Abstract
Cardiac magnetic resonance (CMR) is the gold standard for the diagnostic classification and risk stratification in most patients with cardiac disorders. The aim of the present study was to investigate the ability of Strain-encoded MR (SENC) for the prediction of major adverse cardiovascular events (MACE). A systematic review and meta-analysis was performed according to the PRISMA Guidelines, including patients with or without cardiovascular disease and asymptomatic individuals. Myocardial strain by HARP were used as pulse sequences in 1.5 T scanners. Published literature in MEDLINE (PubMed) and Cochrane’s databases were explored before February 2023 for studies assessing the clinical utility of myocardial strain by Harmonic Phase Magnetic Resonance Imaging (HARP), Strain-encoded MR (SENC) or fast-SENC. In total, 8 clinical trials (4 studies conducted in asymptomatic individuals and 4 in patients with suspected or known cardiac disease) were included in this systematic review, while 3 studies were used for our meta-analysis, based on individual patient level data. Kaplan–Meier analysis and Cox proportional hazard models were used, testing the ability of myocardial strain by HARP and SENC/fast-SENC for the prediction of MACE. Strain enabled risk stratification in asymptomatic individuals, predicting MACE and the development of incident heart failure. Of 1332 patients who underwent clinically indicated CMR, including SENC or fast-SENC acquisitions, 19 patients died, 28 experienced non-fatal infarctions, 52 underwent coronary revascularization and 86 were hospitalized due to heart failure during median 22.4 (17.2–28.5) months of follow-up. SENC/fast-SENC, predicted both all-cause mortality and MACE with high accuracy (HR = 3.0, 95% CI = 1.2–7.6, p = 0.02 and HR = 4.1, 95% CI = 3.0–5.5, respectively, p < 0.001). Using hierarchical Cox-proportional hazard regression models, SENC/fast-SENC exhibited incremental value to clinical data and conventional CMR parameters. Reduced myocardial strain predicts of all-cause mortality and cardiac outcomes in symptomatic patients with a wide range of ischemic or non-ischemic cardiac diseases, whereas in asymptomatic individuals, reduced strain was a precursor of incident heart failure.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Cardiovascular magnetic resonance (CMR) is the established reference standard for the identification of functional and structural abnormalities of the heart and for myocardial tissue characterization1,2,3,4. Hereby, a plethora of clinical questions such as the detection of myocardial ischemia due to coronary artery disease (CAD), differentiation of hypertrophy of unclear etiology, detection of subtle myocardial damage due to cardiotoxicity or infiltrative disorders can be addressed5,6,7,8,9.
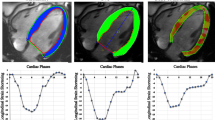
Harmonic phase strain analysis (HARP) for measuring myocardial strain was introduced in 200010,11. However, HARP is time-consuming and has limited spatial resolution. Strain-encoded MR (SENC) arose within the last two decades, providing higher spatial resolution compared to HARP12. More recently, fast-SENC has been introduced, offering strain analysis within a single heartbeat, without the need for breath holding13. This has advantages in patients with arrhythmias and in those who cannot perform breathholds (Fig. 1). Experimental and clinical studies highlighted the role of SENC/fast-SENC in patients with different cardiac disorders14.
Recently, myocardial strain gained scientific and clinical interest for risk stratification of patients, i.e., the prediction of major adverse cardiovascular events (MACE)14, which constitutes a common endpoint in large randomized controlled trials (RCTs)15. While such ‘megatrials’ require the inclusion of large patient sample sizes to achieve a significantly altered primary endpoint, myocardial strain may serve as a surrogate marker, reducing the number of patients needing to be included16. We sought to systematically review clinical trials, investigating the prognostic role of strain by HARP, SENC and fast-SENC.
