Abstract
The blood–brain barrier (BBB) provides optimal conditions for the functioning of brain neurons. The barrier properties of the cerebrovascular (CV) endothelium are determined by the claudin family of proteins and occludin, which are major molecular determinants of selective intercellular transport. Plasma membrane lipids play an important role in forming the structure of tight junctions, being combined with these proteins into lipid rafts. The development of neurodegenerative diseases and psychiatric disorders correlates with changes in claudin levels in the CV endothelium. We studied the effect of methyl-beta-cyclodextrin (MbCD), which causes destabilization of the plasma membrane lipid phase, on claudin-1, -5 and occludin levels in rat brain tissue. Male rats were injected intravenously with MbCD at a dose of 5 mg/kg of body weight, and 30 min later, brain tissue was prepared for Western blot and immunohistochemistry. MbCD induced a significant decrease in the claudin-5 level in the frontal lobes of the rat brain, while claudin-1 and occludin levels remained unchanged. Immunohistochemistry and confocal laser microscopical image analysis confirmed that changes in claudin-5 localization are confined to the CV endothelium. Since claudin-5 is a major contributor to the impermeability of the CV endothelium, we can assume that a change in the orderliness of the raft lipid phase may lead to an increase in intercellular permeability. The results suggest that the lipid environment is an important molecular component of the tight junction in the rat brain CV endothelium, which may be implicated in the maintenance and regulation of the barrier properties of the BBB.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
The cerebrovascular (CV) endothelium is one of the key structures of the blood–brain barrier (BBB), which provides regulated transport of ions, water, and various substances between blood plasma and the brain parenchyma [1, 2]. The functioning of the CV endothelium leads to the stabilization and maintenance of neuronal functions [3, 4], while the disruption of the BBB integrity is involved in the pathogenesis of various neurodegenerative diseases [5, 6]. Tight junctions represent an apical complex of proteins located in the plasma membranes of neighboring endotheliocytes and interacting with each other in the intercellular space. A variety of proteins composing this complex, primarily proteins of the claudin family, provides selective paracellular permeability [7, 8]. The molecular architectonics of tight junctions is represented as clusters of interrelated proteins [9], which form a signaling platform via the adaptor proteins and protein kinases [10]. Lipids, combined into lipid rafts together with claudins, play an important role in the clustering of tight junctional proteins [11]. The role of the lipid environment in the functional regulation of plasma membrane protein clusters is studied using the derivatives of the cyclic oligosaccharide cyclodextrin. These compounds cause destabilization of the plasma membrane lipid phase, leading to an increase in the fluidity and permeability of these membranes under model conditions [12, 13]. In vivo studies in animal models of neurodegenerative diseases predominantly use 2-hydroxypropyl-beta-cyclodextrin [14]. Methyl-beta-cyclodextrin (MbCD) is used to analyze the role of the lipid environment in the tight junctions [15, 16].
Data on the role of the lipid environment in the formation of the endothelial barrier of brain blood vessels in animals are limited. Claudin-5 is known to be the main contributor to the non-permeability of the CV endothelium [17]. The development of psychiatric disorders correlates with changes in claudin-5 of the CV endothelium [18, 19]. Data on the effect of MbCD on the CV endothelium are confined to a single article in which the immunohistochemical method showed a decrease in the claudin-5 signal in endotheliocytes upon exposure to this compound [15]. These data require confirmation using other methods of assessing protein levels in endotheliocytes. It is worth noting that various claudins are represented in the tight junction of the CV endothelium. The presence of claudin-1 has been demonstrated in primary cultures of CV endotheliocytes of mice and rats [20, 21]. The removal of cholesterol from lipid rafts in Caco-2 cell line by MbCD indicates that this protein, unlike other claudins, does not respond to destabilization of the lipid environment [16]. This fact suggests a differential response of tight junctional proteins in the CV endothelium to MbCD, which may be fundamentally important in predicting changes in the permeability of the CV endothelial barrier. While claudins affect epithelial permeability for ions, occludin is required to restrict the paracellular transport of organic molecules [22]. Occludin is present in the tight junction of human CV endothelial cells and is involved in the permeability regulation of these cells under the effect of various factors [23, 24]. This study aimed to investigate the levels of claudin-1, -5, and occludin in the rat CV endothelium during intravenous injection of methyl-beta-cyclodextrin.
