intermingled with neural progenitor cells (ascl1+) in the more medial regions, suggesting a different organisation of the neurogenic niche (Figs 2 and 3) [79].
Fate specification and neuronal cell migration in the developing cephalopod brain. A. Fate map of the cephalopod lateral lip region (depiction on a Stage XIII Octopus vulgaris embryo), based on lineage tracing studies in octopus and squid embryos. Abbreviations: SEM: supra-esophageal mass, PEM: peri-esophageal mass; PVL: palliovisceral lobe; PL: pedal lobe. B. Schematic coronal section overview of a developing cephalopod in the organogenesis stage (O. vulgaris – Stage XIII). Green areas express ascl1, neural progenitor marker, red areas depict postmitotic neuronal elav expression, and dark red dots indicate neuroD expression. C. Cell migration. In the retina, neuroepithelial progenitors (brown) display interkinetic nuclear migration (IKNM): in the M phase, nuclei are at the apical surface where they undergo mitosis, generating a postmitotic cell (yellow) and a progenitor. The nucleus of this progenitor migrates to the basal side, where it spends the S phase. The lateral lip (LL) does not have a pseudostratified epithelial structure, and neurogenesis generates postmitotic neurons (yellow) that migrate away to the brain along different trajectories (dashed arrows)
Lineage tracing in Doryteuthis pealeii and Octopus vulgaris has shown that different areas in the lateral lips contribute cells to different regions of the central brain (Fig. 3A, Table1) [79, 84]. These findings indicate that the progenitor zone is patterned and the destination of neurons is determined while the cells are in the lateral lips. Cephalopods thus follow a similar strategy of spatial patterning of the progenitor zone compared to vertebrates. Deryckere et al. also found preliminary evidence for temporal patterning, as the optic lobe cortex cells were generated proportionally more at early stages than the optic lobe medulla cells, and the palliovisceral lobe could only be traced at earlier stages [79]. More in-depth studies are needed to reveal whether specific cell types are born at specific time points during development.
The development and maturation of neurons and processes in the coleoid cephalopod brain during embryogenesis is asynchronous [27, 79, 90]. For instance, at hatching, the optic lobes have a structure with a clearly laminated cortex and a cauliflower-organised medulla reminiscent of the adult optic lobe, while other brain lobes are far less developed than their adult counterpart [75, 78, 79, 91, 92]. Moreover, the cell density and the size of the brain lobes at the end of embryonic neurogenesis also differ across different coleoid species. These variations are linked to the species-specific differences in the life cycle, the embryonic developmental time, egg size, and behaviour repertoire that hatchlings need to possess to survive post-hatching. Thus, relative brain maturity at hatching differs between cephalopod species. For example, many of the brain lobes in Todarodes pacificus are immature before hatching, and the hatchlings are known to be suspension feeders and not active predators [76]. At what embryonic stage coordinated neuronal activity arises in the cephalopod brain is also unclear. As an example, a holobenthic cephalopod species, Sepia officinalis, shows distinctive signs of visual learning related to food imprinting at least a week before hatching, indicating this axis is already operational during embryonic development [93, 94], whereas another holobenthic cephalopod species Octopus berrima does not [95].
Neuronal migration
Since neurons in the cephalopod brain are not born locally, it was hypothesised that they display migratory behaviour from the lateral lips to their final destination in the brain. Migratory behavior of cephalopod cells was first observed by Marthy and Aroles in Loligo vulgaris, where cells seemed to migrate out of "oculo-ganglionar complex" explants containing retina, lateral lips and optic lobe tissue [96]. Because the explant contained different tissues, it was difficult to understand where these cells originated, yet they seemed to obtain bipolar and multipolar neuronal shapes [96]. More recent lineage studies using CFDA-SE in Octopus vulgaris embryos labelled cells along a trajectory spanning the lateral lips, posterior transition zone (PTZ) and optic lobe, providing further evidence that after being born, neurons travel over long distances to their final destination [79]. Different routes of migration are delineated by tissue boundaries that arise within the embryo, but molecular guidance cues have not yet been described (Fig. 3B,C). Besides long-distance neuronal migration, nuclear migration has been observed in the cephalopod retina [97]. There, neurogenic progenitors are organised in a typical pseudostratified neuroepithelial layer that generates photoreceptors and supporting cells. Nuclei of neuroepithelial stem cells migrate in a coordinated fashion with the cell cycle, a process known as interkinetic nuclear migration (Fig. 3B,C) [97]. Intriguingly, both long-distance migration and interkinetic nuclear migration are mechanisms occurring during the development of large brain structures such as the mammalian cerebral cortex, suggesting they were adopted independently and might be required for the growth of large nervous systems.
Molecular mechanisms regulating embryonic brain development
The exploration of the cephalopod neurogenic toolkit started over two decades ago by studying the temporospatial expression patterns of specific intrinsic (cytoplasmic or nuclear factors such as transcription factors and post-transcriptional regulators) and extrinsic (components of signaling pathways located outside of cells) factors.
Intrinsic factors
Most of these molecular studies have focused on one specific gene family of transcription factors, examining the temporospatial patterning of the identified orthologs of specific genes throughout embryogenesis. In addition, there have been attempts to investigate the neural regionalisation of the brain by studying either a specific region (anterior/posterior part of the brain) or patterning of the neural axis (for instance, along the mediolateral axis discussed in Buresi et al., 2016 [98]). Since the visual system is seen as an extension of the nervous system, the majority of eye development and visual system studies on cephalopods have also provided information about the expression patterns of the investigated genes in the brain. Almost all these studies have been done on squid species (Idiosepius notoides, Idiosepius paradoxus, Sepia officinalis, Euprymna scolopes, Loligo opalescens, and Doryteuthis pealeii). Considering these conditions and to the best of our knowledge, we attempted to provide tables summarising the spatial and temporal expression pattern of the intrinsic factors expressed in CNS studied in coleoid cephalopods over development (Refer to Table 2 for spatial expression of TFs, Table 3 for temporal expression of TFs).
Among the intrinsic factors, the paired-box and homeodomain families of transcription factors (TFs) are the most extensively studied gene families in coleoid cephalopods [100, 110,111,112, 115].
