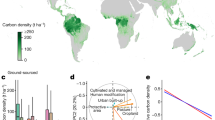
Abstract
The uptake of carbon dioxide (CO2) by terrestrial ecosystems is critical for moderating climate change1. To provide a ground-based long-term assessment of the contribution of forests to terrestrial CO2 uptake, we synthesized in situ forest data from boreal, temperate and tropical biomes spanning three decades. We found that the carbon sink in global forests was steady, at 3.6 ± 0.4 Pg C yr−1 in the 1990s and 2000s, and 3.5 ± 0.4 Pg C yr−1 in the 2010s. Despite this global stability, our analysis revealed some major biome-level changes. Carbon sinks have increased in temperate (+30 ± 5%) and tropical regrowth (+29 ± 8%) forests owing to increases in forest area, but they decreased in boreal (−36 ± 6%) and tropical intact (−31 ± 7%) forests, as a result of intensified disturbances and losses in intact forest area, respectively. Mass-balance studies indicate that the global land carbon sink has increased2, implying an increase in the non-forest-land carbon sink. The global forest sink is equivalent to almost half of fossil-fuel emissions (7.8 ± 0.4 Pg C yr−1 in 1990–2019). However, two-thirds of the benefit from the sink has been negated by tropical deforestation (2.2 ± 0.5 Pg C yr−1 in 1990–2019). Although the global forest sink has endured undiminished for three decades, despite regional variations, it could be weakened by ageing forests, continuing deforestation and further intensification of disturbance regimes1. To protect the carbon sink, land management policies are needed to limit deforestation, promote forest restoration and improve timber-harvesting practices1,3.
Similar content being viewed by others
Main
Atmospheric CO2 concentration surpassed 420 ppm in 2023 (ref. 4) and climate change is approaching potential tipping points that portend considerable future impacts1 unless urgent action is taken5,6. Humanity has converged on the goal of achieving net zero greenhouse-gas emissions by 2050 (ref. 7). One of the most challenging elements is the need for large-scale ‘negative emissions’ of up to 6 Pg C yr−1 to compensate for the inability to eliminate all emissions from fossil fuels8. The land sector has the capacity to sequester and store more carbon because historically it has lost 180 Pg of stored carbon through changes in land use, and this former carbon reservoir can be restored to some extent5,9,10. Because forests are the dominant component of the land carbon sink11, we need to know how much atmospheric carbon the world’s forests have been sequestering, where it is stored and whether recent trends are consistent with the desired strengthening of Earth’s land sink.
Advances in remote sensing, modelling and computation can map and model Earth’s land sinks at high temporal and spatial resolution, but they have difficulty in generating long-term baselines and may diverge substantially in some regions and timeframes12. By contrast, the extensive ground-based and historical information from forest inventories and ecological studies enables the analysis of forest dynamics (growth, harvest and mortality) by region or country, because they are all ultimately based on tree-by-tree measurement of size, species and biomass. Whether regional13 or global11, these data provide a unique perspective on Earth’s forests and how they are changing, and are highly complementary to top-down or model-driven approaches. The length, quantity and consistency of such records now enable a three-decade perspective on Earth’s global and regional forest carbon balance and fluxes to span the entire period of land-use change, shifting forest dynamics and accelerating climate change since the first assessment report of the Intergovernmental Panel on Climate Change (IPCC) in 1990 (ref. 14).
We analysed three decades of ground-based measurements by the global forest community (Supplementary Table 1), combined with forest area estimates based on remote sensing in national forest inventories and other types of land survey, to evaluate the recent magnitude, trend, impact factors and locations of the global forest carbon sink. We constructed a global record of forest inventory measurements from 1990 to 2019, supplemented with high-quality data from long-term ecosystem-monitoring sites. Our estimates of the forest land carbon sink are largely independent of other approaches, including atmospheric CO2 observations and inverse models15, dynamic global vegetation models (DGVMs)16 and mass-balance assessments2. The uncertainty of our estimated global forest carbon sink is around 0.4 Pg C yr−1, whereas other estimated terrestrial sinks2 have uncertainties in the range 0.5–1.8 Pg C yr−1. We call for investment in specific research and monitoring priorities to reduce uncertainties in forest carbon assessments.
Global forest areas, carbon stocks and sinks
The world’s forest area declined by 5% from 1990 to 2020, from 4,022 Mha to 3,812 Mha (−210 Mha) (Extended Data Table 1). This net decline in forest land is driven by losses in the tropics (−273 Mha, −13%). By contrast, temperate-forest area increased (+52 Mha, +7%) and boreal-forest area was stable (+12 Mha, +1%). In the tropics, 467 Mha (26%) of intact forest was lost but the area of regrowth forests expanded (+194 Mha, +56%).
The carbon stock in the world’s forests in 2020 was 870 ± 61 Pg C (Extended Data Table 2). In boreal, temperate and tropical regrowth forests, stocks increased by 74 Pg C over three decades. Meanwhile, deforestation reduced intact tropical-forest carbon stocks by 149 Pg, but remaining intact tropical forests sequestered 32 Pg C to make up some of the losses (Extended Data Fig. 1). Most of the 2020 global forest carbon stock is in live biomass (43%) and soils (45%), with smaller proportions in dead wood (8%) and litter (4%). The fraction of total carbon in living biomass increases towards the Equator, but the proportion in soils shows the opposite pattern: boreal forests stored 20% of their carbon in living biomass and 64% in soils; temperate forests stored 38% in living biomass and 54% in soils; and tropical forests stored 57% in living biomass and 32% in soils. Total carbon stocks were highest in the tropics, lowest in temperate forest and intermediate in boreal forests.
The carbon density (Mg C ha−1) increased from 1990 to 2020 in each biome (Extended Data Fig. 2c). This indicates that global forests overall continued to gain carbon nearly everywhere, which is consistent with rising CO2 concentrations increasing photosynthetic rates globally17,18. Other factors, such as warmer temperatures and increased nitrogen deposition, may also enhance forest carbon densities regionally (Supplementary Table 2). Nevertheless, the average global forest carbon density barely changed. This apparent paradox is due to the loss of high-density intact tropical forests and their partial replacement by much lower-carbon-density regrowth forests, resulting in the average global forest carbon density remaining almost constant despite density increases in each category (Supplementary Table 3).
The carbon sink in Earth’s forests was estimated to be 3.59 ± 0.34, 3.57 ± 0.36 and 3.53 ± 0.41 Pg C yr−1 for the 1990s, 2000s and 2010s (Table 1), which is statistically stable over the decades (Supplementary Fig. 1). These stable global totals mask large biome-scale changes: an increased sink in temperate (+30%) and tropical regrowth forests (+29%) but a decline in boreal (−36%) and tropical intact (−31%) forest sinks. Furthermore, the carbon sink in global established forests (excluding tropical regrowth forests) declined by 19% from 2.32 ± 0.21 to 1.89 ± 0.24 Pg C yr−1 over 30 years (Table 1). After accounting for carbon emissions from tropical deforestation, the net carbon sink (see the definition in Box 1) in Earth’s forests was still positive, being 0.93 ± 0.63, 1.66 ± 0.56 and 1.39 ± 0.69 Pg C yr−1 in the 1990s, 2000s and 2010s, respectively (Table 1).
