Abstract
UBE3A is an E3 ubiquitin ligase encoded by the neurally imprinted UBE3A gene. The abundance and subcellular distribution of UBE3A has been the topic of many previous studies as its dosage and localization has been linked to neurodevelopmental disorders including Autism, Dup15q syndrome, and Angelman syndrome. While commercially available antibodies have been widely employed to determine UBE3A localization, an extensive analysis and comparison of the performance of different UBE3A antibodies has not been conducted. Here we evaluated the specificities of seven commercial UBE3A antibodies in two of the major experimental models used in UBE3A research, mouse and human pluripotent stem cell-derived neural cells and tissues. We tested these antibodies in their two most common assays, immunofluorescence and western blot. In addition, we also assessed the ability of these antibodies to capture dynamic spatiotemporal changes of UBE3A by utilizing human cerebral organoid models. Our results reveal that among the seven antibodies tested, three antibodies demonstrated substantial nonspecific immunoreactivity. While four antibodies show specific localization patterns in both mouse brain sections and human cerebral organoids, these antibodies varied significantly in background signals and staining patterns in undifferentiated human pluripotent stem cells.
Similar content being viewed by others
Introduction
UBE3A is a HECT domain E3 ubiquitin ligase that targets substrate proteins, including itself, for proteasomal degradation1. In addition to this role, UBE3A also has a putative role in transcriptional regulation where it co-activates steroid hormone receptors such as progesterone and estrogen2. Although biallelic in most tissues, UBE3A is expressed exclusively from the maternal allele in neurons due to tissue specific paternal imprinting3. Mutations or deletions of maternal UBE3A, leads to loss of UBE3A in neurons and Angelman syndrome (AS), a severe neurodevelopmental disorder characterized by delayed development, speech impairment, ataxia, and intellectual disability4. On the other hand, increased levels of UBE3A activity due to gene duplication or mutation has been associated with Dup15q syndrome and Autism Spectrum Disorder (ASD)5,6,7.
In addition to its imprinted expression, one of the key molecular features of UBE3A is its distinct subcellular localization patterns, potentially regulated by its three isoforms8,9. Although it has a cytoplasmic component, UBE3A is predominantly nuclear in human and mouse neurons10,11,12,13; however, its roles in these subcellular compartments in different cell types and developmental stages have not been fully uncovered. Previous reports show that while mice lacking the nuclear UBE3A isoform (mIso3) exhibit electrophysiological and behavioral deficits similar to other AS model mice13, the cytoplasmic UBE3A isoform (mIso2) is important for polarized dendrite morphogenesis14. Taken together, these findings indicate that both cytoplasmic and nuclear components of UBE3A may be critical for proper brain development and underscore the importance of studying its subcellular distribution and abundance.
The primary methods to determine UBE3A localization have been the use of commercially available antibodies in immunoassays such as western blot, subcellular fractionation followed by immunoblotting, or immunostaining of fixed cell culture or tissue samples. Although a few commercial UBE3A antibodies have been rigorously validated in the published literature for mouse brain tissue12 and human stem cell derived neurons9, others are less well documented. Furthermore, except for the mouse monoclonal antibody from Sigma-Aldrich (SAB1404508)9 and rabbit polyclonal antibody from Bethyl Laboratories (A300-351A)15, the specificities of most antibodies in human samples remain unknown, and even when validated, were tested only in limited cell types. With the growing use of human pluripotent stem cell-derived neuronal cultures and three-dimensional cerebral organoids as models to study human UBE3A biology, it will be important to assess the specificities of UBE3A antibodies in human stem cell-derived systems as well as mouse models. Human stem cells and cerebral organoids are therefore the major focus of this work, with the use of mouse tissues to provide comparisons to prior work and to identify potential species-specific differences.
In this work we evaluated seven commercial UBE3A antibodies including those most commonly used in the field. We tested antibody specificities in immunofluorescence (IF) and western blotting (WB) assays using AS model mice and the H9UBE3A m−/p− hESC line as negative controls. In addition to specificity, we also evaluated the ability of these antibodies to capture important disease relevant features of UBE3A through the use of whole brain human cerebral organoid (hCO) models generated from a large panel of human pluripotent stem cell (hPSC) lines including isogenic wild-type (H9WT), AS (H9UBE3A m−/p+), and double knockout models (H9UBE3A m−/p−), two additional WT hPSC lines (H1WT and an hiPSC line derived from a neurotypical donor, hiPSCWT), and two patient-derived AS hiPSC lines (derived from a patient with a large chromosome15: q11-13 deletion, hiPSCASdel, and F583S point mutation, hiPSCASmut).