Methods
Eligibility criteria and study selection
We conducted our research in line with the recommendations of the Cochrane Collaboration Handbook and according to the PRISMA 2020 (Preferred Reporting Items for Systematic Reviews and Meta-Analyses, Supplementary Information) guidelines17. “CMR” and “Strain” combined with the AND/OR as Boolean comprised our search terms. PubMed, Google scholar and Cochrane’s search engine were utilized for our search. Our research strategy’s inclusion criteria were the following: (i) publications written and published in English language, (ii) patient populations consisting of adult patients with (symptomatic) or without (asymptomatic) cardiovascular disease undergoing CMR, including myocardial strain assessment using HARP, SENC or fast-SENC, (iii) studies reporting on MACE, including cardiovascular death, myocardial infarction, coronary revascularization and hospitalizations due heart failure and their association with CMR-based myocardial strain.
Data extraction and statistical analysis
Based on individual patient data, which were available from 3 studies, investigating the role of SENC or fast-SENC on clinical outcomes18,19,20, we sought to determine the ability of myocardial strain for the prediction of all-cause mortality and MACE. All 3 studies included patients who underwent CMR due to clinical reasons, whereas studies including asymptomatic patients were not included. Statistics were performed using the dedicated statistical software (MedCalc 20.009, Mariakerke, Belgium). Continuous normally distributed variables were expressed as mean ± standard deviation, whereas non-normally distributed variables were reported as medians with interquartile range (IQR). Normal distribution was assessed using the Shapiro–Wilk test. Categorical variables were reported as numbers and proportions. For survival analysis the endpoints (i) all-cause mortality, and (ii) a composite endpoint, including all-cause mortality, myocardial infarction, coronary revascularization, and hospitalization due to heart failure were used. Survival curves were estimated by the Kaplan–Meier method to estimate the distribution of cardiac events as a function of the follow-up duration and comparisons were performed using log-rank tests. Cox proportional hazard models were used to evaluate the incremental value of myocardial strain to clinical and conventional CMR parameters. Furthermore, Cox proportional hazard models using the following hierarchic steps: (1) clinical data, (2) conventional CMR markers (wall motion abnormalities (WMA), late gadolinium enhancement (LGE) and LV-ejection fraction) and (3) myocardial strain by SENC or fast-SENC were applied. Model χ2 values were compared to each other for each incremental step. Differences were considered statistically significant at p < 0.05.
Quality and risk of bias assessment
Bias assessment was performed according to the PROBAST tool, which is specialized for assessing the bias and applicability of studies developing prediction models21. Due to the character of the studies and the lack of validation in the general population, all studies include significant amounts of biases in the outcome domain. Moreover, the shrinking population especially for studies developing prediction models, may introduce additional biases in the analysis domain of the questionnaire. Therefore, the amount of bias insertion was evaluated as relatively high according to PROBAST21. On the other hand, the applicability of all studies was assessed with a reduced amount of bias for all studies since the software and hardware used for the studies is commercially available and such measures can be conducted in departments with available CMR equipment. The results of the risk of bias assessment are provided in Table 1.
Results
Initially, 568 studies were identified based on predefined criteria. After exerting all inclusion and exclusion criteria 8 studies remained18,19,20,22,23,24,25,26. The selection strategy is depicted in the PRISMA Flowchart in Fig. 2. In total, 8349 patients were enrolled in these 8 studies, which were included in the final analysis. The follow-up duration ranged from 1.9–8.3 years (median = 2.3, IQR = 2.0–7.4 years). The basic characteristics of each study including population characteristics, endpoints, and CMR software and hardware are presented in Table 1. In addition, an overview of univariate and multivariable predictors of MACE and of the corresponding cut-off values for strain used in each study is provided in Tables 2, 3.
Systematic review of studies including asymptomatic patients
Three studies analyzed data from the Multi-Ethnic Study of Atherosclerosis (MESA)27. Choi et al. investigated the CMR findings of 1768 individuals from the MESA cohort22. Circumferential strain (Ecc) predicted incident HF independent of clinical and CMR imaging parameters.
Similarly, time to peak circumferential systolic strain (SDTPEcc), a marker of LV-dyssynchrony, was predictive for MACE, including myocardial infarction, stroke, and death in asymptomatic female but not in male individuals (1392 included in total)23.