Forest carbon sinks by region, biome and pool
Boreal forests
The boreal carbon sink declined from 508 ± 63 Tg C year−1 to 324 ± 41 Tg C yr−1 from the 1990s to the 2010s (Extended Data Table 3) and was strongly affected by Asian Russian forests, which account for 57% of the boreal forest area (Extended Data Table 1). The carbon sink in Asian Russian forests declined by 42% over the three decades, with the greatest reduction occurring in the late 2010s, primarily resulting from an increased severity of wildfires, insect outbreaks and increased logging, both legal and illegal19 (Fig. 1). Notably, living biomass contributed a large carbon sink in the 1990s (145 Tg C yr−1) but switched to being a source in the 2010s (−20 Tg C yr−1); meanwhile the dead-wood sink increased20 by 44%. Alaska Interior managed forests were a small carbon sink in the 1990s but reduced by 76% in the 2010s, probably because of soil warming and increasing wildfires21. Canadian managed forests were approximately carbon neutral in the 1990s and small sources in the 2000s and 2010s (Extended Data Table 3). The much greater source from living biomass in the 2000s (−55 Tg C yr−1) was caused by increased outbreaks of insects and wildfires22. In the 2010s, living biomass, dead wood and litter pools all became carbon sources and the soil sink was reduced by 35%, reflecting increased impacts of disturbances, warming and droughts22.
Green bars represent established forests (boreal, temperate and tropical intact forests) and brown bars represent tropical regrowth forests. All values are in Pg C yr−1. Positive values (with downward bars) indicate carbon sinks and negative values (upward bars) show carbon sources. Detailed uncertainties of sinks and sources are shown in Extended Data Table 3. We grouped a few regions and countries together so there are fewer categories to prevent the graphic getting too cluttered. These include: Europe (Europe temperate and other Europe); Japan and Korea; South Asia (India and other South Asia); and Mexico and Central America (Extended Data Table 3).
Unlike Canadian managed forests, which have become drier, European Russia and European boreal forests have become wetter over the past half-century23. The boreal forests of European Russia had a relatively stable multi-decadal carbon sink, with a slight increase in the 2000s when agricultural lands that were abandoned in the 1990s returned to forest24, followed by a slight decrease in the 2010s, which was probably the result of increased harvesting and disturbances. However, our estimates show that the soil carbon sink decreased by 31% in the 2010s compared with the 2000s, possibly because of soil warming25. European boreal forests showed an increasing carbon sink over time, resulting from improved management and growth enhancements caused by CO2 fertilization and longer growing seasons26. The latest forest inventory updates from Finland27 and Sweden28 indicate a recent sink downturn, responding to a combination of drought, changes in stand age structure, reduced roundwood imports and intensive harvests (Supplementary Information).
Temperate forests
The carbon sink in temperate forests was 526 ± 37 Tg C yr−1 in the 1990s, increasing to 685 ± 50 Tg C yr−1 in the 2010s (Extended Data Table 3). The main driver was the increase in China’s forest area under national-scale afforestation and reforestation programmes during the late 1980s and early 1990s, when those new forests reached their highly productive stages in the 2000s and 2010s, increasing the sink by 86 Tg C yr−1 each decade29.
The carbon sink in US forests decreased by 10% in the 2000s compared with the 1990s and remained at that level in the 2010s (Fig. 1). In the 2000s, US forests experienced increased natural disturbances and summer droughts30. Although the US forest carbon sink did not recover fully in the 2010s, the rate of decline was reduced. The carbon sink in European temperate forests declined by 12% from the 2000s to the 2010s (Extended Data Table 3), probably because large forest areas planted in the 1950s approached carbon saturation as they matured31. More recently, Central European forests suffered increasing damage from bark beetles, triggered by several years of droughts32, which could lead to forests becoming carbon sources at the national level, although droughts alone did not seem to reduce growth33.
In Japan, the carbon sink in living biomass decreased considerably in the 2010s, perhaps because of the ageing forests planted in the 1960s34 (Extended Data Table 3). Australian forests were carbon sources in the 1990s and 2000s and became merely neutral in the 2010s (Extended Data Table 3). This carbon source was due to extensive deforestation for agriculture, which declined in the recent decade because of legislative restrictions on clearing. Carbon was also lost from the harvesting of native, high-carbon-density forests, which were replaced by younger lower-carbon-density regrowth forests. Intensified droughts and wildfires in the 2000s and 2010s also contributed to increased net annual emissions.
Tropical intact forests
The carbon sink in tropical intact forests declined from 1,284 ± 202 Tg C yr−1 in the 1990s to 881 ± 235 Tg C yr−1 in the 2010s (Extended Data Table 3), caused mainly by deforestation that reduced the remaining intact forest area by 26%. The greatest losses proportionally occurred in Southeast Asia, with 53% loss of intact forests (101 Mha) in the past 30 years, largely because of the expansion of oil-palm plantations35. The greatest losses by area were in South America (187 Mha, 22%) and Africa (175 Mha, 29%) (Extended Data Table 1). The carbon contained in deforested lands (149 Pg C) had different fates: about 45% was rapidly emitted to the atmosphere, 17% was lost to processing harvested timber and the use of short-lived wood products such as paper, 2% was stored in long-lived wood products, such as construction materials, and the remaining 36% continued to be stored on the land in the new land-use types, such as ranch-land soils (Extended Data Fig. 1).
Tropical intact-forest carbon sinks declined in Southeast Asia, Africa and South America by 25%, 17% and 42%, respectively (Extended Data Table 3). South America experienced the largest reduction because it lost most intact forest area and because Amazon droughts contributed to increased tree mortality and slowing of tree growth rates36,37,38. Consequently, the 2010s sink in South American intact forests was less than two-thirds of that in the 1990s (Fig. 1). The smallest decline in the forest carbon sink was in Africa, reflecting similar proportional losses of forest area but less impact of drought and warming on forest processes37. The decreased carbon sink in Southeast Asian forests was mainly driven by forest land losses.
Tropical regrowth forests
The carbon sink in tropical regrowth forests increased from 1,273 ± 260 Tg C yr−1 in the 1990s to 1,640 ± 333 Tg C yr−1 in the 2010s. Despite occupying just 20% of the area of intact forest in the 1990s, these forests had a similar carbon sink (Extended Data Table 3) because their carbon-sequestration rates were about five times higher, reflecting the early successional biomass accumulation phase of tropical forests. The regrowth carbon sink increased greatly in the 2000s and 2010s with expanded areas (Extended Data Table 1). Overall, the increasing tropical regrowth-forest carbon sink balanced the declining sink in intact forests across 1990 to 2020, resulting in a nearly constant tropical-forest carbon sink of around 2.5 ± 0.4 Pg C yr−1 for three decades (Table 1). Although the carbon sinks in tropical intact and regrowth forests were large, high emissions resulting from deforestation and degradation counteracted nearly all of these remarkable sinks, making tropical forest lands almost carbon neutral (Extended Data Fig. 3), with a small net sink or source of between −0.1 and 0.6 Pg C yr−1, fluctuating with deforestation intensities in different decades (Table 1).
Necromass and harvested-wood products
We include estimates of carbon stock and sink in different components of forest necromass (non-living organic matter in standing and lying dead wood, litter and soils) to enable reporting of complete forest-ecosystem carbon budgets, even though estimation of these pools has greater uncertainty. Necromass accounted for an average of 58% of total forest carbon stocks (514 ± 52 Pg C), with proportions smallest in tropical forests (45%, 226 ± 42 Pg C), intermediate in temperate forests (64%, 80 ± 9 Pg C) and greatest in boreal forests (80%, 207 ± 10 Pg C) (Extended Data Table 2). The fraction of the carbon sink in necromass was 30% (781 ± 154 Tg C yr−1) of that in living biomass globally, but varied greatly among biomes, averaging 184% (266 ± 48 Tg C yr−1) in boreal forests but just 26% and 20% in temperate (109 ± 16 Tg C yr−1) and tropical (406 ± 105 Tg C year−1) forests, respectively (Extended Data Table 3).
