Abstract
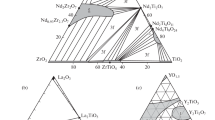
Phase compositions, structural features, and element distributions were studied for samples of the compositions REE2(Zr2−x Ti x )O7, which are potential matrices for immobilization of the rare earth-actinide fraction of high-level waste from spent nuclear fuel reprocessing. Samples with x up to 0.8 consist of pyrochlore, and at higher titanium content, of pyrochlore and monoclinic REE titanate with perovskite-type structure. The monoclinic phase becomes prevalent at x > 1.2. With respect to the content of the incorporated waste, it surpasses pyrochlore matrices by 10 wt %. The radiation resistance of this phase is close to that of titanate pyrochlore, but its amorphization dose is lower than for zirconate and titanate-zirconate pyrochlore. To check the suitability of monoclinic titanate for immobilization of the REE-actinide waste fraction, it is necessary to study its behavior in solutions and the effect of amorphization on the actinide leaching. The matrix can be prepared by the cold pressing-sintering method suggested in the United States for the synthesis of pyrochlore matrices with plutonium. High rate of solid-phase reactions in titanate systems allows the equilibrium to be attained at a moderate temperature (1400°C) within short sintering time (the first hours). One more possible procedure for matrix fabrication is cold crucible induction melting followed by the melt crystallization.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Laverov, N.P., Velichkin, V.I., Omel’yanenko, B.I., et al., Izolyatsiya otrabotavshikh yadernykh materialov (geologo-geokhimicheskie osnovy) (Isolation of Spent Nuclear Materials (Geological and Geochemical Principles)), Moscow: Inst. Fiziki Zemli, Ross. Akad. Nauk. 2008.
Laverov, N.P., Yudintsev, S.V., Stefanovsky, S.V., and Jang, Y.N., Dokl. Earth Sci., 2001, vol. 381A, no. 9, pp. 1053–1056.
Laverov, N.P., Yudintsev, S.V., Livshits, T.S., et al., Geochem. Int., 2010, vol. 48, no. 1, pp. 1–14.
Yudintsev, S.V., Geol. Ore Deposits, 2003, vol. 45, no. 2, pp. 151–165.
Yudintsev, S.V., Stefanovsky, S.V., and Nikonov, V.S., Dokl. Earth Sci., 2014, vol. 454, no. 1, pp. 54–58.
Lumpkin, G.R., Elements, 2006, vol. 2, no. 6, pp. 365–372.
Ewing, R.C., Earth Planet. Sci. Lett., 2005, vol. 229, pp. 165–181.
Hayatt, N.C., Stennet, M., Jenni, A., et al., Mater. Res. Soc. Symp. Proc., 2009, vol. 1193, pp. 61–66.
Ewing, R.C. and Weber, W.J., The Chemistry of the Actinide and Transactinide Elements, Morss, L.R., Edelstein, N.M., Fuger, J., Eds., Dordrecht (Netherlands): Springer, 2010, vol. 6, ch. 35, pp. 3813–3889.
Burakov, B.E., Ojovan, M.I., and Lee, W.E., Crystalline Materials for Actinide Immobilization. Materials for Engineering, London: Imperial College Press. 2011, vol. 1.
Ewing, R.C., Weber, W.J., and Lian, J., J. Appl. Phys., 2004, vol. 95, no. 11, pp. 5949–5971.
Ebbinghaus, B.B., Van Konynenburg, R.A., Vance, E.R., et al., US Patent 6 137 025, 1999.
Strachan, D.M., Scheele, R.D., Buck, E.C., et al., J. Nucl. Mater., 2005, vol. 345, pp. 109–135.
Icenhower, J.P., Strachan, D.M., McGrail, B.P., et al., Am. Mineral., 2006, vol. 91, pp. 39–53.
Ewing, R.C., Can. Mineral., 2005, vol. 43, pp. 2099–2116.
Myasoedov, B.F., in Materialy seminara “Mezhdunarodnoe khranilishche obluchennogo yadernogo topliva” (Proc. Workshop “International Spent Nuclear Fuel Repository”), Moscow, May 14–15, 2003, Moscow: Avangard, 2005, pp. 248–258.
Implications of Partitioning and Transmutation in Radioactive Waste Management, Vienna: IAEA, 2004.
Kopyrin, A.A., Karelin, A.I., and Karelin, V.A., Tekhnologiya proizvodstva i radiokhimicheskoi pererabotki yadernogo topliva (Technology for Production and Radiochemical Reprocessing of Nuclear Fuel), Moscow: Atomenergoizdat. 2006.
Treatment and Recycling of Spent Nuclear Fuel, Paris: CEA, 2008.
National Program in Chemical Partitioning, Paris: NEA OECD, 2010.
Nash, K.L., Madic, Ch., Mathur, J.N., and Lacquement, J., The Chemistry of the Actinide and Transactinide Elements, Morss, L.R., Edelstein, N.M., Fuger, J., Eds., Dordrecht (Netherlands): Springer, 2010, vol. 4, ch. 24, pp. 2622–2798.
Bruno, J. and Ewing, R.C., Elements, 2006, vol. 2, pp. 343–349.
Walker, C.T., Rondinella, V.V., Papaioannou, D., et al., J. Nucl. Mater., 2005, vol. 345, pp. 192–205.
