Abstract
Accumulating evidences have demonstrated that lipopolysaccharide (LPS) represents the important etiologic factor for sepsis. Some previous studies have reported the relationship between common polymorphisms rs4986790 and rs4986791 in the coding gene for this receptor and the susceptibility to sepsis, but there were distinct divergences between those findings. We therefore designed this meta-analysis incorporated 28 published articles containing 6,537 sepsis patients and 8,832 controls for a more comprehensive conclusion on this matter. Odds ratios (ORs) and 95% confidence interval (95% CIs) were calculated to evaluate the association of toll like receptor 4 gene polymorphisms rs4986790 and rs4986791 with sepsis risk. Heterogeneity between included studies was inspected using Q test, and sensitivity analysis was implemented via sequential deletion of each included study to investigate the stability of overall estimates. Funnel plot and Egger’s test were adopted to examine publication bias across selected studies. We found no significant association for either the polymorphism rs4986790 or rs4986791 with sepsis susceptibility in total analysis under any genetic models. Neither did we after combining these two polymorphisms. The results of this meta-analysis suggest that the rs4986790 and rs4986791 polymorphisms in toll like receptor 4 gene may have no statistically significant influence on sepsis susceptibility.
Similar content being viewed by others
Introduction
Sepsis is a systemic inflammatory response syndrome (SIRS) induced by organism infections from pathogenic microorganisms1. Organisms identify pathogen microorganisms through innate immunity system, thereby starting protective inflammatory response to eliminate pathogenic microorganisms, but over-response of systemic inflammation in organism may cause sepsis or septic shock2. Thousands of patients undergo target organ damage and multiple organ dysfunction caused by severe sepsis and septic shock annually, and the death rate of sepsis is still increasing constantly despite the development of medical condition and technology3. Although the precise etiology of sepsis remains unclear, relevant studies have shown that gene polymorphism plays important role in affecting individual susceptibility to sepsis4,5,6. Consequently, key gene polymorphisms involved in immune response pathways have been detected their influence on sepsis onset7,8.
Innate immune system depends on pattern recognition receptors (PRRs) to detect conserved structures of pathogenic microorganisms (like bacteria, virus, fungus and protozoa), which are called pathogen-associated molecular patterns (PAMPs)9. Toll like receptors (TLRs) play an important role in autoimmunity, and one of them, toll like receptor 4, occupies an extremely significant position as a receptor recognizing PAMPs10. Reportedly, polymorphisms in the gene coding for this receptor may change the extracellular structures of the protein and affect the combinations with pathogen ligands, especially the combination with lipopolysaccharide (LPS) of gram negative bacteria11, thereby changing the progression of sepsis. Studies have demonstrated that toll like receptor 4 gene polymorphism rs4986790 (also known as Asp299Gly or +896 A > G) increased the risk of severe sepsis12,13. In addition, Shalhub et al. indicated that apart from the polymorphism rs4986790, another one rs4986791 (also known as Thr399Ile) in this gene was also related to the severity of post-traumatic sepsis14. Whereas Jessen et al. showed that there were no obvious association between polymorphisms in the gene toll like receptor 4 and sepsis caused by gram negative bacteria in their study15. Feterowski et al. found that the morbidity and mortality rate of sepsis didn’t correlate with toll like receptor 4 gene mutations in their study on postoperative sepsis induced by various microorganism infections16.
It is important in terms of clinical value and theoretical significance to get clearer perspective on the relationship of toll like receptor 4 gene polymorphisms rs4986790 and rs4986791 with sepsis risk. Consequently, we systematically analyzed this correlation via the method of meta-analysis in this study.