MATERIALS AND METHODS
Experimental animals and experimental design. Male Wistar rats (180–200 g, n = 5) were used in the experiments. Animals were kept in cages by five individuals in each under stabilized temperature and standard illumination (12 h light/12 h dark). The animals were kept on a standard diet with ad libitum access to food and water. All procedures conducted in the experiments involving animals complied with the ethical standards approved by legislative acts of the Russian Federation, the principles of the Basel Declaration, and the recommendations of the St. Petersburg State University Ethics Committee for Animals.
MbCD was injected intravenously (i.v.) at a dose of 5 mg/kg of animal body weight. Previously, it was found that this dose, when administered intravenously, affects the tight junctional structure of brain blood vessels [15]. After 30 min, the animals were decapitated on a guillotine (OpenScience, Russia). Animals of the control group received i.v. the same volume of physiological solution. The tissue of the frontal lobes was frozen at –80°C for subsequent Western blot analysis, and fixed in 10% buffered formalin for immunohistochemistry. Samples for Western blot and immunohistochemistry were taken in all animals from the same brain region. The section of the brain region located anterior to the central gyrus, which corresponds to the rat cortical motor area, was dissected.
Western blot. For Western blot analysis, brain tissue was homogenized using a Retsch MM 400 mechanical homogenizer (Retsch, Germany) with addition of RIPA lysis buffer (25 mM HEPES (pH 7.6); 2 mM EDTA; 25 mM NaF; 1% SDS) containing protease inhibitors, cOmplete mini EDTA tablets (Roche, Germany). Western blot was performed as previously described [25]. Primary antibodies to claudin-1 (1 : 1000, Invitrogen, USA), claudin-5 (1 : 500, Invitrogen, USA), occludin (1 : 1000, Invitrogen, USA), as well as goat-anti-rabbit and goat-anti-mouse secondary antibodies (1 : 1000, Abcam, USA), were used. The claudin content was normalized to a total protein of the same sample. Protein levels in the control groups were taken as 100%.
Immunohistochemistry. After pre-fixation in 10% buffered formalin, before slicing, brain tissue was placed in a 15% sucrose solution (24 h). Then, the tissue was washed in PBS solution and immersed in cryoprotectant (Frozen Section Compound, Leica Biosystems, USA) for 30 min. Serial slices of the frozen brain tissue (15 µm thick) were obtained on a CM1850UV cryostat microtome (Leica Microsystems, Austria). The slices were heat-treated for 45 min in citrate buffer (T = 80°C, 0.01 M, pH = 6.0) and incubated in a blocking solution (0.2% Triton, 10% BSA, PBS pH = 7.4) for 2 h at 37°C. Primary antibodies to claudin-1 and occludin (1 : 100, Thermo Fisher Scientific, USA, # 71-7800, 71-1500), as well as to claudin-5 (1 : 100, Thermo Fisher Scientific, USA, # 32-2500), were used for a 24-h incubation at 4°C. Secondary antibodies conjugated to Alexa Fluor-488 (1 : 800, Invitrogen, USA) were used for 120 min at 37°C. Finally, the sections were washed in PBS and mounted in a medium with a nuclear dye 4',6-diamidino-2-phenylindole (DAPI, 0.1 μg/mL). The sections were viewed using a Leica TCS SP5 microscope (Leica Microsystems GmbH, Germany). To further confirm the localization of tight junctional proteins specifically in the brain blood vessels, we used primary mouse monoclonal antibodies to PECAM-1 (M-20, 1 : 200, Santa Cruz, USA) and claudin-5 (1 : 200, Thermo Fisher Scientific, USA) followed by secondary antibodies conjugated to Alexa Fluor-488 (1 : 800, Invitrogen, USA) and CF-633 (1 : 800, Sigma Aldrich, USA). To rule out nonspecific fluorescence of secondary antibodies, a negative control was carried out, which showed the absence of background fluorescence.
Composition of solutions and concentrations of physiologically active substances. MbCD and reagents for the preparation of solutions were purchased from Sigma Aldrich (USA).
