Abstract
Promoting brown adipose tissue (BAT) activity innovatively targets obesity and metabolic disease. While thermogenic activation of BAT is well understood, the rheostatic regulation of BAT to avoid excessive energy dissipation remains ill-defined. Here, we demonstrate that adenylyl cyclase 3 (AC3) is key for BAT function. We identified a cold-inducible promoter that generates a 5′ truncated AC3 mRNA isoform (Adcy3-at), whose expression is driven by a cold-induced, truncated isoform of PPARGC1A (PPARGC1A-AT). Male mice lacking Adcy3-at display increased energy expenditure and are resistant to obesity and ensuing metabolic imbalances. Mouse and human AC3-AT are retained in the endoplasmic reticulum, unable to translocate to the plasma membrane and lack enzymatic activity. AC3-AT interacts with AC3 and sequesters it in the endoplasmic reticulum, reducing the pool of adenylyl cyclases available for G-protein-mediated cAMP synthesis. Thus, AC3-AT acts as a cold-induced rheostat in BAT, limiting adverse consequences of cAMP activity during chronic BAT activation.







Similar content being viewed by others
Data availability
Illumina datasets from RT and cold-activated eWAT and iBAT are available at the Gene Expression Omnibus under accession GSE212574. H3K4me3 ChIP–seq data from BAT of chow diet-fed and HFD-fed male C57BL/6N mice housed at 22 °C or exposed to 5 °C for 24 h were downloaded from the Gene Expression Omnibus (GSE20065). snRNA-seq data from Adipoq-tdTomato-positive adipocyte nuclei were downloaded from ArrayExpress (E-MTAB-8562). All immunofluorescence images have been uploaded on figshare https://doi.org/10.6084/m9.figshare.c.6725808.v1Source data are provided with this paper.
References
Kusminski, C. M., Bickel, P. E. & Scherer, P. E. Targeting adipose tissue in the treatment of obesity-associated diabetes. Nat. Rev. Drug Discov. 15, 639–660 (2016).
Cannon, B. & Nedergaard, J. Brown adipose tissue: function and physiological significance. Physiol. Rev. 84, 277–359 (2004).
van Marken Lichtenbelt, W. D. et al. Cold-activated brown adipose tissue in healthy men. N. Engl. J. Med. 360, 1500–1508 (2009).
Cypess, A. M. et al. Identification and importance of brown adipose tissue in adult humans. N. Engl. J. Med. 360, 1509–1517 (2009).
Virtanen, K. A. et al. Functional brown adipose tissue in healthy adults. N. Engl. J. Med. 360, 1518–1525 (2009).
Yoneshiro, T. et al. Recruited brown adipose tissue as an antiobesity agent in humans. J. Clin. Invest. 123, 3404–3408 (2013).
Cypess, A. M. et al. Activation of human brown adipose tissue by a beta3-adrenergic receptor agonist. Cell Metab. 21, 33–38 (2015).
Mueller, E. Browning and graying: novel transcriptional regulators of brown and beige fat tissues and aging. Front. Endocrinol. 7, 19 (2016).
Marti-Solano, M. et al. Combinatorial expression of GPCR isoforms affects signalling and drug responses. Nature 587, 650–656 (2020).
Ceddia, R. P. & Collins, S. A compendium of G-protein-coupled receptors and cyclic nucleotide regulation of adipose tissue metabolism and energy expenditure. Clin. Sci. 134, 473–512 (2020).
Kajimura, S., Spiegelman, B. M. & Seale, P. Brown and beige fat: physiological roles beyond heat generation. Cell Metab. 22, 546–559 (2015).
Blondin, D. P. et al. Human brown adipocyte thermogenesis is driven by beta2-AR stimulation. Cell Metab. 32, 287–300 (2020).
Khannpnavar, B., Mehta, V., Qi, C. & Korkhov, V. Structure and function of adenylyl cyclases, key enzymes in cellular signaling. Curr. Opin. Struct. Biol. 63, 34–41 (2020).
Reverte-Salisa, L., Sanyal, A. & Pfeifer, A. Role of cAMP and cGMP signaling in brown fat. Handb. Exp. Pharmacol. 251, 161–182 (2019).
Wu, L., Shen, C., Seed Ahmed, M., Ostenson, C. G. & Gu, H. F. Adenylate cyclase 3: a new target for anti-obesity drug development. Obes. Rev. 17, 907–914 (2016).
Stergiakouli, E. et al. Genome-wide association study of height-adjusted BMI in childhood identifies functional variant in ADCY3. Obesity 22, 2252–2259 (2014).
Saeed, S. et al. Loss-of-function mutations in ADCY3 cause monogenic severe obesity. Nat. Genet. 50, 175–179 (2018).
Grarup, N. et al. Loss-of-function variants in ADCY3 increase risk of obesity and type 2 diabetes. Nat. Genet. 50, 172–174 (2018).
Toumba, M. et al. Molecular modelling of novel ADCY3 variant predicts a molecular target for tackling obesity. Int. J. Mol. Med. 49, 10 (2022).
Tong, T., Shen, Y., Lee, H. W., Yu, R. & Park, T. Adenylyl cyclase 3 haploinsufficiency confers susceptibility to diet-induced obesity and insulin resistance in mice. Sci. Rep. 6, 34179 (2016).
