Abstract
A concave-shaped maximal expiratory flow-volume (MEFV) curve is a spirometric feature in chronic obstructive pulmonary disease (COPD). The MEFV curve is characterized by an increase in the Obstructive Index, which is defined as a ratio of forced vital capacity to the volume-difference between two points of half of the peak expiratory flow on the MEFV curve. We hypothesized that the Obstructive Index would reflect the severity of emphysema in patients with COPD and asthma-COPD overlap (ACO). Thus, the aim of this retrospective study was to evaluate whether the Obstructive Index on spirometry is associated with the extent of emphysema on computed tomography (CT) in patients with COPD, ACO, and asthma (N = 65, 15, and 53, respectively). The percentage of low-attenuation volume (LAV%) and wall area (WA%) were measured on CT. The Obstructive Index was higher in patients with COPD and ACO than in those with asthma. Spearman correlation showed that a greater Obstructive Index was associated with a higher LAV%, but not WA%. Multivariate analysis showed that Obstructive Index was associated with LAV% (standardized β = 0.43, P < 0.0001) independent of other spirometric indices. The Obstructive Index is a useful spirometric index that reflects the extent of emphysema.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Chronic obstructive pulmonary disease (COPD) is a major concern worldwide, as its prevalence and mortality rate continue to increase, imposing a huge economic burden1,2. The heterogeneity of COPD further complicates its clinical management3. It can be classified into emphysema or non-emphysema phenotypes, which are easily identifiable on computed tomography (CT). Numerous CT studies have shown that the emphysema phenotype is associated with a rapid decline in forced expiratory volume in 1 second (FEV1)4, frequency of exacerbations5, a poor prognosis6, osteoporosis7, occurrence of lung cancer8, loss of skeletal muscle mass9, and reduction in body mass index (BMI)10. These findings emphasize the importance of identifying the emphysema phenotype in the early stage of the disease; however, chest CT scans entail radiation exposure11 and are not always available in primary care facilities. In addition, although it is possible to estimate the extent of emphysema by measuring diffusion capacity, this physiological examination also requires large equipment and it is impractical to routinely perform this examination for all patients suspected of having obstructive lung diseases.
Spirometry is widely used for low-cost management of COPD and non-invasively measures lung function parameters, such as FEV1 and forced vital capacity (FVC). In addition, it allows for the visualization of the maximal expiratory flow-volume (MEFV) curve. MEFV curves with varying concave contours are seen in patients with COPD, and previous reports have shown that this concave shape was partly attributable to loss of elastic recoil12,13.
Therefore, we hypothesized that it would be possible to predict the extent of emphysema among patients with obstructive lung diseases by using a simple spirometry measurement to obtain the Obstructive Index, initially proposed in Japan in 1978, which expresses the concavity of MEFV curves even in patients who have low FVC14. In this study, we explored the usefulness of the Obstructive Index by comparing quantitative CT (QCT) of lung emphysema with visual assessment of MEFV curves in patients with chronic obstructive lung diseases.
Results
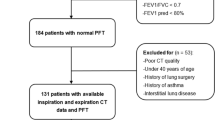
Between January 1, 2015, and March 31, 2017, 10,808 patients had a chest CT scan and 3,875 patients had a spirometry test at the Tsukuba Medical Centre Hospital. Among them, 942 patients had both a chest CT scan and a spirometry test within a 3-month period on the same day or on separate occasions. Four hundred and six patients were excluded because their thin slice digital imaging and communications in medicine (DICOM) data were not available. Furthermore, patients with lung cancer (N = 249), interstitial pneumonia (N = 42), bacterial pneumonia (N = 35) and pleural effusion (N = 14), as well as 62 patients who had surgery were excluded. Finally, one patient with a severe cough during spirometry was excluded. A total of 133 patients who had COPD, asthma–COPD overlap (ACO), or bronchial asthma (BA) were subsequently analyzed for the current study (Fig. 1)
Baseline characteristics
Table 1 shows the characteristics of subjects and the results of univariate analyses of the nonparametric test, including the Obstructive Index in each group. In the Steel-Dwass test, the Obstructive Index in the BA group was lower than those in the COPD and ACO groups (P < 0.0001 and P = 0.0001). The half of the peak expiratory flow (PEF), i.e., the reference point of the Obstructive Index, was larger than the forced expiratory flow at 50% (FEF50), which reflects the concavity of the MEFV curve in COPD or ACO compared to BA, suggesting that half of the PEF is reached faster than the halfway volume of the MEFV curve, FEF50, in COPD or ACO. In terms of QCT measurements, the percentage of low attenuation volume (LAV%) was highest in the COPD group, followed by those in the ACO and BA groups, while there was no difference in the percentage of wall area (WA%) of the right apical segmental bronchus (B1) or right anterior basal segmental bronchus (B8) among the disease groups.
