Abstract
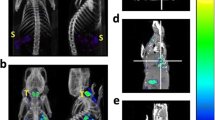
99mTc-ethambutol was preclinically evaluated as an alternative diagnostic tuberculosis agent. Its radiochemical purity, stability in saline and serum, in vitro Mycobacterium tuberculosis (MBT) binding, and biodistribution in mice were studied, and targeted imaging in model rabbits was performed. The following labeling conditions were found to be optimum: 3 mg of EMB, 150 µg of stannous fluoride, 3 mCi of sodium pertechnetate, pH 5.6. The highest product yield was reached in the time interval 30–90 min after reconstitution. The relative content of 99mTc-EMB in normal saline at room temperature decreased to 90 ± 0.25% in 240 min. 99mTc-EMB was stable in vitro in serum at 37°C, with 16.8 ± 0.3% radioimpurities accumulated in 16 h. 99mTc-EMB showed 69.8 ± 1.5% in vitro uptake in live strains of MBT. High in vivo accumulation (15.5 ± 0.8% ID) of the labeled EMB was observed in the infected site of model mice as compared to the normal and inflamed (muscles) sites. High uptake in the MBT infected site of a model rabbit was seen scintigraphically at 120 min after the intravenous administration of the 99mTc-EMB. Stability in normal saline and serum, substantial in vitro MBT uptake, high in vivo accumulation in the infected site, and accurate scintigraphy of the infected site make 99mTc-EMB promising as a novel alternative diagnostic tool for tuberculosis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Liu, X., Cheng, D., Gray, B.D., et al., Nucl. Med. Biol., 2012, vol. 39, pp. 709–714.
Nanni, C., Errani, C., Boriani, L., et al., J. Nucl. Med., 2010, vol. 51, pp. 1932–1936.
Litzler, P.Y., Manrique, A., Etienne, M., et al., J. Nucl. Med., 2010, vol. 51, pp. 1044–1048.
Mirbolooki, M.R., Upadhyay, S.K., Constantinescu, C.C., et al., Nucl. Med. Biol., 2014, vol. 41, pp. 10–16.
Belloli, S., Brioschi, A., Politi, L.S., et al., Nucl. Med. Biol., 2013, vol. 40, pp. 831–840.
Wang, Y., Chen, L., Liu, X., et al., Nucl. Med. Biol., 2013, vol. 40, pp. 89–96.
Liu, Z., Wyffels, L., Barber, C., et al., Nucl. Med. Biol., 2011, vol. 38, pp. 795–805.
Zhang, J., Guo, H., Zhang, S., et al., Bioorg. Med. Chem. Lett., 2008, vol. 18, pp. 5168–5170.
Chattopadhyay, S., Ghosh, M., Sett, S., et al., Appl. Radiat. Isot., 2012, vol. 10, pp. 2384–2387.
Roohi, S., Mushtaq, A., Jehangir, M., et al., J. Radioanal. Nucl. Chem., 2006, vol. 267, pp. 561–566.
Chattopadhyay, S., Das, S.S., Chandra, S., et al. Appl. Radiat. Isot., 2010, vol. 68, pp. 314–316.
Shah, S.Q., Khan, A.U., and Khan, M.R., Appl. Radiat. Isot., 2010, vol. 68, pp. 2255–2560.
Shah, S.Q. and Khan, M.R., J. Radioanal. Nucl. Chem., 2011, vol. 288, pp. 511–516.
Yamada, D., Saiki, S., Furuya, N., et al., Biochem. Biophys. Commun., 2016, vol. 471, pp. 109–116.
Causse, J.E. et al., Radiat. Appl. Instrum., Part A: Appl. Radiat. Isot., 1990, vol. 41, pp. 493–496.
Singh, N. and Bhatnagar, A., Tubercul. Res. Treat., 2010, pp. 1–9.
Shah, S.Q. and Momin, S., J. Glycom. Metab., 2017, vol. 2, pp. 12–23. DOI: https://doi.org/10.14302/issn.2572-5424.jgm-16-1352.
Shah, S.Q. and Alam, M., Infect. Disord. Drug Targets, 2017, vol. 3, pp. 185–191. DOI: 10.2174/1871526517666170606114650.
Welling, M.M., Annema, A.P., Balter, H.S., et al., Eur. J. Nucl. Med., 2000, vol. 27, pp. 292–301.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shah, S.Q., Ullah, N. Preclinical Evaluation of 99mTc-Ethambutol, an Alternative Tuberculosis Diagnostic Tool. Radiochemistry 61, 233–237 (2019). https://doi.org/10.1134/S1066362219020176
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1066362219020176