Materials and Methods
Literature search and selection criteria
We searched potentially relevant studies from MEDLINE, EMBASE, Google Scholar and CNKI up to January 2016. The key terms adopted in search strategy included “toll like receptor 4 or TLR4”, “polymorphism or mutation or variant” and “sepsis”. What’s more, we also searched for additional articles through manually screening the reference lists of relevant articles or reviews to minimize the possibility of missing potent papers. Eligible studies were enrolled in this meta-analysis based on the following criteria: they estimated the correlation of the rs4986790 and/or rs4986791 polymorphisms in toll like receptor 4 gene with sepsis and had a case-control design; they adopted valid genotyping method; and they provided information about genotype distribution in case and control group for calculating pooled odds ratios (ORs) with the corresponding 95% confidence intervals (95% CIs). Meanwhile, publications were excluded if they were case-only studies, with irrelevant title or abstract, without sufficient genotype data or focusing on animals.
Data extraction
The following information were extracted by two investigators from included articles: the first author’s name, publication year, original country, ethnicity, control source, genotyping method, numbers of cases and controls, genotype frequency and P values for Hardy-Weinberg equilibrium (HWE) in control group. All disagreements over extracted data were resolved through discussion between the two investigators to reach a consensus.
Statistical analysis
The ORs and 95% CIs were calculated to assess the association of sepsis susceptibility with toll like receptor 4 rs4986790 polymorphism under five genetic models: GG vs. AA, GG + GA vs. AA, GG vs. GA+AA, allele G vs. allele A, and GA vs. GG, as well as with the polymorphism rs4986791 under the five genetic models: TT vs. CC, TT+CT vs. CC, TT vs. CC+CT, allele T vs. allele C and CT vs. CC. In addition, subgroup analysis was implemented according to ethnicity. Between-study heterogeneity was evaluated through the χ2-based Q test. The fixed-effect model was adopted to calculate the pooled ORs when P > 0.05 in Q test which indicated the lack of heterogeneity; otherwise, the random-effect model was selected. HWE was checked in the control group via χ2 test. Publication bias was examined with Begg’s funnel plot and Egger’s test. Sensitivity analysis was performed to reflect the effect of each individual dataset on pooled results through sequentially omitting each included study. Statistical analysis was conducted using STATA version 12.0 (STATA Corp). P < 0.05 was considered statistically significant for all tests.
Results
Characteristics of studies
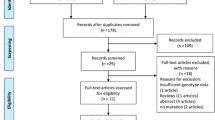
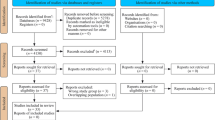
According to the search strategy, 277 potentially relevant studies were initially identified from MEDLINE, EMBASE, Google Scholar and CNKI, and 163 of them were exclude due to irrelevant titles or abstracts. Additionally, consequent screening removed 86 more reports for not concerning on the association between the polymorphism rs4986790 or rs4986791 of toll like receptor 4 and sepsis (71) and insufficient data (15). Therefore, as showed in Fig. 1, this meta-analysis finally included 28 eligible articles, containing 51 independent case-control studies (31 for the polymorphism rs4986790 and 20 for rs4986791)13,14,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41. The characteristics of included studies are summarized in Table 1. Apart from studies not providing sufficient information for χ2 test, genotype distributions in controls were consistent with HWE in all studies for the polymorphism rs4986791 (P > 0.05, with a range of 0.3620 to 0.8560); while such distributions deviated from HWE expectation in two studies from the report by Tellería-Orriols et al.20 for the polymorphism rs4986790 (P = 0.0001), and perfectly fit in with the expectation in the other studies with P values ranging from 0.1690 to 0.9620.
Meta-analysis results
In total analysis, the polymorphism rs4986790 of toll like receptor 4 gene showed no significant association with sepsis susceptibility under genetic models GG vs. AA (OR = 0.87, 95% CI = 0.53–1.43) (Fig. 2), GG + AG vs. AA (OR = 1.03, 95% CI = 0.83-1.29) (Fig. 3), GG vs. AA + AG (OR = 0.85, 95% CI = 0.52–1.41) (Fig. 4), allele G vs. allele A (OR = 1.03, 95% CI = 0.81–1.31) (Fig. 5) and AG vs. AA (OR = 1.05, 95% CI=0.83–1.33) (Fig. 6). Whereas, after stratification analysis by ethnicity, an increasing effect thereof on the sepsis risk was uncovered in other-ethnicity subgroup under GG + AG vs. AA (OR = 1.59 95% CI = 1.15–2.20) (Fig. 7), allele G vs. allele A (OR = 1.54 95% CI = 1.06–2.23) and AG vs. AA (OR = 1.5695% CI = 1.09–2.24) comparisons.
