Abstract
MYC is an oncogenic transcription factor that binds globally to active promoters and promotes transcriptional elongation by RNA polymerase II (RNAPII)1,2. Deregulated expression of the paralogous protein MYCN drives the development of neuronal and neuroendocrine tumours and is often associated with a particularly poor prognosis3. Here we show that, similar to MYC, activation of MYCN in human neuroblastoma cells induces escape of RNAPII from promoters. If the release of RNAPII from transcriptional pause sites (pause release) fails, MYCN recruits BRCA1 to promoter-proximal regions. Recruitment of BRCA1 prevents MYCN-dependent accumulation of stalled RNAPII and enhances transcriptional activation by MYCN. Mechanistically, BRCA1 stabilizes mRNA decapping complexes and enables MYCN to suppress R-loop formation in promoter-proximal regions. Recruitment of BRCA1 requires the ubiquitin-specific protease USP11, which binds specifically to MYCN when MYCN is dephosphorylated at Thr58. USP11, BRCA1 and MYCN stabilize each other on chromatin, preventing proteasomal turnover of MYCN. Because BRCA1 is highly expressed in neuronal progenitor cells during early development4 and MYC is less efficient than MYCN in recruiting BRCA1, our findings indicate that a cell-lineage-specific stress response enables MYCN-driven tumours to cope with deregulated RNAPII function.
Similar content being viewed by others
Main
To study the transcriptional function of MYCN, we expressed a MYCN–ER chimeric protein in SH-EP neuroblastoma cells, which do not express endogenous MYCN (Fig. 1a). RNA sequencing of cells treated with 4-hydroxytamoxifen (4-OHT) revealed that activation of MYCN generated an expression profile that discriminated MYCN-amplified neuroblastoma from non-MYCN-amplified tumours, independent of tumour stage (Extended Data Fig. 1a, b). The strong increase in expression of canonical MYC-activated genes in MYCN-amplified relative to non-amplified tumours of the same stage accounted for the difference (Extended Data Fig. 1c). MYCN-amplified tumours also showed suppression of genes that are specifically expressed in T cells, which probably reflects a low number of T cells in these tumours5 (Extended Data Fig. 1c).
a, Immunoblot documenting MYC and MYCN–ER levels in SH-EP cells. Cells were treated with 200 nM 4-OHT or ethanol for 3 h. CDK2 is used as loading control. n = 3; in all legends, n indicates the number of independent biological replicates. b, Correlation between regulation by MYCN measured by nascent and steady-state RNA levels. In total, 18,213 genes were binned. Data are mean ± 95% confidence interval. n = 3. c, Metagene plots of total RNAPII after 4-OHT treatment (3 h) for the 914 most strongly MYCN-activated (top) and 615 MYCN-repressed genes (bottom). Data are mean ± s.e.m. (indicated by the line and shading throughout). n = 4. EtOH, ethanol; RPM, reads per million mapped reads. TES, transcription end site; TSS, transcription start site. d, Metagene plots of RNAPII(pSer2) for MYCN-activated and MYCN-repressed genes. Data are mean ± s.e.m. n = 4. e, Correlation between transcriptional changes and RNAPII occupancy at promoters and transcription end sites after MYCN–ER activation. In total, 11,093 expressed genes were binned based on log2(fold change in expression). Plots show median ± bootstrapped 95% confidence intervals of 1,000 resamplings. f, Kernel density plot of regulation by MYCN of gene with promoters that have different G/C skews (strong: 4,003; weak: 3,381; no: 1,377). n = 3. P values were calculated with a two-tailed Wilcoxon one-sample signed-rank test with μ = 0.
Activation of MYCN led to both the activation and repression of genes (Fig. 1b). Chromatin immunoprecipitation coupled with sequencing (ChIP–seq) showed that promoters of both activated and repressed genes were bound by MYCN (Extended Data Fig. 1d). MYCN-activated genes bound to more MYCN and showed higher basal expression levels than MYCN-repressed genes6,7,8 (Extended Data Fig. 1d, e). Sequencing of 4-thiouridine-labelled nascent RNA showed that changes in RNA synthesis paralleled mRNA levels at steady state6 (Fig. 1b and Extended Data Fig. 1f). Activation of MYCN caused a shift in overall RNAPII occupancy at actively expressed genes from the promoter into the gene body that has previously been observed in response to MYC3 (Fig. 1c and Extended Data Fig. 2a, b). RNAPII occupancy data were identical whether data were read-normalized or normalized using a spike-in control (Extended Data Fig. 2c). ChIP–seq of RNAPII phosphorylated at Ser2 (RNAPII(pSer2)) confirmed that activation of MYCN caused an increase in transcriptional elongation when averaged over all genes (Extended Data Fig. 2a). Stratifying the alteration in RNAPII occupancy to MYCN activation by the change in mRNA expression showed that the decrease in RNAPII occupancy at the promoter occurred uniformly on activated, repressed and non-regulated genes, which are expressed at a similar level (Fig. 1c and Extended Data Fig. 2d). By contrast, the MYCN-dependent change in RNAPII and RNAPII(pSer2) occupancy in the body of the gene or at the transcription end site was highly regulated: RNAPII(pSer2) increased strongly on MYCN-activated genes, did not change on non-regulated genes and decreased on MYCN-repressed genes (Fig. 1d, e and Extended Data Fig. 2d). Over all genes, the amount of elongating RNAPII paralleled the MYCN-dependent change in mRNA levels (Fig. 1e). A strong G/C-skew9 in promoter-proximal regions correlated closely with positive regulation by MYCN (Fig. 1f).