Another study, included 1544 patients from the MESA cohort, focusing on diastolic dysfunction24. Strain relaxation index (SRI) was predictive for the occurrence of heart failure and atrial fibrillation during follow-up.
The most recent study in asymptomatic patients was conducted by Pezel et al., including 1506 asymptomatic patients from the MESA study’s cohort26,27. Ecc was assessed using the HARP method28,29. Using Cox analyses, strain analysis was predictive for incident heart failure. The prognostic value remained after adjustment for traditional cardiovascular risk factors.
Systematic review of studies including symptomatic patients
The first study in symptomatic patients was published in 201118, included 320 patients, who underwent dobutamine stress CMR due to suspected or known CAD. 175 patients also underwent invasive coronary angiography. Visual and quantitative strain by SENC provided higher accuracy than cine-imaging for the detection of obstructive CAD and predicted MACE, independent of WMA and clinical parameters.
Using Fast-SENC, heart failure patients in stages A and B were prospectively analyzed19. Patients with reduced strain exhibited increased risk for death and hospitalization due to HF and for new onset of HF medications. In addition, strain analysis reclassified a substantial part of presumably healthy individuals at risk for heart failure to Stage B individuals with subclinical LV-dysfunction.
Another study by Steen et al., investigated 111 patients with known or suspected CAD who underwent adenosine stress CMR20. Patients with reduced strain showed higher rates for all-cause mortality, non-fatal MI, and coronary revascularization during follow-up.
The role of Eccglobal for the prediction of MACE in 539 symptomatic patients was investigated by Mordi et al.25 Reasons for referral of the patients were suspected dilative (36.9%) or ischemic cardiomyopathy (19.9%), myocarditis (11.9%), arrhythmias (13.9%), LV hypertrophy (10.9%), or aortic disease (6.5%). Hereby, Eccglobal, LGE, and LVEF were predictive for MACE.
Meta-analysis based on individual data in symptomatic patients
Individual baseline and CMR data were available in 1689 patients from 3 previous studies, (n = 20020, n = 32018, and n = 116919, respectively) who underwent strain analysis by SENC/fast-SENC for ischemic heart disease (n = 985), suspected structural heart disease (n = 203), myocarditis or cardiotoxicity (n = 421), or due to other reasons (n = 80). Patients were 60.0 (47.4–70.8) years old and 976 (57.8%) were male, whereas 262 (15.5%) had diabetes mellitus (Table 4). Notably, the most recent study by Steen-H et al.20, reported on 111 patients with stress CMR and complete follow-up, while baseline data were available in 200 patients, which were included in the present analysis. Healthy volunteers19, were not included.
Follow-up was available in 1332 patients (n = 11120, n = 32018 and n = 90119, respectively) during a median follow-up duration of 22.4 (17.2–28.5) months. During follow-up, 19 (1.4%) patients died, 28 (2.1%) experienced non-fatal infarction, 52 (3.9%) underwent coronary revascularization and 86 (6.5%) were hospitalized due to heart failure.
Conventional CMR parameters such as resting WMA and LGE predicted the composite endpoint, whereas a non-significant trend was observed for all-cause mortality (HR of 2.0, p < 0.001 and of 2.3, p = 0.07, Fig. 3A,B). Myocardial strain by SENC/fast-SENC, on the other hand, predicted both all-cause mortality and the composite endpoint with high accuracy (HR of 3.0, p = 0.02 and of 4.1, p < 0.001, Fig. 3C,D).
Using Cox-proportional hazard regression models, myocardial strain by SENC/fast-SENC provided the most robust prediction of the composite endpoint, independent of clinical and conventional CMR variables (Table 5). Hierarchical Cox-proportional hazard regression models demonstrated the incremental value (i) of CMR paraments compared to clinical data and (ii) of myocardial strain compared to conventional CMR markers (Fig. 4).