Wang, Z. et al. Adult type 3 adenylyl cyclase-deficient mice are obese. PLoS ONE 4, e6979 (2009).
Wong, S. T. et al. Disruption of the type III adenylyl cyclase gene leads to peripheral and behavioral anosmia in transgenic mice. Neuron 27, 487–497 (2000).
Pitman, J. L. et al. A gain-of-function mutation in adenylate cyclase 3 protects mice from diet-induced obesity. PLoS ONE 9, e110226 (2014).
Chen, X. et al. Ablation of type III adenylyl cyclase in mice causes reduced neuronal activity, altered sleep pattern, and depression-like phenotypes. Biol. Psychiatry 80, 836–848 (2016).
Chao, Y. et al. Regulatory roles and mechanisms of alternative RNA splicing in adipogenesis and human metabolic health. Cell Biosci. 11, 66 (2021).
Lin, J. C., Lu, Y. H., Liu, Y. R. & Lin, Y. J. RBM4a-regulated splicing cascade modulates the differentiation and metabolic activities of brown adipocytes. Sci. Rep. 6, 20665 (2016).
Vernia, S. et al. An alternative splicing program promotes adipose tissue thermogenesis. eLife 5, e17672 (2016).
Engelhard, C. A., Khani, S., Derdak, S., Bilban, M. & Kornfeld, J. W. Nanopore sequencing unveils the complexity of the cold-activated murine brown adipose tissue transcriptome. iScience 26, 107190 (2023).
Ruas, J. L. et al. A PGC-1α isoform induced by resistance training regulates skeletal muscle hypertrophy. Cell 151, 1319–1331 (2012).
Jannig, P. R., Dumesic, P. A., Spiegelman, B. M. & Ruas, J. L. SnapShot: regulation and biology of PGC-1α. Cell 185, 1444 (2022).
Li, Y. et al. Comparative transcriptome profiling of cold exposure and beta3-AR agonist CL316,243-induced browning of white fat. Front. Physiol. 12, 667698 (2021).
Omori, K. & Kotera, J. Overview of PDEs and their regulation. Circ. Res. 100, 309–327 (2007).
Granneman, J. G. Expression of adenylyl cyclase subtypes in brown adipose tissue: neural regulation of type III. Endocrinology 136, 2007–2012 (1995).
Son, Y. et al. REEP6 knockout leads to defective beta-adrenergic signaling in adipocytes and promotes obesity-related metabolic dysfunction. Metabolism 130, 155159 (2022).
Sun, W. et al. snRNA-seq reveals a subpopulation of adipocytes that regulates thermogenesis. Nature 587, 98–102 (2020).
Perdikari, A. et al. BATLAS: deconvoluting brown adipose tissue. Cell Rep. 25, 784–797 (2018).
Eguchi, J. et al. Transcriptional control of adipose lipid handling by IRF4. Cell Metab. 13, 249–259 (2011).
Hamann, A., Flier, J. S. & Lowell, B. B. Decreased brown fat markedly enhances susceptibility to diet-induced obesity, diabetes, and hyperlipidemia. Endocrinology 137, 21–29 (1996).
Li, F. et al. Epigenetic interaction between UTX and DNMT1 regulates diet-induced myogenic remodeling in brown fat. Nat. Commun. 12, 6838 (2021).
Schmidt, E. et al. LincRNA H19 protects from dietary obesity by constraining expression of monoallelic genes in brown fat. Nat. Commun. 9, 3622 (2018).
Oliverio, M. et al. Dicer1-miR-328-Bace1 signalling controls brown adipose tissue differentiation and function. Nat. Cell Biol. 18, 328–336 (2016).
Feldmann, H. M., Golozoubova, V., Cannon, B. & Nedergaard, J. UCP1 ablation induces obesity and abolishes diet-induced thermogenesis in mice exempt from thermal stress by living at thermoneutrality. Cell Metab. 9, 203–209 (2009).
Cao, W. et al. p38 mitogen-activated protein kinase is the central regulator of cyclic AMP-dependent transcription of the brown fat uncoupling protein 1 gene. Mol. Cell. Biol. 24, 3057–3067 (2004).
Lindquist, J. M. & Rehnmark, S. Ambient temperature regulation of apoptosis in brown adipose tissue. Erk1/2 promotes norepinephrine-dependent cell survival. J. Biol. Chem. 273, 30147–30156 (1998).
Hattori, K. et al. beta-adrenergic receptor signaling evokes the PKA-ASK axis in mature brown adipocytes. PLoS ONE 15, e0232645 (2020).
Ji, H. et al. CDK7 mediates the beta-adrenergic signaling in thermogenic brown and white adipose tissues. iScience 23, 101163 (2020).
Roh, H. C. et al. Warming induces significant reprogramming of beige, but not brown, adipocyte cellular identity. Cell Metab. 27, 1121–1137 (2018).
Pan, D. et al. Jmjd3-mediated H3K27me3 dynamics orchestrate brown fat development and regulate white fat plasticity. Dev. Cell 35, 568–583 (2015).
Brunmeir, R. et al. Comparative transcriptomic and epigenomic analyses reveal new regulators of murine brown adipogenesis. PLoS Genet. 12, e1006474 (2016).