Harvested-wood products (HWPs) are defined as a carbon sink, related to the amount of timber harvested and the portion that remains in use or in solid waste-disposal sites. Globally, only about 10% of the carbon in harvested timber is counted as HWP39 because about half of the wood is used for fuel and much of the rest is lost during processing into wood products, followed by losses when the products are discarded and decomposed3. The average half-life of pulp and paper products is only two years, whereas for sawn-wood products it is 35 years39. The annual HWP increased by 10% over three decades to 0.21 Pg C yr−1 in the 2010s, implying that more wood was harvested from forests. On average, HWP contributes only 6% of the global carbon sink (7%, 13% and 4% in boreal, temperate and tropical forests, respectively) (Extended Data Table 3), although this estimate does not fully account for the effects of illegal logging on wood-harvesting fluxes.
Status of the global forest carbon sink
Our estimates show a large, long-term persistent sink of 3.56 ± 0.37 Pg C yr−1 in global forests since at least 1990, with a statistically insignificant change, based on Monte Carlo simulations and Cohen’s d (Supplementary Fig. 1). Although stable overall, the contribution to this carbon sink of different forest biomes has fluctuated greatly over time. In the tropics there has been a shift from equal contributions of intact and regrowth tropical forests in the 1990s to 65% of the sink being in regrowth forests in the 2010s, as the intact sink declined and the regrowth sink increased (Table 1). Boreal and temperate forests contributed similar carbon sinks in the 1990s, but by the 2010s the boreal sink had decreased to less than half of the temperate sink (Table 1).
Carbon stock densities (Mg C ha−1) in all forest biomes in all climate zones steadily increased (Extended Data Fig. 2c), indicating that forest ecosystems across the planet continuously sequestered carbon, implying that a universal growth factor, or several factors, was enhancing forest sinks on continental scales. A suite of multidisciplinary evidence indicates that the global carbon-sink persistence and carbon-density increases were in part the result of CO2 fertilization contributing to substantially increased photosynthesis17,18, as well as to longer growing seasons in temperate and boreal regions26. These factors may have outweighed the negative effects on forest carbon from global heating, changing rainfall patterns and changes in the frequency and severity of natural disturbances in the remaining forests1,5.
Regional vulnerability of carbon sinks
The carbon sink in Earth’s forests is vulnerable to deforestation, degradation and disturbances triggered or intensified by climate change. In intact tropical forests, the foremost threats remain ongoing deforestation and degradation, which are the primary causes of the declining carbon sink (Extended Data Fig. 1). More-intense and frequent droughts have also killed millions of trees, contributing to a weaker carbon sink in the Amazon37,40. Given that the combined sink in intact and regrowth forest is stable, the sign of the net sink for tropical forests as a whole is determined largely by the rate of deforestation emissions. Only reducing deforestation and degradation will keep stored carbon out of the atmosphere, and by protecting tropical forests we also protect their biodiversity and sink capacity in the future.
Boreal forests have experienced major impacts from climate change, including greater increases in temperature and variability than in the other regions41. Climate change has disrupted the carbon dynamics in vegetation and soils, and has exacerbated disturbances caused by wildfires, insect outbreaks and droughts. The high carbon stock and sink in boreal forest necromass are threatened by increased decomposition rates and wildfires resulting from dry conditions42. These impacts made Canadian forests a carbon source22, and Asian Russian forests lost 42% of their sink strength over three decades, particularly in the late 2010s25. Future threats for boreal-forest carbon dynamics also include the northward shifting of bioclimatic zones that directly causes thawing of permafrost, triggering megafires such as one in 2020–22, increased risk of large-scale pest outbreaks, and increased rates of legal and illegal logging, all of which result in the release of methane and CO2 (Supplementary Information).
Temperate forests include Earth’s most intensively managed forest ecosystems. The increased carbon sink resulted mainly from past tree planting in China29. Temperate forests that recovered on abandoned agricultural lands or heavily harvested forests in the early-to-mid twentieth century are now approaching the age at which growth rates begin to decline, although growth trajectories and successional dynamics differ in the temperate forest biome31,34,43. Climate change has caused increases in the frequency and intensity of natural disturbances, triggering intensified outbreaks of bark beetles after droughts in some European forests32. Furthermore, increasing temperate-zone tree harvests over the three decades (+17%) caused loss of stocks.
Although asynchronous regional dynamics ensured that the aggregate carbon sink in Earth’s forests was almost constant, our analysis shows how biome- and continental-scale forest carbon sinks were susceptible to multiple environmental changes and timber harvesting. All these factors impact growth, mortality and stocks, and therefore future changes will affect the persistence and strength of the global forest carbon sink. With several strong positive and negative drivers (Supplementary Table 4), each likely to develop differently among biomes and regions, the global forest carbon sink has an uncertain future. We therefore recommend carefully monitoring its future evolution.
Land sinks and the global carbon budget
Our estimates for forests can be placed in the context of terrestrial carbon sinks and sources estimated from the global carbon budget (GCB)2 (Fig. 2). Both the mass balance of the GCB and the mean of 17 DGVM results estimated that the land gross carbon sink grew44, meaning that the contribution of Earth’s total forest carbon sink (around 3.6 Pg C yr−1) to the land gross sink has declined relatively from 75% in the 1990s to 65% in the 2010s (Extended Data Table 4). This also indicates that non-forest lands have been progressively removing more carbon from the atmosphere (Fig. 2). Our results showing relatively stable global forest gross sinks contrast with most carbon model estimates, which show that carbon uptake is increasing in most forest biomes44. This means that the modelled future terrestrial carbon uptake by forests may be overestimated.
Carbon fluxes are presented as sink (+) or source (–) Pg C yr−1. Fossil-fuel emissions (EFOS), ocean carbon sequestration (SOCEAN), land-use-change gross emissions (EGLUC), global land gross sink (SGLAND) and global land net sink (SNLAND) were estimated by or derived from the GCB2. The global forest gross sink (SGFOR), tropical deforestation gross emissions (EDFOR), global forest net sink (SNFOR) and differences (EGLUC − EDFOR and SGLAND − SGFOR) were estimated in this study. Our values for SGFOR and EDFOR, when compared with GCB total land estimates, provide new ground-inventory constraints from which to derive global non-forest land gross emissions and non-forest gross sinks for each decade since 1990 (Extended Data Table 4). Credits: industrial emission, iStock.com/Spicytruffel; car emissions, iStock.com/Maria Dekhnik; broadleaf trees, Clker-Free-Vector-Images; conifer trees, Clker-Free-Vector-Images via Pixabay; tree stump, iStock.com/KajaNi; tree cutting, iStock.com/colematt; fire, iStock.com/bonezboyz; grasses, iStock.com/airdone, iStock.com/belander, iStock/illustrador o artista vectorial Dibujos animados Arte gráfico Abstracto Fondos Diseño Vacaciones; wheat, iStock/Stepanyda, iStock/Diane Labombarbe; corn, iStock.com/Bezvershenko; bushes, iStock.com/ekkawit998, iStock.com/sakdam, iStock.com/Johannesk; sky, iStock.com/zoom-zoom; beach, Clker-Free-Vector-Images.