As for the polymorphism rs4986791, no significant influence thereof was detected in total analysis under the contrasts of TT vs. CC (Fig. 2), TT + CT vs. CC (Fig. 3), TT vs. CC + CT (Fig. 4), allele T vs. allele C (Fig. 5) and CT vs. CC (Fig. 6) (OR = 0.55, 95% CI = 0.20–1.50; OR = 1.05, 95% CI = 0.77–1.41; OR = 0.56, 95% CI = 0.20–1.51; OR = 1.08, 95% CI = 0.79–1.48; OR = 1.12, 95% CI = 0.82–1.52) either. However, this polymorphism significantly elevated the sepsis susceptibility in other-ethnicity subgroup under TT + CT vs. CC (Fig. 8), allele T vs. allele C and CT vs. CC models (OR = 2.10, 95% CI = 1.33–3.30; OR = 2.08, 95% CI = 1.34–3.22; OR = 2.13, 95% CI = 1.35–3.36) after stratified analysis by ethnicity.
In addition, we also examined possible effects of the combination between the polymorphisms rs4986790 and rs4986791 on the sepsis onset. As a result, the combination of these two polymorphism still lacked significant impact on the disease risk under homozygous model (OR = 0.79, 95% CI = 0.51–1.23) (Fig. 2), dominant model (OR = 1.04, 95% CI = 0.87–1.24) (Fig. 3), recessive model (OR = 0.78, 95% CI = 0.50–1.22) (Fig. 4), allele model (OR = 1.05, 95% CI = 0.87–1.26) (Fig. 5) and heterozygous model (OR = 1.07, 95% CI = 0.89–1.29) (Fig. 6).
Test of heterogeneity
Significant heterogeneity was observed for the polymorphism rs4986790 under GG + GA vs. AA model, allele G vs. allele A model and GA vs. GG model (P = 0.001; P < 0.001; P = 0.002) as well as for the polymorphism rs4986791 under TT + CT vs. CC, allele T vs. allele C and CT vs. CC contrasts (P = 0.017; P = 0.008; P = 0.034), thus the random-effects model was applied to calculate pooled ORs in these cases. As for the other comparisons, the fixed-effects model was utilized considering the absence of significant heterogeneity (for rs4986790: P = 0.904 under GG vs. AA; P = 0.935 under GG + AG vs. AA; for rs4986791: P = 0.785 under TT vs. CC; P = 0.812 under TT + CT vs. CC).
As for the OR values for the effects of the combination between the two polymorphism, they were summarized applying the random-effects model under the dominant, allele and heterozygous models (P < 0.001; P < 0.001; P = 0.001) in view of the existence of significant heterogeneity, and pooled with the fixed-effects model under the homozygous and recessive models (P = 0.954; P = 0.972) because of the lack of significant heterogeneity.
Sensitivity analysis and publication bias
We implemented sensitivity analysis to evaluate the stability of pooled results. We excluded each study one by one and observed that no pooled ORs was substantially affected (Fig. 9, with a range of lower CI from 0.42 to 0.56 and of upper CI from 1.30–1.57), indicating high stability of the meta-analysis results. Additionally, Begg’s funnel plot and Egger’s test were employed to evaluate publication bias between included studies. Consequently, all funnel plots seemed symmetrical (Fig. 10), implying no significant publication bias. What’s more, Egger’s test showed statistical evidence for these results (P = 0.674).