Results
Four commercial UBE3A antibodies show specific and similar localization patterns in mouse brain sections
In this study we evaluated seven different commercially available UBE3A antibodies from five vendors (Fig. 1A,B and S1A). The criteria of selection for the antibodies ranged from their previous use in the literature, applications reported as appropriate by the vendors, and the epitopes they were raised against. The epitopes these antibodies were raised against cluster around two functional UBE3A domains: the AZUL domain that was recently implicated in protein localization13 and the E6 oncoprotein binding domain (Figs. 1B and S1A). While six of the selected antibodies target common regions in all protein coding UBE3A isoforms (human isoforms hUBE3A iso1,2,3 and mouse isoforms mUbe3a iso2,3), the Proteintech UBE3A antibody was raised against an epitope which includes the 12 amino acid region that is unique to the long UBE3A isoforms (hUBE3A iso2-3 and mUbe3a iso2) (Figs. 1A,B and S1A).
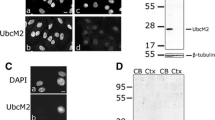
Four commercial UBE3A antibodies show specific and similar localization patterns in mouse brain sections. (A) Details of commercial UBE3A antibodies used in this study. (B) Schematic representation of the UBE3A protein, its known functional domains, epitopes used to raise the antibodies used in this study, and corresponding regions in different protein isoforms. Depicted are the N-terminal extensions of UBE3A mouse isoform 2 (mUBE3A Iso2) and human isoform 2 and 3 (hUBE3A Iso3), the N-terminal zinc-binding AZUL domain, HERC2 binding domain, E6 binding domain and the C-terminal HECT domain. (C) Immunofluorescence analysis from wild-type (Ube3a m+/p+) and Angelman Syndrome (Ube3a m−/p+) mouse models using seven different UBE3A antibodies.
We first evaluated the specificity of the selected antibodies in an IF assay with adult (P191) WT (Ube3am+/p+) and AS (Ube3am−/p+) mouse brain sections. Mouse models are widely used in the field and they have been shown to successfully model some of the core molecular features of UBE3A including imprinting of the paternal allele and nuclear localization of Ube3a in neurons16. They have also been extensively studied using IF, especially with the Sigma-Aldrich antibody (SAB1404508), and would therefore serve as a point of comparison for our IF methodology which closely mirrors prior work12. We focused specifically on cortical regions with a high density of neurons, as indicated by MAP2 staining, that previous work showed exhibits strong nuclear Ube3a signal in WT mice and no Ube3a signal in AS mice10,12 (Figs. 1C and S1B).
Our analysis showed that the antibodies from Bethyl Laboratories performed similarly to the commonly used Sigma-Aldrich antibody (SAB1404508) and exhibited the expected prominent nuclear localization pattern in WT neurons (Fig. 1C). Moreover, all three antibodies showed drastically reduced immunoreactivity in AS model mice (Ube3am−/p+) due to paternal imprinting. Among them, the Sigma-Aldrich antibody (SAB1404508) demonstrated the lowest background signal followed by Bethyl Laboratories A300-352A and Bethyl Laboratories A300-351A (Fig. 1C). In addition to these three antibodies, the other Sigma-Aldrich antibody (E8655) also showed specific, but weaker staining in neuronal nuclei (Fig. 1C). While the Bio-Rad and Novus Biologicals antibodies did not show any specific staining pattern, the Proteintech antibody showed the expected nuclear labeling in WT mouse neurons. Surprisingly, AS sections also showed a very similar nuclear staining pattern with the Proteintech antibody indicating its potential nonspecific labeling (Fig. 1C).
Commercial antibodies vary significantly in background signal in double UBE3A knockout pluripotent stem cells
Mouse models were central to many pivotal discoveries in the field. However, given the 100 million years of divergent evolution between rodents and primates, the patterns of UBE3A expression in humans may not be fully conserved. Recent studies support this notion where differences in the localization patterns and mechanisms of human and mouse isoforms were discovered8. This underscores the importance of investigating the subcellular distribution of UBE3A in human specific models. While important previous studies examined the expression patterns of UBE3A in human cortical samples and hiPSC-derived neurons using the Sigma-Aldrich UBE3A antibody (SAB1404508)11,13, the specificity of this commonly used antibody has not been evaluated in human knockout models until recently9. Here we expanded upon this analysis and evaluated the specificities of the 7 antibodies in IF using H9WT and H9UBE3A m−/p− hESC lines (Fig. 2). Moreover, we also investigated the staining patterns in five additional hPSC lines including three AS models (Figure S2). Since the imprinting of the paternal allele is a characteristic specific to mature neurons not present in these cultures, we expected all undifferentiated stem cell lines including the AS models to express UBE3A.