By contrast, over the three decades, the global forest net sink (1.3 Pg C yr−1) amounted to 91% of the land net sink (1.4 Pg C yr−1) (Fig. 2). The forest net sink we estimated is therefore similar to the land net sink independently estimated using DGVMs of 0.9, 1.2 and 1.5 Pg C yr−1 for the 1990s, 2000s and 2010s, respectively, and is broadly comparable with inverse model estimates and other methods44. Finally, although the magnitude of the global forest net sink is only 17% of that of fossil-fuel emissions, the forest gross carbon sink was of course much greater. The total three-decadal sink of 106.9 Pg C is equivalent to around 46% of fossil-fuel emissions. Even for the 2010s alone, the global forest carbon gain would have amounted to 37% of contemporary fossil–fuel emissions if deforestation had ceased (Extended Data Table 4).
Uncertainties and research priorities
Uncertainty of carbon stock-change estimates (that is, carbon sink or source) varied by biome, with the largest uncertainties in tropical (±27%) and boreal (±13%) biomes, and the smallest in temperate biomes (±7%) (Extended Data Table 3). Countries with well-established national forest inventories based on statistical sampling had the lowest reported uncertainty. Thus, more ground measurements and monitoring are needed, particularly in tropical biomes and countries that currently lack statistical sampling; in soils and dead wood globally; and in areas affected by natural disturbances and logging. For future global analyses based on bottom-up approaches, we recommend several research and monitoring priorities:
-
1.
Increased sampling of below-ground biomass, dead wood, litter and soil carbon. These have much greater uncertainties than does above-ground biomass, although they have smaller impacts on the total uncertainty, except for boreal forests. For instance, if we had increased soil sink uncertainties to 100% in all biomes (Supplementary Table 5), globally it would increase uncertainty in the total carbon sink by only about 1% because sinks in living biomass are the dominant components. Along with more field measurements, scaling up to the region and biome should use detailed maps of forest type to represent the distinct and variable forest conditions that make up the total forest area.
-
2.
Increased research and sampling of under-represented tropical forests, such as Southeast Asian wetland forests and African dry forests. This could be combined with better maps of forest type to mitigate potential biases from uneven sampling. This would require broad-scale support and investment in long-term on-the-ground monitoring of tropical forest biomass, growth and mortality, distributed across all tropical forest types. The enhanced land monitoring would complement, and greatly leverage investment in, space-based forest monitoring, and reduce uncertainties in data about changes and climate sensitivities of Earth’s most productive and diverse biomes.
-
3.
Better information about uncertainty in forest area estimates, which mostly rely on remote sensing or remote-sensing-based forest inventory statistics and are often reported without uncertainty information45. Uncertainty in forest areas is caused by inconsistent remote-sensing data processing methods and definitions of forests, and makes up a considerable proportion of the uncertainty in carbon sink estimates.
Sustaining the forest carbon sink
Our results indicate that the single most important action for sustaining and increasing the forest carbon sink is to stop emissions from deforestation and degradation, along with protecting the large carbon stocks that have accumulated over centuries, such as in tropical forest biomass and in boreal forest soils. Recovery of functions by degraded forests and lands offers further opportunities for enhancing carbon sinks, with many additional benefits, such as protecting biodiversity46. The pathways for stopping global deforestation and degradation will rely on international cooperation, such as the United Nations REDD+ programme. Financial, legislative and other incentives are also needed, particularly in tropical countries. Deforestation-free supply chains and well-managed selective logging can all lower deforestation rates.
Our study demonstrates the considerable impacts of large-scale reforestation and afforestation on enhancing carbon sinks, through either natural recovery or mandated actions. Some countries, such as the United States, have lands suitable for afforestation or improved management but historically have low adoption rates (Extended Data Table 1). Tropical forest regrowth represents another important opportunity to accumulate more carbon on abandoned land. Declining carbon-sink strength caused by forest ageing has become more common in some temperate zones31,34, although most older forests maintain high carbon stocks in the absence of human disturbance, and some remain productive for very long periods43. In the future, management intensity and its effects on forest age dynamics may determine the carbon sink trends of temperate forests.
Strategic planning will help to prioritize forest management approaches to minimize carbon emissions and maximize carbon uptake and its benefits. For instance, adaptive and climate smart forestry practices5, such as reduced-impact logging47, fuel management to increase resistance to wildfires48, optimizing tree species resilience after disturbances and restoring old-growth characteristics, can be highly effective49. Protecting carbon stocks is also essential. For example, our data show that tropical regrowth forests have high rates of carbon sequestration, but recovering carbon density can take many decades to reach the level of intact forest. So replacing tropical intact forests with tropical regrowth forests, which have large carbon sinks but much lower carbon stocks and diminished biodiversity, is highly imprudent.
Long-lived HWPs store carbon but represent only around 10% of the carbon in harvested timber, so switching from short-lived products such as fuel wood or pulp wood to long-lived sawn-wood products could sequester more carbon, provided that the total harvested volume does not increase and reduce ecosystem carbon stocks. Improving wood processing technologies to reduce waste47, developing new long-lived materials and recycling more50 may benefit a sustainable and circular economy, as suggested by the IPCC5.
Our estimates indicate that 107 Pg of carbon has been sequestered from the atmosphere by global forests since 1990, which is equal to 46% of fossil-fuel emissions. Although 63% of this uptake was negated by tropical deforestation emissions, the remaining forests helped to slow climate change. The global forest sequestration rate of around 3.56 Pg C yr−1 (around 13 Gt of CO2-equivalent per year) for 1990–2019 provides a baseline for the IPCC’s ambitious assessment4 that agriculture, forestry and other land-use sectors have a combined potential to mitigate an additional 8–14 Gt CO2-equivalent per year in 2020–2050. Mitigating and adapting to the climate crisis are defining challenges for humanity, and these goals cannot be achieved without both protecting the carbon stocks and sinks in Earth’s forests and reducing emissions from fossil fuels.
Methods
Forest biomes and lands
Estimates of carbon stocks and stock changes are reported for forests partitioned into three biomes: boreal, temperate and tropical (including subtropical); and by carbon component (living biomass, dead wood, litter, soil and harvested wood products). Forests in boreal and temperate biomes include both ‘forest land remaining forest land’ and reforested or afforested lands (collectively ‘new forests’), and tropical forests are separated into remaining forests (intact forests) and regrowth forests. The area of global forests used as a basis for estimating carbon stocks and sinks was around 4.0 billion hectares, representing 95% of global forested land51 (Extended Data Table 1 and Supplementary Table 6). The 5% not covered comprises some remote forest areas, including unmanaged forests in northern Canada and Alaska interior, and some areas of West and Central Asia with sparse forests where we lacked credible ground data. We did not include non-forested peatlands or wetlands, or coastal mangrove forests, which commonly contain high carbon in soil or sediments52.
Definitions of forest carbon pools and stocks
We generally followed the definitions in Table 3.1.2 of the IPCC’s good practice guidance53. Definitions of five main carbon pools are detailed in the Supplementary Information: living biomass, dead wood, litter, soil organic matter and harvested wood products.
Carbon stock is defined as carbon contained in different carbon pools or in all carbon pools.
Carbon stock change (or carbon flux) is defined as change in carbon stocks between time points, and can represent carbon gain (sink) or carbon loss (source).
Overview of data and calculation methods
Sources of data used in this study
This study covers three decades (the 1990s, 2000s and 2010s) using data from 1990 to 2020 (Supplementary Table 1). Since our previous study (for 1990–2007)11, country-scale greenhouse gas inventories in temperate and boreal countries and regions have expanded to include more countries and have been updated. Networks of sample plots in tropical regions of the Amazon, Africa and Southeast Asia have expanded. Our data are not always consistent with what individual countries have reported to the FAO or IPCC (Supplementary Table 1). Unless more credible data is available, we use the FAO data reported in the Global Forest Resources Assessment 2020 (ref. 51) to establish the total forest area by country or region. These data are a credible source for trend information about forest area over decades and across geographies.