Engelhard, C. A. et al. Comprehensive transcriptional profiling and mouse phenotyping reveals dispensable role for adipose tissue selective long noncoding RNA Gm15551. Noncoding RNA 8, 32 (2022).
Darcy, J. & Tseng, Y. H. ComBATing aging-does increased brown adipose tissue activity confer longevity? Geroscience 41, 285–296 (2019).
Crossthwaite, A. J., Ciruela, A., Rayner, T. F. & Cooper, D. M. A direct interaction between the N terminus of adenylyl cyclase AC8 and the catalytic subunit of protein phosphatase 2A. Mol. Pharmacol. 69, 608–617 (2006).
Ding, Q., Gros, R., Chorazyczewski, J., Ferguson, S. S. & Feldman, R. D. Isoform-specific regulation of adenylyl cyclase function by disruption of membrane trafficking. Mol. Pharmacol. 67, 564–571 (2005).
Freeze, H. H. & Kranz, C. Endoglycosidase and glycoamidase release of N-linked glycans. Curr. Protoc. Immunol. 8, 8.15.1–8.15.26 (2010).
Lang, T. Imaging SNAREs at work in ‘unroofed’ cells–approaches that may be of general interest for functional studies on membrane proteins. Biochem. Soc. Trans. 31, 861–864 (2003).
Lin, J., Handschin, C. & Spiegelman, B. M. Metabolic control through the PGC-1 family of transcription coactivators. Cell Metab. 1, 361–370 (2005).
Huang, P. I. et al. PGC-1α mediates differentiation of mesenchymal stem cells to brown adipose cells. J. Atheroscler. Thromb. 18, 966–980 (2011).
Liu, Z. et al. N-terminal truncated peroxisome proliferatoractivated receptor gamma coactivator 1 alpha alleviates phenylephrineinduced mitochondrial dysfunction and decreases lipid droplet accumulation in neonatal rat cardiomyocytes. Mol. Med. Rep. 18, 2142–2152 (2018).
Zhang, J. W., Klemm, D. J., Vinson, C. & Lane, M. D. Role of CREB in transcriptional regulation of CCAAT/enhancer-binding protein beta gene during adipogenesis. J. Biol. Chem. 279, 4471–4478 (2004).
Zhang, Y. et al. Alternative mRNA splicing produces a novel biologically active short isoform of PGC-1α. J. Biol. Chem. 284, 32813–32826 (2009).
Kim, J. et al. NT-PGC-1α deficiency attenuates high-fat diet-induced obesity by modulating food intake, fecal fat excretion and intestinal fat absorption. Sci. Rep. 11, 1323 (2021).
Martinez-Redondo, V., Pettersson, A. T. & Ruas, J. L. The hitchhiker’s guide to PGC-1α isoform structure and biological functions. Diabetologia 58, 1969–1977 (2015).
Finck, B. N. & Kelly, D. P. PGC-1 coactivators: inducible regulators of energy metabolism in health and disease. J. Clin. Invest. 116, 615–622 (2006).
Durham, A. L., Speer, M. Y., Scatena, M., Giachelli, C. M. & Shanahan, C. M. Role of smooth muscle cells in vascular calcification: implications in atherosclerosis and arterial stiffness. Cardiovasc. Res. 114, 590–600 (2018).
Clement, N., Glorian, M., Raymondjean, M., Andreani, M. & Limon, I. PGE2 amplifies the effects of IL-1beta on vascular smooth muscle cell de-differentiation: a consequence of the versatility of PGE2 receptors 3 due to the emerging expression of adenylyl cyclase 8. J. Cell. Physiol. 208, 495–505 (2006).
Keuylian, Z. et al. The Notch pathway attenuates interleukin 1β (IL1β)-mediated induction of adenylyl cyclase 8 (AC8) expression during vascular smooth muscle cell (VSMC) trans-differentiation. J. Biol. Chem. 287, 24978–24989 (2012).
Hewer, R. C., Sala-Newby, G. B., Wu, Y. J., Newby, A. C. & Bond, M. PKA and Epac synergistically inhibit smooth muscle cell proliferation. J. Mol. Cell. Cardiol. 50, 87–98 (2011).
McKean, J. S. et al. The cAMP-producing agonist beraprost inhibits human vascular smooth muscle cell migration via exchange protein directly activated by cAMP. Cardiovasc. Res. 107, 546–555 (2015).
Vallin, B. et al. Novel short isoforms of adenylyl cyclase as negative regulators of cAMP production. Biochim. Biophys. Acta Mol. Cell. Res. 1865, 1326–1340 (2018).
Hu, B., Nakata, H., Gu, C., De Beer, T. & Cooper, D. M. A critical interplay between Ca2+ inhibition and activation by Mg2+ of AC5 revealed by mutants and chimeric constructs. J. Biol. Chem. 277, 33139–33147 (2002).
Montminy, M. Transcriptional regulation by cyclic AMP. Annu. Rev. Biochem. 66, 807–822 (1997).
Li, Y. et al. Clenbuterol upregulates histone demethylase JHDM2a via the beta2-adrenoceptor/cAMP/PKA/p-CREB signaling pathway. Cell. Signal. 24, 2297–2306 (2012).