Discussion
Our study summarizes for the first time in the current literature the value of CMR based strain by HARP, SENC and fast-SENC in the clinical setting, serving as a promising surrogate parameter for the prediction of MACE in patients both with ischemic and non-ischemic cardiac diseases as well as in asymptomatic individuals18,19,20,22,23,24,25,26. By performing meta-analysis of individual patient data, SENC/fast-SENC provided the most robust prediction of the composite endpoint of death, myocardial infarction, coronary revascularization and hospitalization due to heart failure, beyond clinical parameters and conventional CMR parameters, such as WMA, LV-ejection fraction and LGE (HR of 4.46, 95% CI = 2.96–6.73, p < 0.001)18,19,20. In addition, the assessment of myocardial strain by SENC/fast-SENC predicted all-cause mortality (HR of 3.0, 95% CI = 1.2–7.6, p = 0.02).
The MESA study offers a large database with healthy patients undergoing multiple examinations including CMR, whereas long-term follow-up of up to 8 years was available in the studies included in our systematic review22,23,24,26. Various strain variables assessed by HARP emerged as significant predictors of MACE and incident heart failure in asymptomatic individuals from different ethnicities, surpassing the value of clinical parameters and standard CMR variables22,23,24,26. The underlying pathophysiologic mechanism of this observation is not completely understood. Possibly, regional circumferential myocardial dysfunction represents a response to increased wall stress, reflecting local alterations of myocardial properties, such as fibrosis or ischemia due to microvascular disease or CAD. This increased afterload may contribute to the development of progressive myocardial remodeling and dysfunction, triggering poorer outcomes30. In addition, Ecc was significantly related to the LV mass index, which again underlines that the relationship between reduced strain and subclinical heart failure, which may convert to symptomatic disease due to adverse remodeling of the ventricle30,31. Notably, the thresholds provided for strain values in studies including asymptomatic patients have been largely inhomogeneous, ranging between − 10% and − 17%, whereas some studies selected relative cut-off values based on percentiles or tertiles. In addition, follow-up duration largely ranged between 2.2 and 15.9 years in these studies.
Four studies on the other hand, focused on the ability of SENC/fast-SENC for the prediction of outcomes in patients who underwent clinically indicated CMR examinations18,19,20,25. In two of these studies focusing on symptomatic patients with CAD, SENC and fast-SENC respectively, outperformed the ability of WMA for the diagnostic classification and risk stratification of patients with ischemic heart disease18,20. The results were similar although different stressors (dobutamine versus adenosine) were used for pharmacologic stimulation, which underlines the wide applicability of SENC for ischemia detection. In the two further studies, investigating patients who underwent clinically indicated CMR due to suspected ischemic and non-ischemic, structural cardiac diseases, the role HARP and fast-SENC for risk stratification was reestablished19,25. Hereby, patients with normal myocardium > 80% by fast-SENC exhibited better outcomes compared to patients with reduced baseline strain, who experienced higher mortality, higher rates for hospitalization due to heart failure symptoms and significantly more frequent transition rates from subclinical LV-dysfunction to symptomatic heart failure19. In addition, in our meta-analysis, based on individual patient data, SENC and fast-SENC provided the most robust prediction of MACE beyond clinical and conventional CMR parameters, exhibiting incremental value for the risk stratification of patients with a broad spectrum of cardiac diseases18,19,20. In addition, myocardial strain achieved prediction of all-cause mortality, which was not the case with conventional CMR markers.
Comparison to myocardial strain assessment by feature tracking imaging (FTI) and technical considerations
Several previous studies investigated the role of feature tracking imaging (FTI) for the risk stratification of patients with ischemic and non-ischemic heart disease32,33,34,35,36. In this regards, FTI derived GLS exhibited incremental value to CMR variables such as LV-ejection fraction and late gadolinium enhancement (LGE) for the prediction of MACE, including sudden cardiac death, resuscitated cardiac arrest and hospitalization due to heart failure in patients with hypertrophic cardiomyopathy32. In addition, LV strain parameters were independent predictors of MACE beyond clinical and conventional CMR markers, such as LVEF and LGE, in 162 patients with acute myocarditis, analyzed within a multi-center trial, while left atrial and right ventricular strain were less useful in this context33. In the same direction, previous studies underlined the incremental prognostic value of FTI in patients with non-ischemic dilative cardiomyopathy, beyond NYHA classification, LV-ejection fraction and LGE34. This could be confirmed in recent multi-center CMR studies, where FTI derived strain parameters surpassed the value of conventional functional CMR parameters, thus strengthening the body of evidence for the clinical implementation of strain for the risk stratification of patients with non-ischemic heart diseases35,36. Fewer studies, however, have investigated the value of FTI for the diagnostic classification or risk stratification of patients with ischemic heart disease37,38.
