Abstract
Purpose
This study investigated and compared the effects of Gd enhancement on brain tumours with a half-dose of contrast medium at 5.0 T and with a full dose at 3.0 T.
Methods
Twelve subjects diagnosed with brain tumours were included in this study and underwent MRI after contrast agent injection at 3.0 T (full dose) or 5.0 T (half dose) with a 3D T1-weighted gradient echo sequence. The postcontrast images were compared by two independent neuroradiologists in terms of the signal-to-noise ratio (SNR), contrast-to-noise ratio (CNR) and subjective image quality score on a ten-point Likert scale. Quantitative indices and subjective quality ratings were compared with paired Student's t tests, and interreader agreement was assessed with the intraclass correlation coefficient (ICC).
Results
A total of 16 enhanced tumour lesions were detected. The SNR was significantly greater at 5.0 T than at 3.0 T in grey matter, white matter and enhanced lesions (p < 0.001). The CNR was also significantly greater at 5.0 T than at 3.0 T for grey matter/tumour lesions, white matter/tumour lesions, and grey matter/white matter (p < 0.001). Subjective evaluation revealed that the internal structure and outline of the tumour lesions were more clearly displayed with a half-dose at 5.0 T (Likert scale 8.1 ± 0.3 at 3.0 T, 8.9 ± 0.3 at 5.0 T, p < 0.001), and the effects of enhancement in the lesions were comparable to those with a full dose at 3.0 T (7.8 ± 0.3 at 3.0 T, 8.7 ± 0.4 at 5.0 T, p < 0.001). All subjective scores were good to excellent at both 5.0 T and 3.0 T.
Conclusion
Both quantitative and subjective evaluation parameters suggested that half-dose enhanced scanning via 5.0 T MRI might be feasible for meeting clinical diagnostic requirements, as the image quality remains optimal. Enhanced scanning at 5.0 T with a half-dose of contrast agents might benefit patients with conditions that require less intravenous contrast agent, such as renal dysfunction.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Magnetic resonance imaging (MRI) plays a vital role in the diagnosis and characterization of brain tumours due to its high spatial resolution and optimal soft-tissue contrast [1]. However, the key challenge in detecting tumours at an early stage by conventional MRI is its low sensitivity [2]. Therefore, various contrast agents have been developed to improve the sensitivity of MRI [3]. Gadolinium (Gd)-based contrast agents, including Gd-DTPA (Magnevist®), Gd-DO3A-butrol (Gadovist®) and Gd-EOB-DTPA (Primovist®), are the most commonly used contrast agents in clinical practice. Following intravenous administration, Gd-based contrast agents may increase MRI sensitivity for the detection of brain tumours [2, 3]. However, Gd3 + chelates still have low toxicity. The risk of nephrogenic systemic fibrosis (NSF) for patients with impaired renal function and long-term adverse effects due to Gd brain deposition has raised concerns [4,5,6], especially for patients with brain diseases that require longitudinal monitoring after treatment, such as low-grade gliomas or brain metastases. In diagnosing and treating most brain tumours, multiple gadolinium contrast agent-enhanced scans are required to determine the tumour grade, progression, and prognosis during follow-up. Furthermore, the effects of gadolinium deposits in the brain are still unknown; therefore, doses should be kept as low as possible to prevent gadolinium buildup [7]. Thus, designing new Gd3 + chelates and using lower doses of contrast agents have always been popular research topics [2, 8, 9].
The development of high- and ultrahigh-field MRI scanners (≥ 3.0 Tesla (T)) offers the possibility for using fewer contrast agents in the clinic [9,10,11]. Many researchers have conducted comparative studies on injection doses at 1.5 T versus 3.0 T and 3.0 T versus 7.0 T and have shown the feasibility of injecting a reduced amount of contrast agents during higher-field MRI for brain tumours [11, 12]. However, whole-body imaging at 7.0 T has not yet been approved by the Food and Drug Administration (FDA) and is not available in the clinic due to safety issues such as increased specific absorption rate (SAR) distributions in the body and technical issues such as B1 field inhomogeneity [13, 14]. Recently, a 5.0 T clinical MRI scanner was developed that can be used to scan the whole body with good image homogeneity and contrast uniformity while avoiding issues such as a high SAR [15]. Many basic MRI applications could benefit from the increased signal intensity (SI), contrast and spatial resolution of 5.0 T systems. However, the feasibility and image quality of lower-dose contrast-enhanced scanning with 5.0 T systems have not been investigated or compared with those of standard full-dose contrast-enhanced scanning with 3.0 T systems.