Accounting approaches for different forest regions/biomes
There are slightly different accounting approaches used in this paper because the available data have been developed and presented in different ways in inventories, country reports and the scientific literature. Estimates were harmonized between accounting systems by carefully defining land areas and matching these with the sources of data, and by adjusting reported estimates where necessary to account for known inconsistencies. Our calculation methods are summarized in Supplementary Table 1 and described in more detail in the Supplementary Information.
Either the ‘stock change’ or the ‘default’ approaches were used for boreal and temperate biomes, following the guidance from the IPCC53,54. The stock-change approach was also applied for several tropical countries or regions (only intact forests) including India, other South Asian countries, Mexico, Central America and the Caribbean. If there is no land-use change, the stock-change approach is nearly identical to estimating the land–atmosphere CO2 flux, with the exception of ‘lateral transfers’ of carbon, which primarily include river erosion, transport, outgassing and deposition, and harvested wood products. One exception is Canada, which reports carbon stock changes based on the ‘gain–loss’ approach. The default approach commences with a single forest inventory and then adds carbon gains from forest growth and losses from harvest, fires and decomposition without confounding estimates through carbon transfers between land-use categories53.
We accounted for HWPs but not for other lateral transport, which may be responsible for a substantial global carbon sink into coastal oceans from forests that is not reflected in the stock-change method. If there is land-use change, the stock-change accounting overestimates the carbon uptake by forests in proportion to the area of afforestation during the period of change, because existing carbon stocks on new forest land (primarily soil carbon) appear instantaneously in the forest carbon inventory, transferred from the previous land-use category. Conversely, the stock-change approach may underestimate carbon uptake by forests in proportion to the area of deforestation because existing soil carbon may be moved to a non-forest land category and appear as a loss of carbon from forest. We corrected for this apparent loss in our accounting.
For the tropics (Southeast Asia, Africa and South America), carbon sinks and sources (or net fluxes) were estimated using a ‘flow’ approach because most tropical areas lack the repeated national-scale forest inventories that are the basis for the stock-change approach. This approach is similar to the IPCC ‘tier 2’ methods53 that multiply region-specific estimates of carbon density or change in carbon density with the associated areas represented by the region-specific estimates. For intact tropical forests (not affected by land use or change), fluxes were estimated from measured carbon stock changes on permanent sample plots, which is nearly equivalent to forest–atmosphere carbon exchange except for river transport and deposition of carbon. The approach allows accounting for carbon gains in forests, including some impacts of forest degradation affecting rates of carbon gains, but not carbon losses resulting from deforestation because carbon stored in deforested areas is accounted for separately in our global budget (Extended Data Fig. 1).
The effects of land-use change and harvesting on carbon flux were estimated separately using a book-keeping approach55 that keeps track of ecosystem carbon emissions and harvested wood products from deforestation and logging, and ecosystem carbon uptake on regrowing forests.
Estimates of carbon stock changes pertain to ‘forest land remaining forest land’ plus ‘afforested land’ for boreal and temperate forests. For tropical intact and regrowth forests of Southeast Asia, Africa and South America, and also for tropical regrowth forests of Mexico and Central America and the Caribbean, changes in carbon density times the associated areas were used. Estimates of carbon stocks for specific years (Extended Data Table 2) pertain to the total area of forest land in the given year and therefore include carbon stocks lost because of deforestation, which are not included in Extended Data Table 3. Thus, it is not possible to consistently match the estimates between these two tables, which is particularly true for tropical intact forests because they are the only biome that has lost substantial forest area (Extended Data Table 1).
Forest area and area change
Area estimates (Extended Data Table 1) are from country-level forest inventories or reports based on forest inventories. Forest inventories typically use remote sensing combined with ground observations to estimate forest area and area changes following FAO forest definitions, excluding ‘other wooded land’. Where forest inventory data direct from countries are lacking, particularly in the tropics, FAO statistics were used to estimate total forest area for 1990, 2000, 2010 and 2020 (ref. 51). In some regions, particularly the tropics and Russia, the quality of data reported to the FAO is poor and the protocols may be subject to change over time. Because tropical intact forests defined in this study are not the same as primary forests defined in FAO statistics (see the definition in Box 1), we used area estimates of tropical intact forests from published studies for Southeastern Asia, Africa and South America36. The difference between the total tropical forest area from the FAO51 and the area of tropical intact forest for these regions was assumed to be the area of tropical regrowth forest. We attempted to establish good consistency between the change in reported areas from the years 1990, 2000, 2010 and 2020, and estimated areas of afforestation and deforestation from inventories, country reports and analyses of emissions from land-use changes.
Carbon stocks and carbon-stock changes
Where available, carbon stock and density estimates are from country-level forest inventories or reports based on national forest inventories. Most countries in temperate and boreal zones have established national forest inventories with repeated measurement of permanent sample plots. Generally, sample plots are randomly located across all areas of the country and measurements taken on those plots that are located on forest land. Thus, the inventory is an unbiased sample of the population of trees in the country, and the precision of estimates may be calculated. The re-measurement interval is typically between five and ten years. At each sample plot, individual trees were selected for measurement of diameter, height, species and condition. Re-measurement determines the basic tree population dynamics: growth, mortality and harvest. Extra measurements may be taken to include understorey vegetation, woody debris, litter and soils. For some temperate or boreal countries where direct access to inventory data is not available, we used a ‘biomass expansion factor’ approach, which converts estimates of growing stock volume to estimates of biomass or carbon stocks. The measured data may be used to estimate the carbon stocks and carbon-stock changes using a variety of country-specific methods (Supplementary Information), but generally following guidelines provided by the IPCC53,54. For example, the basic tree measurements of diameter and height are used to estimate tree biomass and carbon using allometric models and conventional statistical methods.
For tropical intact forests in Southeast Asia, Africa and South America, we used data from repeated long-term measurements of networks of ecological research plots, upscaled to the regions to estimate biomass and other carbon pools for the region’s forest areas36,37,38 For tropical regrowth forests, which lack sufficient ground-based data, we followed the book-keeping approach56, which is based on a literature review of regrowth rates (Supplementary Table 7) and carbon stocks and knowledge of forest areas and conditions, averaged over different ecozones (tropical wet, moist and dry forests) for each region57. These methods are described in more detail in the Supplementary Information.
Data from regions, countries or continents were aggregated to global biomes: boreal, temperate and tropical forests. For countries and regions that do not allow access to original data, data from the FRA regional reports51 were used to fill the data gaps (Supplementary Table 1b). Available data allowed carbon stock and area estimates to be compiled for 1990, 2000, 2010 and 2020, and annual changes in carbon stocks (sometimes referred to as ‘sink’ if there was a carbon gain and as ‘source’ if there was a carbon loss) to be estimated for three time periods: 1990–99, 2000–09 and 2010–19.