From a technical point of view FTI is based on pattern matching techniques across multiple images in a cardiac cycle39. By FTI, pixels are identified in one frame and followed in the next frames, enabling tracking of myocardial deformation with conventional cine images40. This is the foremost advantage of FTI since it does not require additional image acquisition and can estimate myocardial strain using clinical SSFP cine images. Different software packages with FTI however, use different algorithms for the calculation of strain, which results in different numerical values. These values are also different from CMR based HARP or SENC and fast-SENC and considerations have been raised, regarding strain over- or underestimation with FTI, which may be less sensitive in terms of disease detection41,42. In addition, strain reproducibility may be lower by FTI, compared to SENC, which may allow more comprehensive assessment of regional myocardial strain compared to FTI43,44. Such differences may be decisive for the diagnostic classification or risk stratification of patients with ischemic heart disease38,45. In this regard, FTI based strain exhibited lower precision than fast-SENC for the identification of segments with regional myocardial dysfunction due to ischemic heart disease46.
Considering the practical advantages of fast-SENC compared to HARP and SENC, it should be noted, that fast-SENC can be acquired during free-breathing of the patients, within a single heartbeat and high heart rates under inotropic stress CMR (> 150 bpm), which is of clinical importance, especially in patients with symptomatic heart failure, arrhythmias and chronic obstructive lung disease47. In addition, post-processing analysis with fast-SENC requires much lower time spent, compared to earlier sequences like HARP, thus increasing the potential of fast-SENC for translation into the clinical realm44,47. Finally, the use of artificial intelligence (AI) in CMR imaging protocols evaluating potential clinical predictors in patients with cardiovascular diseases continuously increases48. Incorporating AI in future studies may also increase the precision of strain algorithms for the risk stratification of patients, simultaneously reducing the required time spent for quantification analysis.
Limitations
Our study has some limitations. Thus, a classical meta-analysis was not possible due to substantial heterogeneity in the definition of outcomes between trials. We therefore performed individual patient data analysis in only 3 of the studies. In this regard, the strain sequences and acquisitions differed between the 3 studies, preventing the selection of a binary illustration and a universal cut-off value. In addition, studies were conducted with different scanners and different image quality can be anticipated, which may have affected the resultant image quality and the acquired strain values. However, our study cohort included patients with cardiac diseases based on largely heterogeneous etiologies, so that it may add important evidence for the value of myocardial strain for the risk stratification of symptomatic patients across a wide range of cardiac disorders. In addition, WMA and LGE were assessed visually in studies building the base for our meta-analysis, whereas data on T1 and T2 mapping and extracellular volume fractions (ECV) were not available. However, myocardial strain was also treated as a categorical variable in our statistical analysis, although quantification or semi quantification analysis has been available in the individual studies. In addition, T1 and T2 mapping techniques, although meanwhile established for the diagnostic work of patients with non-ischemic cardiomyopathies8,9,49, were not widely used in studies performed more than one decade ago.