This study aimed to compare the enhancing effects of half-dose enhanced scanning at 5.0 T and full-dose enhanced scanning at 3.0 T in brain tumours. The enhancement effects were assessed using quantitative indices, including the signal-to-noise ratio (SNR) and contrast-to-noise ratio (CNR), differences in SI before and after enhanced scans, as well as subjective image quality scores. Then, the feasibility of using half-dose contrast agents on 5.0 T MRI for brain tumour diagnosis was evaluated.
Materials and methods
Patient characteristics
This prospective study was approved by the ethics committee of our hospital (approval no. 2021110). Between 11/2021 and 01/2023, patients suspected of having brain tumours (gliomas, meningiomas or brain metastases) were enrolled, and all participants signed an informed consent form. The inclusion criteria were as follows: 1. 18–80 years old; 2. suspected brain tumours; 3. no safety-related contraindications to undergoing magnetic resonance imaging (MRI) or receiving intravenous contrast agent; and 4. glomerular filtration rate (GFR) > 60 mL/min. The exclusion criterion was poor image quality so that subjective evaluation and image analysis could not be carried out. The participants included in this study underwent both 3.0 T and 5.0 T MRI scans (time interval between the two scans > = 24 h). The study flowchart is shown in Fig. 1. Finally, a total of 12 patients with clinically diagnosed brain tumours were recruited.
Image acquisition
All MR scans were performed on a clinical 3.0 T scanner (uMR 790, United Imaging Healthcare) and a clinical 5.0 T scanner (uMR Jupiter, United Imaging Healthcare), with a 48-channel head coil on 5.0 T and a 24-channel head coil on 3.0 T, respectively. A 3D gradient echo (3D-GRE) sequence was acquired in the sagittal plane and reconstructed in the axial and coronal planes. The axial images were used for analysis. A clinically acceptable acquisition time (approximately three and a half min) was used at both 3.0 T and 5.0 T. Under the constraint of this short scanning duration, the sequence parameters were adjusted and optimized to meet the clinical resolution limits at both 3.0 T (0.9 × 0.9 × 0.9 mm3) and 5.0 T (0.7 × 0.7 × 0.7 mm3) (Table 1). Acceleration techniques such as partial-Fourier k-space filling; compression sensing; and parallel RF transmission (pTx for 5.0 T) and parallel acquisition, which are similar to GRAPPA and SENSE [16], were used for both 3.0 T and 5.0 T imaging.
The sequence parameters at 5.0 T were as follows: Repetition time(TR)/Echo time(TE)/Inversion time(TI) = 8.1/2.6/940 ms; FOV = 220 × 248 mm2; matrix size = 312 × 352; in-plane resolution = 0.70 × 0.70 mm2; slice thickness = 0.7 mm; flip angle = 9°; number of slices = 208; receiver bandwidth = 180 Hz/pixel; acceleration factor = 3.5; averages = 1; and total acquisition duration = 224 s.
The sequence parameters for 3.0 T were as follows: TR/TE/TI = 7.6/3.3/810 ms; FOV = 220 × 244 mm2; matrix size = 245 × 272; in-plane resolution = 0.90 × 0.90 mm2; slice thickness = 0.9 mm; flip angle = 9°; number of slices = 186; receiver bandwidth = 230 Hz/pixel; acceleration factor = 3.0; averages = 1; and total acquisition duration = 204 s.
The contrast medium administration protocol was as follows: all subjects first underwent intravenous gadolinium contrast-enhanced scans under 3.0 T MRI, with an injection dose of 0.10 mmol/kg (full dose). Gd-DO3A-butrol (Gadovist, Bayer Schering Pharma AG) contrast medium was used. Using a high pressure injector (Insight M12, Jusha Display Technology Co., Ltd., Nanjing) that was 5.0T compliant, the contrast medium was automatically injected at 5.0T. Gadolinium-based MRI contrast agents serve as catalysts for water proton relaxation, and their efficacy is quantified by a rate constant referred to as relaxivity [17]. Gadovist is eliminated from plasma with a mean terminal half-life of 1.81 h (range 1.33–2.13 h) [18]. After 24 h, another contrast-enhanced scan was performed at 5.0 T, with an injection dose of 0.05 mmol/kg (half dose).