Our results showed that commercial antibodies have different staining patterns in H9WT hESCs and vary significantly in their background signal in H9UBE3A m−/p− hESCs (Fig. 2). While Bethyl Laboratories antibodies showed a diffused staining pattern across nuclei and cytosol, the immunoreactivity of the Sigma-Aldrich antibodies was consistently enriched primarily in the cytosol across all hPSCs tested in this study (Figs. 2 and S2). Surprisingly, both Sigma-Aldrich UBE3A antibodies demonstrated significant and strong background signal in H9UBE3A m−/p− hESCs (Fig. 2). In addition, the Bethyl Laboratories antibody targeting the E6-binding region (A300-352A) showed weaker labeling in H9WT hESCs and higher nonspecific signal in H9UBE3A m−/p− hESCs when used at the same concentration as the Bethyl Laboratories antibody targeting the region near the AZUL domain (A300-351A) (Figs. 1A,B, 2 and S2).
The Bio-Rad, Proteintech, and Novus Biologicals antibodies showed variable staining patterns. The Bio-Rad antibody was diffuse across nuclei and cytosol. The Proteintech antibody was primarily nuclear. The Novus Biologicals antibody had a stronger signal in the cytosol with a diffuse nuclear labeling (Figs. 2 and S2). All three of these antibodies also revealed strong staining signal in H9UBE3A m−/p− hESCs (Fig. 2). Among the tested antibodies, Bethyl Laboratories UBE3A antibody (A300-351A) showed the lowest background in H9UBE3A m−/p− hESCs compared to its signal in H9WT hESCs (Fig. 2).
Four commercial antibodies capture similar neurodevelopmental localization changes in differentiated hCOs
In addition to hPSCs, we and others have previously used human stem cell-derived neurons17,18 and hCOs15,19 as promising experimental models for AS research. In particular, in a recent study our group showed that hCOs can recapitulate the important disease relevant molecular changes surrounding UBE3A including paternal imprinting and nuclear localization in neurons15. Here, we conducted IF experiments to evaluate not only antibody specificities, but also the ability of the selected antibodies to capture these salient molecular features of UBE3A using 12 week hCOs generated from seven different hPSC lines including three AS models (Figs. 3, S3 and S4).
Four commercial antibodies capture similar neurodevelopmental localization patterns in differentiated hCOs. (A,B) Arrow heads: Neurons (MAP2+/SOX2−). Double arrows: Progenitor cells (SOX2+). Arrows: DAPI+ cells that are MAP2−/SOX2−/UBE3A−. (A) Immunofluorescence results from 12 week hCOs generated from H9 wild type (H9WT) and H9 double UBE3A knockout (H9UBE3 m−/p−) hESCs using seven different UBE3A antibodies. (B) Immunofluorescence results using antibody A300-351A on 12 week hCOs generated from H9 maternal UBE3A knockout hESCs (H9UBE3A m−/p+), wild type H1 hESCs (H1WT), hiPSCs derived from a neurotypical donor (hiPSCWT), hiPSCs derived from a patient with a large deletion in UBE3A region (hiPSCASdel), and hiPSCs derived from a patient with a point mutation (F583S) in UBE3A (hiPSCASmut).
In contrast to their different staining patterns observed during the pluripotent stage, both Bethyl Laboratories and both Sigma-Aldrich antibodies showed the expected nuclear staining pattern in neurons (MAP2+/SOX2−) of H9WT, hiPSCWT and H1WT hCOs (Figs. 3 and S3). Among these four antibodies, SAB1404508 showed the lowest background signal in H9UBE3A m−/p− hCOs, A300-352A showed the highest background, while the nuclear localization pattern was the cleanest across all WT cell lines with A300-351A (Figs. 3 and S3A).
In addition, both Bethyl Laboratories antibodies showed reduced signal similar to their background levels in the neuronal regions (MAP2+/SOX2−) of both AS model hCOs (H9UBE3A m−/p+ and hiPSCASdel) indicating imprinting and silencing of the paternal allele, as was previously observed15 (Figs. 3B and S3B). The signal from the Sigma-Aldrich antibodies in H9UBE3A m−/p+ and hiPSCASdel hCOs was stronger than their background levels, but it was reduced compared to WT hCOs, and the staining pattern appeared to be nonspecific particularly in neuronal regions (MAP2+/SOX2−) (Figure S3B). Collectively these results indicate that these four antibodies can capture the paternal imprinting of UBE3A in the neurons of hCOs.