Relationship of MEFV curve concavity on emphysematous change
The classification of MEFV curves is shown in Fig. 2. We found that patients with the airway collapse (AC) pattern had the most severe emphysema, followed by those with the intermediate (Int), curvilinear (C), and normal (N) patterns (Table 2). There was no difference in the WA% between the types of MEFV curves. Each type of MEFV curve was distinguished by the Obstructive Index values. We also examined the presence or absence of the inflection point in the descending limb of the MEFV curves. Nominal logistic regression analysis showed that LAV% was the significant factor associated with the presence of an inflection point on MEFV curves (Table 3).
Schematic representation of MEFV curves. The AC (a) has an abrupt decrease in flow rate and an inflection point that occurs at less than 50% of the PEF and within the first 25% of the FVC12. The Int (b,c) is like the AC but meets only 1 of the AC criteria13. The C (d) exhibits a gradual decrease in the descending limb of the MEFV curve. N (e) is the normal MEFV curve. The visual patterns of the MEFV curves were established by a consensus reading by 3 respiratory physicians (FM, KF, and TS). AC, airway collapse; C, curvilinear; FEF25, forced expiratory flow at 25%, FVC, forced vital capacity; Int, intermediate; MEFV, maximal expiratory flow-volume; N, normal, PEF, peak expiratory flow.
Bivariate correlation analysis
The Obstructive Index was significantly correlated with the LAV% (ρ = 0.56, P < 0.0001), but not with the WA%. FEV1 (ρ = −0.34, P < 0.0001), FEV1% predicted (ρ = −0.36, P < 0.0001), forced expiratory flow between 25 and 75% (FEF25–75), (ρ = −0.47, P < 0.0001), and FEF25–75/FVC (ρ = −0.56, P < 0.0001) were also related to the LAV%. FEV1 and FEV1% of the predicted were weakly correlated with the right B1 WA% (ρ = −0.32, P = 0.0002, and ρ = −0.22, P = 0.012, respectively) (Table 4).
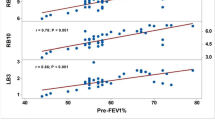
The relationship between Obstructive Index, FEV1% predicted and FEF25–75/FVC (Fig. 3)
A linear correlation was found between the Obstructive Index and FEV1% predicted overall; the Obstructive Index values were particularly high in COPD patients who had lower FEV1% predicted and higher LAV% (Fig. 3a). The relationship between the Obstructive Index and FEF25–75/FVC was hyperbolic (Fig. 3b). The Obstructive Index values were again particularly high in COPD patients with lower FEF25–75/FVC and higher LAV% values.
Correlation of the Obstructive Index with FEV1% predicted and FEF25–75/FVC. (a) Although a linear correlation was found between the Obstructive Index and FEV1% predicted in patients who had high FEV1% predicted, the linearity disappeared and the Obstructive Index was high in COPD patients who had low FEV1% predicted. (b) The relationship between the Obstructive Index and FEF25–75/FVC32 was hyperbolic. COPD, chronic obstructive pulmonary disease; FEV1%, forced expiratory volume in 1 second; FEF25–75, forced expiratory flow between 25 and 75%; FVC, forced vital capacity.