Image analysis and evaluation
The regions of interest (ROIs) were delineated for each subject at locations with normal grey matter and white matter tissues and enhanced tumour lesions with the interactive software tool ITK-SNAP. The in plane size of ROIs placed on the turmous lesions were around 25 to 100 mm2. The ROIs of grey matter and white matter were drawn on the slice of the corpus callosum. When drawing the ROIs of the tumours, to ensure the comparability of the measurement data, all the ROIs were placed in the same position (grey matter, white matter and lesion) for measurement on 3.0 T and 5.0 T images by one neuroradiologist (Dr. Jiang, who has 10 years of experience). In instances where there was a discrepancy in image quality between the 3.0 T and 5.0 T images, we employed 3D reconstruction postprocessing techniques to align the results and ensure that the delineated regions of interest (ROIs) were consistently positioned. The quantitative indices, including the SNR and CNR, were calculated as follows.
where \({SI}_{tissue}\) is the SI of tissues (grey matter, white matter and tumour lesions), \({SD}_{background}\) is the standard deviation of SI at the four corners of the background, \({SI}_{Lesion}\) is the SI of tumour tissues, and SIg/w is the SI of grey matter or white matter on the contralateral side. All the calculations were completed in MATLAB (2018a, MathWorks), while the open-source toolbox “MRIqual” was used to calculate the SNR (https://github.com/elayden/MRIqual).
We measured the SI of the grey matter, white matter and lesions at 3.0 T and 5.0 T with the same ROI in the T1 image before and after contrast agent injection, subtracted the SI before enhancement from the SI after enhancement, and analysed the changes in SI after enhancement. Concurrently, we assessed the dimensions of the lesions.
The subjective evaluation was performed by two independent neuroradiologists (Dr. Yu, who has 6 years of experience, and Dr. Jiang, who has 10 years of experience). During the evaluation, the MRI scanner information and patient name were anonymized. A ten-point Likert scale was used, and the following parameters were evaluated: lesion-related parameters (contrast agent enhancement, internal structure, and delineation), image quality (grey‒white differentiation and homogeneity), artefacts (motion artefacts, vessel pulsation artefacts, and susceptibility artefacts) and overall sequence quality [11]. All subjective evaluations pertain to the quality of images.
To compare differences in quantitative indices and subjective image quality scores between 3.0 T and 5.0 T scans, paired Student's t tests were conducted. Interreader agreement of the subjective evaluation parameters was assessed by using the intraclass correlation coefficient (ICC) with a two-way random effects model. All the statistical analyses were performed using SPSS 24.0 for Windows. P value less than 0.05 was considered to indicate statistical significance.
Results
Patient characteristics
As shown in Table 2, the mean age of the participants was 53.58 ± 11.65 years (range: 35–74), with 8 males and 4 females. The mean height was 1.67 ± 0.62 m, the mean weight was 64.23 ± 4.91 kg, and the mean body mass index (BMI) was 22.91 ± 1.17 kg/m2. The diagnosis of brain tumours was based on the World Health Organization (WHO) 2016 guidelines [19]. The clinical diagnosis results were as follows: four cases of newly diagnosed meningioma; two cases of newly diagnosed astrocytoma; two cases of suspected brain metastasis (metastatic squamous cell carcinoma of lung origin); one case of anaplastic oligodendroglioma (WHO grade III) recurrence after 7 years of surgery; one case of glioblastoma (WHO grade IV) recurrence 3 years after surgery; one case of left temporal occipital lobe brain tumour (Patient 8, suspected glioblastoma, with nonsurgical treatment); and one case of left occipital brain tumour (Patient 11, suspected glioblastoma, with nonsurgical treatment). A total of 16 lesions were identified across 12 patients.