Discussion
Sepsis is caused by infections and characterized by acute onset, rapid progression and high fatality rate, being a common postoperative complication of severe traumas and burns. SIRS may develop to septic stock and multiple organ dysfunction syndrome (MODS), and studies on SIRS, sepsis, severe sepsis, septic stock and MODS indicate significant differences in different individuals. Specifically, inflammatory responses may develop easily in some people, which is difficult to be controlled and then become MODS, while different results may occur in other people with the same SIRS. Moreover, people with similar SIRS or sepsis can show different prognosis when they adopt same therapeutic measures. All these evidence indicate that sepsis is affected by genetic factors as well. Therefore, it’s important to explore the roles of sepsis-related genes, which provides theoretical basis to understand sepsis pathogenesis.
TLRs, a group of main PRRs, can recognize pathogenic microorganisms through PAMPs, activate intracellular signal transduction pathways and induce the generation of innate immunity. Therefore, TLRs are involved in the pathogenic courses of numerous diseases and closely correlated with communicable diseases, tumors, cardiovascular diseases, autoimmune diseases and allergy. According to their positions in chromosome, genetic structures and amino acid sequences, 11 members of TLRs family are divided to 5 subfamilies. Among members in this family, toll like receptor 4, first found and reported by Medzhitov et al. in 199742, is a transmembrane protein producing homologization to drosophila toll protein which is composed of extracellular region, transmembrane domain and intracellular region of leucine-rich-repeat (LRR), distributing mainly on the surface of cells (such as monocyte). This protein can recognize LPS of gram negative bacteria, mannan of fungus and soluble components of mycobacterium tuberculosis, and is the endogenous ligand of some heat shock proteins and fibronectins. Therefore, polymorphisms in coding gene for the protein may greatly affect systemic inflammation and immunoreaction.
For example, Chen et al. found that the polymorphism T-2242C in toll like receptor 4 gene might be related to higher sepsis morbidity rate and organ dysfunction43. Besides, Mansur et al. put forward in their study that the polymorphism rs11536889 in this gene was associated with renal, coagulation and hepatic organ failure in sepsis patients44. In addition, a study by Nachtigall et al. reported that the polymorphism rs4986790 might shorten the time-to-onset of severe sepsis or septic shock in patients permitted to intensive care units45. In the study by Child et al., the polymorphism rs4986790 was demonstrated to be involved in the severity of SIRS46. Additionally, this polymorphism was found to be correlated with septic shock induced by gram-negative bacteria33. However, there was research suggesting that such mutation might not influence the incidence of postoperative sepsis16. Such an inconclusive status was also true for another common polymorphism in the toll like receptor 4 gene, rs4976891. And these discrepancies might be attributed to but not limited to such aspects as various genetic backgrounds, different selection criteria for participants and uneven sample sizes.
Considering those discrepancies, we performed this meta-analysis to comprehensively analyze the associations of the rs4986790 and rs4986791 polymorphisms in toll like receptor 4 gene with sepsis susceptibility involving 51 relevant case-control studies. The results of this study showed that our studied polymorphisms, overall, had no significant association with the susceptibility of sepsis, but that both polymorphisms were related to increased risk of developing sepsis in other-ethnicity subgroups under corresponding genetic models after stratified analysis by ethnicity. Based on 6,537 sepsis patients and 8,832 controls, our findings had certain reliability, and sensitivity analysis also verified their stability. Compared to the previous individual case-control studies, our meta-analysis had a larger sample size and a more detailed stratification analysis by ethnicity, so our results were more reliable and comprehensive. In a recent meta-analysis about rs4986790 polymorphsism and sepsis susceptibility published by Zhu et al. in 201247, 17 eligible articles were included, while our meta-analysis collected 28 related ones. Therefore, the present meta-analysis was an up-dated one with more eligible studies, suggesting our results could represent latest research results. However, there were some limitations in this meta-analysis, which might affect the accuracy of the result. For example, source limitation in literature searching might miss some relevant articles in other sources, thus leading to possible publication bias not detected even with Begg’s funnel plot or Egger’s test. In addition, possible effects from other relevant factors and gene-gene or gene-environment interactions were not incorporated in this study due to the insufficient data. Therefore, the results of the present meta-analysis should be applied with cautious.