Factors associated with emphysema on multivariate regression analysis
The most striking association was found between LAV% and the Obstructive Index (standardized β = 0.43, P < 0.0001), which was independent of FEV1% predicted, FEF25–75/FVC, and lower FEV1/FVC, as judged by the lower limit of normal for the FEV1/FVC ratio. BMI was significantly inversely associated with the extent of emphysema (standardized β = −0.28, P < 0.0001). These correlations of LAV% with the Obstructive Index and BMI remained significant for a subgroup consisting of patients with both COPD and with ACO (standardized β = 0.32; P = 0.0025, β = −0.32; P < 0.0001, respectively), and for a subgroup consisting of only patients with COPD only (standardized β = 0.33; P = 0.0059, β = −0.27; P = 0.0019, respectively) (Table 5).
Comparison of QCT measurements by degree of FEV1% predicted
We also compared the LAV% or WA% of the right B1 or right B8 according to the degree of FEV1% predicted and cut-off level of Obstructive Index (Fig. 4a–f). There was a significant difference in LAV% at the cut-off level of 7.0 Obstructive Index in patients who had an FEV1% predicted ≤40% (P = 0.007, Fig. 4a), and either at a cut-off level of 3.0 Obstructive Index in patients with a high FEV1% predicted ≥70% (P = 0.009, Fig. 4d). Regarding WA%, a significant difference was observed in the right B8 at a cut-off level of 3.0 Obstructive Index in patients with a high FEV1% predicted ≥70% (P = 0.005, Fig. 4f).
Comparison of QCT measurements at the cut-off value of Obstructive Index by the degree of airway obstruction. Bars represent LS mean ± SEM. Values are adjusted by age, sex, height, smoking index, smoking status, and CT scanner type. There was a difference between LAV% and the Obstructive Index in patients who had either a low FEV1% predicted (≤40%, a–c) or a high FEV1% predicted (≥70%, d–f). The Obstructive Index <7.0, N = 11 (ACO = 5, BA = 2, COPD = 4); the Obstructive Index ≥7.0, N = 12 (ACO = 1, BA = 0, COPD = 11); the Obstructive Index <3.0, N = 36 (ACO = 0, BA = 31, COPD = 5), the Obstructive Index ≥3.0, N = 24 (ACO = 2, BA = 8, COPD = 14) ACO, asthma–COPD overlap; BA, bronchial asthma; COPD, chronic obstructive pulmonary disease; CT, computed tomography; FEV1, forced expiratory volume in 1 second; LAV, low attenuation volume; LS, least squares; QCT, quantitative CT; SEM, standard error of the mean; WA%, the percentage of wall area.
Optimal Obstructive Index cut-off values for predicting the presence of emphysema
To determine the optimal Obstructive Index cut-off values for predicting the presence of emphysema, the area under the curves (AUCs) for receiver operating characteristic (ROC) curves by levels of LAV% were calculated (Fig. 5 and Table 6). We evaluated four stages of LAV%: 10%, 20%, 30%, and 40%. The AUC at a LAV% of 30% was 0.819 (95% CI; 0.736–0.901) and the corresponding cut-off value of the Obstructive Index for predicting emphysema was 4.38 (Table 6).
Comparison of LAV of total lungs for predicting the presence of emphysema by ROC analysis. We evaluated emphysema based on four stages of LAV%:10%, 20%, 30%, and 40%. ROC analysis showed that a LAV of 30% and an Obstructive Index of 4.38 were the optimal cut-off values for determining the presence of emphysema in patients with obstructive lung diseases (Table 6). LAV, low attenuation volume; ROC, receiver operating characteristic.
Discussion
This study investigated whether the Obstructive Index, measured spirometrically, could be used to predict the extent of emphysema, as measured on CT, using cross-sectional data of patients with COPD, ACO, or BA; this has not been reported previously. Previous physiological studies showed that concave MEFV curves on spirometry are common in patients with COPD and that this shape reflects the loss of elastic recoil; however, no report to date has investigated its association with the severity of emphysema. Therefore, the present study substantially extended this body of knowledge by revealing that Obstructive Index is closely correlated with the extent of emphysematous change on CT, independently of FEV1% predicted, FEF25–75/FVC, and a lower FEV1/FVC ratio. This association was preserved even when the same analyses were performed in a subgroup including patients with COPD only or a subgroup including patients with both COPD and with ACO or with BA (Supplementary Tables S1–S5 and Figs S1–3). Furthermore, the Obstructive Index closely linked visual assessment of concavity of the MEFV curve likewise the extent of emphysema.