Within the paired-box genes, Pax6 seems to be a prominent TF involved in the development of the visual and nervous systems in coleoid cephalopods, similar to vertebrates and Drosophila [100, 116, 117]. It is predominantly expressed anteriorly in the cerebral cord (later on SEM) and optic cords (later on PEM) [100]. Furthermore, it is known in vertebrates that Pax6 negatively regulates Shh and is involved in the specification of dorsal identity in the developing nervous system [118,119,120,121,122]. In S. officinalis, it has been shown that the expression patterns of Pax6 and Shh do not overlap since Shh is expressed at the cord borders, whereas Pax6 is expressed in the entire cerebral and optic cords [101]. This could be an indication that Pax6 and Shh also interact with each other in a regulatory way in cephalopods.
Homeodomain genes are known to play a crucial role in the formation and patterning of the central nervous system [123]. Although initial studies found the HOX cluster to be in different scaffolds [10, 124], more recent assemblies found that genes of the HOX cluster are organised on the same chromosome, albeit over a larger genic distance [125]. As expected, their expression marks different brain regions along the A-P and D-V axis, but there seem to be differences between cephalopods and between stages (Table 2, 3) [110, 112, 126]. For the moment, it is still difficult to distil a clear picture, and a clear definition of orthologs and a more systematic approach will be needed before we can conclude whether the observed differences among species are an indication of evolutionary and developmental differences among coleoid cephalopods.
Extrinsic factors
Even though there have been a few studies exploring the involvement of extrinsic factors in coleoid cephalopod embryogenesis (Notch signalling in eye development [84], Hedgehog signalling in the mantle and its coexpression with Pax6 [101, 127], Wnt Pathway in the cephalopod lens [128], Hedgehog, BMP and Wnt Pathways in the limb [129]), only the Notch signalling pathway has clearly been implemented in (retinal) neurogenesis [97]. Notch signalling is known to maintain neural progenitor identity and regulate cell cycle and differentiation in vertebrates and Drosophila. Inhibition of Notch signalling using small molecule inhibitors like DAPT in the squid retina leads to premature cell cycle exit, failure to differentiate into a photoreceptor cell and disorganisation of the retinal layers [84]. In the retina, neuro-epithelial progenitors have to make a binary choice to generate photoreceptor cells that start expressing EphR, or supporting cells that express SoxB1. Upon Notch inhibition by DAPT, EphR expression was increased at the expense of SoxB1, suggesting a fate switch towards photoreceptors. The mature photoreceptor marker rhodopsin was never expressed, indicating a block of cell differentiation. Notch signalling thus acts as the regulator of the cell cycle exit, differentiation and cell fate determination and conserves the progenitor identity in the squid retina [97]. To our knowledge, it is still unclear whether Notch plays similar roles in neurogenesis in the lateral lips. Also, for other morphogen pathways, no information is yet available on the role extrinsic factors play in controlling neurogenesis or neural migration in the brain.
Molecularly defined neural cell types
Neural cell subtypes are defined by morphological, molecular and functional characteristics [130]. While adult neural cell morphologies have been extensively documented [5, 131], for embryonic neuronal cell types in coleoid cephalopods the current characterisation relies mainly on the expression of markers for gene expression, including those indicative of neurotransmitter and -peptide usage. In the studied coleoid cephalopod hatchlings, the most prevalent neurotransmitters present in CNS are glutamate, dopamine and acetylcholine. GABA, serotonin and octopamine-producing cells have also been identified, but they appear less in the cephalopod CNS (Fig. 4) [132, 133].
Neurotransmitters in the brain of the cephalopod hatchling. A Schematic depiction of a horizontal section through a brain of the hatchling. B Spatial depiction of gene expression of tyrosine hydroxylase (th) for dopaminergic neurons, vesicular acetylcholine transporter (vacht) for cholinergic neurons, vesicular glutamate transporter (vglut) for glutamatergic neurons, tyramine beta-hydroxylase (tbh) for octopaminergic neurons, glutamate decarboxylase (gad) for GABAergic neurons and tryptophan hydroxylase 2 (tph2) for serotonergic neurons. Abbreviations: BL: basal lobe; FU: funnel; OL: optic lobe; PL: pedal lobe; ST: statocyst; SVL: subvertical lobe; VL: vertical lobe. Reproduced from Styfhals et al., 2022 [132]
Glutamate is the predominant neurotransmitter, exhibiting a widespread occurrence throughout the entire brain of the hatchling Octopus vulgaris and pre-hatchling Loligo vulgaris. Expression of the glutamate marker, vglut (vesicular glutamate transporter), was observed from the outer granular layer in the optic lobe all the way to the basal lobe (Fig. 4) [132, 133]. Acetylcholine is the second most prominent neurotransmitter, as evidenced by the expression pattern of vacht (vesicular acetylcholine transporter), the corresponding marker. In O. vulgaris, this expression spanned the whole brain with the exception of the optic lobe's outer granular layer and, to a more limited extent, in the SEM (Fig. 4) [132]. Conversely, th (tyrosine hydroxylase), the marker for dopaminergic cells, displayed robust expression in the optic lobe cortex and, to a lesser extent, in the medulla and central brain (Fig. 4) [132]. A dual-transmitter cell type, expressing both dopaminergic and glutamatergic markers, was predominantly situated in the inner granular layer of the optic lobe. A dual dopaminergic and glutamatergic cell type is also observed in the brains of Drosophila larvae and vertebrate embryos [132, 134].
The cells expressing other cephalopod neurotransmitters appear in smaller populations. A lower number of serotonergic cells, marked by the sodium-dependent serotonin transporter sert and tryptophan hydroxylase 2 (tph2), are found in the optic lobe medulla and throughout the central brain which is in line with the embryonic appearance of serotonin (5-HT) in cephalopods [132, 133, 135, 136]. GABAergic neurons represented only a small population of cells spread over different regions of the central brain. In O. vulgaris, the expression of gad (glutamate decarboxylase) indicates the presence of clusters of GABAergic neurons in the SUB. Additionally, GABAergic neurons were observed in the optic lobe medulla [132, 133]. Octopaminergic neurons expressing tbh (tyramine B-hydroxylase) reside in the outer granule layers of the optic lobes in hatchlings and juvenile octopuses, which suggests that cell types in the brain of the hatchling remain present throughout life [137]. Indeed, Songco-Casey et al. discovered a laminated pattern of different cell types throughout the juvenile optic lobe cortex. Several had strong molecular similarities with cell types already present at hatching in Octopus vulgaris. Besides octopaminergic cells, dopaminergic, cholinergic as well as glutamatergic and dual dop/glut cell types were present, suggesting that the optic lobe cellular build-up and cell types seem largely conserved at the molecular level, although the repertoire might still expand after hatching [137]. For a more extensive review of the cephalopod optic lobe cell types and function, we refer readers to [138].