Furthermore, all four antibodies also showed a striking nuclear localization pattern in the neurons of hiPSCASmut hCOs which was an anticipated result (Figs. 3B and S3B). Previously it was shown that UBE3A localization is regulated by the N-terminal extension and the AZUL domain8,13. The hiPSCASmut line was derived from a patient with a point mutation (F583S) on the N-terminal region of the N lobe of HECT domain, and previous reports indicate that this variant does not exhibit protein folding issues20. Furthermore, immunogens of the antibodies tested in this study do not directly align with the mutated region, with one exception being mouse monoclonal Sigma-Aldrich antibody (E8655) that was raised against full length human recombinant UBE3A (isoform1).
Bio-Rad, Proteintech and Novus Biologicals antibodies showed similar staining patterns and signal intensities across all hCO models including the H9UBE3A m−/p− and AS models. Interestingly, while the staining patterns of the Bio-Rad and Novus Biologicals antibodies appeared to be diffuse and nonspecific, the Proteintech antibody exhibited strong nuclear labeling in neuronal regions of all hCO models (Figs. 3A and S3B). To ensure the elevated background signal observed with this antibody in H9UBE3A m−/p− hCOs was not due to high antibody concentration, we conducted another IF assay using serial dilutions of this antibody in H9WT and H9UBE3A m−/p− hCOs. The results showed that even at low concentrations, the Proteintech UBE3A antibody showed similar labeling intensity between H9WT and H9UBE3A m−/p− hCOs (Figure S4A). Importantly, none of the secondary antibodies showed significant background labeling when used without the primary antibodies (Figure S4B).
Four commercial antibodies show specific labeling of UBE3A in western blotting
Another commonly used method in the field to probe UBE3A expression is WB. While several important previous studies showed the specificity of Bethyl Laboratories and Sigma-Aldrich antibodies using adult AS mouse models12,13,21,22,23, the human specific activity of these antibodies in WB assays had not been tested previously with a double knockout cell line.
Here we used whole cell lysates from H9WT and H9UBE3A m−/p− hESCs to evaluate the specificity of the selected UBE3A antibodies in WB (Fig. 4). In addition to these cell lines, we also used WT adult mouse cerebellum (mCER) samples as controls (Fig. 4). Our results showed that both Bethyl Laboratories and both Sigma-Aldrich antibodies labeled specific bands in WT samples at ~ 100 kDa which is the predicted molecular weight for UBE3A (Uniprot: Q05086-1, 2, 3). While the Sigma-Aldrich antibody (E8655) showed a faint band in H9UBE3A m−/p− hESCs lysate, when this condition was repeated in another blot, this band was much weaker and appeared as a smear in the membrane (Figs. 4A, S4C,D). In addition, low molecular weight, faint nonspecific bands were observable in the Bethyl Laboratories antibodies, particularly in the mCER samples (Fig. 4A). The Sigma-Aldrich antibody (SAB1404508) also showed weak, low molecular weight, nonspecific bands in H9WT hESC samples. When used at the same concentration, Bethyl Laboratories antibody A300-352A showed significantly weaker staining compared to A300-351A which is in agreement with the IF results (Fig. 4).
Four commercial antibodies show specific labeling of UBE3A in western blots. (A) Immunoblot analysis of UBE3A in whole cell lysates from H9 wild type (H9WT) hESCs, H9 double UBE3A knockout (H9UBE3A m−/p−) hESCs and wild-type mouse cerebellum (mCER) using seven different UBE3A antibodies. Full gels are depicted; vertical lines are drawn only for clarity. (B) Quantitative analysis of the UBE3A bands, normalized to GAPDH.
The Bio-Rad and Novus Biologicals antibodies showed a similar band pattern to each other, with two higher molecular weight nonspecific bands in human cell lysates. The largest band was missing in mCER samples highlighting the species-specific differences in antibody reactivity (Fig. 4A). The Proteintech antibody also demonstrated nonspecific labeling around 75 kDa (Fig. 4A). The signal strength in the nonspecific bands observed with these three antibodies appeared to be comparable to the expected bands around 100 kDa, indicating that the Bio-Rad, Novus Biologicals and Proteintech antibodies have similar affinities towards UBE3A as they do to other off-target proteins.
Discussion
During the last two decades, commercial antibodies have been widely used in scientific reports aimed to determine the abundance and subcellular distribution of UBE3A/Ube3a. While individual studies validated specific antibodies using AS and double knockout models in mice12 and human neurons9, the present work provides the first broad analysis dedicated to the evaluation of multiple commercial UBE3A antibodies in IF and WB assays using both mouse and human specific models that include multiple neural cell types.