The concavity in the descending limb of the MEFV curve is a recognized feature of obstructive lung diseases14,15,16,17,18,19. The earliest change in small airways are thought to be reflected in the terminal portion of the MEFV curve20 and quantitatively, the FEF25–75 was reported to reflect concavity and small airway narrowing20,21,22.
The relationship between FEF25–75/FVC and Obstructive Index is shown in Fig. 3b; each parameter could describe different obstructive impairment features. In bivariate analysis, FEF25–75, FEF25–75/FVC, and Obstructive Index were associated with emphysema, while in multivariate analysis, the Obstructive Index was the only significant factor related to the extent of emphysema among spirometric indices. This suggests that the Obstructive Index would be a more appropriate measurement than FEF25–75 or FEF25–75/FVC for evaluating the extent of emphysema.
As COPD progresses, timed segments of spirometry, i.e., FEV1, decreases; FEV1 is used as an index of the severity of airway obstruction. We also examined the relationship between the Obstructive Index and the extent of emphysema by the degree of FEV1% predicted (Fig. 4) and found a persistent association between emphysema and the Obstructive Index, irrespective of the level of FEV1% predicted. Therefore, it is possible to differentiate patients with the phenotypic expression of emphysema from those with obstructive lung diseases, who have similar degrees of airway obstruction, by using the Obstructive Index.
We also explored the relationship between visual assessment of the MEFV curve and the extent of emphysema. The presence of an inflection point in the descending part of MEFV curves was associated with the extent of emphysema. Mechanistically, when airflow obstruction becomes more severe, the expiratory flow decreases more abruptly, to the level of half of the PEF, due to loss of elastic recoil, reduced central airway support, and increased peripheral resistance and pleural pressures during forced expiration12,13. In fact, although FEF50 also reflects the concavity of the MEFV curve16, we found that the half-PEF values were larger than the FEF50 values in patients with COPD or ACO (Table 1), suggesting that focusing on the MEFV curve at the half-PEF level is a rational approach for detecting earlier collapse of the MEFV curve. In addition, as the Obstructive Index is a ratio of FVC relative to “a” (Fig. 6), it could express the concavity more appropriately, even in patients who have decreased FVC, such as those with severe airflow obstruction.
Schematic illustration of Obstructive Index. The Obstructive Index14 is defined as the numerator (FVC) divided by the denominator (“a”), which is the volume between the points at half-PEF on the MEFV curve. The Obstructive Index is reported to be correlated with the area ratio of S to triangle ABC even in patients with both obstructive and restrictive impairments, while the FEV1/FVC ratio is not correlated with the area ratio to triangle ABC in patients with restrictive impairments14. The S is defined as the area under the descending limb of the MEFV curve. The triangle ABC is formed by the top of the PEF (“A”) and the line perpendicular to X-axis “B” and the point of residual volume, “C.” This illustration is a modified reprint from Jpn. J. Chest Dis. 37, Ohsugi, T. et al., 956, A new index of the maximal expiratory flow-volume curve, 1978, with permission from KOKUSEIDO PUBLISHING. Copyright © KOKUSEIDO PUBLISHING. FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; MEFV, maximal expiratory flow-volume; PEF, peak expiratory flow.