Muglia, L. M. et al. The 5′-flanking region of the mouse adenylyl cyclase type VIII gene imparts tissue-specific expression in transgenic mice. J. Neurosci. 19, 2051–2058 (1999).
Chao, J. R. et al. Characterization of the mouse adenylyl cyclase type VIII gene promoter: regulation by cAMP and CREB. Eur. J. Neurosci. 16, 1284–1294 (2002).
Chinsomboon, J. et al. The transcriptional coactivator PGC-1alpha mediates exercise-induced angiogenesis in skeletal muscle. Proc. Natl Acad. Sci. USA 106, 21401–21406 (2009).
Cong, L. et al. Multiplex genome engineering using CRISPR/Cas systems. Science 339, 819–823 (2013).
Zhang, M. et al. Recessive cardiac phenotypes in induced pluripotent stem cell models of Jervell and Lange-Nielsen syndrome: disease mechanisms and pharmacological rescue. Proc. Natl Acad. Sci USA 111, 5383–5392 (2014).
Stutz, A., Horvath, G. L., Monks, B. G. & Latz, E. ASC speck formation as a readout for inflammasome activation. Methods Mol. Biol. 1040, 91–101 (2013).
Patro, R., Duggal, G., Love, M. I., Irizarry, R. A. & Kingsford, C. Salmon provides fast and bias-aware quantification of transcript expression. Nat. Methods 14, 417–419 (2017).
Soneson, C., Love, M. I. & Robinson, M. D. Differential analyses for RNA-seq: transcript-level estimates improve gene-level inferences. F1000Res 4, 1521 (2015).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Yu, G., Wang, L. G., Han, Y. & He, Q. Y. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16, 284–287 (2012).
Li, H. Minimap2: pairwise alignment for nucleotide sequences. Bioinformatics 34, 3094–3100 (2018).
Hahne, F. & Ivanek, R. Visualizing genomic data using Gviz and Bioconductor. Methods Mol. Biol. 1418, 335–351 (2016).
Sieckmann, K. et al. AdipoQ-a simple, open-source software to quantify adipocyte morphology and function in tissues and in vitro. Mol. Biol. Cell 33, br22 (2022).
Pichlo, M. et al. High density and ligand affinity confer ultrasensitive signal detection by a guanylyl cyclase chemoreceptor. J. Cell Biol. 206, 541–557 (2014).
Jespersen, N. Z. et al. A classical brown adipose tissue mRNA signature partly overlaps with brite in the supraclavicular region of adult humans Cell Metab. 17, 798–805 (2013).
Letunic, I. & Bork, P. Interactive Tree Of Life (iTOL) v5: an online tool for phylogenetic tree display and annotation. Nucleic Acids Res. 49, W293–W296 (2021).
Meredith, R. W. et al. Impacts of the cretaceous terrestrial revolution and KPg extinction on mammal diversification. Science 334, 521–524 (2011).
Perelman, P. et al. A molecular phylogeny of living primates. PLoS Genet. 7, e1001342 (2011).
Swanson, M. T., Oliveros, C. H. & Esselstyn, J. A. A phylogenomic rodent tree reveals the repeated evolution of masseter architectures. Proc. Biol. Sci. 286, 20190672 (2019).
Pradas-Juni, M. et al. A MAFG-lncRNA axis links systemic nutrient abundance to hepatic glucose metabolism. Nat. Commun. 11, 644 (2020).
Tang, A. D. et al. Full-length transcript characterization of SF3B1 mutation in chronic lymphocytic leukemia reveals downregulation of retained introns. Nat. Commun. 11, 1438 (2020).
Acknowledgements
We thank J. Alber and A.-B. Marcher for indirect calorimetry (TSE Phenomaster) support. Sequencing was carried out at the Functional Genomics and Metabolism Research Unit, University of Southern Denmark. We thank T. P. Mortensen, M. Wishoff and R. Nielsen for sequencing assistance. We thank C. Baitzel and A. Lietzau, Max Planck Institute for Metabolism Research, for assistance in generating Adcy3∆AT mice. J.-W.K. and B.D.M.L. received funding from European Research Council Starting Grant (ERC StG) TransGenRNA (675014), Sygeforsikring Denmark Healthcall, the University of Southern Denmark, and the Danish Diabetes Academy, which is funded by the Novo Nordisk Foundation. J.-W.K., A.J. and A.R.T. received support from Challenge (33444) and Bioscience and Basic Biomedicine Programs of the NNF (28416). H.T. was supported by a research grant from the Danish Diabetes Academy, which is funded by the Novo Nordisk Foundation, grant number NNF17SA0031406. S.K. was supported by a German Academic Exchange Service (DAAD) PhD scholarship (A/12/97620). We thank the Microscopy Core Facility of the Medical Faculty at the University of Bonn for providing help, services, and devices funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation; project nos. 169331223 and 388159768). Research in the laboratory of D.W. was supported by grants from the DFG SFB 1454 (project number 432325352; TRR83 – 112927078, TRR333/1 – 450149205), under Germany’s Excellence Strategy (EXC2151 – 390873048), SPP1926, SPP1726, FOR2743, as well as intramural funding from the University of Bonn. A.B. appreciates funding from DFG SFB1123-B10 and ERC StG PROTEOFIT. A.P. and T.G. are funded by the DFG (450149205-TRR333/1; A.P.: P10; T.G.: P11).