Conclusions
Reduced myocardial strain derived from myocardial tagging by HARP, SENC or fast-SENC is a predictor of all-cause mortality and adverse outcomes in symptomatic patients with a wide range of ischemic or non-ischemic cardiac diseases as well as in asymptomatic individuals. Its value in terms of diagnostic classification and risk stratification is shown across multiple scanner and software vendors. SENC and fast-SENC provide the most robust prediction of MACE, beyond clinical parameters and conventional CMR parameters, such as WMA, LV-ejection fraction, and LGE. Especially in patients where contrast agent administration may be problematic, fast-SENC, which allows single heartbeat acquisitions of myocardial strain, and may represent a valuable alternative for the quantitative characterization of underlying myocardial pathologies.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Korosoglou, G., Giusca, S., Gitsioudis, G., Erbel, C. & Katus, H. A. Cardiac magnetic resonance and computed tomography angiography for clinical imaging of stable coronary artery disease. Diagnostic classification and risk stratification. Front. Physiol. 5, 291 (2014).
Salvador, D. B. Jr. et al. Diabetes and myocardial fibrosis: A systematic review and meta-analysis. JACC Cardiovasc. Imaging 15(5), 796–808 (2022).
Hudsmith, L. E. & Neubauer, S. Magnetic resonance spectroscopy in myocardial disease. JACC Cardiovasc. Imaging 2(1), 87–96 (2009).
Ferreira, V. M. et al. Cardiovascular magnetic resonance in nonischemic myocardial inflammation: Expert recommendations. J. Am. Coll. Cardiol. 72(24), 3158–3176 (2018).
Antiochos, P. et al. Prognostic value of stress cardiac magnetic resonance in patients with known coronary artery disease. JACC Cardiovasc. Imaging 15(1), 60–71 (2022).
Lipinski, M. J., McVey, C. M., Berger, J. S., Kramer, C. M. & Salerno, M. Prognostic value of stress cardiac magnetic resonance imaging in patients with known or suspected coronary artery disease: A systematic review and meta-analysis. J. Am. Coll. Cardiol. 62(9), 826–838 (2013).
Giusca, S. et al. Multi-parametric assessment of left ventricular hypertrophy using late gadolinium enhancement, T1 mapping and strain-encoded cardiovascular magnetic resonance. J. Cardiovasc. Magn. Reson. 23(1), 92 (2021).
Giusca, S. et al. Multiparametric early detection and prediction of cardiotoxicity using myocardial strain, T1 and T2 mapping, and biochemical markers: A longitudinal cardiac resonance imaging study during 2 years of follow-up. Circ. Cardiovasc. Imaging 14(6), e012459 (2021).
Korosoglou, G. et al. Diagnostic work-up of cardiac amyloidosis using cardiovascular imaging: Current standards and practical algorithms. Vasc. Health Risk Manag. 17, 661–673 (2021).
Garot, J. et al. Fast determination of regional myocardial strain fields from tagged cardiac images using harmonic phase MRI. Circulation 101(9), 981–988 (2000).
Sampath, S., Derbyshire, J. A., Atalar, E., Osman, N. F. & Prince, J. L. Real-time imaging of two-dimensional cardiac strain using a harmonic phase magnetic resonance imaging (HARP-MRI) pulse sequence. Magn. Reson. Med. 50(1), 154–163 (2003).
Osman, N. F., Sampath, S., Atalar, E. & Prince, J. L. Imaging longitudinal cardiac strain on short-axis images using strain-encoded MRI. Magn. Reson. Med. 46(2), 324–334 (2001).
Pan, L. et al. Real-time imaging of regional myocardial function using fast-SENC. Magn. Reson. Med. 55(2), 386–395 (2006).
Korosoglou, G. et al. Strain-encoded magnetic resonance: A method for the assessment of myocardial deformation. ESC Heart Fail. 6(4), 584–602 (2019).
Anker, S. D. et al. Empagliflozin in heart failure with a preserved ejection fraction. N. Engl. J. Med. 385(16), 1451–1461 (2021).
Korosoglou, G., Giusca, S. & Kelle, S. SGLT2 inhibition in HFpEF. Do we need more quantitative and load independent metrics to understand the results of the EMPEROR-preserved trial?. Front. Cardiovasc. Med. 8, 822968 (2021).
Page, M. J. et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 372, n71 (2021).