Statistical data processing. Statistical processing was carried out conventional statistical methods and the GraphPad Prism 8 computer statistical program (GraphPad, San Diego, CA, USA). One-way ANOVA with Dunnett’s correction were used for data processing. A normal data distribution was tested using the Shapiro–Wilk test. The significance level of p < 0.05 was taken as statistically significant. Data are presented as means ± standard error of the mean (M ± m).
RESULTS
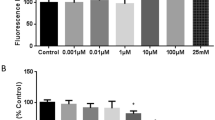
Exposure to MbCD for 30 min at a dose of 5 mg/kg body weight i.v. led to changes in claudin levels in the tissue of the rat frontal lobes. A significant 75% decrease in the level of claudin-5 was found compared to the control group. The claudin-1 and occludin levels remained unchanged (Fig. 1a).
PECAM-1 is one of the main proteins of intercellular contacts between endothelial cells, so that it has been referred to the marker of these cells since the earliest studies [26]. Another marker of the vascular endothelium in various brain regions is claudin-5 [27]. Double immunohistochemical labeling on control samples demonstrated identical distribution of PECAM-1 and claudin-5 fluorescence signals in brain tissue, indicating their localization in blood vessel (Fig. 2). Moreover, the extended pattern of signal distribution and the oval shape of the nuclei suggest that immunolabeling is confined to the area of vascular endothelial cells. Claudin-1, -5, and occludin signals were detected in all samples. An image analysis shows that MbCD exposure led to no redistribution of these proteins across the brain tissue. Both the decrease and increase in their levels were endothelium-specific because they were localized within the blood vessels (Fig. 1b).
Expression and localization of tight junctional proteins in frontal lobe tissue of the rat brain in the Control and after intravenous injection of methyl-beta-cyclodextrin (MbCD) at a dose of 5 mg/kg for 30 min. (a) Representative immunoblots (upper panel) and densitometry (lower panel) of tight junctional proteins (n = 4 in both groups). One-way ANOVA with Dunnett’s correction: **p < 0.01 vs. control. (b) Localization of tight junctional proteins in the vascular region of the frontal lobes. Distribution of claudins (green channel) and the oval shape of endothelial cell nuclei (DAPI, blue channel) coincide with the characteristic topographic pattern of blood vessels in brain tissue (scale bar, 50 µm).
DISCUSSION
The results of this study show that destabilization of the plasma membrane lipid composition in the rat CV endothelium affects the structure of tight junctions between endothelial cells. Intravenous administration of MbCD at a dose of 5 mg/kg for 30 min alters the level of tight junctional proteins. MbCD decreases the expression of claudin-5, which is the main tight junctional protein in the vascular endothelium, while claudin-1 and occludin levels remained unchanged. As it is well known that claudin-5 is the main barrier-forming protein of tight junctions in brain endotheliocytes [17], we assume that changes in the orderliness of the raft lipid phase may lead to an increased BBB permeability.
MbCD is a xenobiotic for the mammalian organism. However, in the blood plasma, there are metabolites that can also affect membrane lipids. A decrease of the epithelial barrier properties under the effect of sodium caprate (fatty acid, C10) may result from the lipid raft destruction [28], suggesting a possible alteration in the state of the BBB when metabolic changes occur in the blood plasma biochemical profile.
The immunohistochemical method has previously shown an MbCD-induced decrease in claudin-5 in the CV endothelium [15]. In our experiments, Western blotting confirmed a decrease in the claudin-5 level when the same dose of MbCD was applied, as well as no changes in claudin-1 and occludin levels. It is worth noting that the results obtained on animals agree with the experiments on claudin distribution in lipid rafts during the incubation of MDCK line cells with sodium caprate. Sodium caprate application results in the displacement of claudin-5 from lipid rafts but does not affect claudin-1 distribution [28]. Perhaps this is due to the fact that rafts have different lipid compositions [5]. Cyclodextrins show a selective activity toward lipid extraction. MbCD removes predominantly phospholipids and cholesterol from the plasma membrane [29]. These observations may indicate the selective interaction of individual claudins, which have different amino acid compositions, with raft lipids, as well as their heterogeneous distribution in the structure of tight junction.