Our analysis in mouse brain sections with the commonly used Sigma-Aldrich antibody (SAB1404508) showed that the IF protocol used in this study produced comparable results to previously published work10,12. Among the seven UBE3A antibodies tested, the Bethyl Laboratories antibodies (A300-351A and -352A) and another Sigma-Aldrich (E8655) antibody showed similar specific staining patterns in mouse brain tissue. However, while Bio-Rad and Novus Biologicals antibodies were raised against the same epitope sequence as the established Sigma-Aldrich antibody (SAB1404508), these antibodies revealed different patterns of reactivity with significant nonspecific signal in AS mice, potentially due to the differences in selected antibody clones (3E5 for Sigma-Aldrich SAB1404508, 2F6 for Bio-Rad and Novus Biologicals). While consistent with each other, Bio-Rad and Novus Biologicals antibodies also produced a distinct nonspecific band pattern in WB analysis with human hPSC lysates. These results indicate that these antibodies recognize unknown proteins of diverse molecular sizes other than UBE3A and highlight the importance of clone selection and validation in monoclonal antibody production.
Among the tested antibodies, the Proteintech antibody yielded the most unusual results. When tested in IF with adult WT mouse brain and WT hCO samples, it demonstrated the expected nuclear staining pattern in neuronal regions. However, it also showed the identical staining pattern and comparable signal strength in AS mouse sections and double knockout hCO sections. Consistent with this result, even though the WB analysis for the 100 kDa UBE3A target appeared as expected in WT lysate while being absent in the double UBE3A knockout samples, the Proteintech antibody also showed another band with similar signal strength to the UBE3A bands which appeared in both WT and double knockout cell lysates. These results indicate that the immunoreactivity observed with the Proteintech antibody is not solely related to the presence or absence of the UBE3A and that this antibody reacts with another distinct unknown protein. Although basic local alignment search tool (BLAST) analysis did not yield another potential target, the comparison of the epitope sequence used to raise this antibody with the protein coding UBE3A isoform sequences showed that it does not have 100% sequence homology with the most common UBE3A variant (hUBE3A iso1 and mUbe3a iso3) (Figs. 1A, B and S1A)8,9. This may imply a reduced affinity for the majority of the UBE3A protein expressed in a cell. Moreover, the polyclonal nature of this antibody may also contribute to its cross-reactivity with other targets. Due to its misleading IF staining pattern in WT models that looks like a specific signal, future studies that plan to utilize this antibody to determine UBE3A expression and localization should make sure the proper negative controls are employed.
Our IF analysis specifically in human hPSCs in their undifferentiated states revealed distinct staining patterns between the antibodies. These differences were stark, particularly between the polyclonal Bethyl Laboratories antibodies (diffuse cytoplasmic and nuclear staining) and the monoclonal Sigma-Aldrich antibodies (strongly cytoplasmic staining). In contrast, all four antibodies performed similarly in mouse brain sections and differentiated hCOs. Our analysis with the Sigma-Aldrich (SAB1404508) antibody also showed differences in staining pattern and background signal compared to a recently published study using the same double knockout hPSC line9. This discrepancy between the two studies could be due to differences in culture conditions (i.e. media reagents, feeder vs feeder-free conditions), where the morphology and gene expression state of pluripotent cells can vary substantially. This possible explanation is also supported by the results from hCOs where both Bethyl Laboratories and both Sigma-Aldrich antibodies demonstrated the expected nuclear staining pattern in the neurons of WT hCOs, captured indications of paternal imprinting in AS hCOs, and showed significantly reduced signal in hCOs generated from the double knockout hESC line.
This study highlights the importance of testing multiple UBE3A antibodies in different detection methods. The two Sigma-Aldrich antibodies have been the primary choice in the majority of the literature. Based on our results they are suitable for both IF and WB assays, albeit with a still unknown nonspecific signal in undifferentiated double UBE3A knockout hESCs. Both Bethyl Laboratories antibodies have previously been used in WB of mouse, hPSCs, and hPSC-derived neuronal lysates9,24,25; however, IF analysis with these antibodies was limited to a few studies in mouse models21,26. The present work indicates that the Bethyl A300-351A is a viable antibody option with performance at least on par with the Sigma-Aldrich antibodies and could be used for future IF and WB assays in both mouse and human model systems. Finally, it is not advised to use the Bio-Rad, Novus Biologicals, and Proteintech antibodies as they exhibit considerable non-specific staining in both IF and WB applications.