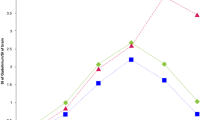
SNR and CNR
A total of 16 enhanced tumour lesions were detected and compared between 3.0 T and 5.0 T images. As shown in Table 3, for tumour lesions, the SNR was 30 ± 8 at 3.0 T and 48 ± 15 at 5.0 T (P < 0.001); for white matter, the SNR was 18 ± 3 at 3.0 T and 18 ± 3 at 5.0 T (P < 0.001); for grey matter, the SNR was 11 ± 2 at 3.0 T and 22 ± 5 at 5.0 T (P < 0.001). The CNR for lesion/white matter was 36 ± 18 at 3.0 T and 69 ± 43 at 5.0 T (P < 0.001); the CNR for lesion/grey matter was 40 ± 17 at 3.0 T and 103 ± 49 at 5.0 T (P < 0.001); and the CNR for grey matter/white matter was 15 ± 2 at 3.0 T and 33 ± 4 at 5.0 T (P < 0.001). The SNR of the tumour lesions was significantly greater with a half-dose at 5.0 T than with a full dose at 3.0 T (Fig. 2).
SI difference between postcontrast and precontrast images
The difference distribution diagram of the SI before and after enhancement is shown in Fig. 3. When comparing the SI differences before and after Gd enhancement scans between 3.0 T and 5.0 T, there was no significant difference in white matter or grey matter (white matter, P = 0.068 ;gray matter, P = 0.115), while the difference was obvious in tumour lesions (P < 0.001) (Table 3).
Distribution of SI differences (postcontrast SI—precontrast SI). In grey matter and white matter, there was no significant difference in SI, and in tumour lesions, the difference in SI in the 5.0 T group was significantly greater than that in the 3.0 T group. The yellow line represents the 25%-75% confidence interval. The yellow line depicts a range within the 1.5 interquartile range. The white dot represents the median
Subjective evaluation
As shown in Table 4, the average observer ratings were classified as very good or excellent for all the evaluation parameters. Images of different types of tumours acquired at 5.0 T all showed significantly better lesion contours and internal structures than those acquired at 3.0 T, as shown in Figs. 4, 5 and 6. Interobserver agreement was excellent, as assessed by the ICC (ICC > = 0.89, p < 0.01) and Pearson’s correlation coefficient (r = 0.80, p < 0.01), as shown in Fig. 7.
Atypical meningioma, WHO Grade II, 35-year-old male. (a1) T1w MRI without contrast enhancement at 3.0 T and (b1) T1w MRI without contrast enhancement at 5.0 T. (a2) and (a3) Two different slices of full-dose enhanced T1w MRI at 3.0 T. (b2) and (b3) Two different slices of half-dose enhanced T1w MRI at 5.0 T. A 5.0 T MRI displayed a clearer boundary and internal structure of the lesion and showed a stronger inhibitory effect than did 3.0 T MRI; at the same time, in the half-dose 5.0 T images, the degree and range of edge enhancement in the lesion were stronger than those in the full-dose 3.0 T images
Brain metastases (lung origin), 64-year-old male. (a1) T1w MRI without contrast enhancement at 3.0 T and (b1) T1w MRI without contrast enhancement at 5.0 T. (a2) Full-dose enhanced T1w MRI at 3.0 T and (b2) half-dose enhanced T1w MRI at 5.0 T. As depicted within the red circle, the augmentation of the lesion parenchyma exhibits a slightly greater intensity at 3.0 T compared to 5.0 T. However, in terms of delineating the intricacies of the lesion, 5.0 T surpasses 3.0 T due to its employment of a thinner scanning layer thickness and higher resolution
Anaplastic oligodendroglioma, WHO Grade III, 49-year-old male, relapsed after surgery in 2014. (a1) T1w MRI without contrast enhancement at 3.0 T and (b1) T1w MRI without contrast enhancement at 5.0 T. (a2) and (a3) Two different slices of full-dose enhanced T1w MRI at 3.0 T. (b2) and (b3) Two different slices of half-dose enhanced T1w MRI at 5.0 T. For lesions with low-level blood‒brain barrier (BBB) leakage (green circle), the enhancement effect was similar between 3.0 T and 5.0 T, but for lesions with high-level BBB leakage (red circle), the enhancement effect with a half-dose at 5.0 T was significantly better than that with a full-dose at 3.0 T
Bland–Altman plot and scatter plot assessing interobserver pairwise agreement for image quality ratings. (a) Bland‒Altman plot. The x-axis represents the mean values, and the y-axis represents the difference between the two. The red dashed line shows the mean bias, and the dashed lines show the 95% CI based on the standard deviation of the distribution. (b) Scatter plot. The x-axis represents the ratings from observer 1, and the y-axis represents the ratings from observer 2
Discussion
The clinical routine for diagnosing brain tumours involves a full-dose contrast-enhanced MRI scan at 3.0 T. In this study, the Gd-based contrast enhancement of brain tumours using a half-dose at 5.0 T and a full-dose at 3.0 T were compared. Both quantitative and subjective evaluation results indicated that 5.0 T MRI with a half-dose of contrast enhancement may be a feasible option to meet the diagnostic requirements in the clinic.
