Abstract
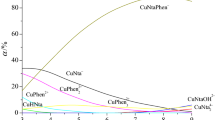
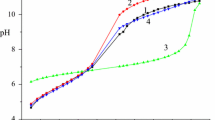
The formation of mixed-ligand complexes in the M(II)–Ida–L systems (M = Cu, Ni, L = His, Orn, Lys), where Ida is the iminodiacetic acid residue, was studied by pH-metry, calorimetry, and spectrophotometry. The thermodynamic parameters (logK, Δ r G 0, Δ r H, Δ r S) of formation of the complexes were determined at 298.15 K and the ionic strength I = 0.5 (KNO3). The most probable mode of coordination of the chelating agent and the amino acid in the mixed-ligand complexes was elucidated.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
G. Arena, S. Musumeci, E. Rizzarelli, and S. Sammartano, Trans. Met. Chem. 10, 399 (1985).
H. Block, B. Maertens, A. Spriestersbach, et al., Meth. Enzymol. 463, 439 (2009).
G. Sharma and J. P. Tandon, Talanta 18, 1163 (1971).
P. C. Parikh and P. K. Bhattacharya, J. Ind. Chem. Soc. 50, 804 (1973).
M. V. Chidambaram and P. K. Bhattacharya, Acta Chim. Hung. 75, 123 (1975).
O. Yamauchi, T. Sakurai, and A. Nakahara, J. Am. Chem. Soc. 101, 4164 (1979).
E. V. Kozlovskii, D. F. Pyreu, and T. B. Khochenkova, Russ. J. Inorg. Chem. 53, 1158 (2008).
I. Kiseleva, D. Pyreu, T. Krivonogikh, et al., Polyhedron 51, 10 (2013).
V. A. Borodin, V. P. Vasil’ev, and E. V. Kozlovskii, Mathematical Problems of Chemical Thermodynamics, (Nauka, Novosibirsk, 1985) [in Russian].
G. Anderegg, F. Arnaud-Neu, R. Delgado, et al., Pure Appl. Chem. 77, 1445 (2005).
L. D. Pettit, Pure Appl. Chem. 56, 247 (1984).
O. Yamauchi and A. Odani, Pure Appl. Chem. 68, 469 (1996).
V. P. Vasil’ev, Thermodynamic Properties of Electrolyte Solutions (Vysshaya Shkola, Moscow, 1982) [in Russian].
V. A. Garavin, Candidate’s Dissertation in Chemistry (Ivanovo Inst. of Chem. Technol., Ivanovo, 1983).
A. Gergely, E. Farkas, I. Nagypál, and E. Kas, J. Inorg. Nucl. Chem. 40, 1709 (1978).
J. G. Mesu, T. Visser, F. Soulimani, et al., Inorg. Chem. 45, 1960 (2006).
V. G. Shtyrlin, Y. I. Zyavkina, E. M. Gilyazetdinov, et al., Dalton Trans. 41, 1216 (2012).
C. Conato, A. Contino, G. Maccarrone, et al., Thermochim. Acta 362, 13 (2000).
A. Ya. Fridman, E. V. Kozlovskii, V. V. Serdyukov, and E. N. Kalachev, Zh. Neorg. Khim. 37, 1611 (1992).
G. Sharma and J. P. Tandon, Z. Naturforsch. 25, 22 (1970).
P. Amico, G. Arena, P. Daniele, et al., Inorg. Chem. 20, 772 (1981).
T. Kiss, I. Sovago, and A. Gergely, Pure Appl. Chem. 63, 597 (1991).
S. Bandyopadhyay and G. N. Mukherjee, Proc. Indian Acad. Sci. Sect. A 115, 249 (2003).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © D.F. Pyreu, M.A. Bazanova, S.N. Gridchin, E.V. Kozlovskii, 2016, published in Zhurnal Neorganicheskoi Khimii, 2016, Vol. 61, No. 7, pp. 950–958.
Rights and permissions
About this article
Cite this article
Pyreu, D.F., Bazanova, M.A., Gridchin, S.N. et al. Thermochemical study of the complex formation of copper(II) and nickel(II) iminodiacetates with amino acids in aqueous solutions. Russ. J. Inorg. Chem. 61, 909–917 (2016). https://doi.org/10.1134/S0036023616070159
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023616070159