Besides using neurotransmitters, it has become clear that cephalopod brains, like many invertebrates, make use of neuropeptides for wireless neurotransmission and neuromodulation. Neuropeptidergic cell types seem prevalent throughout the brain, and the neuropeptide repertoire seems extended [139, 140]. The most studied neuropeptide is FMRFamide, which appears restricted to the palliovisceral cord in early embryonic development in Idiosepius notoides [141]. During mid organogenesis, it is found in the middle and posterior SUB, the optic lobes and the posterior basal as well as superior buccal lobes in Octopus vulgaris and Argonauta hians and in the superior and inferior buccal lobes in Idiosepius notoides [136, 141]. Furthermore, fmrf expression was observed in the SEM and distributed throughout the optic lobes in Loligo vulgaris [133].
Whereas the picture of neuronal diversity is gaining molecular clarity, the types of glial cells remained less studied. In cephalopods, glial cells in the brain have been suggested to contribute to the blood-brain barrier, and phagocytosis of apoptotic cells [142, 143]. Based on recent transcriptomic analysis, three glial subtypes were identified in brain of O. vulgaris hatchling, localised mainly in the neuropil tissue, but also in between neurons and in an ependymal-like layer surrounding the brain [132]. One of these subtypes displayed the presence of the neurotransmitter GABA, distinguished by the expression of gat1 (GABA transporter 1), while the other subtypes did not display neurotransmitter- or peptide characteristics. However, all glial cells exhibited elevated expression levels of gs2 (glutamine synthetase 2) and eaat1 (excitatory amino acid transporter 1), both involved in glutamate clearance [132]. This aligns with the expression patterns observed in glial cells in Drosophila [144, 145]. In contrast to mammals, where the number of glial cells generally surpasses that of neurons, the brain of the O. vulgaris hatchling exhibits a different ratio, with approximately 10% of all brain cells being glial cells, which is similar to other invertebrate brains [132, 146].
In conclusion, the single-cell atlases of coleoid cephalopods have played a crucial role in elucidating the molecular identities of neural cell subtypes. The observed array of neurotransmitters underscores the intricate and diverse nature of neural cell subtypes in these species, shedding light on the complexity of their neural circuitry. Moreover, a parallel characterisation in the juvenile Octopus bimaculoides corroborates these findings and indicates that neuronal diversity still increases after hatching [137].
Conclusions and future directions
The molecular and cell biological study of neurogenesis, neural migration and patterning in coleoid cephalopods is still in its infancy. Recent studies have revealed that mechanisms known from vertebrate model species, such as the use of pro-neural transcription factors, long-distance neuronal migration, and interkinetic nuclear migration, have also been observed in cephalopods [79, 97]. What remains unclear is the cellular organisation of the periocular neurogenic niche and what factors steer the spatial and temporal patterning. One might expect that neurogenic progenitors generate intermediate progenitors as a means to increase neuronal output, but evidence is still lacking. The picture that emerges from the spatial expression of intrinsic transcription factors is still fragmentary, and focused on a single species, time point or tissue.
The fact that neurons migrate long distances seems to indicate that extrinsic signalling molecules and guidance cues play a role, but these are still unknown. Furthermore, knowledge on other neurogenic zones present in the cephalopod body remains very limited. Whether neurogenesis in the developing arm, or stellate and other ganglia present in the mantle or gastrointestinal tract follow a similar temporospatial patterning is still a mystery.
Many of these aspects will become more clear once a more concerted comparative effort can be made using next-generation sequencing methods and molecular tools that have become available to the cephalopod field [147]. A comprehensive approach that combines single-cell RNA sequencing (scRNA-seq) and in situ hybridisation or spatial transcriptomics in combination with morphological technologies into 2D and 3D browsable atlases might spur new hypotheses [58]. Now that novel methods have been established [148, 149], functional analysis of the transcription factors driving neurogenesis by genome editing will bring new insights into their roles in cell type specification.
Overall, our current knowledge about the cellular and molecular mechanisms involved in coleoid cephalopod neurogenesis barely scratched the surface of how these incredible animals evolved their way of generating neurons and developing the largest invertebrate nervous system. With this review, we aimed to bring together the available knowledge on this topic and raise questions that can help the cephalopod developmental neurobiology field.
Availability of data and materials
Not applicable.
Abbreviations
- A:
-
Anterior
- A. hians :
-
Argonauta hians
- ar:
-
Arm
- asc:
-
Achaete-Scute
- ascl1:
-
Achaete-Scute homolog 1
- ATZ:
-
Anterior Transition Zone
- bHLH:
-
Basic Helix-Loop-Helix transcription factors
- BL:
-
Basal Lobe
- BLC:
-
Basal Lobe Complex
- BrLC:
-
Brachial Lobe Complex
- CC:
-
Cerebral Cord
- CFDA-SE:
-
Carboxyfluorescein Diacetate Succinimidyl Ester
- CNS:
-
Central Nervous System
- D:
-
Dorsal
- D. pealeii (Dp):
-
Doryteuthis pealeii
- Drosophila :
-
Drosophila melanogaster
- E. scolopes (Esc):
-
Euprymna scolopes
- eaat1:
-
Excitatory Amino Acid Transporter 1
- elav:
-
Embryonic Lethal, Abnormal Vision
- es:
-
Esophagus
- ey:
-
Eye
- fu:
-
Funnel
- gad:
-
Glutamate Decarboxylase
- gat1:
-
GABA Transporter 1
- gs2:
-
Glutamine Synthetase 2
- I. notoides (Ino):
-
Idiosepius notoides
- I. paradoxus (Ip):
-
Idiosepius paradoxus
- IGL:
-
Inner Granular Layer
- IKNM:
-
Interkinetic Nuclear Migration
- ISH:
-
in situ Hybridization
- L:
-
Left
- L. opalescens (Lo):
-
Loligo opalescens
- L. vulgaris :
-
Loligo vulgaris
- LL:
-
Lateral Lips
- MCLC:
-
Magnocellular Lobe Complex
- med:
-
Medulla
- mo:
-
Mouth
- neuroD:
-
Neuron Differentiation Marker
- neurog:
-
Neurogenin
- NM:
-
Not Mentioned
- O. berrima :
-
Octopus berrima
- O. bimaculoides :
-
Octopus bimaculoides
- O. vulgaris (Ov):
-
Octopus vulgaris
- OC:
-
Optic Cord
- OGL:
-
Outer Granular Layer
- OL:
-
Optic Lobe
- OTC:
-
Optic Tract Complex
- P:
-
Posterior
- PC:
-
Pedal Cord
- pcna:
-
Proliferating Cell Nuclear Antigen
- PEM:
-
Periesophageal Mass
- PL:
-
Pedal Lobe
- pl:
-
Plexiform Layer
- PLC:
-
Pedal Lobe Complex
- PNS:
-
Peripheral Nervous System
- PTZ:
-
Posterior Transition Zone
- PVC:
-
Palliovisceral Cord
- PVLC:
-
Palliovisceral Lobe Complex
- R:
-
Right; Ref: References
- S. lessoniana :
-
Sepioteuthis lessoniana
- S. officinalis (Sof):
-
Sepia officinalis
- scRNA-seq:
-
Single-Cell RNA Sequencing
- SEM:
-
Supraesophageal Mass
- sert:
-
Sodium-Dependent Serotonin Transporter
- si:
-
Sinus Ophthalmicus
- ST:
-
Statocyst; SUB: Subesophageal Mass
- SVL:
-
Subvertical Lobe
- T. pacificus :
-
Todarodes pacificus
- tbh:
-
Tyramine B-Hydroxylase
- TFs:
-
Transcription Factors
- th:
-
Tyrosine Hydroxylase
- tph2:
-
Tryptophan Hydroxylase 2
- V:
-
Ventral
- vacht:
-
Vesicular Acetylcholine Transporter
- vglut:
-
Vesicular Glutamate Transporter
- VL:
-
Vertical Lobe
- VLC:
-
Vertical Lobe Complex
References
Williamson R, Chrachri A. Cephalopod neural networks. NeuroSignals. 2004;13(1–2):87–98.