The results indicated that the tumour-to-brain contrast, as reflected by the CNR of lesion/white matter and lesion/grey matter, was significantly greater with a half-dose at 5.0 T than with a full dose at 3.0 T. This finding was consistent with previous studies that compared lesion enhancement between high-field and low-field magnetic resonance imaging (MRI) systems [11]. Moreover, we observed that the CNR increase in some patients and tumour lesions was even greater than the increase in magnetic field strength (i.e., more than 1.7-fold). This might be due to the following two reasons. On the one hand, the effectiveness of the T1-shortening effect of a Gd-based contrast agent increases nonlinearly with the field strength [12]. In vitro experiments indicated that the r1 relativity of Gd-based contrast agents exhibited minimal variations across diverse field strengths [20, 21], and the increased baseline tissue T1 relaxation times at higher fields amplify the relaxation-modifying effect of contrast agents [12]. An increase in tissue T1 values with increasing field strength results in a corresponding increase in relative contrast enhancement. This is due to the combined effects of protein binding, which leads to increased field strength and solvent dependencies, ultimately resulting in notable changes in T1 relaxivity values at higher magnetic field strengths [22]. Prior studies have also shown that the augmentation of channel head coils at elevated field strengths is advantageous for enhancing the signal-to-noise ratio and image resolution of voxels [23, 24]. A 3D-GRE sequence was used in this study, and a similar TR and TE and the same flip angle were set for both 3.0 T and 5.0 T sequences. Therefore, when imaging with similar sequence parameters at both field strengths, grey and white matter may not relax completely and may exhibit a lower-than-expected SI increase at 5.0 T. Since the extensive invasion of glioma may impair the integrity of the blood‒brain barrier (BBB) [25], tumour lesions with more severe BBB disruption would relax almost completely and exhibit high SI at 5.0 T. In addition, a previous study demonstrated that a 3D-GRE sequence was clinically more suitable for detecting brain tumours than other sequences [26]. Hence, the augmented field strength and refined protocols employed in this study resulted in a substantial enhancement in the contrast between tumour tissue and brain tissue at 5.0 T.
In addition to the improved tumour-to-brain contrast, the SNR and CNR of grey matter, white matter, and tumour lesions were significantly greater with half-dose imaging at 5.0 T than with full-dose imaging at 3.0 T. This could be attributed to the greater magnetic field strength (5.0 T vs. 3.0 T) and the greater number of channels of the receiving coils (48 channels at 5.0 T vs. 24 channels at 3.0 T) [27, 28]. Theoretically, a thinner slice or smaller voxel size would result in poorer (lower) SNR and CNR in the same magnetic field due to a decreased amount of aligning protons within the small voxel. The increased SNR and CNR obtained at 5.0 T even with a thinner slice and smaller voxel size indicates the extraordinary benefits of higher magnetic field strength, including clearer images at higher resolutions, which is beneficial for clinical applications [12, 13]. Furthermore, the enhancement of SI at ultrahigh fields, together with the modification of transverse and longitudinal relaxation times, produces enhanced image contrasts that are useful in anatomical MRI applications. We also observed an increase in the grey matter/white matter contrast at 5.0 T versus 3.0 T, which was consistent with previous findings comparing 3.0 T with 1.5 T [29]. A previous study revealed an increased CNR in T1w images at 3.0 T compared with 1.5 T [29]; however, this increase was not found in a previous comparison study of 7.0 T versus 3.0 T [11]. Since B1 field inhomogeneity might influence the CNR [30], this finding at 7.0 T might be because the B1 field inhomogeneity at 7.0 T was more severe than that at 5.0 T and 3.0 T, thus leading to decreased grey matter/white matter contrast [13]. A previous simulation study also revealed that the variation in B1 magnitude was nearly twofold greater at 7.0 T than at 4.0 T [16]. Together with this previous simulation study [31], our findings indicated that 5.0 T might have better B1 field uniformity than 7.0 T and similar uniformity to 3.0 T, thus leading to good grey matter/white matter contrast. Therefore, from a practical perspective, we can conclude that 5.0 T might be superior to 3.0 T in brain tumour imaging, not only because of the greater SNR of brain tissues but also because of better grey matter/white matter contrast than 3.0 T.