The absence of changes in the content of occludin, which is important for the stabilization of tight junction in epithelial tissues [30], also supports the heterogeneous effect of MbCD on the structure of tight junctions in the vascular endothelium. MbCD-induced changes in levels of various tight junctional proteins may occur in the activation of different cell signaling systems. Integral proteins of the plasma membrane, which include claudins and occludin, interact through adaptor proteins with the apical actomyosin complex and various signaling proteins [10]. MbCD exposure leads to cholesterol destabilization in the plasma membrane, changes in its mechanical properties, and hence to cytoskeleton rearrangement and actin polymerization, which triggers intracellular signaling [31]. Remodeling of the apical actomyosin ring causes molecular reorganization of tight junctional composition and structure, as well as changes in epithelial permeability, including for sodium ions [32]. In experiments on cell lines, it was shown that a decrease in plasma membrane cholesterol leads to a decrease in the expression of the Na,K-ATPase alpha-1 subunit upon activation of the Src-dependent signaling pathway [33]. The use of the specific Na,K-ATPase ligand ouabain confirms the functional interaction between Na,K-ATPase and modulation of the expression claudins in tight junction via Src-signaling [25]. Thus, the results obtained in this study indicate that the lipid environment is an important molecular component of the tight junction in the rat CV endothelium, which and may be involved in the maintenance and regulation of the barrier properties of the BBB.
REFERENCES
Sheikov N, McDannold N, Sharma S, Hynynen K (2008) Effect of focused ultrasound applied with an ultrasound contrast agent on the tight junctional integrity of the brain microvascular endothelium. Ultrasound Med Biol 34:1093–1104. https://doi.org/10.1016/j.ultrasmedbio.2007.12.015
Ahishali B, Kaya M (2021) Evaluation of Blood-Brain Barrier Integrity Using Vascular Permeability Markers: Evans Blue, Sodium Fluorescein, Albumin-Alexa Fluor Conjugates, and Horseradish Peroxidase. Methods Mol Biol 2367:87–103. https://doi.org/10.1007/7651_2020_316
Abbott NJ, Patabendige AA, Dolman DE, Yusof SR, Begley DJ (2010) Structure and function of the blood-brain barrier. Neurobiol Dis 37:13–25. https://doi.org/10.1016/j.nbd.2009.07.030
Nico B, Ribatti D (2012) Morphofunctional aspects of the blood-brain barrier. Curr Drug Metab 13:50–60. https://doi.org/10.2174/138920012798356970
Petrov AM, Kasimov MR, Zefirov AL (2016) Brain Cholesterol Metabolism and Its Defects: Linkage to Neurodegenerative Diseases and Synaptic Dysfunction. Acta Naturae 8(1):58–73.
Anchisi L, Dessì S, Pani A, Mandas A (2013) Cholesterol homeostasis: A key to prevent or slow down neurodegeneration. Front Physiol 3:486. https://doi.org/10.3389/fphys.2012.00486
Günzel D, Yu AS (2013) Claudins and the modulation of tight junction permeability. Physiol Rev 93:525–569. https://doi.org/10.1152/physrev.00019.2012
Tsukita S, Tanaka H, Tamura A (2019) The Claudins: From Tight Junctions to Biological Systems. Trends Biochem Sci 44:141–152. https://doi.org/10.1016/j.tibs.2018.09.008
Markov AG, Aschenbach JR, Amasheh S (2015) Claudin clusters as determinants of epithelial barrier function. IUBMB Life 67:29–35. https://doi.org/10.1002/iub.1347
Zahraoui A, Louvard D, Galli T (2000) Tight junction, a platform for trafficking and signaling protein complexes. J Cell Biol 151:F31–36. https://doi.org/10.1083/jcb.151.5.f31
Lee DB, Jamgotchian N, Allen SG, Abeles MB, Ward HJ (2008) A lipid-protein hybrid model for tight junction. Am J Physiol Renal Physiol 295:F1601–1612. https://doi.org/10.1152/ajprenal.00097.2008