Budelmann BU. The cephalopod nervous system: what evolution has made of the molluscan design. Nervous Syst Invertebrates. 1995;72(2):115–38.
Nixon M, Young JZ. The brains and lives of cephalopods. Oxford: Oxford University Press; 2003.
Vidal EAG, Shea EK. Cephalopod ontogeny and life cycle patterns. Front Marine Sci. 2023;10:1–24.
Young JZ. The anatomy of the nervous system of Octopus vulgaris. Oxford: Oxford University Press; 1971.
Young JZ. The number and sizes of nerve cells in Octopus. Proc Zool Soc. 1963;140:229–54.
Jardim-Messeder D, Lambert K, Noctor S, Pestana FM, de Castro Leal ME, Bertelsen MF, et al. Dogs have the most neurons, though not the largest brain: Trade-off between body mass and number of neurons in the cerebral cortex of large carnivoran species. Front Neuroanat. 2017;11:296229.
Hanlon RT, Messenger JB. Cephalopod behaviour. Cambridge University Press; 1996.
Naef A. The Embryonic Development of the Octopodids. In: Die Cephalopoden. 1921. p. 268–447.
Albertin CB, Simakov O, Mitros T, Wang ZY, Pungor JR, Edsinger-Gonzales E, et al. The octopus genome and the evolution of cephalopod neural and morphological novelties. Nature. 2015;524(7564):220–4.
Albertin CB, Medina-Ruiz S, Mitros T, Schmidbaur H, Sanchez G, Wang ZY, et al. Genome and transcriptome mechanisms driving cephalopod evolution. Nat Commun. 2022;13(1):2427.
Rosenthal JJC, Seeburg PH. A-to-I RNA Editing: Effects on Proteins Key to Neural Excitability. Neuron. 2012;74(3):432-439.
Getting PA. Neural Control of Behavior in Gastropods. In: The Mollusca. 1985.
Subaraja M, Vanisree AJ. Counter effects of Asiaticosids-D through putative neurotransmission on rotenone induced cerebral ganglionic injury in Lumbricus terrestris. IBRO Reports. 2019;6:160-75.
Le Marrec-Croq F, Drago F, Vizioli J, Sautière PE, Lefebvre C. The leech nervous system: A valuable model to study the microglia involvement in regenerative processes. Clin Dev Immunol. 2013; 2013.
Brown DDR, Pearson BJ. A brain unfixed: Unlimited neurogenesis and regeneration of the adult planarian nervous system. Front Neurosci. 2017;11(MAY).
Quiroga SY, Carolina Bonilla E, Marcela Bolaños D, Carbayo F, Litvaitis MK, Brown FD. Evolution of flatworm central nervous systems: Insights from polyclads. Genet Mol Biol. 2015;38(3):233–48.
Hobert O. Neurogenesis in the nematode Caenorhabditis elegans. WormBook: the online review of C. elegans biology. 2010;1-24.
Pfeffer S, Wolf H. Arthropod spatial cognition. Anim Cogn. 2020;23:1041–9.
Naumann RK, Anjum F, Roth-Alpermann C, Brecht M. Cytoarchitecture, areas, and neuron numbers of the Etruscan Shrew cortex. J Comp Neurol. 2012;520(11):2512-30.
Naumann RK. Even the smallest mammalian brain has yet to reveal its secrets. Brain Behav Evol. 2015;85:1-3.
Zupanc GKH, Sîrbulescu RF. Adult neurogenesis and neuronal regeneration in the central nervous system of teleost fish. Eur J Neurosci. 2011;34(6):917–29.
Stiefel KM, Barrett GA. Sea urchins as an inspiration for robotic designs. J Marine Sci Eng. 2018;6:112.
Sprecher SG. Neural Cell Type Diversity in Cnidaria. Front Neurosci. 2022; 16.
Leys SP, Farrar N. Porifera. In: Structure and Evolution of Invertebrate Nervous Systems. 2016.
Yoshida MA, Ogura A, Ikeo K, Shigeno S, Moritaki T, Winters GC, et al. Molecular evidence for convergence and parallelism in evolution of complex brains of cephalopod molluscs: insights from visual systems. Integr Comp Biol. 2015;55(6):1070–83.
Shigeno S, Andrews PLR, Ponte G, Fiorito G. Cephalopod brains: An overview of current knowledge to facilitate comparison with vertebrates. Front Physiol. 2018;9(JUL):1–16.
Cayre M, Malaterre J, Scotto-Lomassese S, Strambi C, Strambi A. The common properties of neurogenesis in the adult brain: From invertebrates to vertebrates. In: Comparative Biochemistry and Physiology - B Biochemistry and Molecular Biology. 2002.
Chapouton P, Jagasia R, Bally-Cuif L. Adult neurogenesis in non-mammalian vertebrates. BioEssays. 2007;29:745-57.
Ohtsuka T, Kageyama R. Regulation of temporal properties of neural stem cells and transition timing of neurogenesis and gliogenesis during mammalian neocortical development. Semin Cell Dev Biol. 2019;95:4-11.