One limitation of this study is that only 12 subjects with 16 enhanced lesions were included, and some subjects had only mild BBB leakage. We hypothesized that patients with severe BBB disruption might benefit more from a half-dose at 5.0 T, but the CNR was only slightly improved in lesions with low-level BBB leakage. A previous dynamic contrast-enhanced imaging study suggested that low-level BBB leakage might induce systematic errors in the calculation of measured parameters [32]. In some 3.0 T studies, a double dose is recommended for patients with brain metastases [33]. Thus, our results suggested that the doses of contrast agent might need to be modified for patients with subtle BBB breakdown. In the future, more subjects with different types of brain tumours should be included to increase the generalization of our conclusions. In addition, only one contrast agent (Gadovist) and one type of T1-weighted sequence (GRE 3D) were examined in the current study; thus, our findings might not be applicable to other contrast agents and MRI sequences. However, a previous study suggested that Gadovist is a recommended contrast agent in routine MRI protocols for brain tumours [34], the GRE sequence might be superior to fast spin‒echo sequences [21]. Moreover, we only examined brain tumours, and future studies of tumours in various organs of the body, especially the abdominal area, should be conducted to investigate the full potential of low-dose contrast agent-enhanced MRI on 5.0 T systems. Another limitation of this study is that 3.0 T scanning was performed before 5.0 T scanning for all the subjects due to ethical considerations, and retention of the contrast agent in subsequent scans due to the leakage of contrast agent in tumours would be possible, despite the presence of at least a 24-h gap between the two contrast injection sessions. To rule out this limitation, a precontrast scan was performed to ensure little or no retention of the contrast agent, and the postcontrast images were subtracted from the precontrast images to calculate the SI difference. The precontrast images on the second scan (5.0 T ) showed no significant enhancement. The subtracted SI value was still significantly greater at 5.0 T, as shown in Table 3. Thus, we believe that the cumulative effect of contrast agents on tumour lesions is minimal. In the future, an earlier imaging session of 5.0T may be necessary to support the results of subsequent imaging studies.
Conclusions
In conclusion, the results of this study suggest that administering a half-dose of intravenous contrast agent in conjunction with 5.0 T MRI could yield improved tumour-to-brain contrast and SNR in enhanced tumour lesions compared to the full dose at 3.0 T MRI. These findings indicate the feasibility of using a reduced dose of contrast agent for the diagnosis of brain tumours in high-field MRI systems.
Availability of data and materials
The datasets generated and/or analysed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- MRI:
-
Magnetic resonance imaging
- SNR:
-
Signal-to-noise ratio
- CNR:
-
Contrast-to-noise ratio
- ROI:
-
Region of interest
- BBB:
-
Blood‒brain barrier
- SAR:
-
Specific absorption rate
- SI:
-
Signal intensity
- WHO:
-
World Health Organization
- TR:
-
Repetition time
- TE:
-
Echo time
- TI:
-
Inversion time
- GRE:
-
Gradient echo
References
Young RJ, Knopp EA. Brain MRI: tumor evaluation. J Magn Reson Imaging. 2006;24(4):709–24.
Cheng W, Ping Y, Zhang Y, Chuang KH, Liu Y. Magnetic resonance imaging (MRI) contrast agents for tumor diagnosis. J Healthc Eng. 2013;4(1):23–45.
Zhou Z, Lu ZR. Gadolinium-based contrast agents for magnetic resonance cancer imaging. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2013;5(1):1–18.
Kanal E, Tweedle MF. Residual or retained gadolinium: practical implications for radiologists and our patients. Radiology. 2015;275(3):630–4.
Beam AS, Moore KG, Gillis SN, Ford KF, Gray T, Steinwinder AH, et al. GBCAs and risk for nephrogenic systemic fibrosis: a literature review. Radiol Technol. 2017;88(6):583–9.