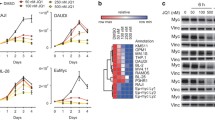
We reasoned that activation of MYCN–ER would increase the dependence on genes that control the transcriptional functions of MYCN and screened for short hairpin RNAs (shRNAs) that decrease the fitness of MYCN–ER cells in the presence—but less in the absence—of 4-OHT (Extended Data Fig. 3a). Out of a total of 12,931 shRNAs, this screen identified 104 shRNAs targeting 99 genes that were more-strongly depleted upon growth in the presence of 4-OHT (Supplementary Table 1). Notably, activation of MYCN strongly enhanced the selection against three out of the six shRNAs targeting BRCA1 that were present in the screen (Extended Data Fig. 3b). Independent shRNAs targeting BRCA1 suppressed colony formation more strongly in the presence of 4-OHT (Extended Data Fig. 3c, d). The expression of genes targeted by hits from this screen was enhanced in MYCN-amplified neuroblastomas (Extended Data Fig. 3e). Indeed, BRCA1 expression closely correlated with MYCN amplification (Extended Data Fig. 3f) and was a strong indicator of poor prognosis (Extended Data Fig. 3g). Available datasets of copy-number variation did not show increases in BRCA1 in MYCN-amplified tumours (data not shown). By contrast, genome-wide DNA-methylation data identified BRCA1 as the most significant differentially methylated gene in high-risk and MYCN-amplified neuroblastoma (Fig. 2a). Specifically, the BRCA1 promoter was significantly hypomethylated in patients with high-risk neuroblastoma, whereas hypermethylation was observed in patients with low-risk neuroblastoma (Extended Data Fig. 3h), indicating that there is selective pressure for high BRCA1 expression in MYCN-amplified neuroblastoma.
a, BRCA1 genomic region with average methylation status of 67 primary neuroblastomas and organoids. UTR, untranslated region. b, Genome browser tracks of the NCL locus after 3 h (RNAPII and MYCN) and 5 h (BRCA1) treatment with 4-OHT. n = 2. c, BRCA1 occupancy in promoter-proximal regions. In total, 7,812 expressed genes (log2(CPM) > 1.28; CPM, counts per million) with a BRCA1 peak in the promoter were binned. Data are mean ± 95% confidence interval. P values for the difference in slopes between without 4-OHT and with 4-OHT were calculated using a linear model and analysis of covariance (ANCOVA). d, Position of BRCA1, MYCN and RNAPII on chromatin relative to the transcription start site. The heat map is sorted by the position of the MYCN-binding site. e, Quantification of proximity ligation assays (PLAs) that shows complex formation of BRCA1 with unphosphorylated, and Ser5- and Ser2-phosphorylated RNAPII upon activation of MYCN–ER (4 h). P values were calculated using a two-tailed Wilcoxon rank-sum test (n = 3). In the box plot, the central line reflects the median and the borders of the boxes show the interquartile range of the plotted data. The whiskers extend to 1.5× the interquartile range and outliers are shown as dots. f, BRCA1 occupancy after treatment with 4-OHT or ethanol (5 h) in DMSO-treated (top) and flavopiridol-treated (bottom) cells. Data are mean ± s.e.m.