More data are available for live biomass and biomass changes than for other carbon pools. Some forest inventories and many ecological studies also collect and report data for dead wood and litter, although less consistently than for biomass, so empirical models were often the source of estimates for these carbon pools. Inventories of forest soil carbon across the landscape are scarcer than inventories of biomass or other ecosystem carbon pools, and sampling methods include varying soil depths for sampling among regions and countries. There are existing soil surveys in different countries, but very rarely with periodic revisits, and rarely associated with documented information about above-ground forest vegetation. To evaluate forest soil carbon change over time is particularly difficult because the formation and respiration of soil carbon is affected by various biological, environmental and geographical factors, as well as land-use history, and is not always correlated with more easily observable vegetation traits. In almost every region, empirical modelling methods were used to combine data from soil surveys and field studies for developing estimates of soil carbon.
HWPs
HWPs are defined as a component of the carbon sink in this study and included in the carbon stock change category. Where available, estimates of carbon in HWPs are from country-level inventory reports. Otherwise, harvested roundwood data were derived from FAO annual statistics (Supplementary Information). Generally, estimates of carbon in HWPs account for the temporary exclusion from the atmosphere, which includes both the wood products in use and discarded wood products remaining in landfills or dumps. For countries that lacked reported estimates of HWP, we derived a simple conversion factor from the countries that did report: the ratio of carbon flux in HWPs (Tg C yr−1) to the quantity of harvested roundwood (million cubic metres) according to FAO reports51, which is 0.095.
Approaches to estimate uncertainty
We report the standard error for estimates of carbon stocks and changes in carbon stocks using the 95% confidence level. Values presented as y ± x should be interpreted to mean that we are 95% certain that the actual value is between y − x and y + x. The 95% boundary was chosen to communicate the high degree of certainty that the actual value was in the reported range and the low likelihood (5% or less) that it was outside that range. This characterization is not, however, a statistical property of the estimate, and should not be confused with statistically defined 95% confidence intervals.
We report uncertainty using two approaches, depending on the availability of uncertainty estimates from data sources: quantitative estimates and expert opinion. Quantitative estimates are based on remote sensing and sampling combined with empirical models, using either error propagation methods or Monte Carlo simulation approaches to combine all carbon pools, and including the uncertainty of area estimates. The expert-opinion approach is based on that adopted by the IPCC for reporting in global assessments (described in Supplementary Information). Quantitative estimates are more commonly available for data derived from national forest inventories or extensive sampling-plot networks, whereas expert opinion is used where quantitative estimates are unavailable and has been used in previous large-scale analyses58 (Supplementary Information). In applying these approaches, we ensured that estimates based on expert opinion were not overly optimistic compared with estimates from similar countries or regions that reported quantitative estimates.
Evaluating major uncertainties in different biomes and carbon components
We reported uncertainties for the aggregated sums of individual carbon pools (such as litter and dead wood), but not for each individual pool, because this detailed information is not regularly included in the publicly available estimates, even though the uncertainty of each individual carbon pool is included in the aggregated estimates of carbon stocks and stock changes that are estimated using error propagation approaches.
Uncertainty estimates for stock change in boreal forests are around ±13% and possibly more considering the uncertainty in soil carbon estimates. The largest stock change by far is in Russian boreal forests, and the uncertainty is particularly important because of the large sink estimated in this region. The main reasons for the uncertainty of boreal-forest estimates involve incomplete sampling of large areas of Alaska, Canada and Russia combined with poor data on soil carbon and wildfires, particularly in the Asian part of Russia.
Uncertainty estimates for stock change in temperate forests are about ±7%, representing the lowest value among all biomes. This is mainly because most temperate countries have strong and repeated forest inventory sampling programmes that cover most of the forest area. The greatest uncertainty in temperate forests is for changes in soil carbon, which is not monitored as easily or as often as the other carbon pools.
Uncertainty estimates for stock change in tropical intact forests are about ±27% in the most recent period, largely because the estimates are based on a relatively small number of intensively monitored sites in which data are individually quite accurate but are not conducive to scaling because representation of the larger population of forests by the collection of sites is unknown. This uncertainty is particularly notable because the largest component of the global forest carbon sink is in tropical forests. The effects of disturbances, particularly drought, are difficult to quantify, and there are relatively scant data about the carbon pools apart from live biomass.
Uncertainty estimates for stock change in tropical regrowth forests are about ±20%, a little lower than estimates for intact forests. The area of tropical regrowth forests is not well known, and there is relatively little sampling done. The error estimates, based on expert opinion, probably underestimate the true uncertainty of this increasingly important component of the global budget.
The uncertainties of stock-change estimates for soil carbon, dead wood, litter and HWP are high in boreal regions and the tropics. However, the size of the sink in these pools is relatively small compared with living biomass, except boreal forests, so the contribution to overall uncertainty is also small. As shown by uncertainty experiments (Supplementary Table 5), although ignoring soil carbon sinks would reduce the estimated global forest carbon sink by around 400 Tg C yr−1, it would have minimal impact on the global and biome-level temporal trends. Increasing 100% uncertainties in soil sinks, the total carbon sinks in boreal, temperate and tropical forests increased their uncertainties by 15%, 2% and less than 1%, respectively, but with error propagation it increased uncertainty in total global carbon sink by around 1%.
Additional sources of uncertainty are described in the Supplementary Information.
Ground data versus modelling and remote-sensing approaches
Remote-sensing and modelling estimates of the forest sector are subject to considerable uncertainty and inconsistency between different studies59,60,61 compared with ground data, which are based on more standard definitions and protocols51,53,54. Different representations and complexity of regional ecological processes and limited calibration with data for parameterization are often the cause of inconsistencies in model results62,63. Indeed, remote sensing and modelling approaches are dependent on summarized ‘standard’ per-hectare biomass estimates derived from field studies. Ground data have improved greatly, and multiple carbon pools are measured and monitored more often. Our estimates represent a credible complement to the remote-sensing and model-based estimates used for the land part of the GCB2,44, with terrestrial data in the GCB being based on an average of models44,62. It is important to use multiple methods to contrast and compare calculations to improve overall estimates of land carbon sinks.
Data availability
The data used for synthesis and analysis are derived from more-detailed measurements and are available in spreadsheets with embedded formulae for access at https://doi.org/10.2737/RDS-2023-0051. Our results can be replicated using these spreadsheets. The estimates used for tables and figures of the main text and Extended Data are also in the data repository. The repository also includes original measurements for a few countries and the source data information for others, along with DOIs and websites for accessing original data. Because policies for data sharing vary from country to country, some sources include original measurement data from sampling with fully open access, while some include only aggregated data. Most original data are publicly available through direct access, but in a few cases for which the data are not publicly available, the data can be requested from the regional authors. Full descriptions of regional datasets and estimation approaches, including links, are provided in the Supplementary Information. Source data are provided with the paper.
Change history
06 August 2024
A Correction to this paper has been published: https://doi.org/10.1038/s41586-024-07897-w
References
IPCC Working Group II. Climate Change 2022: Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change (eds Pörtner, H. O. et al.) (Cambridge Univ. Press, 2023),
Friedlingstein, P. et al. Global carbon budget 2021. Earth Syst. Sci. Data 14, 1917–2005 (2022).
Peng, L., Searchinger, T. D., Zionts, J. & Waite, R. The carbon costs of global wood harvests Nature 620, 110–115 (2023).
NOAA. Monthly Average Mauna Loa CO2. Trends in CO2, CH4, N2O, SF6 (Global Monitoring Laboratory, accessed 15 May 2024); https://gml.noaa.gov/ccgg/trends/.
Nabuurs, G.-J. et al. in Climate Change 2022: Mitigation of Climate Change. Contribution of Working Group III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change (eds Shukla, P. R. et al.) Ch. 7 (Cambridge Univ. Press, 2022).
McKay, D. I. A. et al. Exceeding 1.5°C global warming could trigger multiple climate tipping points Science 377, eabn7950 (2022).