This study also has some limitations. Firstly, although the expiratory effort could affect the PEF and FVC, which are components of the Obstructive Index, we did not assess the impact of the FVC maneuver on Obstructive Index computation. Jayamanne et al.12 assessed the effects of the graded effort vital capacity maneuver on patients with an AC pattern. They reported that with regard to the submaximal effort, defined by 50 to 60% of the maximal pleural pressure, there was no difference in the flow-volume curve when compared with the MEFV curve, whereas a minimal effort <40% of the pleural pressure resulted in the disappearance of the inflection point in the descending limb of the MEFV curve. In this study, as the spirometry was performed by well-trained technicians following the statement of American Thoracic Society (ATS) and the European Respiratory Society (ERS) guideline23, we believe that the quality of the spirometry was superior. Secondly, we did not assess total expiratory time in our patients with airway obstruction. We acknowledge that vital capacity in many obstructed patients is a function of total expiratory time and variations in expiratory time may have influenced our computations. Thirdly, we did not include healthy persons or patients who have combined pulmonary fibrosis and emphysema24 (CPFE). Future studies, using larger numbers of patients with obstructive lung diseases, should examine the utility of the Obstructive Index for predicting the presence and severity of emphysema, and should further explore the effects of fibrosis on the concavity of the MEFV curve. Fourthly, although we used multivariate analysis to adjust for the effects of the use of two different CT scanners with different slice thicknesses, it cannot be denied that measurements of LAV might have been influenced by the differences between these scanners. However, we also did the same analysis excluding patients who were scanned using the VCT, and we confirmed that the Obstructive Index was still a significant factor associated with LAV% (Supplementary Table S6). Lastly, there may be cases of bronchiolitis obliterans syndrome25 with severe airway obstruction and air trapping, or endobronchial tumors26 affecting the Obstructive Index.
In conclusion, Obstructive Index was the strongest predictor of the extent of emphysema, independently of other spirometric measurements, in patients with obstructive lung diseases in a stable condition. Although the Obstructive Index is not a reflection of the entire information that can be deduced from the shape of the MEFV curve, it can be easily calculated from the MEFV curve and we believe that this parameter could be used for evaluating the extent of emphysema in clinical practice.
Methods
Ethics
This retrospective study complied with the requirements of the Declaration of Helsinki, as well as with the Ethical Guidelines for Medical and Health Research Involving Human Subjects, which was issued in Japan in December of 2014 and revised in February of 2017. The latter waives the need for obtaining informed consent for retrospective collection of anonymized data and stipulates that subjects be notified of the information required for the study. Approval was granted by the ethics committee for clinical research of Tsukuba Medical Centre Hospital (IRB 2016-054, 2017-008).
Patients
We retrospectively screened patients who underwent both chest CT and spirometry, on separate occasions within a 3-month period, and included 133 patients who were diagnosed with COPD (N = 65), ACO (N = 15), or BA (N = 53) with varying levels of airflow obstruction (Fig. 1). All patients were evaluated and treated by board-certified respiratory physicians. At the time of examination, all patients were in a stable condition, with appropriate treatments, such as long-acting muscarinic antagonist, long-acting β agonist, and/or inhaled corticosteroids according to the treatment guidelines27,28,29. The definition of each obstructive lung disease and smoking status are shown in Supplementary Appendix 1. The patients were all Japanese and were covered by public medical insurance.
Spirometry and definition of each index
Spirometry was performed with an automated electronic spirometer SYSTEM 21 (Minato Medical Science Co., Ltd, Osaka, Japan), with the patient in a sitting position; bronchodilators were withheld before spirometry. These tests were conducted and evaluated by well-trained technicians following the statement of the ATS/ERS guideline23. Patients performed 3 forced expiratory maneuvers and the best curve was selected for all parameters. FEV1% of predicted, FVC% of predicted, and lower limit of normal of FEV1/FVC were determined using Japanese spirometric reference values calculated with the lambda-mu-sigma (LMS) methods published by the Japanese Respiratory Society30. As the variability in an individual’s lung volume and airway size could affect the concavity of the MEFV curve31, we included FEF25–75/FVC32, which is an index of dysanapsis31,33, to evaluate the curvilinearity of the MEFV curve.
The Obstructive Index was calculated by dividing FVC by the volume-difference between two points of half of the peak expiratory flow on the MEFV curve (Fig. 6)14.