Ivanets, D.V., Tananaev, I.G., and Sarychev, G.A., Tsvetn. Met., 2010, no. 3, pp. 66–72.
Collins, E.D., Jubin, R.T., DelCul, G.D., et al., in Proc. Int. Conf. “Global 2009,” Paris (France), Sept. 06–11, 2009, pp. 2595–2602.
Subramanian, M.A., Aravamudan, G., and Subba Rao, G.V., Prog. Solid State Chem., 1983, vol. 15, pp. 55–143.
Shoup, S.S. and Bamberger, C.E., Mater. Res. Soc. Symp. Proc., 1997, vol. 465, pp. 379–386.
Laverov, N.P., Yudintsev, S.V., Stefanovsky, S.V., et al., Dokl. Earth Sci., 2002, vol. 383, no. 2, pp. 190–193.
Stefanovsky, S.V., Yudintsev, S.V., and Nikonov, B.S., Fiz. Khim. Obrab. Mater., 2004, no. 2, pp. 68–77.
Gong, W. and Zhang, R., J. Alloys Compd., 2013, vol. 548, pp. 216–221.
Skapin, S.D., Kolar, D., and Suvorov, D., Solid State Sci., 1999, vol. 1, pp. 245–255.
Skapin, S.D., Kolar, D., and Suvorov, D., J. Eur. Ceram. Soc., 2000, vol. 20, pp. 1179–1185.
Preuss, A. and Gruehn, R., J. Solid State Chem., 1994, vol. 110, pp. 363–369.
Gong, W. and Zhang, R., Thermochim. Acta, 2012, vol. 534, pp. 28–32.
Shoup, S.S., Bamberger, C.E., Tyree, J.L., and Anovitz, L.M., J. Solid State Chem., 1996, vol. 127, pp. 231–239.
Shoup, S.S., Bamberger, C.E., Haverlock, T.J., and Peterson, J.R., J. Nucl. Mater., 1997, vol. 240, pp. 112–117.
Harvey, E.J., Whittle, K.R., Lumpkin, G.R., et al., J. Solid State Chem., 2005, vol. 178, no. 3, pp. 800–810.
Smith, K.L., Blackford, M.G., Lumpkin, G.R., et al., Microsci. Microanal., 2006, vol. 12, suppl. 2, pp. 1094–1095.
Whittle, K.R., Lumpkin, G.R., Smith, K.L., et al., Mater. Res. Soc. Symp. Proc., 2007, vol. 985, CD-version, paper 0985-NN08-09.
Whittle, K.R., Lumpkin, G.R., Blackford, M.G., et al., J. Solid State Chem., 2010, vol. 183, pp. 2416–2420.
Laverov, N.P., Yudintsev, S.V., Stefanovsky, S.V., and Ewing, R.Ch., Dokl. Earth Sci., 2012, vol. 443, no. 2, pp. 526–531.
Shannon, R.D., Acta Crystallogr., Sect. A, 1976, vol. 32, no. 5, pp. 751–767.
PDFWIN-2, Int. Centre for Diffraction Data, Newton Square, PA (USA), 1999.
Begg, B.D., Vance, E.R., Day, R.A., et al., Mater. Res. Soc. Symp. Proc., 1997, vol. 465, pp. 325–332.
Begg, B.D. and Vance, E.R., Mater. Res. Soc. Symp. Proc., 1997, vol. 465, pp. 333–340.
Begg, B.D., Vance, E.R., and Lumpkin, G.R., Mater. Res. Soc. Symp. Proc., 1998, vol. 506, pp. 79–86.
Fortner, J.A., Kropf, A.J., Bakel, A.J., et al., Mater. Res. Soc. Symp. Proc., 2000, vol. 608, pp. 401–406.
GOST (State Standard) R 50926-96: Solidified High-Level Waste. General Technical Requirements, Moscow: Izd. Standartov, 1996.
Wang, S.X., Begg, B.D., Wang, L.M., et al., J. Mater. Res., 1999, vol. 14, no. 12, pp. 4470–4473.
Laverov, N.P., Yudintsev, S.V., Yudintseva, T.S., et al., Geol. Ore Deposits, 2003, vol. 45, no. 6, pp. 423–451.
Sykora, R.E., Raison, P.E., and Haire, R.G., J. Solid State Chem., 2005, vol. 178, pp. 578–583.
Lumpkin, G.R., Pruneda, M., Rios, S., et al., J. Solid State Chem., 2007, vol. 180, pp. 1512–1518.
Laverov, N.P., Yudintsev, S.V., Velichkin, V.I., et al., Radiochemistry, 2009, vol. 51, no. 5, pp. 529–536.
Stefanovsky, S.V., Yudintsev, S.V., and Myasoedov, B.F., Dokl. Chem., 2012, vol. 447, no. 2, pp. 296–299.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © S.V. Yudintsev, S.V. Stefanovsky, B.S. Nikonov, M.S. Nikol’skii, T.S. Livshits, 2015, published in Radiokhimiya, 2015, Vol. 57, No. 2, pp. 161–171.
Rights and permissions
About this article
Cite this article
Yudintsev, S.V., Stefanovsky, S.V., Nikonov, B.S. et al. Potential matrices for immobilization of the rare earth-actinide fraction of high-level waste in the REE2Zr2O7-REE2Ti2O7 system. Radiochemistry 57, 187–199 (2015). https://doi.org/10.1134/S1066362215020125
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1066362215020125