Oberst P, Agirman G, Jabaudon D. Principles of progenitor temporal patterning in the developing invertebrate and vertebrate nervous system. Curr Opin Neurobiol. 2019;56:185–93. https://doi.org/10.1016/j.conb.2019.03.004.
Guillemot F. Spatial and temporal specification of neural fates by transcription factor codes. Development. 2007;134:3771–80.
Zhang X, Leavey P, Appel H, Makrides N, Blackshaw S. Molecular mechanisms controlling vertebrate retinal patterning, neurogenesis, and cell fate specification. Trends Genet. 2023;39:736-57.
Holguera I, Desplan C. Neuronal specification in space and time. Science. 2018;362(6411):176-80.
Kohwi M, Doe CQ. Temporal fate specification and neural progenitor competence during development. Nat Rev Neurosci. 2013;14(12):823-38.
Cremisi F, Philpott A, Ohnuma SI. Cell cycle and cell fate interactions in neural development. Curr Opin Neurobiol. 2003;13(1):26-33.
Isshiki T, Pearson B, Holbrook S, Doe CQ. Drosophila neuroblasts sequentially express transcription factors which specify the temporal identity of their neuronal progeny. Cell. 2001;106(4):511-21.
Brody T, Odenwald WF. Cellular diversity in the developing nervous system: A temporal view from Drosophila. Development. 2002;129(16):3763-70.
Kay JN, Finger-Baier KC, Roeser T, Staub W, Baier H. Retinal ganglion cell genesis requires lakritz, a zebrafish atonal homolog. Neuron. 2001;30(3):725-36.
Ohnuma SI, Hopper S, Wang KC, Philpott A, Harris WA. Co-ordinating retinal histogenesis: Early cell cycle exit enhances early cell fate determination in the Xenopus retina. Development. 2002;129(10):2435-46.
Konstantinides N, Holguera I, Rossi AM, Escobar A, Dudragne L, Chen YC, et al. A complete temporal transcription factor series in the fly visual system. Nature. 2022;604(7905):316-22.
Doe CQ. Temporal patterning in the drosophila CNS. Annu Rev Cell Dev Biol Annu Rev Cell Dev Biol. 2017;33:219–40.
Seuntjens E, Nave K-A, Goebbels S, Nityanandam A, Huylebroeck D, Debruyn J, et al. Sip1 regulates sequential fate decisions by feedback signaling from postmitotic neurons to progenitors. Nat Neurosci. 2009;12(11):1373–80.
Zappaterra MW, Lehtinen MK. The cerebrospinal fluid: Regulator of neurogenesis, behavior, and beyond. Cell Mol Life Sci. 2012;69(17):2863-78.
Gao P, Postiglione MP, Krieger TG, Hernandez L, Wang C, Han Z, et al. Deterministic progenitor behavior and unitary production of neurons in the neocortex. Cell. 2014;159(4):775-88.
Vaid S, Huttner WB. Progenitor-based cell biological aspects of neocortex development and evolution. Front Cell Dev Biol. 2022;10:892922.
Taverna E, Götz M, Huttner WB. The cell biology of neurogenesis: toward an understanding of the development and evolution of the neocortex. Annu Rev Cell Dev Biol. 2014;30:465–502.
Schmid A, Chiba A, Doe CQ. Clonal analysis of Drosophila embryonic neuroblasts: Neural cell types, axon projections and muscle targets. Development. 1999;126(21):4653-89.
Bossing T, Udolph G, Doe CQ, Technau GM. The embryonic central nervous system lineages of Drosophila melanogaster. I. Neuroblast lineages derived from the ventral half of the neuroectoderm. Dev Biol. 1996;179(1):41-64.
Kriegstein AR, Noctor SC. Patterns of neuronal migration in the embryonic cortex. Trends Neurosci. 2004;27(7):392-9.
Nadarajah B, Parnavelas JG. Modes of neuronal migration in the developing cerebral cortex. Nat Rev Neurosci. 2002;3(6):423-32.
Malin J, Desplan C. Neural specification, targeting, and circuit formation during visual system assembly. Proc Natl Acad Sci USA. 2021;118(28):e2101823118.
Hasegawa E, Kitada Y, Kaido M, Takayama R, Awasaki T, Tabata T, et al. Concentric zones, cell migration and neuronal circuits in the Drosophila visual center. Development. 2011;138(5):983-93.
Mira H, Morante J. Neurogenesis From Embryo to Adult – Lessons From Flies and Mice. Front Cell Dev Biol. 2020;8:533.
Park ST, Kim J. Trends in next-generation sequencing and a new era for whole genome sequencing. Int Neurourol J. 2016;20(Suppl 2):S76-83.
van der Oost J, Patinios C. The genome editing revolution. Trends Biotechnol. 2023;41(3):396-409.
Aldridge S, Teichmann SA. Single cell transcriptomics comes of age. Nat Commun. 2020;11(1):4307.
Montague TG, Rieth IJ, Gjerswold-Selleck S, Garcia-Rosales D, Aneja S, Elkis D, et al. A brain atlas for the camouflaging dwarf cuttlefish Sepia bandensis. Curr Biol. 2023;33(13):2794-2801.e3.
Shigeno S, Parnaik R, Albertin CB, Ragsdale CW. Evidence for a cordal, not ganglionic, pattern of cephalopod brain neurogenesis. Zool Lett. 2015;1(1):1–13.
Young JZ. Computation in the learning system of cephalopods. Biol Bull. 1991;180(2):200–8.
Shomrat T, Turchetti-Maia AL, Stern-Mentch N, Basil JA, Hochner B. The vertical lobe of cephalopods: an attractive brain structure for understanding the evolution of advanced learning and memory systems. J Comp Physiol A Neuroethol Sensory Neural Behav Physiol. 2015;201(9):947–56.
Koizumi M, Shigeno S, Mizunami M, Tanaka NK. Three-dimensional brain atlas of pygmy squid, Idiosepius paradoxus, revealing the largest relative vertical lobe system volume among the cephalopods. J Comp Neurol. 2016;524(10):2142–57.
Young JZ. The Nervous System of Loligo: V. The Vertical Lobe Complex. R Soc London Philos Trans Ser B. 1979;285:311–54.
Boycott B. The functional organization of the brain of the cuttlefish Sepia officinalis. Proc R Soc London Ser B Biol Sci. 1961;153(953):503–34.