In conclusion, the results of this meta-analysis showed that toll like receptor 4 gene rs4986790 and rs4986791 polymorphisms might not have independent association with sepsis susceptibility. In view of the advantages and disadvantages in this meta-analysis, the results should be further verified by better-designed studies based on larger sample size and more consideration of gene-gene and gene-environment interactions.
Additional Information
How to cite this article: Liu, R. et al. The relationship between toll like receptor 4 gene rs4986790 and rs4986791 polymorphisms and sepsis susceptibility: A meta-analysis. Sci. Rep. 6, 38947; doi: 10.1038/srep38947 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Dombrovskiy, V. Y., Martin, A. A., Sunderram, J. & Paz, H. L. Rapid increase in hospitalization and mortality rates for severe sepsis in the United States: a trend analysis from 1993 to 2003. Crit Care Med 35, 1244–50 (2007).
Liang, S. Y. & Kumar, A. Empiric antimicrobial therapy in severe sepsis and septic shock: optimizing pathogen clearance. Curr Infect Dis Rep 17, 493 (2015).
Gatewood, M. O., Wemple, M., Greco, S., Kritek, P. A. & Durvasula, R. A quality improvement project to improve early sepsis care in the emergency department. BMJ Qual Saf 24, 787–95 (2015).
Baghel, K. et al. TNF-alpha, IL-6, and IL-8 cytokines and their association with TNF-alpha-308 G/A polymorphism and postoperative sepsis. J Gastrointest Surg 18, 1486–94 (2014).
Jaskula, E. et al. NOD2/CARD15 single nucleotide polymorphism 13 (3020insC) is associated with risk of sepsis and single nucleotide polymorphism 8 (2104C > T) with herpes viruses reactivation in patients after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 20, 409–14 (2014).
Feng, B., Mao, Z. R., Pang, K., Zhang, S. L. & Li, L. Association of tumor necrosis factor alpha -308G/A and interleukin-6 -174G/C gene polymorphism with pneumonia-induced sepsis. Journal of critical care, doi: 10.1016/j.jcrc.2015.04.123 (2015).
Kimura, T. et al. Autophagy-related IRGM polymorphism is associated with mortality of patients with severe sepsis. PLoS One 9, e91522 (2014).
Adamzik, M. et al. The NFKB1 promoter polymorphism (-94ins/delATTG) alters nuclear translocation of NF-kappaB1 in monocytes after lipopolysaccharide stimulation and is associated with increased mortality in sepsis. Anesthesiology 118, 123–33 (2013).
Akira, S. & Hemmi, H. Recognition of pathogen-associated molecular patterns by TLR family. Immunol Lett 85, 85–95 (2003).
Chebrolu, C. et al. Species and mediator specific TLR4 antagonism in primary human and murine immune cells by betaGlcN(1<–>1)alphaGlc based lipid A mimetics. Mol Immunol 67, 636–41 (2015).
Nunez Miguel, R. et al. A dimer of the Toll-like receptor 4 cytoplasmic domain provides a specific scaffold for the recruitment of signalling adaptor proteins. PLoS One 2, e788 (2007).
Barber, R. C. et al. Innate immunity SNPs are associated with risk for severe sepsis after burn injury. Clin Med Res 4, 250–5 (2006).
Barber, R. C. et al. TLR4 and TNF-alpha polymorphisms are associated with an increased risk for severe sepsis following burn injury. J Med Genet 41, 808–13 (2004).
Shalhub, S. et al. Variation in the TLR4 gene influences the risk of organ failure and shock posttrauma: a cohort study. J Trauma 66, 115–22, discussion 122–3 (2009).