United Nations/Framework Convention on Climate Change. Adoption of the Paris Agreement. FCCC/CP/2015/L.9/Rev.1 (United Nations, 2015).
Fuss, S. et al. Negative emissions—part 2: costs, potentials and side effects. Environ. Res. Lett. 13, 063002 (2018).
IPCC Working Group I. Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change (eds Stocker, T. F. et al.) (Cambridge Univ. Press, 2013).
Walker, W. S. et al. The global potential for increased storage of carbon on land. Proc. Natl Acad. Sci. USA 119, e2111312119 (2022).
Pan, Y. et al. A large and persistent carbon sink in the world’s forests. Science 333, 988–993 (2011).
Araza, A. et al. Past decade above-ground biomass change comparisons from four multi-temporal global maps. Int. J. Appl. Earth Obs. Geoinf. 118, 103274 (2023).
Suarez, D. R. et al. Estimating aboveground net biomass change for tropical and subtropical forests: refinement of IPCC default rates using forest plot data. Glob. Chang. Biol. 25, 3609–3624 (2019).
IPCC. Climate Change: The IPCC 1990 and 1992 Assessments. https://www.ipcc.ch/site/assets/uploads/2018/05/ipcc_90_92_assessments_far_full_report.pdf (1992).
Schimel, D., Stephens, B. B. & Fisher, J. B. Effect of increasing CO2 on the terrestrial carbon cycle. Proc. Natl. Acad. Sci. USA 112, 436–441 (2014).
Sitch, S. et al. Recent trends and drivers of regional sources and sinks of carbon dioxide. Biogeosciences 12, 653–679 (2015).
Pan, Y. et al. Contrasting responses of woody and grassland ecosystems to increased CO2 as water supply varies. Nat. Ecol. Evol. 6, 315–323 (2022).
Walker, A. P. et al. Integrating the evidence for a terrestrial carbon sink caused by increasing atmospheric CO2. New Phytol. 229, 2413–2445 (2021).
Shvetsov, E. G., Kukavskaya, E. A., Shestakova, T. A., Laflamme, J. & Rogers, B. M. Increasing fire and logging disturbances in Siberian boreal forests: a case study of the Angara region. Environ. Res. Lett. 16, 115007 (2021).
Fan, L. et al. Siberian carbon sink reduced by forest disturbances. Nat. Geosci. 16, 56–62 (2023).
Wang, J. A., Baccini, A., Farina, M., Randerson, J. T. & Friedl, M. A. Disturbance suppresses the aboveground carbon sink in North American boreal forests. Nat. Clim. Change 11, 435–441 (2021).
Kurz, W. A. et al. Carbon in Canada’s boreal forest — a synthesis. Environ. Rev. 21, 260–292 (2013).
Dai, A. Increasing drought under global warming in observations and models. Nat. Clim. Change 3, 52–58 (2013).
Gutman, G. & Radeloff, V. (eds) Land-cover and Land-use Changes in Eastern Europe after the Collapse of the Soviet Union in 1991 (Springer, 2017).
Walker, X. J. et al. Increasing wildfires threaten historic carbon sink of boreal forest soils. Nature 572, 520–523 (2019).
Kauppi, P. E. et al. Managing existing forests can mitigate climate change. For. Ecol. Manage. 513, 120186 (2022).
Henttonen, H. M., Nöjd, P. & Mäkinen, H. Environment-induced growth changes in forests of Finland revisited – a follow-up using an extended data set from the 1960s to the 2020s. For. Ecol. Manage. 551, 121515 (2024).
Korosuo, A. et al. The role of forests in the EU climate policy: are we on the right track? Carbon Balance Manage. 18, 15 (2023).
Yang, C. et al. Updated estimation of forest biomass carbon pools in China, 1977–2018. Biogeosciences 19, 2989–2999 (2022).
Domke, G. C. et al. in Second State of the Carbon Cycle Report (SOCCR2): A Sustained Assessment Report (eds Cavallaro, N. et al.) Ch. 9 (U.S. Global Change Research Program, 2018). https://doi.org/10.7930/SOCCR2.2018.Ch9.
Nabuurs, G.-J. et al. First signs of carbon sink saturation in European forest biomass. Nat. Clim. Change 3, 792–796 (2013).
Hlásny, T. et al. Living With Bark Beetles: Impacts, Outlook and Management Options. From Science to Policy 8 (European Forest Institute, 2019).
Salomón, R. L. et al. The 2018 European heatwave led to stem dehydration but not to consistent growth reductions in forests. Nat. Commun. 13, 28 (2022).
Forestry Agency, Japan. State of Japan’s Forests and Forest Management: 3rd Country Report of Japan to the Montreal Process https://www.maff.go.jp/e/policies/forestry/attach/pdf/index-8.pdf (2019).
Vijay, V., Pimm, S. L., Jenkins, C. N. & Smith, S. J. The impacts of oil palm on recent deforestation and biodiversity loss. PLoS ONE 11, e0159668 (2016).
Hubau, W. et al. Asynchronous carbon sink saturation in African and Amazonian tropical forests. Nature 579, 80–87 (2020).
Phillips, O. L. et al. Drought sensitivity of the Amazon rainforest. Science 323, 1344–1347 (2009).
Lewis, S. L., Brando, P. M., Phillips, O. L., van der Heijden, G. M. F. & Nepstad, D. The 2010 Amazon drought. Science 331, 554 (2011).
IPCC. 2019 Refinement to the 2006 IPCC Guidelines for National Greenhouse Gas Inventories https://www.ipcc.ch/report/2019-refinement-to-the-2006-ipcc-guidelines-for-national-greenhouse-gas-inventories/ (2019).
Brienen, R. J. W. et al. Long-term decline of the Amazon carbon sink. Nature 519, 344–348 (2015).
IPCC. Climate Change 2014–Impacts, Adaptation and Vulnerability. Part B: Regional Aspects: Working Group II contribution to the IPCC Fifth Assessment Report (eds Barros, V. R. et al.) (Cambridge Univ. Press, 2015); https://doi.org/10.1017/CBO9781107415386
Phillips, C. A. et al. Escalating carbon emissions from North American boreal forest wildfires and the climate mitigation potential of fire management. Sci. Adv. 8, eabl7161 (2022).
Law, B. E. et al. Land use strategies to mitigate climate change in carbon dense temperate forests. Proc. Natl. Acad. Sci. USA 115, 3663–3668 (2018).
Friedlingstein, P. et al. Global carbon budget 2022. Earth Syst. Sci. Data 14, 4811–4900 (2022).
Hansen, M. C. et al. High-resolution global maps of 21st-century forest cover change. Science 342, 850–853 (2013).
Arneth, A. et al. Restoring degraded lands. Ann. Rev. Environ. Resour. 46, 569–599 (2021).
Sasaki, N. et al. Sustainable management of tropical forests can reduce carbon emissions and stabilize timber production. Front. Environ. Sci. https://doi.org/10.3389/fenvs.2016.00050 (2016).
Hurteau, M. D., North, M. P., Koch, G. W. & Hungate, B. A. Managing for disturbance stabilizes forest carbon. Proc. Natl. Acad. Sci. USA 116, 10193–10195 (2019).
Thom, D. et al. The climate sensitivity of carbon, timber, and species richness covaries with forest age in boreal–temperate North America. Global Change Biol. 25, 2446–2458 (2019).
Birdsey, R. et al. Climate, economic, and environmental impacts of producing wood for bioenergy. Environ. Res. Lett. 13, 050201 (2018).