Young JZ. The nervous system of Loligo. III. Higher motor centres: the basal supraoesophageal lobes. Philos Trans R Soc London B, Biol Sci. 1977;276(948):351-98.
Young JZ. The nervous system of Loligo. II. Suboesophageal centres. Philos Trans R Soc Lond B Biol Sci. 1976;274(930):101-67.
Young JZ. The central nervous system of Loligo. I. The optic lobe. Philos Trans R Soc Lond B Biol Sci. 1974;267(885):263-302.
Young JZ. The nervous system of Loligo IV. The peduncle and olfactory lobes. Philos Trans R Soc London B Biol Sci. 1979;285(1008):275-309.
Messenger JB. The effects on locomotion of lesions to the visuo-motor system in octopus. Proc R Soc London Ser B Biol Sci. 1967;167(8):252-81.
Messenger JB. The peduncle lobe: a visuo-motor centre in octopus. Proc R Soc London Ser B Biol Sci. 1967;167(8):225-51.
Richter S, Loesel R, Purschke G, Schmidt-Rhaesa A, Scholtz G, Stach T, et al. Invertebrate neurophylogeny: suggested terms and definitions for a neuroanatomical glossary. Front Zool. 2010;7:29.
Wright WG. Behavior and its Neural Control in Gastropod Molluscs. Comp Biochem Physiol Part A Mol Integr Physiol. 2003;136(3):969–72.
Page LR. Developmental analysis reveals labial and subradular ganglia and the primary framework of the nervous system in nudibranch gastropods. J Neurobiol. 1993;24(11):1443-59.
Nezlin LP, Voronezhskaya EE, Tyurin SA. Neuronal development in larval chiton Ischnochiton hakodadensis (Mollusca: Polyplacophora). J Comp Neurol. 2002;444(1):25-38.
Marquis VF. Die Embryonalentwicklung des Nervensystem von Octopus vulgaris Lam. (Cephalopoda, Octopoda), eine histologische Analyse. Verhandlungen der Naturforschenden Gesellschaft Basel. 1989;99(1):23–75.
Shigeno S, Kidokoro H, Tsuchiya K, Segawa S, Yamamoto M. Development of the brain in the oegopsid squid, Todarodes pacificus: an atlas up to the hatching stage. Zoolog Sci. 2001;18(4):527–41.
Shigeno S, Tsuchiya K, Segawa S. Embryonic and paralarval development of the central nervous system of the loliginid squid Sepioteuthis lessoniana. J Comp Neurol. 2001;437(4):449–75.
Yamamoto M, Shimazaki Y, Shigeno S. Atlas of the embryonic brain in the pygmy squid Idiosepius paradoxus. Zoolog Sci. 2003;20(2):163–79.
Deryckere A, Styfhals R, Elagoz AM, Maes GE, Seuntjens E. Identification of neural progenitor cells and their progeny reveals long distance migration in the developing octopus brain. Elife. 2021;10:e69161.
Young JZ. The central nervous system of Nautilus. Philos Trans R Soc Lond B Biol Sci. 1965;249(754):1-25.
Deryckere A, Styfhals R, Vidal EAG, Almansa E, Seuntjens E. A practical staging atlas to study embryonic development of Octopus vulgaris under controlled laboratory conditions. BMC Dev Biol. 2020;20(1):1–18.
Meister G. Organogenese von Loligo vulgaris Lam. Zool Jahrb für Anat. 1972;89:247–300.
Butler-Struben HM, Brophy SM, Johnson NA, Crook RJ. In vivo recording of neural and behavioral correlates of anesthesia induction, reversal, and euthanasia in cephalopod molluscs. Front Physiol. 2018;9:1–18.
Koenig KM, Sun P, Meyer E, Gross JM. Eye development and photoreceptor differentiation in the cephalopod Doryteuthis pealeii. Development. 2016;143(17):3168–81.
Shigeno S, Kidokoro H, Goto T, Tsuchiya K, Segawa S. Early ontogeny of the Japanese Common Squid Todarodes pacificus (Cephalopoda, Ommastrephidae) with special reference to its characteristic morphology and ecological significance. Zoolog Sci. 2001;18(7):1011-26.
Focareta L, Cole AG. Analyses of Sox-B and Sox-E family genes in the cephalopod sepia officinalis: revealing the conserved and the unusual. PLoS One. 2016;11(6):e0157821.
Elagoz AM, Styfhals R, Maccuro S, Masin L, Moons L, Seuntjens E. Optimization of whole mount RNA multiplexed in situ hybridization chain reaction with immunohistochemistry, clearing and imaging to visualize octopus embryonic neurogenesis. Front Physiol. 2022;13:1–12.
Paridaen JT, Huttner WB. Neurogenesis during development of the vertebrate central nervous system. EMBO Rep. 2014;15(4):351–64.
Mescher A. Junqueira’s Basic Histology: Text and Atlas. 13th ed. McGraw Hill; 2013.
Buresi A, Canali E, Bonnaud L, Baratte S. Delayed and asynchronous ganglionic maturation during cephalopod neurogenesis as evidenced by Sof-elav1 expression in embryos of Sepia officinalis (Mollusca, Cephalopoda). J Comp Neurol. 2013;521(7):1482–96.
Shigeno S, Yamamoto M. Organization of the nervous system in the pygmy cuttlefish, Idiosepius paradoxus Ortmann (Idiosepiidae, Cephalopoda). J Morphol. 2002;254(1):65–80.
Shigeno S, Kidokoro H, Tsuchiya K, Segawa S, Yamamoto M. Development of the brain in the oegopsid squid, todarodes pacificus: an atlas from hatchling to juvenile. Zoolog Sci. 2001;18(8):1081–96.
Romagny S, Darmaillacq AS, Guibé M, Bellanger C, Dickel L. Feel, smell and see in an egg: emergence of perception and learning in an immature invertebrate, the cuttlefish embryo. J Exp Biol. 2012;215(23):4125–30.
Darmaillacq AS, Lesimple C, Dickel L. Embryonic visual learning in the cuttlefish, Sepia officinalis. Anim Behav. 2008;76(1):131-4.
Hua QQH, Nande M, Doubleday ZA, Gillanders BM. Innate response to first feeding in Octopus berrima hatchlings despite embryonic food imprinting. Behaviour. 2023;160(7):635-60.
Marthy HJ, Aroles L. In vitro culture of embryonic organ and tissue fragments of the squid Loligo vulgaris with special reference to the establishment of a long term culture of ganglion-derived nerve cells. Zool Jahrbücher Abteilung für Allg Zool und Physiol der Tiere. 1987;91(2):189–202.