Methods
Cell culture and human cerebral organoid generation
Feeder-independent H9 and H1 hESCs (WA09 and WA01) were obtained from WiCell. Neurotypical hiPSCs were purchased from Systems Biosciences (Cat# SC102-A1). Angelman Syndrome (AS) hiPSCs with a class II deletion (ASdel) were developed by the laboratories of Stormy J. Chamberlain and Marc Lalande at the University of Connecticut27 and obtained from Kerafast. H9UBE3A m−/p− and H9UBE3A m−/p+ cells with a 66 kb deletion (chr15: 25338949–25405676) and AS point mutation hiPSC line (F583S) (ASmut) were provided by Dr. Stormy Chamberlain (UCONN). The H9UBE3Am−/p− hESC cell line was generated and validated by Sirois and colleagues9. Our group also confirmed the deletion in these cells in an earlier study15. All cell lines were maintained in 6-well tissue culture dishes (Fisher Scientific) coated with 0.5 mg/cm2 Vitronectin (VTN-N) (Thermo Fisher) in E8 medium (Thermo Fisher) and passaged using standard protocols. Whole brain human cerebral organoids (hCOs) were generated and maintained using the same protocols as described28. Cells and hCOs were maintained in a humid incubator at 37 °C with 5% CO2.
Animals
All methods to generate the original animal sections were carried out in accordance with the relevant guidelines and regulations of the University of North Carolina Institutional Animal Care and Use Committee. All experimental protocols using vertebrate animals to generate the original animal sections were approved by the University of North Carolina Institutional Animal Care and Use Committee. This study was carried out in compliance with the Animal Research: Reporting of in vivo Experiments (ARRIVE) guidelines. Wild-type (WT) (Ube3am+/p+) and Angelman Syndrome (AS) (Ube3am−/p+) mouse brain sections were residual samples from unrelated experiments and were provided by Dr. Ben Philpot (UNC). Mice carrying the Ube3a knock-out allele were originally generated by the laboratory of Dr. Arthur Beaudet29 and back-crossed to a congenic C57BL/6J background. AS model mice were generated by mating WT male mice to female mice with paternal inheritance of the Ube3a knock-out allele (Ube3am+/p−), which themselves are phenotypically normal29. Genotyping regarding the AS model mice has previously been described10,12. The sections used in this study are from a single perfused animal for each genotype (Ube3am+/p+ and Ube3am−/p+).
Histology and immunofluorescence
Tissues and cells were fixed and immunostained as previously described15. Briefly, tissues and cells were fixed in 4% paraformaldehyde for 15 min at 4 °C followed by 3 × 10 min PBS washes. Tissues were allowed to sink in 30% sucrose overnight and then embedded in 10% gelatin/7.5% sucrose. Embedded tissues were frozen in an isopentane bath between − 50 and − 30 °C and stored at − 80 °C. Frozen blocks were cryosectioned to 30 µm. For immunohistochemistry, sections were blocked and permeabilized in 0.2% Triton X-100 and 5% normal donkey serum in PBS. Sections were incubated with primary antibodies in 0.2% Triton X-100, 5% normal donkey serum in PBS overnight at 4 °C in a humidity chamber. Sections were then incubated with secondary antibodies in 0.2% Triton X-100, 5% normal donkey serum in PBS for 2 h at room temperature, and nuclei were stained with DAPI (Invitrogen). Slides were mounted using ProLong Antifade Diamond (Thermo Fisher Scientific). Secondary antibodies used were donkey Alexa Fluor 488, 546 and 647 conjugates (Invitrogen, 1:250). Details on primary antibodies can be found below. Images were taken using a Nikon A1R confocal microscope (Nikon Instruments). High magnification images were captured using thin (1.5 μm) optical sections. All samples immunostained with the same antibody were processed at the same time, imaged using the same microscope settings, and adjusted identically. UBE3A antibody information including citations and antibody specifications provided by the vendors have been downloaded and stored on the Keung Lab GitHub page to provide access in the future if any of the antibodies used in this study are discontinued. The link to the publicly available GitHub repository is http://github.com/keung-lab/UBE3A_Antibodies.