Visual assessment of MEFV curve concavity
The shape of the MEFV curves was visually divided into four groups according to Jayamanne’s12 and Healy’s criteria13: AC, Int, C, and N, (Fig. 2). The AC has an abrupt decrease in flow rate and an inflection point that occurs at less than 50% of the PEF and within the first 25% of the FVC. The Int is similar to the AC but meets only 1 of the AC criteria. The C exhibits a gradual decrease in the descending limb of the MEFV curve. The visual patterns of the MEFV curves were assessed by 3 respiratory physicians reaching a consensus.
CT scan protocols
CT examinations were performed during a breath-hold at deep inspiration. Of the patients, 103 were scanned with an Aquilion™ CT device (Canon Medical Systems Corporation, Otawara, Tochigi, Japan), and 30 were examined using a Light Speed VCT™ (GE Healthcare, Waukesha, WI). Images were obtained at a slice thickness of 2.0 mm (Aquilion) or 1.25 mm (VCT), with a scan time of 400 or 500 milliseconds (Aquilion) or 400 milliseconds (VCT), tube voltage of 120 kV, and with autoexposure. The CT images were reconstructed with a smoothing kernel (FC10 and Standard).
CT measurements of the lung
The DICOM formatted data of each patient were used to analyze lung measurements. Using SYNAPSE VINCENT™ ver 4.6 (FUJIFILM Medical Corp., Tokyo, Japan), LAV% at −960 Hounsfield units, divided by the whole-lung volume, and WA% of the right B1 and the right B8 were measured5,6,7,9,10,34.
Statistical analyses
All statistical analyses were performed using JMP™ version 14.1.0 for Windows (SAS Institute Inc., Cary, NC). Differences in characteristics of subjects and QCT measurements among obstructive lung diseases were analyzed using omnibus tests (Kruskal-Wallis Tests or Chi-square tests), followed by nonparametric comparisons for all pairs using the Steel-Dwass method. Spearman’s rank correlation was used to evaluate the relationship between QCT measures and spirometric indices. To investigate the factors associated with LAV%, multivariate regression analysis was performed. To determine the optimal cut-off values of the Obstructive Index for predicting the presence of emphysema, we constructed ROC curves and estimated AUC by levels of LAV%. The Youden index35 was used to identify the Obstructive Index cut-off values that maximized sensitivity and specificity. P values less than 0.05 were considered significant.
Date Availability
The datasets analyzed during the study are available from the corresponding author upon reasonable request.
References
Collaborators, G. B. D. C. R. D. Global, regional, and national deaths, prevalence, disability-adjusted life years, and years lived with disability for chronic obstructive pulmonary disease and asthma, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Respir. Med. 5, 691–706, https://doi.org/10.1016/S2213-2600(17)30293-X (2017).
Guarascio, A. J., Ray, S. M., Finch, C. K. & Self, T. H. The clinical and economic burden of chronic obstructive pulmonary disease in the USA. Clinicoecon. Outcomes Res. 5, 235–245, https://doi.org/10.2147/CEOR.S34321 (2013).
Houben-Wilke, S. et al. COPD stands for complex obstructive pulmonary disease. Eur. Respir. Rev. 27, https://doi.org/10.1183/16000617.0027-2018 (2018).
Nishimura, M. et al. Annual change in pulmonary function and clinical phenotype in chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 185, 44–52, https://doi.org/10.1164/rccm.201106-0992OC (2012).
Tanabe, N. et al. Impact of exacerbations on emphysema progression in chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 183, 1653–1659, https://doi.org/10.1164/rccm.201009-1535OC (2011).
Haruna, A. et al. CT scan findings of emphysema predict mortality in COPD. Chest 138, 635–640, https://doi.org/10.1378/chest.09-2836 (2010).
Ohara, T. et al. Relationship between pulmonary emphysema and osteoporosis assessed by CT in patients with COPD. Chest 134, 1244–1249, https://doi.org/10.1378/chest.07-3054 (2008).
Smith, B. M. et al. Emphysema detected on computed tomography and risk of lung cancer: a systematic review and meta-analysis. Lung Cancer 77, 58–63, https://doi.org/10.1016/j.lungcan.2012.02.019 (2012).