Food and Agriculture Organization Global Forest Resource Assessment 2020: Main Report https://doi.org/10.4060/ca9825en (2020).
Murdiyarso, D., Kauffman, J. B. & Verchot, L. Climate change mitigation strategies should include tropical wetlands. Carbon Manage. 4, 491–499 (2014).
IPCC. Good Practice Guidance for Land Use, Land-Use Change and Forestry (eds Penman, J. et al.) http://www.ipcc-nggip.iges.or.jp/public/gpglulucf/gpglulucf.html (Institute for Global Environmental Strategies, 2003).
IPCC. IPCC Guidelines for National Greenhouse Gas Inventories http://www.ipcc-nggip.iges.or.jp/public/2006gl/index.html (Institute for Global Environmental Strategies, 2006).
Houghton, R. A. & Castanho, A. Annual emissions of carbon from land use, land-use change, and forestry from 1850 to 2020. Earth Syst. Sci. Data 15, 2025–2054 (2023).
Houghton, R. A. Terrestrial fluxes of carbon in GCP carbon budgets. Global Change Biol. https://doi.org/10.1111/gcb.1505 (2020).
Cook-Patton, S. C. et al. Mapping carbon accumulation potential from global natural forest regrowth. Nature 585, 545–550 (2020).
King, A. W. et al. (eds) The First State of the Carbon Cycle Report (SOCCR): The North American Carbon Budget and Implications for the Global Carbon Cycle (National Oceanic and Atmospheric Administration/National Climatic Data Center, 2007).
Lang, N. et al. Global canopy height regression and uncertainty estimation from GEDI LIDAR waveforms with deep ensembles. Remote Sens. Environ. 268, 112760 (2022).
Bastos, A. et al. Sources of uncertainty in regional and global terrestrial CO2 exchange estimates. Global Biogeochem. Cycles 34, e2019GB006393 (2020).
Ciais, P. et al. Definitions and methods to estimate regional land carbon fluxes for the second phase of the Regional Carbon Cycle Assessment and Processes Project (RECCAP-II). Geosci. Model Dev. 15, 1289–1316 (2022).
O’Sullivan, M. et al. Process-oriented analysis of dominant sources of uncertainty in the land carbon sink. Nat. Commun. 13, 4781 (2022).
Fatichi, S., Pappas, C., Zscheischler, J. & Leuzinger, S. Modelling carbon sources and sinks in terrestrial vegetation. New Phytol. 221, 652–668 (2019).
Acknowledgements
We thank G. Domke for information on interior Alaska; K. McCullough for making the map in Fig. 1; S. P. Wang and J. P. Brown for help with statistical analyses for Supplementary Fig. 1; and H. F. Hoen and A. Nordin for leading data compilation for Norway and Sweden, respectively. For supporting this work, we thank the US Forest Service and the Embassy Science Program (to Y.P.); Woodwell Climate Research Center (R.A.B., R.A.H. and A.C.); the European Research Council (advanced grant TFORCES GA291585), the Royal Society (International collaboration award ICA\R1\180100), NERC (NE/S011811/1 ARBOLES and NE/X014347/1 AMSINK) and the World Resources Institute (Sustaining Tropical Forest Monitoring) (O.L.P); the National Natural Science Foundation of China (grant 31988102) (J.F.); JSPS KAKENHI (grant JP21H03580) (A.I. and S.H.); JSPS KAKENHI (JP21H05318) (A.I.); European Union projects H2020-Verify and H2020 Superb (G.-J.N. and B.L.); the European Space Agency (CCI Biomass 4000123662/18/I-NB) and the European Union’s Horizon Europe research and innovation programme (EYE-CLIMA 10108139) (A.S. and D.S.); and the US Agency for International Development (grant MTO 069018). This study is a product of the global forest carbon working group.
Author information
Authors and Affiliations
Contributions
Y.P. and R.A.B. were lead authors, synthesized the data and drafted the manuscript; O.L.P, R.A.H. and J.F. provided critical concepts and substantial editing; Y.P., R.A.B., O.L.P., R.A.H., J.F., P.E.K., H.K., W.A.K., A.I., S.L.L., G.-J.N. and A.S. contributed primary datasets and analyses, and led regional estimates and writing of the Methods; S.H., B.L., A.C., D.S. and D.M. contributed regional estimates and methodology documentation. All authors contributed in writing, discussions or comments.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Ivan Janssens, Maxime Réjou-Méchain and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Why have tropical intact forests lost carbon stocks yet also remained a carbon sink?.
From 1990 to 2019, tropical intact forests that remain intact continued to sequester carbon by 32.0 Pg C (Table 1). Deforestation reduced the area of tropical intact forests by 467 Mha (containing C stocks of 149.4 Pg C). About 45% of C stocks in the deforested lands was emitted to the atmosphere shortly after the deforestation (mainly due to the slash-and-burning practice for agricultural land conversion), 36% was transferred to other land-uses such as agricultural lands (mostly as soil carbon), 17% was lost in processing harvested timber such as via wood shavings or stored in short-lived products such as fuelwood and paper, and 2% was retained in harvested wood products (HWP) such as long-lived construction materials. Because the remaining intact forests had provided a 32.0 Pg C sink, the net C stock loss from the intact forests was 117.5 Pg C. Credits: forest canopy, iStock.com/Rhet Ayers Butler - Mongabay; deforested area, Kids Encyclopedia Facts, CC BY 3.0; livestock, iStock.com/edsongrandisoli; industrial woodchipper, iStock.com/EBREHИЙ XaИTOHOB; stack of logs, iStock.com/Pandavector; cabin, Clker-Free-Vector-Images.
Extended Data Fig. 2 Forest areas, carbon stocks, and carbon stock changes in the global forest and forest biomes.
(a) forest areas; (b) carbon stocks; (c) carbon stock densities; (d) carbon stocks by pool; (e) carbon stock change (sinks); and (f) carbon stock change per hectare. The error bars represent standard deviations. For (a) we assumed 10% uncertainty in forest areas due to lack of documented uncertainty in remotely-sensed data; for (d) the uncertainty values of individual carbon pools were not included with most data sources, so we assumed that deadwood, litter and soil carbon pools have twice the uncertainty of the biomass pool, and estimated the uncertainty values of the individual carbon pools from the total carbon stock uncertainty. Uncertainties in the remaining charts are calculated based on data in Extended Data Table 2 and Extended Data Table 3.
Extended Data Fig. 3 Carbon sinks and sources in global forests.
The C sink and source (Pg C yr−1) are expressed as the mean annual rate across the full three-decadal period 1990 to 2019. Positive values represent carbon sinks, while negative (red) values carbon sources. Because carbon fluxes estimated in temperate and boreal forests were based on the “stock change” method, which included carbon gains and losses (from temporarily harvested forests), the C sink estimated was a net sink. Because carbon fluxes estimated in tropical forests were based on the “flux” method, C sinks estimated were gross sinks. Tropical deforestation emissions were estimated by a book-keeping model. The tropical forest net sink, therefore, was the balance of C sinks and emissions (see Methods for concepts and details). Credit: forest fire, iStock/Blueastro.
Supplementary information
Supplementary Information
Supplementary text and data including Supplementary Tables 1–7 and Supplementary Fig. 1 (see contents for details).
Rights and permissions
About this article
Cite this article
Pan, Y., Birdsey, R.A., Phillips, O.L. et al. The enduring world forest carbon sink. Nature 631, 563–569 (2024). https://doi.org/10.1038/s41586-024-07602-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-024-07602-x
- Springer Nature Limited