Author information
Authors and Affiliations
Contributions
S.K., H.T., R.K., A.R.T., A.J., B.D.L.M., M.J.G., P.L., N.M.E., A.S.G., N.S., P.M.M.R., I.G., N.R.H., E.S., P.K., L.-M.V., R.S., F.D., J.-H.K., A.B., C.A.E., S.G., T.G. and M.P.-J. performed the experiments. H.T., S.K., P.F., A.R.-I., T.J.S., F.T.W., A.P., A.B. and M.J. contributed discussions and performed training. S.K., H.T., D.W. and J.-W.K. conceived the experiments. D.W. and J.-W.K. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Metabolism thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editors: Revati Dewal and Isabella Samuelson, in collaboration with the Nature Metabolism team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 cAMP biosynthesis in adipose tissue after cold.
Transcriptional regulation of cAMP biosynthesis in adipose tissue after cold exposure. a–c, Principal component analysis (PCA) of transcriptomic changes in BAT (a), iWAT (b), and eWAT (c) after 24 h (a,c) or 72 h (b) of 5 °C cold exposure (n = 3–5 per condition). Dots represent individual, chow diet fed, male C57BL/6N animals with n = 3–5 animals per condition. d–f, mRNA expression of phosphodiesterases (Pde) in BAT (d), iWAT (e) and eWAT (f) as determined by mRNA-seq and depicted in transcripts per million (TPM). Bar graphs represent mean + SEM with all data points plotted. Unpaired, two-tailed, and non-parametric Mann-Whitney tests were performed to assess statistical significance.
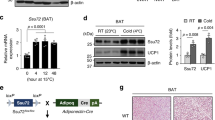
Extended Data Fig. 2 mAC3 is required for cAMP biosynthesis in BAT.
mAC3 is required for cAMP biosynthesis in BAT and required for cold adaptation in cold. a,b, mRNA expression of transcriptional BAT regulators (a) and BAT-specific genes (b) in BAT from LoxP and Adcy3-AdcKO mice (at 22 °C or after 24 h of 5 °C cold exposure). Expression has been determined by mRNA-seq and depicted in transcripts per million (TPM); n = 4. The upper/lower whiskers represent largest/smallest observation less/greater than upper/lower hinge+1.5 × interquartile range (IQR). Central median represents 50% quantile. c,d, qPCR analysis of indicated BAT mRNA markers measured in 1°BA derived from SVF of LoxP or Adcy3-AdcKO mice, injected with 100,00 multiplicity of infection (MOI) of AAV8-CMV-Cre (AAV8-Cre) or AAV8-CMV-GFP (AAV-GFP), and stimulated for 6 h with 10 µM CL316,243 (CL), n = 4. Bar graphs represent mean + SEM with all data points plotted. To test for statistical significance, non-parametric (ranked) Kruskal–Wallis one-way analysis of variance (ANOVA) tests with Dunn’s correction for multiple testing was performed.
Extended Data Fig. 3 mAC3 is required for cAMP biosynthesis in BAT.
mAC3 is required for cAMP biosynthesis in BAT and required for cold adaptation in cold. a–d, Relative (body-weight corrected) tissue weights from indicated adipose tissue depots and liver of LoxP (n = 4 for 22 °C, n = 8 for 5 °C) and Adcy3-AdcKO (n = 4 for 22 °C, n = 8 for 5 °C) mice either fed chow diet (a,b) or HFD (c,d) at 22 °C (a,c) or exposed for 24 h to 5 °C (b,d). Bar graphs represent mean + SEM with all data points plotted. Unpaired, two-tailed, and non-parametric Mann-Whitney tests were performed to assess statistical significance between both genotypes within each tissue. e, Western blot analysis of BAT from chow-diet fed, male LoxP (n = 3 for 22 °C, n = 4 for 5 °C) and Adcy3-AdcKO mice (n = 4 for 22 °C, n = 5 for 5 °C) at 22 °C or exposed to 5 °C for 6 days. Anti-phospho-PKA substrate antibodies were used for detection of PKA phosphorylation substrates. Anti-UCP1 antibodies probed for UCP1 protein levels. anti-HSC70 antibody served as loading control. f–h, Analysis of STK signaling activity in mouse BAT. Kinome trees depict the mean kinase statistic of differentially activated STKs measured by Pam Gene Array technology in LoxP at 22 °C compared to 5 °C for 24 h (f), LoxP at 22 °C compared to 5 °C for 6 days (g) and Adcy3-AdcKO compared to LoxP at 22 °C (h). i, Inserts show zoom-ins of relevant areas of the kinome tree (blue box: down-regulated kinases; red box: up-regulated kinases). ii, Mean kinase statistic of 15 most significantly differentially activated kinases is represented in bar graphs. iii, Venn diagram depicting differences in differentially activated kinases of the respective comparison. Data represents the mean of n = 4 biological replicates for all conditions.
Extended Data Fig. 4 Activated adipocytes express truncated Adcy3.