Ramalho J, Semelka RC, Ramalho M, Nunes RH, AlObaidy M, Castillo M. Gadolinium-based contrast agent accumulation and toxicity: an update. AJNR Am J Neuroradiol. 2016;37(7):1192–8.
Guo BJ, Yang ZL, Zhang LJ. Gadolinium deposition in brain: current scientific evidence and future perspectives. Front Mol Neurosci. 2018;20(11):335.
Oliveira AC, Filipe HAL, Ramalho JPP, Salvador A, Geraldes CFGC, Moreno MJ, et al. Modeling Gd3+ complexes for molecular dynamics simulations: toward a rational optimization of MRI contrast agents. Inorg Chem. 2022;61(30):11837–58.
Trattnig S, Pinker K, Ba-Ssalamah A, Nöbauer-Huhmann IM. The optimal use of contrast agents at high field MRI. Eur Radiol. 2006;16(6):1280–7.
Hagberg GE, Scheffler K. Effect of r1 and r2 relaxivity of gadolinium-based contrast agents on the T1-weighted MR signal at increasing magnetic field strengths. Contrast Media Mol Imaging. 2013;8(6):456–65.
Noebauer-Huhmann IM, Szomolanyi P, Kronnerwetter C, Widhalm G, Weber M, Nemec S, et al. Brain tumors at 7T MRI compared to 3T–contrast effect after half and full standard contrast agent dose: initial results. Eur Radiol. 2015;25(1):106–12.
Krautmacher C, Willinek WA, Tschampa HJ, Born M, Träber F, Gieseke J, et al. Brain tumors: full- and half-dose contrast-enhanced MR imaging at 3.0 T compared with 1.5 T-initial experience. Radiology. 2005;237(3):1014–9.
Vaughan JT, Snyder CJ, DelaBarre LJ, Bolan PJ, Tian J, Bolinger L, et al. Whole-body imaging at 7T: preliminary results. Magn Reson Med. 2009;61(1):244–8.
Yetisir F, Poser BA, Grant PE, Adalsteinsson E, Wald LL, Guerin B. Parallel transmission 2D RARE imaging at 7T with transmit field inhomogeneity mitigation and local SAR control. Magn Reson Imaging. 2022;93:87–96.
Shi Z, Zhao X, Zhu S, Miao X, Zhang Y, Han S, et al. Time-of-flight intracranial MRA at 3 T versus 5 T versus 7 T: visualization of distal small cerebral arteries. Radiology. 2022;30:220114.
Aja-Fernández S, Vegas-Sánchez-Ferrero G, Tristán-Vega A. Noise estimation in parallel MRI: GRAPPA and SENSE. Magn Reson Imaging. 2014;32(3):281–90.
Hao D, Ai T, Goerner F, Hu X, Runge VM, Tweedle M. MRI contrast agents: basic chemistry and safety. J Magn Reson Imaging. 2012;36(5):1060–71.
The pharmacokinetic properties of Gadobutrol. https://www.bayer.com/sites/default/files/GADOVIST_EN_PI.pdf.
Wesseling P, Capper D. WHO 2016 Classification of gliomas. Neuropathol Appl Neurobiol. 2018;44(2):139–50.
Shen Y, Goerner FL, Snyder C, Morelli JN, Hao D, Hu D, et al. T1 relaxivities of gadolinium-based magnetic resonance contrast agents in human whole blood at 1.5, 3, and 7 T. Invest Radiol. 2015;50(5):330–8.
Kalavagunta C, Michaeli S, Metzger GJ. In vitro Gd-DTPA relaxometry studies in oxygenated venous human blood and aqueous solution at 3 and 7 T. Contrast Media Mol Imaging. 2014;9(2):169–76.
Rohrer M, Bauer H, Mintorovitch J, Requardt M, Weinmann HJ. Comparison of magnetic properties of MRI contrast media solutions at different magnetic field strengths. Invest Radiol. 2005;40(11):715–24.
Parikh PT, Sandhu GS, Blackham KA, Coffey MD, Hsu D, Liu K, Jesberger J, Griswold M, Sunshine JL. Evaluation of image quality of a 32-channel versus a 12-channel head coil at 1.5T for MR imaging of the brain. AJNR Am J Neuroradiol. 2011;32(2):365–73.