Additional primary antibodies used in immunofluorescence assays:
Antigen | Host | Supplier | Cat. No | RRID | Dilution |
|---|---|---|---|---|---|
SOX2 | Goat | R&D systems | AF2018 | AB_355110 | 1:20 |
MAP2 | Mouse | Millipore | M1406 | AB_477171 | 1:250 |
MAP2 | Rabbit | Millipore | AB5622 | AB_91939 | 1:500 |
Western blot
Whole cell lysates from H9WT, H9UBE3A m−/p−, and mouse cerebellum (mCER) derived from C57BL/6 mice were prepared as previously described30. Proteins separated in PAGE gel were transferred to PVDF membranes and treated with UBE3A antibodies (Figs. 1A and S1A). GAPDH was used as a loading control. The antigen–antibody complexes were visualized after incubation with the corresponding HRP-conjugated secondary antibody (anti-mouse-HRP and anti-rabbit-HRP) and ECL substrate (Bio-Rad, Clarity Western ECL Substrate). Signals from enhanced chemiluminescence were detected with a Licor Odyssey Fc imaging system using the Chemi channel after 10 min (UBE3A) and 5 min (GAPDH) exposures. The quantitative analysis was performed using Image Studio 5.2 Software (LI-COR Biosciences). While the membrane was cut vertically to allow for treatment with different UBE3A antibodies, they were treated together with ECL solution and imaged together using same exposure settings. Details on primary and secondary antibodies can be found below.
Primary and secondary antibodies used in immunoblotting assays:
Antigen | Host | Supplier | Cat. No | RRID | Dilution |
|---|---|---|---|---|---|
GAPDH | Mouse | Calbiochem | CB1001 | AB_2107426 | 2 μg/ml |
m-IgGκ BP-HRP | mouse IgGκ light chain | Santa Cruz Biotechnology | sc516102 | AB_2687626 | 1:2500 |
r-IgG-HRP | Mouse anti-rabbit | Santa Cruz Biotechnology | sc2357 | AB_628497 | 1:2500 |
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Huibregtse, J. M., Scheffner, M. & Howley, P. M. Cloning and expression of the cDNA for E6-AP, a protein that mediates the interaction of the human papillomavirus E6 oncoprotein with p53. Mol. Cell. Biol. 13, 775–784 (1993).
Ramamoorthy, S. & Nawaz, Z. E6-associated protein (E6-AP) is a dual function coactivator of steroid hormone receptors. Nucl. Recept. Signal. 4, e006 (2007).
Rougeulle, C., Glatt, H. & Lalande, M. The Angelman syndrome candidate gene, UBE3A/E6-AP, is imprinted in brain. Nat. Genet. 17, 14–15 (1997).
Kishino, T., Lalande, M. & Wagstaff, J. UBE3A/E6-AP mutations cause Angelman syndrome. Nat. Genet. 15, 70–73 (1997).
Yi, J. J. et al. The autism-linked UBE3A T485A mutant E3 ubiquitin ligase activates the Wnt/β-catenin pathway by inhibiting the proteasome. J. Biol. Chem. 292, 12503–12515 (2017).
Hogart, A., Wu, D., LaSalle, J. M. & Schanen, N. C. The comorbidity of autism with the genomic disorders of chromosome 15q11.2–q13. Neurobiol. Dis. 38, 181–191 (2010).
Urraca, N. et al. The interstitial duplication 15q11.2–q13 syndrome includes autism, mild facial anomalies and a characteristic EEG signature. Autism Res. 6, 268–279 (2013).
Zampeta, I. F. et al. Conserved UBE3A subcellular distribution between human and mice is facilitated by non-homologous isoforms. Hum. Mol. Genet. https://doi.org/10.1093/hmg/ddaa194 (2020).
Sirois, C. L. et al. Abundance and localization of human UBE3A protein isoforms. Hum. Mol. Genet. https://doi.org/10.1093/hmg/ddaa191 (2020).
Burette, A. C. et al. Subcellular organization of UBE3A in neurons. J. Comp. Neurol. 525, 233–251 (2017).
Burette, A. C. et al. Subcellular organization of UBE3A in human cerebral cortex. Mol. Autism 9, 54 (2018).
Judson, M. C., Sosa-Pagan, J. O., Del Cid, W. A., Han, J. E. & Philpot, B. D. Allelic specificity of Ube3a expression in the mouse brain during postnatal development. J. Comp. Neurol. 522, 1874–1896 (2014).
Avagliano Trezza, R. et al. Loss of nuclear UBE3A causes electrophysiological and behavioral deficits in mice and is associated with Angelman syndrome. Nat. Neurosci. 22, 1235–1247 (2019).
Miao, S. et al. The Angelman syndrome protein Ube3a is required for polarized dendrite morphogenesis in pyramidal neurons. J. Neurosci. 33, 327–333 (2013).
