Abstract
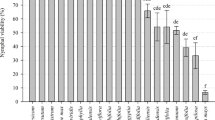
Araujia mosaic virus (ArjMV) has previously been confirmed to have a narrow host range and is considered a potential biological control agent for moth plant (Araujia hortorum), a troublesome environmental weed in northern regions of New Zealand. In this study, the virus was tested on 14 plant species, including species of importance to New Zealand to explore its possible use as a biocontrol agent for moth plant. Mosaic symptoms and occasional distortion were observed on leaves of the three Araujia species tested. Mosaic symptoms were seen on Morrenia odorata and Oxypetalum caeruleum, while mosaic symptoms, chlorotic spots and leaf distortion were observed on all three of the Gomphocarpus species tested. No symptoms were observed on Asclepias curassavica, Catharanthus roseus, Hoya carnosa, Mandevilla × amabilis, Nerium oleander and Vinca major. Because ArjMV infected the three Araujia species causing stunting, this virus would significantly reduce flowering and seed production in these species. The monarch butterfly (Danaus plexippus) relies on Gomphocarpus fruticosus for successful development and is highly valued in New Zealand. It may be possible to genetically modify ArjMV to prevent transmission by insects but it is not an option because genetically modified organisms are not widely supported by the New Zealand public. The research efforts are now focusing on other potential agents for biocontrol of moth plant in New Zealand.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Amin L, Jahi JM, Nor ARM, Osman M, Mahadi NM (2007) Public Acceptance of Modern Biotechnology. Asia-Pacific Journal of Molecular Biology and Biotechnology 15, 39–51.
Anderson WP (1983) Weed Science Principles and Applications, 3rd edn. (West Publishing: St Paul, MN, USA)
Atreya CD, Raccah B, Pirone TP (1990) A point mutation in the coat protein abolishes aphid transmissibility of a potyvirus. Virology 178, 161–165. doi: 10.1016/0042-6822(90)90389-9
Charudattan R, Zettler FW, Cordo HA, Christie RG (1976) Susceptibility of the Florida milkweed vine, Morrenia odorata, to a potyvirus from Araujia angustifolia. Proceedings of the American Phytopathological Society 3, 272.
Charudattan R, Zettler FW, Cordo HA, Christie RG (1980) Partial characterization of a potyvirus infecting the milkweed vine, Morrenia odorata. Phytopathology 70, 909–913. doi: 10.1094/Phyto-70-909
Cook AJ, Fairweather JR, Sattersfield T, Hunt LM (2004) New Zealand Public Acceptance of Biotechnology. Available at http://www.lincoln.ac.nz/story_images/617_RR269AC_s2665.pdf [Verified July 5 2009]
Elliott MS, Zettler FW, Zimmerman MT, Barnett OW Jr, LeGrande MD (1996) Problems with interpretation of serological assays in a virus survey of orchid species from Puerto Rico, Ecuador, and Florida. Plant Disease 80, 1160–1164.
Endress ME, Bruyns PV (2000) A revised classification of the Apocynaceae s.l. Botanical Review 66, 1–56. doi: 10.1007/BF02857781
Endress ME, Liede-Schumann S, Meve U (2007) Advances in Apocynaceae: The enlightenment, an introduction. Annals of the Missouri Botanical Garden 94, 259–267. doi: 10.3417/0026-6493(2007)94[259:AIATEA] 2.0.CO;2
Flasinski S, Cassidy BG (1998) Potyvirus aphid transmission requires helper component and homologous coat protein for maximal efficiency. Archives of Virology 143, 2159–2172. doi: 10.1007/s007050050449
Forster PI, Bruyns PV (1992) Clarification of Synonymy for the Common Moth-Vine Araujia sericifera (Asclepiadaceae). Taxon 41, 746–749. doi: 10.2307/1222403
Gal-On A, Antignus Y, Rosner A, Raccah B (1992) A mutation in the coat protein gene of zucchini yellow mosaic virus restored its aphid transmissibility but had no effect on its multiplication. The Journal of General Virology 73, 2183–2187. doi: 10.1099/0022-1317-73-9-2183
Hiebert E, Charudattan R (1984) Characterization of Araujia mosaic virus by in vitro translation analyses. Phytopathology 74, 642–646. doi: 10.1094/Phyto-74-642
Kawakami K, Fuji S, Miyoshi K (2007) Endangered wild populations of endemic Calanthe orchids on an isolated Japanese island tested for viruses. Australian Journal of Botany 55, 831–836. doi: 10.1071/BT07093
Massey B, Cui X, Hiebert E, Elliott MS, Waipara N, Hayes L, Charudattan R (2007) Partial sequencing of the genomic RNA of Araujia mosaic virus and the comparison of the coat protein sequence with other potyviruses. Archives of Virology 152, 2125–2129. doi: 10.1007/s00705-007-1044-8
Medal JC, Sudbrink D, Gandolfo D, Ohashi D, Cuda JP (2002) Gratiana boliviana, a potential biocontrol agent of Solanum viarum: quarantine host-specificity testing in Florida and field surveys in South America. Biological Control 47, 445–461. Monarch Butterfly NZ Trust (2009) Available at www.monarch.org.nz/monarch/members/items-for-sale/seeds/ [Verified 5 July, 2009]
Ramsay GW (1964) Food of monarch butterfly (Danaus plexippus [L.]) larvae in New Zealand. Tuatara 12, 154.
Siegmann BJ, Elliott MS, Zettler FW (1998) Relative incidences of CYMMV, ORSV, and CYMRSV in commercial private and public orchid collections. Proceedings of the Florida State Horticultural Society 111, 41–43.
Sutula CL, Gillett JM, Morrissey SM, Ramsdell DC (1986) Interpreting ELISA data and establishing the positive — negative threshold. Plant Disease 70, 722–726. doi: 10.1094/PD-70-722
Waipara N (2006) Don’t cry for moth plant in Argentina. What’s New In Biocontrol of Weeds? Issue 36: 1–3, ISSN 1173-762X. Available at www.landcareresearch.co.nz/publications/newsletters/weeds/index.asp [Verified 5 July, 2009]
Waipara NW, Winks CJ, Gianotti AF, Villamil CB, Villamil SC, Delhey R, Kiehr M, Traversa MG, Carpintero DL (2006) Surveys for potential biocontrol agents for moth plant in New Zealand and Argentina. New Zealand Plant Protection 59, 18–22.
Winks CJ, Fowler SV (2000) Prospects for biological control of moth plant, Araujia sericifera. Unpublished Landcare Research Contract Report LC9900/100. Lincoln, New Zealand: Landcare Research. 18 p.
Wise KAJ (1963) Food-plants of ‘monarch’ butterfly larvae. Tuatara 11, 81–82. Available at http://www.nzetc.org/tm/scholarly/tei-Bio11-Tuat02-t1-body-d5.html
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Elliott, M.S., Massey, B., Cui, X. et al. Supplemental host range of Araujia mosaic virus, a potential biological control agent of moth plant in New Zealand. Australasian Plant Pathology 38, 603–607 (2009). https://doi.org/10.1071/AP09046
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1071/AP09046