Albrecht J, Burke M, Haegler K, Schöpf V, Kleemann AM, Paolini M, Wiesmann M, Linn J. Potential impact of a 32-channel receiving head coil technology on the results of a functional MRI paradigm. Clin Neuroradiol. 2010;20(4):223–9.
Pacioni S, D’Alessandris QG, Buccarelli M, Boe A, Martini M, Larocca LM, et al. Brain invasion along perivascular spaces by glioma cells: relationship with blood-brain barrier. Cancers (Basel). 2019;12(1):18.
Kakeda S, Korogi Y, Hiai Y, Ohnari N, Moriya J, Kamada K, et al. Detection of brain metastasis at 3T: comparison among SE, IR-FSE and 3D-GRE sequences. Eur Radiol. 2007;17(9):2345–51.
Collins CM, Smith MB. Signal-to-noise ratio and absorbed power as functions of main magnetic field strength, and definition of "90 degrees " RF pulse for the head in the birdcage coil. Magn Reson Med. 2001;45(4):684–91.
Kim KN, Hernandez D, Seo JH, Noh Y, Han Y, Ryu YC, et al. Quantitative assessment of phased array coils with different numbers of receiving channels in terms of signal-to-noise ratio and spatial noise variation in magnetic resonance imaging. PLoS ONE. 2019;14(7):e0219407.
Fushimi Y, Miki Y, Urayama S, Okada T, Mori N, Hanakawa T, et al. Gray matter-white matter contrast on spin-echo T1-weighted images at 3 T and 1.5 T: a quantitative comparison study. Eur Radiol. 2007;17(11):2921–5.
Brink WM, Webb AG. High permittivity pads reduce specific absorption rate, improve B1 homogeneity, and increase contrast-to-noise ratio for functional cardiac MRI at 3 T. Magn Reson Med. 2014;71(4):1632–40.
Vaughan JT, Garwood M, Collins CM, Liu W, DelaBarre L, Adriany G, et al. 7T vs. 4T: RF power, homogeneity, and signal-to-noise comparison in head images. Magn Reson Med. 2001;46(1):24–30.
Manning C, Stringer M, Dickie B, Clancy U, Valdés Hernandez MC, Wiseman SJ, et al. Sources of systematic error in DCE-MRI estimation of low-level blood-brain barrier leakage. Magn Reson Med. 2021;86(4):1888–903.
Ochi T, Taoka T, Matsuda R, Sakamoto M, Akashi T, Tamamoto T, et al. Comparison between two separate injections and a single injection of double-dose contrast medium for contrast-enhanced MR imaging of metastatic brain tumors. Magn Reson Med Sci. 2014;13(4):221–9.
Koenig M, Schulte-Altedorneburg G, Piontek M, Hentsch A, Spangenberg P, Schwenke C, et al. Intra-individual, randomised comparison of the MRI contrast agents gadobutrol versus gadoteridol in patients with primary and secondary brain tumors, evaluated in a blinded read. Eur Radiol. 2013;23(12):3287–95.
Acknowledgements
Not applicable
Funding
None.
Author information
Authors and Affiliations
Contributions
Contributions ZJ: conceptualization, data curation, writing-original draft preparation, methodology, writing-reviewing and editing. WS: conceptualization, data curation, writing-original draft preparation, methodology, software, and validation. DX: data curation. HM: data curation. JY: conceptualization, data curation. XS: data analysis/interpretation, writing-original draft preparation, writing-reviewing and editing. CM: guarantor of the integrity of the entire study, recruitment of volunteers, methodology, writing, reviewing and editing. HX: guarantor of the integrity of the entire study, data curation, methodology, writing, reviewing and editing. All the authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board (Ethics Committee) of Zhongnan Hospital of Wuhan University (protocol code: No. 2021110 and date of approval: November 2021) for studies involving humans. Informed consent was obtained from all individual participants included in the study.
Consent for publication
The authors affirm that the human research participants provided informed consent for the publication of all images in the figures.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Jiang, Z., Sun, W., Xu, D. et al. The feasibility of half-dose contrast-enhanced scanning of brain tumours at 5.0 T: a preliminary study. BMC Med Imaging 24, 88 (2024). https://doi.org/10.1186/s12880-024-01270-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12880-024-01270-z