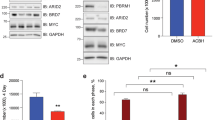
Because BRCA1 stabilizes replication forks and promotes homologous recombination10, we tested whether depletion of BRCA1 interferes with DNA replication or causes double-strand breaks in a MYCN-dependent manner. Depletion of BRCA1 slowed cell-cycle progression and led to an increase in cells in the G1 phase of the cell cycle (Extended Data Fig. 4a, b). Staining with 5-bromo-2′-deoxyuridine (BrdU) showed that BRCA1 depletion reduced the percentage of cells in S phase and that activation of MYCN–ER stimulated S-phase entry in both control and BRCA1-depleted cells (Extended Data Fig. 4b). Depletion of BRCA1 did not induce apoptosis before or after MYCN–ER activation (Extended Data Fig. 4c). DNA fibre assays showed that depletion of BRCA1 reduced the average speed of replication fork progression, but activation of MYCN–ER had no significant effect (Extended Data Fig. 4d, e). Depletion of BRCA1 also led to an increase in the number of γH2A.X and 53BP1 foci, which are indicators of double-stranded DNA breaks (Extended Data Fig. 4f), whereas prolonged activation of MYCN (24 h) led to only a small and statistically insignificant increase in the number of foci. This indicated that MYCN-induced perturbation of DNA replication or double-stranded breaks is unlikely to be the main reason for the increased dependence on BRCA1.
No association of BRCA1 was found with promoter-proximal regions in control cells (Fig. 2b). Activation of MYCN–ER led to a strong increase in the association of BRCA1 with promoter-proximal regions, and the extent of recruitment was equivalent to the expression of the downstream gene (Fig. 2b, c). In addition, MYCN recruited BRCA1 to intergenic sites (Extended Data Fig. 5a). BRCA1-binding sites closely overlapped with MYCN-binding sites (Fig. 2b, d). Consistently, a consensus MYC-binding sequence (E-box) was enriched around BRCA1-binding sites (Extended Data Fig. 5b) and a de novo search for DNA-binding motifs in BRCA1-binding sites identified this sequence as the most strongly enriched DNA motif upon activation of MYCN (Extended Data Fig. 5c). Both short interfering RNA (siRNA)-mediated and shRNA-mediated depletion of BRCA1 reduced the signal observed in ChIP experiments (Extended Data Fig. 5d, e). In addition, ChIP experiments using a second anti-BRCA1 antibody confirmed the validity of the results (Extended Data Fig. 5f). Stable constitutive expression of MYCN also recruited BRCA1 to promoter-proximal regions (Extended Data Fig. 5g, h). By contrast, addition of 4-OHT to SH-EP cells that did not express MYCN–ER had no effect on chromatin occupancy of BRCA1 (Extended Data Fig. 5i).
MYCN activation strongly increased the association between BRCA1 and RNAPII phosphorylated at Ser5, whereas the association between BRCA1 and unphosphorylated RNAPII or RNAPII(pSer2) was only slightly increased11 (Fig. 2e). Inhibition of the CDK9 kinase that phosphorylates RNAPII at Ser2 further enhanced recruitment of BRCA1 to promoters by MYCN (Fig. 2f and Extended Data Fig. 6a), and decreased recruitment to joint MYCN–BRCA1 sites that were not localized in promoters, which indicates a competition for limited pools of BRCA111 (Extended Data Fig. 6b). Inhibition of the CDK7 kinase that phosphorylates RNAPII at Ser5 led to only a small increase in recruitment (Extended Data Fig. 6c). Inhibition of CDK9 in IMR-5 neuroblastoma cells, which contain amplification of MYCN, enhanced recruitment of BRCA1 in a MYCN-dependent manner (Extended Data Fig. 6d). We conclude that stalling of RNA polymerase at the pause site strongly enhances recruitment of BRCA1 by MYCN12.
Depletion of BRCA1 altered the response of RNAPII to MYCN activation (Fig. 3a and Extended Data Fig. 7a). In BRCA1-depleted cells, MYCN activation led to an accumulation of RNAPII at the pause site instead of promoting pause release (Fig. 3a and Extended Data Fig. 7a). This was not due to BRCA1-dependent changes in phosphorylation of RNAPII (Extended Data Fig. 7b). Consistently, activation of MYCN reduced NELF-E promoter occupancy in control cells, but increased it in BRCA1-depleted cells (Extended Data Fig. 7c). As a consequence, activation of MYCN increased rather than decreased the travelling ratio of RNAPII in BRCA1-depleted cells (Extended Data Fig. 7d). A bin plot confirmed that MYCN activation broadly decreased RNAPII association within promoter-proximal regions in control cells but increased the association in BRCA1-depleted cells (Fig. 3b). RNAPII(pSer2) showed an increase at the termination site upon stimulation of MYCN–ER in control cells, but a decrease in BRCA1-depleted cells (Extended Data Fig. 7e). Consistent with these observations, depletion of BRCA1 attenuated activation of multiple MYCN target gene sets and overall changes in gene expression in response to activation of MYCN (Extended Data Fig. 7f,g).