Jessen, K. M., Lindboe, S. B., Petersen, A. L., Eugen-Olsen, J. & Benfield, T. Common TNF-alpha, IL-1 beta, PAI-1, uPA, CD14 and TLR4 polymorphisms are not associated with disease severity or outcome from Gram negative sepsis. BMC Infect Dis 7, 108 (2007).
Feterowski, C. et al. Effects of functional Toll-like receptor-4 mutations on the immune response to human and experimental sepsis. Immunology 109, 426–31 (2003).
Ahmad-Nejad, P. et al. The presence of functionally relevant toll-like receptor polymorphisms does not significantly correlate with development or outcome of sepsis. Genet Test Mol Biomarkers 15, 645–51 (2011).
Carregaro, F. et al. Polymorphisms IL10-819 and TLR-2 are potentially associated with sepsis in Brazilian patients. Mem Inst Oswaldo Cruz 105, 649–56 (2010).
Sampath, V. et al. Toll-like receptor genetic variants are associated with Gram-negative infections in VLBW infants. J Perinatol 33, 772–7 (2013).
Telleria-Orriols, J. J., Garcia-Salido, A., Varillas, D., Serrano-Gonzalez, A. & Casado-Flores, J. TLR2-TLR4/CD14 polymorphisms and predisposition to severe invasive infections by Neisseria meningitidis and Streptococcus pneumoniae. Med Intensiva 38, 356–62 (2014).
Schnetzke, U. et al. Polymorphisms of Toll-like receptors (TLR2 and TLR4) are associated with the risk of infectious complications in acute myeloid leukemia. Genes Immun 16, 83–8 (2015).
Nakada, T. A. et al. Influence of toll-like receptor 4, CD14, tumor necrosis factor, and interleukine-10 gene polymorphisms on clinical outcome in Japanese critically ill patients. J Surg Res 129, 322–8 (2005).
Mensah, N. Y. et al. Toll-like receptor 4 polymorphisms and risk of gram-negative bacteremia after allogeneic stem cell transplantation. A prospective pilot study. Biol Blood Marrow Transplant 15, 1130–3 (2009).
Yoon, H. J. et al. Lack of Toll-like receptor 4 and 2 polymorphisms in Korean patients with bacteremia. J Korean Med Sci 21, 979–82 (2006).
Kompoti, M. et al. Genetic polymorphisms of innate and adaptive immunity as predictors of outcome in critically ill patients. Immunobiology 220, 414–21 (2015).
Elkilany, A. et al. Toll-like receptors (TLR) 2, 3, and 4 gene polymorphisms in critically ill patients. Archives of Biological Sciences 67, 261–273 (2015).
Bronkhorst, M. W. et al. Single-nucleotide polymorphisms in the Toll-like receptor pathway increase susceptibility to infections in severely injured trauma patients. J Trauma Acute Care Surg 74, 862–70 (2013).
Rodriguez-Osorio, C. A. et al. Genetic variations in toll-like receptor 4 in Mexican-Mestizo patients with intra-abdominal infection and/or pneumonia. Immunol Lett 153, 41–6 (2013).
Horcajada, J. P. et al. Polymorphic receptors of the innate immune system (MBL/MASP-2 and TLR2/4) and susceptibility to pneumococcal bacteremia in HIV-infected patients: a case-control study. Curr HIV Res 7, 218–23 (2009).
Özgür, T. T. et al. Lack of association between TLR4 polymorphism and severe gram-negative bacterial infection in neonates. Turk J Med Sci 39, 423–427 (2009).
Read, R. C. et al. A functional polymorphism of toll-like receptor 4 is not associated with likelihood or severity of meningococcal disease. J Infect Dis 184, 640–2 (2001).
Agnese, D. M. et al. Human toll-like receptor 4 mutations but not CD14 polymorphisms are associated with an increased risk of gram-negative infections. J Infect Dis 186, 1522–5 (2002).