Korosoglou, G. et al. Strain-encoded cardiac magnetic resonance during high-dose dobutamine stress testing for the estimation of cardiac outcomes: Comparison to clinical parameters and conventional wall motion readings. J. Am. Coll. Cardiol. 58(11), 1140–1149 (2011).
Korosoglou, G. et al. Fast strain-encoded cardiac magnetic resonance for diagnostic classification and risk stratification of heart failure patients. JACC Cardiovasc. Imaging 14(6), 1177–1188 (2021).
Steen, H. et al. Fast-strain encoded cardiac magnetic resonance during vasodilator perfusion stress testing. Front. Cardiovasc. Med. 8, 765961 (2021).
Moons, K. G. M. et al. PROBAST: A tool to assess risk of bias and applicability of prediction model studies: Explanation and elaboration. Ann. Intern. Med. 170(1), W1–W33 (2019).
Choi, E. Y. et al. Prognostic value of myocardial circumferential strain for incident heart failure and cardiovascular events in asymptomatic individuals: The Multi-Ethnic Study of Atherosclerosis. Eur. Heart J. 34(30), 2354–2361 (2013).
Sharma, R. K. et al. Prognostic implications of left ventricular dyssynchrony for major adverse cardiovascular events in asymptomatic women and men: The Multi-Ethnic Study of Atherosclerosis. J. Am. Heart Assoc. 3(4), e000975 (2014).
Ambale-Venkatesh, B. et al. Diastolic function assessed from tagged MRI predicts heart failure and atrial fibrillation over an 8-year follow-up period: The multi-ethnic study of atherosclerosis. Eur. Heart J. Cardiovasc. Imaging 15(4), 442–449 (2014).
Mordi, I., Bezerra, H., Carrick, D. & Tzemos, N. The combined incremental prognostic value of LVEF, late gadolinium enhancement, and global circumferential strain assessed by CMR. JACC Cardiovasc. Imaging 8(5), 540–549 (2015).
Pezel, T., Bluemke, D. A., Wu, C. O., Lima, J. A. C. & Ambale, V. B. Regional strain score as prognostic marker of cardiovascular events from the Multi-Ethnic Study of Atherosclerosis (MESA). Front. Cardiovasc. Med. 9, 870942 (2022).
Bild, D. E. et al. Multi-ethnic study of Atherosclerosis: Objectives and design. Am. J. Epidemiol. 156(9), 871–881 (2002).
Koos, R. et al. Layer-specific strain-encoded MRI for the evaluation of left ventricular function and infarct transmurality in patients with chronic coronary artery disease. Int. J. Cardiol. 166(1), 85–89 (2013).
Neizel, M. et al. Strain-encoded MRI for evaluation of left ventricular function and transmurality in acute myocardial infarction. Circ. Cardiovasc. Imaging 2(2), 116–122 (2009).
Kass, D. A. Ventricular arterial stiffening: Integrating the pathophysiology. Hypertension 46(1), 185–193 (2005).
Rosen, B. D. et al. Left ventricular concentric remodeling is associated with decreased global and regional systolic function: The Multi-Ethnic Study of Atherosclerosis. Circulation 112(7), 984–991 (2005).
Negri, F. et al. Prognostic role of global longitudinal strain by feature tracking in patients with hypertrophic cardiomyopathy: The STRAIN-HCM study. Int. J. Cardiol. 345, 61–67 (2021).
Vos, J. L. et al. Comprehensive cardiovascular magnetic resonance-derived myocardial strain analysis provides independent prognostic value in acute myocarditis. J. Am. Heart Assoc. 11(19), e025106 (2022).
Buss, S. J. et al. Assessment of myocardial deformation with cardiac magnetic resonance strain imaging improves risk stratification in patients with dilated cardiomyopathy. Eur. Heart J. Cardiovasc. Imaging 16(3), 307–315 (2015).
Tang, H. S. et al. Prognostic utility of cardiac MRI myocardial strain parameters in patients with ischemic and nonischemic dilated cardiomyopathy: A multicenter study. AJR Am. J. Roentgenol. 220, 524–538 (2023).