Martín Del Valle EM (2004) Cyclodextrins and their uses: A review. Process biochemistry 39:1033–1046. https://doi.org/10.1016/S0032-9592(03)00258-9
Dos Santos AG, Bayiha JC, Dufour G, Cataldo D, Evrard B, Silva LC, Deleu M, Mingeot-Leclercq MP (2017) Changes in membrane biophysical properties induced by the Budesonide/Hydroxypropyl-β-cyclodextrin complex. Biochim Biophys Acta Biomembr 1859:1930–1940. https://doi.org/10.1016/j.bbamem.2017.06.010
Du J, Liu X, Zhang Y, Han X, Ma C, Liu Y, Guan L, Qiao L, Lin J (2022) The Effects of Combined Therapy With Metformin and Hydroxypropyl-beta-Cyclodextrin in a Mouse Model of Niemann-Pick Disease Type C1. Front Pharmacol 12:825425. https://doi.org/10.3389/fphar.2021.825425
Atışa M, Akcana U, Yılmazb CU, Orhanc N, Düzgüna P, Ceylana UD, Arıcand N, Karahüseyinoğlue S, Şahine GN, Ahıshalıf B, Kaya M (2019) Effects of methyl-beta-cyclodextrin on blood-brain barrier permeability in angiotensin II-induced hypertensive rats. Brain Res 1715:148–155. https://doi.org/10.1016/j.brainres.2019.03.024
Lambert D, O’Neill CA, Padfield PJ (2007) Methyl-beta-cyclodextrin increases permeability of Caco-2 cell monolayers by displacing specific claudins from cholesterol rich domains associated with tight junctions. Cell Physiol Biochem 20:495–506. https://doi.org/10.1159/000107533
Nitta T, Hata M, Gotoh S, Seo Y, Sasaki H, Hashimoto N, Furuse M, Tsukita S (2003) Size-selective loosening of the blood-brain barrier in claudin-5-deficient mice. J Cell Biol 161:653–660. https://doi.org/10.1083/jcb.200302070
Greene C, Hanley N, Campbell M (2020) Blood-brain barrier associated tight junction disruption is a hallmark feature of major psychiatric disorders. Transl Psychiatry 10:373. https://doi.org/10.1038/s41398-020-01054-3
Dudek KA, Dion-Albert L, Lebel M, LeClair K, Labrecque S, Tuck E, Ferrer Perez C, Golden SA, Tamminga C, Turecki G, Mechawar N (2020) Molecular adaptations of the blood–brain barrier promote stress resilience vs. depression. Proc Natl Acad Sci USA 117:3326–3336. https://doi.org/10.1073/pnas.1914655117
Díaz-Coránguez M, Segovia J, López-Ornelas A, Puerta-Guardo H, Ludert J, Chávez B, Meraz-Cruz N, González-Mariscal L (2013) Transmigration of neural stem cells across the blood brain barrier induced by glioma cells. PLoS One 8:e60655. https://doi.org/10.1371/journal.pone.0060655
Velandia-Romero ML, Calderón-Peláez MA, Castellanos JE (2016) In Vitro Infection with Dengue Virus Induces Changes in the Structure and Function of the Mouse Brain Endothelium. PLoS One 11:e0157786. https://doi.org/10.1371/journal.pone.0157786
Buschmann MM, Shen L, Rajapakse H, Raleigh DR, Wang Y, Wang Y, Lingaraju A, Zha J, Abbott E, McAuley EM, Breskin LA, Wu L, Anderson K, Turner JR, Weber CR (2013) Occludin OCEL-domain interactions are required for maintenance and regulation of the tight junction barrier to macromolecular flux. Mol Biol Cell 24:3056–3068. https://doi.org/10.1091/mbc.E12-09-0688
Zhang Y, Ding X, Miao C, Chen J (2019). Propofol attenuated TNF-α-modulated occludin expression by inhibiting Hif-1α/ VEGF/ VEGFR-2/ ERK signaling pathway in hCMEC/D3 cells. BMC Anesthesiol 19:127. https://doi.org/10.1186/s12871-019-0788-5
Tian J, Shi R, Liu T, She R, Wu Q, An J, Hao W, Soomro MH (2019) Brain Infection by Hepatitis E Virus Probably via Damage of the Blood-Brain Barrier Due to Alterations of Tight Junction Proteins. Front Cell Infect Microbiol 9:52. https://doi.org/10.3389/fcimb.2019.00052
Markov AG, Fedorova AA, Kravtsova VV, Bikmurzina AE, Okorokova LS, Matchkov VV, Cornelius V, Amasheh S, Krivoi II (2020) Circulating Ouabain Modulates Expression of Claudins in Rat Intestine and Cerebral Blood Vessels. Int J Mol Sci 21:5067. https://doi.org/10.3390/ijms21145067