a, Metagene plots of total RNAPII after 4-OHT treatment (3 h) in control and BRCA1-depleted cells treated with BRCA1 shRNA 2 for 914 MYCN-activated genes (n = 2). Data are mean ± s.e.m. b, Correlation between gene expression (n = 3) and RNAPII occupancy at promoters in control and BRCA1-depleted cells treated with BRCA1 shRNA 2. Genes were grouped into 30 bins (300 genes per bin) and the mean for each bin is plotted with 95% confidence interval. c, Strand-specific DNA–RNA immunoprecipitation coupled to high-throughput sequencing data stratified by position of the MYCN-binding site. d, DNA–RNA immunoprecipitation documenting R-loops at the indicated loci upon depletion of BRCA1 and activation of MYCN (4 h). Data are normalized to control (scramble shRNA, ethanol). Data are mean ±s.d. of technical triplicates. n = 4. e, Density plot of DCP1A occupancy after 4-OHT treatment (5 h) in control or BRCA1-depleted cells treated with BRCA1 shRNA 2. Data are mean ± s.e.m. f, Genome browser tracks of the TFAP4 locus show chromatin association of DCP1A in cells treated as in e.
Because the G/C skew of promoters correlates closely with their ability to form R-loops9 and because BRCA1 can suppress R-loop formation at promoter-proximal regions13 and at transcription end sites14, the data suggested that BRCA1 might suppress R-loop formation upon MYCN activation. Available datasets15 showed that 85% of genes annotated to have promoter-proximal R-loops have joint MYCN–BRCA1 sites in promoter-proximal regions, whereas only 50% of genes with distal R-loops do (proximal: 1,328 out of 1,749 promoters; >2 kb: 1,987 out of 3,978 genes; P < 0.0001). Notably, promoters in which MYCN is localized 5′ of the transcription start site often display an R-loop on the antisense strand, whereas this is not observed if the MYCN–BRCA1 site is localized 3′ of the transcription start site (Fig. 3c), which suggests that there is selective pressure for the presence of a MYCN-binding site in the immediate vicinity of an R-loop. We chose 22 R-loop-containing promoters and validated the presence of an R-loop on 14 of them by DNA–RNA immunoprecipitation using the well-characterized R-loops in the ACTB gene as positive control14 (Fig. 3d and Extended Data Fig. 8a, b). On 13 out of 14 of these, activation of MYCN suppressed R-loop formation in control cells. In BRCA1-depleted cells, MYCN-dependent R-loop suppression was strongly compromised on all promoters and activation of MYCN enhanced R-loop formation on 8 out of 14 promoters.
BRCA1 promotes R-loop resolution at transcription end sites by recruiting senataxin, an R-loop-specific DNA/RNA helicase14. However, depletion of BRCA1 enhanced senataxin binding to promoter-proximal regions (Extended Data Fig. 8c). Two termination pathways can limit promoter-proximal accumulation of RNAPII: mRNA decapping, which occurs at the pause site16, and promoter-proximal poly-adenylation17. Upon BRCA1 depletion, activation of MYCN promotes RNAPII accumulation at the pause site, upstream of the most 5′-located polyadenylation site (Extended Data Fig. 8d). Global run-on sequencing showed a drop in nascent transcription close to the pause site (Extended Data Fig. 8e). BRCA1 binds to EDC4, an assembly factor for mRNA decapping complexes18. ChIP–seq of DCP1A, which forms catalytically active decapping complexes with DCP2 upon EDC4-mediated assembly19, showed that depletion of BRCA1 had no effect on the association of DCP1A with chromatin in control cells. Activation of MYCN led to a moderate decrease in DCP1A–chromatin association in control cells, but to a complete loss of DCP1A from promoter-proximal regions in BRCA1-depleted cells, which indicates that recruitment of BRCA1 by MYCN enables mRNA decapping when RNAPII stalls (Fig. 3e, f).