Napoli FR, Daly CM, Neal S, McCulloch KJ, Zaloga AR, Liu A, et al. Cephalopod retinal development shows vertebrate-like mechanisms of neurogenesis. Curr Biol. 2022;32(23):5045-5056.e3.
Buresi A, Andouche A, Navet S, Bassaglia Y, Bonnaud-Ponticelli L, Baratte S. Nervous system development in cephalopods: How egg yolk-richness modifies the topology of the mediolateral patterning system. Dev Biol. 2016;415(1):143–56.
Wollesen T, Rodríguez Monje SV, Todt C, Degnan BM, Wanninger A. Ancestral role of Pax2/5/8 in molluscan brain and multimodal sensory system development. BMC Evol Biol. 2015;15:231.
Navet S, Buresi A, Baratte S, Andouche A, Bonnaud-Ponticelli L, Bassaglia Y. The Pax gene family: Highlights from cephalopods. PLoS One. 2017;12(3):1–23.
Navet S, Andouche A, Baratte S, Bonnaud L. Shh and Pax6 have unconventional expression patterns in embryonic morphogenesis in Sepia officinalis (Cephalopoda). Gene Expr Patterns. 2009;9(7):461–7.
Navet S, Baratte S, Bassaglia Y, Andouche A, Buresi A, Bonnaud L. Neurogenesis in cephalopods: “Eco-evo-devo” approach in the cuttlefish sepia officinalis (mollusca-cephalopoda). J Mar Sci Technol. 2014;22(1):15–24.
Hartmann B, Lee PN, Kang YY, Tomarev S, de Couet HG, Callaerts P. Pax6 in the sepiolid squid Euprymna scolopes: evidence for a role in eye, sensory organ and brain development. Mech Dev. 2003;120:177–83.
Tomarev SI, Callaerts P, Kos L, Zinovieva R, Halder G, Gehring W, et al. Squid Pax-6 and eye development. Proc Natl Acad Sci U S A. 1997;94(6):2421–6.
Yoshida MA, Yura K, Ogura A. Cephalopod eye evolution was modulated by the acquisition of Pax-6 splicing variants. Sci Rep. 2014;4:1–6.
Buresi A, Baratte S, Da Silva C, Bonnaud L. orthodenticle/otx ortholog expression in the anterior brain and eyes of Sepia officinalis (Mollusca, Cephalopoda). Gene Expr Patterns. 2012;12(3–4):109–16.
Wollesen T, McDougall C, Degnan BM, Wanninger A. POU genes are expressed during the formation of individual ganglia of the cephalopod central nervous system. Evodevo. 2014;5(1):1–15.
Nomaksteinsky M, Kassabov S, Chettouh Z, Stoeklé HC, Bonnaud L, Fortin G, et al. Ancient origin of somatic and visceral neurons. BMC Biol. 2013;11:1-11.
Wollesen T, Rodríguez Monje SV, McDougall C, Degnan BM, Wanninger A. The ParaHox gene Gsx patterns the apical organ and central nervous system but not the foregut in scaphopod and cephalopod mollusks. Evodevo. 2015;6(1):1–18.
Lee PN, Callaerts P, De Couet HG, Martindale MQ. Cephalopod Hox genes and the origin of morphological novelties. Nature. 2003;424(6952):1061–5.
Focareta L, Sesso S, Cole AG. Characterization of homeobox genes reveals sophisticated regionalization of the central nervous system in the European cuttlefish sepia officinalis. PLoS One. 2014;9(10):e109627.
Barrera Grijalba CC, Rodríguez Monje SV, Gestal C, Wollesen T. Octopod Hox genes and cephalopod plesiomorphies. Sci Rep. 2023;13(1):1–15. https://doi.org/10.1038/s41598-023-42435-0.
McCulloch KJ, Koenig KM. Krüppel-like factor/specificity protein evolution in the Spiralia and the implications for cephalopod visual system novelties: KLF/SP Evolution and the Cephalopod Eye. Proc R Soc B Biol Sci. 1937;2020(287):0–9.
Farfán C, Shigeno S, Nödl MT, De Couet HG. Developmental expression of apterous/Lhx2/9 in the sepiolid squid Euprymna scolopes supports an ancestral role in neural development. Evol Dev. 2009;11(4):354–62.
Baratte S, Andouche A, Bonnaud L. Engrailed in cephalopods: a key gene related to the emergence of morphological novelties. Dev Genes Evol. 2007;217(5):353–62.
Osumi N, Shinohara H, Numayama-Tsuruta K, Maekawa M. Concise Review: Pax6 Transcription Factor Contributes to both Embryonic and Adult Neurogenesis as a Multifunctional Regulator. Stem Cells. 2008;26(7):1663-72.
Callaerts P, Halder G, Gehring WJ. Pax-6 in development and evolution. Ann Rev Neurosci. 1997;20(1):483-532.
Ericson J, Rashbass P, Schedl A, Brenner-Morton S, Kawakami A, Van Heyningen V, et al. Pax6 controls progenitor cell identity and neuronal fate in response to graded Shh signaling. Cell. 1997;90(1):169–80.
Donner AL, Maas RL. Conservation and non-conservation of genetic pathways in eye specification. Int J Dev Biol. 2004;48(8–9):743–53.
Warren N, Caric D, Pratt T, Clausen JA, Asavaritikrai P, Mason JO, et al. The transcription factor, Pax6, is required for cell proliferation and differentiation in the developing cerebral cortex. Cereb Cortex. 1999;9(6):627-35.
Caballero IM, Manuel MN, Molinek M, Quintana-Urzainqui I, Mi D, Shimogori T, et al. Cell-autonomous repression of Shh by transcription factor Pax6 regulates diencephalic patterning by controlling the central diencephalic organizer. Cell Rep. 2014;8(5):1405-18.
Echelard Y, Epstein DJ, St-Jaques B, Shen L, Mohler J, Mcmahon JA, et al. Sonic Hedgehog, a member of a family of putative signaling molecules, is implicated in the regulation of CNS polarity. Cell. 1993;75:1417–30.
Reilly MB, Cros C, Varol E, Yemini E, Hobert O. Unique homeobox codes delineate all the neuron classes of C. elegans. Nature. 2020;584(7822):595-601.
Belcaid M, Casaburi G, McAnulty SJ, Schmidbaur H, Suria AM, Moriano-Gutierrez S, et al. Symbiotic organs shaped by distinct modes of genome evolution in cephalopods. Proc Natl Acad Sci. 2019;201817322.