Sen, D., Voulgaropoulos, A., Drobna, Z. & Keung, A. J. Human cerebral organoids reveal early spatiotemporal dynamics and pharmacological responses of UBE3A. Stem Cell Rep. https://doi.org/10.1016/j.stemcr.2020.08.006 (2020).
LaSalle, J. M., Reiter, L. T. & Chamberlain, S. J. Epigenetic regulation of UBE3A and roles in human neurodevelopmental disorders. Epigenomics 7, 1213–1228 (2015).
Fink, J. J. et al. Disrupted neuronal maturation in Angelman syndrome-derived induced pluripotent stem cells. Nat. Commun. 8, 15038 (2017).
Hsiao, J. S. et al. A bipartite boundary element restricts UBE3A imprinting to mature neurons. Proc. Natl. Acad. Sci. 116, 2181–2186 (2019).
Sun, A. X. et al. Potassium channel dysfunction in human neuronal models of Angelman syndrome. Science 366, 1486–1492 (2019).
Offensperger, F. et al. Identification of small-molecule activators of the ubiquitin ligase E6AP/UBE3A and Angelman syndrome-derived E6AP/UBE3A variants. Cell Chem. Biol. 27, 1–11 (2020).
Sato, M. & Stryker, M. P. Genomic imprinting of experience-dependent cortical plasticity by the ubiquitin ligase gene Ube3a. Proc. Natl. Acad. Sci. 107, 5611–5616 (2010).
Meng, L. et al. Truncation of Ube3a-ATS unsilences paternal Ube3a and ameliorates behavioral defects in the Angelman syndrome mouse model. PLoS Genet. 9, e1004039 (2013).
Bailus, B. J. et al. Protein delivery of an artificial transcription factor restores widespread Ube3a expression in an Angelman syndrome mouse brain. Mol. Ther. 24, 548–555 (2016).
Gustin, R. M. et al. Tissue-specific variation of Ube3a protein expression in rodents and in a mouse model of Angelman syndrome. Neurobiol. Dis. 39, 283–291 (2010).
Lee, H. M. et al. Characterization and structure-activity relationships of indenoisoquinoline-derived topoisomerase i inhibitors in unsilencing the dormant Ube3a gene associated with Angelman syndrome. Mol. Autism 9, 45 (2018).
Meng, L. et al. Towards a therapy for Angelman syndrome by targeting a long non-coding RNA. Nature 518, 409–412 (2014).
Chamberlain, S. J. et al. Induced pluripotent stem cell models of the genomic imprinting disorders Angelman and Prader-Willi syndromes. Proc. Natl. Acad. Sci. 107, 17668–17673 (2010).
Lancaster, M. A. et al. Cerebral organoids model human brain development and microcephaly. Nature 501, 373–379 (2013).
Jiang, Y. H. et al. Mutation of the Angelman ubiquitin ligase in mice causes increased cytoplasmic p53 and deficits of contextual learning and long-term potentiation. Neuron 21, 799–811 (1998).
Drobná, Z., Jaspers, I., Thomas, D. J. & Stýblo, M. Differential activation of AP-1 in human bladder epithelial cells by inorganic and methylated arsenicals. FASEB J. 17, 67–69 (2003).
Acknowledgements
This work was supported by a Simons Foundation SFARI Explorers Grant (495112), the NCSU Faculty Research and Professional Development Program, an NCSU Research Innovation Seed Fund Grant, the NSF Emerging Frontiers in Research and Innovation program (NSF-1830910), an NIH Avenir Award (DP1-DA044359), Foundation for Angelman Syndrome Therapeutics Targeted Research to Advance a Cure Grant (FT2020-003), and a fellowship from the American Association of University Women (to DS). We thank Drs. Stormy Chamberlain (University of Connecticut Genetics and Genome Sciences, Farmington, CT), Matthew Judson and Benjamin Philpot (University of North Carolina School of Medicine, Chapel Hill, NC) for graciously gifting the H9UBE3A m−/p−, H9UBE3A m−/p+ and AS point mutation (F583S) cell lines and WT and AS mouse brain sections, respectively.
Author information
Authors and Affiliations
Contributions
D.S. and A.J.K. conceived the study. D.S. planned and performed the wet lab experiments with guidance from A.J.K. and experimental support from Z.D. D.S. and A.J.K. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sen, D., Drobna, Z. & Keung, A.J. Evaluation of UBE3A antibodies in mice and human cerebral organoids. Sci Rep 11, 6323 (2021). https://doi.org/10.1038/s41598-021-85923-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-85923-x
- Springer Nature Limited
This article is cited by
-
The porcine islet-derived organoid showed the characteristics as pancreatic duct
Scientific Reports (2024)