Albelda SM, Muller WA, Buck CA, Newmanll PJ (1991) Molecular and cellular properties of PECAM-1 (endoCAM/CD31): a novel vascular cell-cell adhesion molecule. J Cell Biol 114(5):1059–68. https://doi.org/10.1083/jcb.114.5.1059
Morita K, Sasaki H, Furuse M, Tsukita S (1999) Endothelial claudin: claudin-5/TMVCF constitutes tight junction strands in endothelial cells. J Cell Biol 147:185–194. https://doi.org/10.1083/jcb.147.1.185
Sugibayashi K, Onuki Y, Takayama K (2009) Displacement of tight junction proteins from detergent-resistant membrane domains by treatment with sodium caprate. Eur J Pharm Sci 36:246–253. https://doi.org/10.1016/j.ejps.2008.09.011
Monnaert V, Tilloy S, Bricout H, Fenart L, Cecchelli R, Monflier E (2004) Behavior of α-, β-, and γ-Cyclodextrins and Their Derivatives on an in Vitro Model of Blood-Brain Barrier. J Pharm Experim Ther 310:745–751. https://doi.org/10.1124/jpet.104.067512
Saito AC, Higashi T, Fukazawa Y, Otani T, Tauchi M, Higashi AY, Furuse M, Chiba H (2021) Occludin and tricellulin facilitate formation of anastomosing tight-junction strand network to improve barrier function. Mol Biol Cell 32(8):722–738. https://doi.org/10.1091/mbc.E20-07-0464
Hissa B, Pontes B, Roma PM, Alves AP, Rocha CD, Valverde TM, Aguiar PH, Almeida FP, Guimarães AJ, Guatimosim C, Silva AM, Fernandes MC, Andrews NW, Viana NB, Mesquita ON, Agero U, Andrade LO (2013) Membrane Cholesterol Removal Changes Mechanical Properties of Cells and Induces Secretion of a Specific Pool of Lysosomes. PLoS ONE 8:e82988. https://doi.org/10.1371/journal.pone.0082988
He WQ, Wang J, Sheng JY, Zha JM, Graham WV, Turner JR (2020) Contributions of Myosin Light Chain Kinase to Regulation of Epithelial Paracellular Permeability and Mucosal Homeostasis. Int J Mol Sci 21:993. https://doi.org/10.3390/ijms21030993
Zhang J, Li X, Yu H, Larre I, Dube PR, Kennedy DJ, Tang WHW, Westfall K, Pierre SV, Xie Z, Chen Y (2020) Regulation of Na/K-ATPase expression by cholesterol: isoform specificity and the molecular mechanism. Am J Physiol Cell Physiol 319:C1107–C1119. https://doi.org/10.1152/ajpcell.00083.2020
Funding
This work was funded by the Russian Science Foundation (RSF), grant no. 18-15-00043. The studies were carried out using the facilities of the “Center for Molecular and Cellular Technologies” Resource Center (project no. 109-14629) and the “Chromas” Center for Collective Use of Equipment (project no. 101-14625) at the St. Petersburg State University.
Author information
Authors and Affiliations
Contributions
Conceptualization and methodology (A.G.M., I.I.K.); data collection and processing (A.E.B., A.A.F.), manuscript writing and editing (A.G.M., A.E.B., A.A.F., I.I.K.).
Corresponding author
Ethics declarations
CONFLICT OF INTEREST
The authors declare that they have neither evident nor potential conflict of interest related to the publication of this article. A.G.M. is a member of the editorial board of the Russian Journal of Physiology.
Additional information
Translated by A. Polyanovsky
Russian Text © The Author(s), 2022, published in Rossiiskii Fiziologicheskii Zhurnal imeni I.M. Sechenova, 2022, Vol. 108, No. 5, pp. 677–685https://doi.org/10.31857/S0869813922050089.
Rights and permissions
About this article
Cite this article
Markov, A.G., Bikmurzina, A.E., Fedorova, A.A. et al. Methyl-beta-Cyclodextrin Alters the Level of Tight Junction Proteins in the Rat Cerebrovascular Endothelium. J Evol Biochem Phys 58, 849–855 (2022). https://doi.org/10.1134/S0022093022030188
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022093022030188