Da Fonseca RR, Couto A, Machado AM, Brejova B, Albertin CB, Silva F, et al. A draft genome sequence of the elusive giant squid, Architeuthis dux. Gigascience. 2020;9(1):giz152.
Wollesen T, Rodríguez Monje SV, de Oliveira AL, Wanninger A. Staggered Hox expression is more widespread among molluscs than previously appreciated. Proc R Soc B. 2018;285:20181513.
Grimaldi A, Tettamanti G, Acquati F, Bossi E, Guidali ML, Banfi S, et al. A hedgehog homolog is involved in muscle formation and organization of Sepia officinalis (Mollusca) mantle. Dev Dyn. 2008;237(3):659–71.
Neal S, McCulloch KJ, Napoli FR, Daly CM, Coleman JH, Koenig KM. Co-option of the limb Patterning Program in cephalopod eye development. BMC Biol. 2022;20:1-11.
Tarazona OA, Lopez DH, Slota LA, Cohn MJ. Evolution of limb development in cephalopod mollusks. Elife. 2019;8:1–19.
Zeng H, Sanes JR. Neuronal cell-type classification: Challenges, opportunities and the path forward. Nat Rev Neurosci. 2017;18:530-46.
Young JZ. The optic lobes of Octopus vulgaris. Philos Trans R Soc Lond B Biol Sci. 1962;245(718):19-58.
Styfhals R, Zolotarov G, Hulselmans G, Spanier KI, Poovathingal S, Elagoz AM, et al. Cell type diversity in a developing octopus brain. Nat Commun. 2022;13(1):7392.
Duruz J, Sprecher M, Kaldun JC, Al-Soudy AS, Lischer HEL, van Geest G, et al. Molecular characterization of cell types in the squid Loligo vulgaris. Elife. 2023;12:1–27.
Eskenazi D, Malave L, Mingote S, Yetnikoff L, Ztaou S, Velicu V, et al. Dopamine Neurons That Cotransmit Glutamate, From Synapses to Circuits to Behavior. Front Neural Circuits. 2021;15:665386.
Wollesen T, Degnan BM, Wanninger A. Expression of serotonin (5-HT) during CNS development of the cephalopod mollusk, Idiosepius notoides. Cell Tissue Res. 2010;342(2):161-78.
Wollesen T, Sukhsangchan C, Seixas P, Nabhitabhata J, Wanninger A. Analysis of neurotransmitter distribution in brain development of benthic and pelagic octopod cephalopods. J Morphol. 2012;273(7):776-90.
Songco-Casey JO, Coffing GC, Piscopo DM, Pungor JR, Kern AD, Miller AC, et al. Cell types and molecular architecture of the Octopus bimaculoides visual system. Curr Biol. 2022;32(23):5031-44.
Pungor JR, Niell CM. The neural basis of visual processing and behavior in cephalopods. Curr Biol. 2023;33(20):R1106–18.
Zatylny-Gaudin C, Cornet V, Leduc A, Zanuttini B, Corre E, Le Corguillé G, et al. Neuropeptidome of the cephalopod sepia officinalis: identification, tissue mapping, and expression pattern of neuropeptides and neurohormones during egg laying. J Proteome Res. 2016;15(1):48–67.
Wollesen T, Nishiguchi MK, Seixas P, Degnan BM, Wanninger A. The VD1/RPD2 α1-neuropeptide is highly expressed in the brain of cephalopod mollusks. Cell Tissue Res. 2012;348(3):439–52.
Wollesen T, Cummins SF, Degnan BM, Wanninger A. FMRFamide gene and peptide expression during central nervous system development of the cephalopod mollusk, Idiosepius notoides. Evol Dev. 2010;12(2):113-30.
Abbott NJ, Pichon Y. The glial blood-brain barrier of crustacea and cephalopods: a review. Journal de Physiologie. 1987;82:304-13.
Ibrahim G, Luisetto M, Latyshev O. Glial cells in the posterior sub-esophageal mass of the brain in Sepia officinalis (Linnaeus, 1758) (decapodiformes–sepiida): ultrastructure and cytochemical studies. Invertebr Neurosci. 2020;20(4):16.
Avalos CB, Brugmann R, Sprecher SG. Single cell transcriptome atlas of the drosophila larval brain. Elife. 2019;8:e50354.
Davie K, Janssens J, Koldere D, De Waegeneer M, Pech U, Kreft Ł, et al. A single-cell transcriptome atlas of the aging drosophila brain. Cell. 2018;174(4):982-98.
Verkhratsky A, Butt A. The history of the decline and fall of the glial numbers legend. Neuroglia. 2018;1(1):188-92.
Baden T, Briseño J, Coffing G, Cohen-Bodénès S, Courtney A, Dickerson D, et al. Cephalopod-omics: emerging fields and technologies in cephalopod biology. Integr Comp Biol. 2023;63(6):1226-39.
Ahuja N, Hwaun E, Pungor JR, Rafiq R, Nemes S, Sakmar T, et al. Creation of an albino squid line by CRISPR-Cas9 and its application for in vivo functional imaging of neural activity. Curr Biol. 2023;33(13):2774-83.
Crawford K, Diaz Quiroz JF, Koenig KM, Ahuja N, Albertin CB, Rosenthal JJC. Highly efficient knockout of a squid pigmentation gene. Curr Biol. 2020;30(17):3484-3490.e4.
Acknowledgements
The authors would like to thank the members of the Seuntjens and Arckens labs for critical discussions and Maxence Lanoizelet for proofreading the manuscript.
Funding
This work was supported by Research Foundation - Flanders (FWO) (FR/11D4120N to AME, FR/11L1923N to ML, G040124N to ES) and KU Leuven (C14/21/065 to ES, ID-N/20/007 to ES).
Author information
Authors and Affiliations
Contributions
AME wrote the first draft and created Figs. 1 and 2 and all the tables. MVD helped with writing and revising the text. ML contributed to writing and prepared the scheme for Fig. 3A. ES contributed to writing and creating Figs. 3 and 4. AME and ES reviewed and edited the manuscript's content. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Figure 2 is reproduced from Deryckere et al., 2021 [79], and Fig. 4 is reproduced from Styfhals et al., 2022 [132], which are open-access articles distributed under the terms of the Creative Commons CC BY license.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Elagoz, A.M., Van Dijck, M., Lassnig, M. et al. Embryonic development of a centralised brain in coleoid cephalopods. Neural Dev 19, 8 (2024). https://doi.org/10.1186/s13064-024-00186-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13064-024-00186-2