Activated thermogenic adipocytes express a truncated Adcy3 (Adcy-AT) transcript and protein isoform. a, UCSC genome browser tracks depicting changes in mRNA expression (grey) and H3K4me3 (magenta) in BAT from chow diet-fed, male C57BL/6N mice housed either at 22 °C or after 24 h of 5 °C cold exposure as determined by RNA-Seq (grey) and H3K4me3 ChiP-Seq (magenta). Red and blue transcripts depict contiguous Adcy3 and Adcy3-at transcripts, respectively, and Adcy3 isoform specific transcriptional start sites (TSS). b, ChIP-qPCR of H3K4me3 at the TSS of Adcy3 (blue), Adcy3-at (red) and exon 3, which is shared by Adcy3 and Adcy3-at (grey). Replicates represent H3K4me3 immunoprecipitations perform in individual animals (n = 3). Bar graphs represent mean + SEM with all data points plotted and unpaired, two-tailed, and non-parametric Mann-Whitney tests were performed to assess statistical significance. P values are indicated within the panel. c,d, Expression of Adcy3 (c) and Adcy3-at (d) in indicated tissues of chow diet fed, male C57BL/6N mice. Data points represent individual mice (n = 4 BAT, n = 2 iWAT, n = 3 eWAT, n = 3 pancreas, n = 3 kidneys, n = 2 skeletal muscle, n = 3 liver). e, Absolute expression (Cq value) of Adcy, Adcy3-at and housekeeping genes Hprt and Gapdh in hypothalamy of lean C57BL/6N male mice (n = 3 as determined by qPCR analysis. ND = Not detected. f–h, Absolute mRNA expression (Ct threshold levels) of Adcy3-at (f), Adcy3 (g) and Ucp1 (h) in BAT-derived SVF at indicated stages of brown adipogenesis and after stimulation with 1 mM db-cAMP or 10 µM CL316,243 as determined by qPCR. Replicates depict experiments performed in SVF isolated from individual mice (n = 4). Bar graphs represent mean + SEM with all data points. i,j, Expression of indicated BAT marker mRNAs in BAT (i) or iWAT (j) of chow diet or HFD-fed male C57BL/6N mice, each housed either at 22 °C or exposed to 24 h for 5 °C cold exposure as determined by qPCR. k, Expression of Adcy3-at in BAT of chow diet fed male C57BL/6N mice at 2.5 months or 25 months of age (n = 8). Bar graphs represent mean + SEM with all data points plotted. i–k, Bar graphs represent mean + SEM with all data points plotted. To test for statistical significance, non-parametric (ranked) Kruskal–Wallis one-way analysis of variance (ANOVA) tests with Dunn’s correction for multiple testing were performed. P values are indicated in the panel.
Extended Data Fig. 5 Activated adipocytes express truncated Adcy3.
Activated thermogenic adipocytes express a truncated Adcy3 (Adcy-AT) transcript and protein isoform. a–c, Relative expression of Adcy (blue) and Adcy3-at (red) as determined by qPCR analysis in BAT (a), iWAT (b) and eWAT (c) from chow diet fed, male C57BL/6N mice housed either at 28 °C (thermoneutrality) or exposed to 5 °C for 24 h and 7 days (n = 8). Bar graphs represent mean + SEM with all data points plotted. To test for statistical significance, non-parametric (ranked) Kruskal–Wallis one-way analysis of variance (ANOVA) tests with Dunn’s correction for multiple testing were performed. P values are indicated in the panel.
Extended Data Fig. 6 Generation of mice deficient for Adcy3-at.
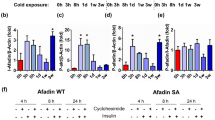
Generation of transgenic mice deficient for Adcy3-at at exon 1 and TSS using CRISPR/CAS9 gene. a, Illustration of CRISPR-Cas9 mediated excision of Adcy3-at specific Exon 2b, including Adcy3-at TSS (light blue), yielding Adcy3-at specific knockout (Adcy3∆AT) mice. b, USCS genome browser track depicting mRNA (grey) and H3K4me3 signals (magenta) in the murine Adcy3 locus from chow diet fed, male C57BL/6N mice either housed at 22 °C (light grey) and exposed to 5 °C for 24 h (dark grey) and from HFD-fed, male C57BL/6N mice housed at 22 °C (light orange) and exposed to 5 °C for 24 h (dark orange) as determined by RNA-Seq and H3 K4me3 ChiP-Seq. Location and sequence of sgRNAs used for CRISPR/Cas9 mediated 978-nt deletion of Adcy3-at TSS are indicated. c, Southern blot of a control (C57BL/6N wildtype) and from Adcy3∆AT embryonic stem cell (ESC) genomic DNA. The asterisk depicts a heterozygous Adcy3∆AT/wild-type ESC clone. d, PCR from genomic DNA isolated from wild-type and Adcy3∆AT ESC genomic DNA. e,f, Relative expression of Adcy3-at (e) and Adcy3 (f) as determined by qPCR analysis in BAT, iWAT, and eWAT from chow diet fed, male LoxP (n = 4 for BAT and iWAT, n = 2 for eWAT) and Adcy3∆AT mice (n = 4) housed at 22 °C. Bar graphs represent mean + SEM with all data points plotted. An unpaired, two-tailed, and non-parametric Mann-Whitney test was performed to assess statistical significance. P values are indicated within the panel. Created with BioRender.com.
Extended Data Fig. 7 mAC3-AT inhibits oxidative metabolism and protects from obesity.