Tanimura, K. et al. Quantitative Assessment of Erector Spinae Muscles in Patients with Chronic Obstructive Pulmonary Disease. Novel Chest Computed Tomography-derived Index for Prognosis. Ann. Am. Thorac. Soc. 13, 334–341, https://doi.org/10.1513/AnnalsATS.201507-446OC (2016).
Ogawa, E. et al. Body mass index in male patients with COPD: correlation with low attenuation areas on CT. Thorax 64, 20–25, https://doi.org/10.1136/thx.2008.097543 (2009).
Mathews, J. D. et al. Cancer risk in 680,000 people exposed to computed tomography scans in childhood or adolescence: data linkage study of 11 million Australians. BMJ 346, f2360, https://doi.org/10.1136/bmj.f2360 (2013).
Jayamanne, D. S., Epstein, H. & Goldring, R. M. Flow-volume curve contour in COPD: correlation with pulmonary mechanics. Chest 77, 749–757, https://doi.org/10.1378/chest.77.6.749 (1980).
Healy, F., Wilson, A. F. & Fairshter, R. D. Physiologic correlates of airway collapse in chronic airflow obstruction. Chest 85, 476–481, https://doi.org/10.1378/chest.85.4.476 (1984).
Ohsugi, T. et al. A new index of the maximal expiratory flow volume curve. Jpn. J. Chest Dis. 37, 956–962 (Japanese) (1978).
Kapp, M. C., Schachter, E. N., Beck, G. J., Maunder, L. R. & Witek, T. J. Jr. The shape of the maximum expiratory flow volume curve. Chest 94, 799–806 (1988).
Landau, L. I., Taussig, L. M., Macklem, P. T. & Beaudry, P. H. Contribution of inhomogeneity of lung units to the maximal expiratory flow-volume curve in children with asthma and cystic fibrosis. Am. Rev. Respir. Dis. 111, 725–731, https://doi.org/10.1164/arrd.1975.111.6.725 (1975).
Mead, J. Analysis of the configuration of maximum expiratory flow-volume curves. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 44, 156–165, https://doi.org/10.1152/jappl.1978.44.2.156 (1978).
Topalovic, M. et al. Computer quantification of airway collapse on forced expiration to predict the presence of emphysema. Respir. Res. 14, 131, https://doi.org/10.1186/1465-9921-14-131 (2013).
Wang, W., Xie, M., Dou, S., Cui, L. & Xiao, W. Computer quantification of “angle of collapse” on maximum expiratory flow volume curve for diagnosing asthma-COPD overlap syndrome. Int. J. Chron. Obstruct. Pulmon. Dis. 11, 3015–3022, https://doi.org/10.2147/COPD.S118415 (2016).
Pellegrino, R. et al. Interpretative strategies for lung function tests. Eur. Respir. J. 26, 948–968, https://doi.org/10.1183/09031936.05.00035205 (2005).
Riley, C. M. et al. Clinical Implications of Having Reduced Mid Forced Expiratory Flow Rates (FEF25-75), Independently of FEV1, in Adult Patients with Asthma. PLoS One 10, e0145476, https://doi.org/10.1371/journal.pone.0145476 (2015).
West, W. W., Nagai, A., Hodgkin, J. E. & Thurlbeck, W. M. The National Institutes of Health Intermittent Positive Pressure Breathing trial–pathology studies. III. The diagnosis of emphysema. Am. Rev. Respir. Dis. 135, 123–129, https://doi.org/10.1164/arrd.1987.135.1.123 (1987).
Miller, M. R. et al. Standardisation of spirometry. Eur. Respir. J. 26, 319–338, https://doi.org/10.1183/09031936.05.00034805 (2005).
Cottin, V. et al. Combined pulmonary fibrosis and emphysema: a distinct underrecognised entity. Eur. Respir. J. 26, 586–593, https://doi.org/10.1183/09031936.05.00021005 (2005).
Robson, K. S. & West, A. J. Improving survival outcomes in lung transplant recipients through early detection of bronchiolitis obliterans: Daily home spirometry versus standard pulmonary function testing. Can. J. Respir. Ther. 50, 17–22 (2014).