Ochs, A. et al. Myocardial mechanics in dilated cardiomyopathy: Prognostic value of left ventricular torsion and strain. J. Cardiovasc. Magn. Reson. 23(1), 136 (2021).
Zhao, L., Zhang, C., Tian, J., DeLano, M. & Ma, X. Myocardial deformation assessed by MR feature tracking in groups of patients with ischemic heart disease. J. Magn. Reson. Imaging 54(3), 808–815 (2021).
Pierpaolo, P. et al. Left ventricular global myocardial strain assessment: Are CMR feature-tracking algorithms useful in the clinical setting?. Radiol. Med. 125(5), 444–450 (2020).
Salerno, M. Feature tracking by CMR: A “Double Feature”?. JACC Cardiovasc. Imaging 11(2 Pt 1), 206–208 (2018).
Ruijsink, B. et al. Fully automated, quality-controlled cardiac analysis from CMR: Validation and large-scale application to characterize cardiac function. JACC Cardiovasc. Imaging 13(3), 684–695 (2020).
Augustine, D. et al. Global and regional left ventricular myocardial deformation measures by magnetic resonance feature tracking in healthy volunteers: Comparison with tagging and relevance of gender. J. Cardiovasc. Magn. Reson. 15(1), 8 (2013).
Wehner, G. J. et al. Comparison of left ventricular strains and torsion derived from feature tracking and DENSE CMR. J. Cardiovasc. Magn. Reson. 20(1), 63 (2018).
Wu, L. et al. Feature tracking compared with tissue tagging measurements of segmental strain by cardiovascular magnetic resonance. J. Cardiovasc. Magn. Reson. 16(1), 10 (2014).
Giusca, S. et al. Reproducibility study on myocardial strain assessment using fast-SENC cardiac magnetic resonance imaging. Sci. Rep. 8(1), 14100 (2018).
Cao, J. J. et al. A comparison of both DENSE and feature tracking techniques with tagging for the cardiovascular magnetic resonance assessment of myocardial strain. J. Cardiovasc. Magn. Reson. 20(1), 26 (2018).
El-Saadi, W. et al. A head-to-head comparison of myocardial strain by fast-strain encoding and feature tracking imaging in acute myocardial infarction. Front. Cardiovasc. Med. 9, 949440 (2022).
Korosoglou, G. et al. Real-time fast strain-encoded magnetic resonance imaging to evaluate regional myocardial function at 3.0 Tesla: Comparison to conventional tagging. J. Magn. Reson. Imaging 27(5), 1012–1018 (2008).
Backhaus, S. J. et al. Artificial intelligence fully automated myocardial strain quantification for risk stratification following acute myocardial infarction. Sci. Rep. 12(1), 12220 (2022).
ausdemSiepen, F. et al. T1 mapping in dilated cardiomyopathy with cardiac magnetic resonance: Quantification of diffuse myocardial fibrosis and comparison with endomyocardial biopsy. Eur. Heart J. Cardiovasc. Imaging 16(2), 210–216 (2015).
Acknowledgements
Open Access funding was enabled and organized by Projekt DEAL (Charité—Universitätsmedizin Berlin, Germany).
Author information
Authors and Affiliations
Contributions
All authors wrote the parts of the main manuscript text, prepared figures and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
H.S., G.K. and S.K. received research grants and honoraria from Myocardial Solutions, S.K. received funding from the DZHK (German Centre for Cardiovascular Research) and the BMBF (German Ministry of Education and Research), is supported by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation)—SFB-1470—B06 and received an unrestricted research grant from Philips Health Care, Germany. S.K. and H.S. own stock options of Myocardial Solutions. All other authors declare that they have no competing interests regarding this manuscript.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Korosoglou, G., Sagris, M., André, F. et al. Systematic review and meta-analysis for the value of cardiac magnetic resonance strain to predict cardiac outcomes. Sci Rep 14, 1094 (2024). https://doi.org/10.1038/s41598-023-50835-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-50835-5
- Springer Nature Limited