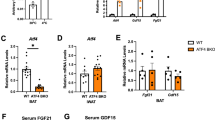
mAC3-AT inhibits oxidative metabolism in vitro and in vivo and protects from diet-induced obesity. a,b, Extracellular acidification rates in 1°iBA derived from SVF of LoxP (n=5) or Adcy3∆AT (n = 5) and stimulated with oligomycin (O), FCCP (F) and Antimycin A plus Rotenone (A/R) (a), or 10 µM CL316,243 (CL) (b) at the indicated time points. The numbers of measured wells are indicated in the panel. c,d, Oxygen consumption rates in 1°iWA derived from SVF of LoxP (n = 5) or Adcy3∆AT (n = 5) and stimulated with oligomycin (O), FCCP (F) and Antimycin A plus Rotenone (A/R) (c) or 10 µM CL316,243 (CL) (d) at the indicated time points. The numbers of measured wells are indicated in the panel. e,f, Extracellular acidification rates in 1°iWA derived from SVF of LoxP (n = 5) or Adcy3∆AT (n = 5) and stimulated with oligomycin (O), FCCP (F) and Antimycin A plus Rotenone (A/R) (e), or 10 µM CL316,243 (CL) (f) at the indicated time points. The numbers of measured wells are indicated in the panel. a–f, Bar graphs represent mean + SEM. Unpaired, two-tailed Student’s T-tests were performed to assess statistical significance. P values are indicated within the panel. Genotype P = 0.0072 (a), P = 0.218 (b), P = 0.007 (c), P = 0.1213 (d), P = 0.032 (e), P = 0.138 (f). g, Body weights in chow diet-fed, male LoxP (n = 7) and Adcy3∆AT (n = 9) mice. h, Blood glucose levels during intraperitoneal glucose tolerance tests in chow diet-fed, male LoxP (n = 7) and Adcy3∆AT (n = 9) mice. i, Blood glucose levels during intraperitoneal insulin tolerance tests in chow diet-fed, male LoxP (n = 7) and Adcy3∆AT (n = 9) mice. j, Indirect calorimetry mediated quantification of cumulative food intake in chow diet-fed, male LoxP (n = 7) and Adcy3∆AT (n = 9) mice. g–j, Graphs represent mean + SEM. Statistical significance was determined by performing Two-Way ANOVA with repeated measurements for x values (mixed models). Post-hoc P value correction to account for multiple testing was performed using Bonferroni adjustment. The source of variation, % of the variation and exact P values are given in the table insets. Genotype P = 0.749 (g), P = 0.48 (h), P = 0.66 (i), P = 0.003 (j). k, Indirect calorimetry measurement of oxygen consumption in chow-diet fed, male LoxP (n = 7) and Adcy3∆AT (n = 9) mice. l, Indirect calorimetry determination of mean daily oxygen consumption in chow diet fed, male LoxP (n = 7) and Adcy3-AdcKO (n = 8) mice. Bar graphs represent mean + SEM with all data points plotted and parametric, unpaired, two-tailed Student’s t-tests were performed to assess statistical significance. P values are indicated within the panel.
Extended Data Fig. 8 mAC3-AT inhibits oxidative metabolism and protects from obesity.
mAC3-AT inhibits oxidative metabolism in vitro and in vivo and protects from diet-induced obesity. a, relative expression of indicated thermogenic BAT marker mRNAs in chow diet-fed, male LoxP (n = 5 for 22 °C, n = 6 for 5 °C) and Adcy3∆AT (n = 3 for 22 °C, n = 4 for 5 °C) mice. Bar graphs represent mean + SEM with all data points plotted and unpaired, two-tailed, non-parametric Mann-Whitney tests were performed for indicated comparisons to assess statistical significance. P values are given within the panel. b,c, Relative expression of indicated thermogenic BAT marker mRNAs in 1°BA (b) and 1°iWA (c) from LoxP (n = 4 individual SVF preparations) and Adcy3∆AT (n = 4 individual SVF preparations). Primary adipocytes were stimulated with 10 μM CL316,243 for 6 h. Bar graphs represent mean + SEM with all data points plotted. To test for statistical significance, non-parametric, ranked Kruskal–Wallis one-way analysis of variance (ANOVA) tests with Dunn’s correction for multiple testing were performed. P values are indicated in the panel. d, Images of HE-stained adipose tisue, extracted from male LoxP and Adcy3ΔAT mice fed CD or HFD. Scale bars: 100 µm. e, Adipocyte size distribution quantified using images as shown in (d). iWAT adipocytes size distribution was analyzed for n = 7 LoxP and n = 3 Adcy3ΔAT animals. gWAT adipocyte size distribution was analyzed for n = 8 LoxP and n = 3 Adcy3ΔAT animals. Mean ± SD of the distributions from all mice per group; P values indicated for unpaired, two-sided t tests with Welch correction between LoxP and Adcy3ΔAT at the size range; *p ≤ 5 × 10–2; **p.