Lorenz, E., Mira, J. P., Frees, K. L. & Schwartz, D. A. Relevance of mutations in the TLR4 receptor in patients with gram-negative septic shock. Arch Intern Med 162, 1028–32 (2002).
Smirnova, I. et al. Assay of locus-specific genetic load implicates rare Toll-like receptor 4 mutations in meningococcal susceptibility. Proc Natl Acad Sci USA 100, 6075–80 (2003).
Guarner-Argente, C. et al. Toll-like receptor 4 D299G polymorphism and the incidence of infections in cirrhotic patients. Aliment Pharmacol Ther 31, 1192–9 (2010).
Ahrens, P. et al. Mutations of genes involved in the innate immune system as predictors of sepsis in very low birth weight infants. Pediatr Res 55, 652–6 (2004).
Van der Graaf, C. A. et al. Toll-like receptor 4 Asp299Gly/Thr399Ile polymorphisms are a risk factor for Candida bloodstream infection. Eur Cytokine Netw 17, 29–34 (2006).
Everett, B. et al. Polymorphisms in Toll-like receptors-2 and -4 are not associated with disease manifestations in acute Q fever. Genes Immun 8, 699–702 (2007).
Yuan, F. F. et al. Clinical relevance of TLR2, TLR4, CD14 and FcgammaRIIA gene polymorphisms in Streptococcus pneumoniae infection. Immunol Cell Biol 86, 268–70 (2008).
Henckaerts, L. et al. Polymorphisms in innate immunity genes predispose to bacteremia and death in the medical intensive care unit. Crit Care Med 37, 192–201, e1-3 (2009).
Kumpf, O. et al. Influence of genetic variations in TLR4 and TIRAP/Mal on the course of sepsis and pneumonia and cytokine release: an observational study in three cohorts. Crit Care 14, R103 (2010).
Medzhitov, R., Preston-Hurlburt, P. & Janeway, C. A., Jr. A human homologue of the Drosophila Toll protein signals activation of adaptive immunity. Nature 388, 394–7 (1997).
Chen, K. et al. Functional significance of the Toll-like receptor 4 promoter gene polymorphisms in the Chinese Han population. Crit Care Med 38, 1292–9 (2010).
Mansur, A. et al. The regulatory toll-like receptor 4 genetic polymorphism rs11536889 is associated with renal, coagulation and hepatic organ failure in sepsis patients. J Transl Med 12, 177 (2014).
Nachtigall, I. et al. Polymorphisms of the toll-like receptor 2 and 4 genes are associated with faster progression and a more severe course of sepsis in critically ill patients. J Int Med Res 42, 93–110 (2014).
Child, N. J. et al. Polymorphisms in Toll-like receptor 4 and the systemic inflammatory response syndrome. Biochem Soc Trans 31, 652–3 (2003).
Zhu L., Li, X. & Miao, C. Lack of association between TLR4 Asp299Gly and Thr399lle polymorphisms and sepsis susceptibility: a meta-analysis. Gene. Jun 15; 501(2), 213–8 (2012).
Acknowledgements
The General Financial Grant from the China Postdoctoral Science Foundation: No. 2015M572749. The Science and Technology Foundation of Chongqing, China: No. cstc2014jcyjA1217 and cstc2012gg-yyjs10010. The Major State Basic Research Development Program of China (973 Program): No. 2014CB541605.
Author information
Authors and Affiliations
Contributions
R.L. and L.L. conceived and designed the experiments; Y.M. and H.W. conceived and performed the experiments; Y.T. and X.W. prepared figures. M.D. and H.Y. wrote the main manuscript text. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Liu, R., Mo, YY., Wang, HL. et al. The relationship between toll like receptor 4 gene rs4986790 and rs4986791 polymorphisms and sepsis susceptibility: A meta-analysis. Sci Rep 6, 38947 (2016). https://doi.org/10.1038/srep38947
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep38947
- Springer Nature Limited