Lutz, J. C. & Nugent, K. M. Flow volume loops in diagnosis. Chest 97, 504–505, https://doi.org/10.1378/chest.97.2.504 (1990).
Global Initiative for Chronic Obstructive Lung Disease: Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease, http://goldcopd.org/gold-2017-global-strategy-diagnosis-management-prevention-copd/ (Updated 2015).
Global Initiative for Asthma: Global strategy for asthma management and prevention Updated, https://ginasthma.org/wp-content/uploads/2016/01/GINA_Report_2015_Aug11-1.pdf (2015).
Diagnosis of Diseases of Chronic Airflow Limitation: Asthma COPD and Asthma - COPD Overlap Syndrome (ACOS) based on the Global Strategy for Asthma Management and the Global Strategy for the Diagnosis, Management and Prevention of Chronic obstructive Pulmonary Diseases, https://goldcopd.org/asthma-copd-asthma-copd-overlap-syndrome/ (2015).
Kubota, M. et al. Reference values for spirometry, including vital capacity, in Japanese adults calculated with the LMS method and compared with previous values. Respir. Investig. 52, 242–250, https://doi.org/10.1016/j.resinv.2014.03.003 (2014).
Green, M., Mead, J. & Turner, J. M. Variability of maximum expiratory flow-volume curves. J. Appl. Physiol. 37, 67–74, https://doi.org/10.1152/jappl.1974.37.1.67 (1974).
Tager, I. B., Weiss, S. T., Munoz, A., Welty, C. & Speizer, F. E. Determinants of response to eucapneic hyperventilation with cold air in a population-based study. Am. Rev. Respir. Dis. 134, 502–508, https://doi.org/10.1164/arrd.1986.134.3.502 (1986).
Mead, J. Dysanapsis in normal lungs assessed by the relationship between maximal flow, static recoil, and vital capacity. Am. Rev. Respir. Dis. 121, 339–342, https://doi.org/10.1164/arrd.1980.121.2.339 (1980).
Terada, K. et al. Impact of gastro-oesophageal reflux disease symptoms on COPD exacerbation. Thorax 63, 951–955, https://doi.org/10.1136/thx.2007.092858 (2008).
Youden, W. J. Index for rating diagnostic tests. Cancer 3, 32–35 (1950).
Acknowledgements
This study was partly supported by a research grant from Fujifilm Medical Corp (Tokyo, Japan). The sponsor had no role in the design of the study, data collection and analysis, or the preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
F.M. is the guarantor of the content of the manuscript, including the data and analysis. F.M. and H.I. had full access to all the data in the study, take responsibility for the integrity of the data and accuracy of the data analysis. F.M., H.I., N.T., S.S., S.M. and N.H. finalized the manuscript. H.I., J.K., Y.Y., H.M., T.S. and N.H. contributed to the study concept, design, and interpretation of data. A.W. and M.S. contributed to radiology data collection and interpretation. K.F., T.S. and H.I.S. contributed to the lung function data collection and interpretation. N.T., S.S. and S.M. contributed to the development of volumetric software and interpretation of data. All authors contributed to and approved the final manuscript. The authors thank Koichi Kurishima, Koji Kanemoto, Junichi Fujita, and Kazuki Obara at the Department of Respiratory Medicine, Tsukuba Medical Centre; and Yuka Hasegawa at Fujifilm Medical Corporation for technical advice for setting volumetric software on the PC, and Editage (www.editage.jp) for English language editing.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mochizuki, F., Iijima, H., Watanabe, A. et al. The Concavity of the Maximal Expiratory Flow–Volume Curve Reflects the Extent of Emphysema in Obstructive Lung Diseases. Sci Rep 9, 13159 (2019). https://doi.org/10.1038/s41598-019-49591-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-49591-2
- Springer Nature Limited
This article is cited by
-
Principal component analysis of flow-volume curves in COPDGene to link spirometry with phenotypes of COPD
Respiratory Research (2023)
-
Pulmonary function testing in COPD: looking beyond the curtain of FEV1
npj Primary Care Respiratory Medicine (2021)