Extended Data Fig. 9 mAC3-AT alters mAC3 localization and limits cAMP.
mAC3-AT alters mAC3 subcellular localization and, thereby, limits cAMP bio synthesis. a,b, analysis of STK activity in Adcy3∆AT BAT. Kinome trees depict the mean kinase statistic of differentially activated STKs in Adcy3 vAT versus LoxP (22 °C) (a) and Adcy3∆AT (22 °C) versus Adcy3∆AT (5 °C, 6 days) (b). c,d, Comparison of Adcy3∆AT versus LoxP at 22 °C (c) and Adcy3∆AT versus LoxP after 6 days at 5 °C (d). At 22 °C, a set of kinases is inversely regulated in BAT of Adcy3∆AT and Adcy3AdcKO mice compared to LoxP mice. The mean kinase statistic of these kinases is shown for 22 °C (c) and for 5 °C (d). Data includes n = 4 biological replicates for all conditions. e, CHO cells stably expressing AC3-HA and transiently transfected with mCherry-CAAX were sonicated and labeled with a Calnexin antibody (ER-marker, magenta) and an HA antibody (green). mCherryCAAX fluorescence is indicated in red. Single channels are shown at the top, merged images at the bottom row. White boxes are shown as magnified view at the bottom-right. Arrows point to endoplasmic reticulum-plasma membrane (ER-PM) contact sites, asterixis indicate contact-free parts within the membrane sheet. Scale bar: 10 μm. f, See (e) for CHO cells stably expressing AC3-AT-HA. The images represent at least four independent experiments, with 5-10 cells analysed per condition per experiment. g, Quantification of the average HA-signal intensity within the PM (co-localization with mCherry-CAAX) and the ER (co-localization with calnexin). Data shown as mean + SEM (empty vector: n = 4; AC3-AT: n = 8, AC3: n = 10). h, Ratio PM/ER distribution (empty vector: n = 4; mAC3-AT: n = 8, mAC3: n = 10). Bar graphs represent mean + SEM with all data points plotted. An unpaired, two-tailed, and non-parametric Mann-Whitney tests were performed to assess statistical significance; p-values are indicated within the panel. i,j, Quantification of intracellular cAMP levels. CHO cells stably expressing mAC3-AT or mAC3, or empty vector transfected CHO cells were stimulated with buffer or 2 μM Forskolin for 30 min (i) or buffer or 1 μM Isoproterenol for 15 min (j). Values were normalized to the respective buffer control and to the respective amount of total protein (empty vector: n = 4; AC3-AT: n = 5, AC3-FL: n = 5). g–j, Data is shown as mean + SEM with all data points plotted. To test for statistical significance, non-parametric (ranked) Kruskal–Wallis one-way analysis of variance (ANOVA) tests with Dunn’s correction for multiple testing were performed; p-values are indicated in the panel.
Extended Data Fig. 10 Conservation of Adcy3-at and Ppargc1a-at.
Evolutionary conservation of Adcy3-at and Ppargc1a-at isoforms. a, Mammalian phylogeny from Meredith et al.89 modified in iTOL88, with silhouettes in black indicating representative species that were selected for comparative analyses of PPARGC1A-AT. b, Genomic conservation among representative rodent species (rodentia) of Adcy3-at identified in the mouse. Color gradient indicates percent nucleotide identity of the Adcy3-at relative to the mouse. Phylogeny based on Swanson et al.91. c, Genomic conservation among representative primates (primates) of Adcy3-at identified in human. Color gradient indicates percent nucleotide identity of Adcy3-at relative to the human. Phylogeny based on Perelman et al.90. d, Nucleotide sequence alignment of Ppargc1a-at (Exon 1b) from various mammalian species. e, Percent identity matrix of Ppargc1a-at among 14 representative mammals (exon 1b). Reproduced from ref. 88 under a Creative Commons license CC BY 4.0.
Supplementary information
Supplementary Tables 1–6
Multi-tabs workbook with Supplementary Tables 1–6. Supplementary Table 1: Adipose depot-specific RNA-seq from cold-exposed C57BL/6 mice. Supplementary Table 2: Gene accession numbers for species conservation analyses. Supplementary Table 3: List of sgRNAs and primers for the generation of Adcy3∆AT mice. Supplementary Table 4: List of SYBR qPCR primers. Supplementary Table 5: List of LNA GapmeR inhibitors. Supplementary Table 6: Alignment mAC3/mAC3-AT.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 6
Statistical source data.
Source Data Fig. 7
Statistical source data.
Unprocessed blots main figures
Uncropped blots for Figs. 1–7.
Unprocessed blots extended data figures
Uncropped blots for Extended Data Figs. 1–9
Source Data Extended Data Fig./Table 1
Statistical source data.
Source Data Extended Data Fig./Table 2
Statistical source data.
Source Data Extended Data Fig./Table 3
Statistical source data.
Source Data Extended Data Fig./Table 4
Statistical source data.
Source Data Extended Data Fig./Table 5
Statistical source data.
Source Data Extended Data Fig./Table 6
Statistical source data.
Source Data Extended Data Fig./Table 7
Statistical source data.
Source Data Extended Data Fig./Table 8
Statistical source data.
Source Data Extended Data Fig./Table 9
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Khani, S., Topel, H., Kardinal, R. et al. Cold-induced expression of a truncated adenylyl cyclase 3 acts as rheostat to brown fat function. Nat Metab 6, 1053–1075 (2024). https://doi.org/10.1038/s42255-024-01033-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-024-01033-8
- Springer Nature Limited