Proteomic analyses did not show an interaction between BRCA1 and MYCN, which suggests that BRCA1 recruitment is indirect20. BRCA1 can be recruited to chromatin by the ubiquitin-specific protease USP11 in a cell-cycle-dependent manner21. MYCN recruited BRCA1 to promoter-proximal regions during S and G2 phases, but not in G1 (Extended Data Fig. 9a), and the association between MYCN and BRCA1 followed a similar regulation (Extended Data Fig. 9b–d). USP11 is present in MYCN complexes20 (Fig. 4a). USP11 heterodimerizes with USP722, which also associates with MYCN23 (Extended Data Fig. 9e). shRNA-mediated depletion showed that USP11 promotes MYCN-dependent recruitment of BRCA1 (Fig. 4b). Indeed, depletion of USP11 strongly reduced expression of both BRCA1 and MYCN (Fig. 4c), arguing that USP11 stabilizes MYCN–BRCA1 complexes. Incubation with the proteasome inhibitor MG-132 reverted the decrease in MYCN levels and partially reverted the decrease in BRCA1 levels in response to USP11 depletion, which suggests that the deubiquitinating activity of USP11 contributes to the stabilization of MYCN and BRCA1 (Extended Data Fig. 9f). Conversely, MYCN promoted complex formation of endogenous USP11 with BRCA1 (Fig. 4d). Consistent with the observation that USP11 stabilizes MYCN, depletion of BRCA1 reduced the association of with MYCN (Fig. 4e). The data show that USP11, MYCN and BRCA1 form a stable complex in neuroblastoma cells.
a, Immunoblot of anti-MYCN immunoprecipitates (IP) from MYCN-amplified (IMR32) neuroblastoma cells. Where indicated, ethidium bromide (EtBr) (1 mg ml−1) was added to the lysate to disrupt DNA-mediated interactions. The input corresponds to 1% of the total protein amount. n = 3. b, Left, ChIP of BRCA1 in SH-EP MYCN–ER cells that express either scramble shRNA or USP11 shRNA upon 4-OHT treatment (5 h). Data are mean + s.d. of technical triplicates. n = 2. Right, immunoblot of cells used for the ChIP experiment. Actin is used as loading control. n = 2. c, Immunoblots of indicated proteins after knockdown of USP11 in MYCN-amplified neuroblastoma cells. Asterisk denotes an unspecific band. Tubulin is used as loading control. n = 3. d, Immunoblots of anti-USP11 immunoprecipitates from control cells or cells that express constitutively active MYCN. CDK2 is used as loading control. The input corresponds to 2% of the total protein amount. n = 2. e, Density plot of MYCN occupancy after 4-OHT treatment (3 h) in control (top) or BRCA1-depleted (bottom) cells treated with BRCA1 shRNA 2 around transcription start sites. f, Immunoblots of anti-USP11 and anti-MYCN immunoprecipitates from SH-EP cells that stably express wild-type (WT) MYCN or MYCN(T58A/S62A) (MUT), as indicated. CDK2 is used as loading control. The input corresponds to 1% of the total protein amount. n = 3.
Pause release of RNAPII by MYC requires the proteasomal turnover of MYC24, which suggests that the turnover of MYCN limits accumulation of the MYCN–BRCA1–USP11 complex. Turnover of MYCN is controlled by phosphorylation of two residues, Ser62 and Thr58, in a highly conserved domain called MYCBoxI; phosphorylated Thr58 is recognized by the FBXW7 ubiquitin ligase, which targets MYCN for degradation25. Alternatively, Thr58 can be dephosphorylated by EYA phosphatases, preventing FBXW7-dependent turnover26. Mass spectrometry demonstrated that all four possible phosphorylated forms—including MYCN that is unphosphorylated or monophosphorylated at Ser62—are present in neuroblastoma cells (Extended Data Fig. 9g). Binding of USP11 to MYCN was enhanced by a Thr58/Ser62 double alanine substitution (Fig. 4f) and by individual substitutions of Ser62 and Thr58 (Extended Data Fig. 9h). Because phosphorylation at Ser62 primes phosphorylation at Thr58 by GSK3, the S62A-mutated allele of MYCN shows reduced phosphorylation at Thr58, but not vice versa (Extended Data Fig. 9i). The data show that dephosphorylation of MYCN at Thr58 is critical for binding of USP11; consistently, proteasomal turnover of MYCN limited the accumulation of MYCN–BRCA1 complexes (Extended Data Fig. 9j).
BRCA1 is part of the cellular stress response, which suggests that transcription-induced stress contributes to BRCA1 recruitment. Inhibition of ATR using VE-821 had no effect on the recruitment of BRCA1, and suppressed phosphorylation of CHK1 at Ser345 (Extended Data Fig. 10a, b). MYCN interacts with poly-ADP-ribose polymerase (PARP1), and PARP1 regulates BRCA1 function in homologous recombination20,27. However, inhibition of PARP1 had no effect on BRCA1 recruitment (Extended Data Fig. 10c). Together with the observation that BRCA1 is not recruited only in S phase, these findings indicate that replication–transcription conflicts are not the major cause for BRCA1 recruitment. Transcriptional activation induces torsional stress28. MYCN interacts with TOPO2A and may relieve torsional stress at promoters20. Indeed, inhibition of type II topoisomerases induced BRCA1 accumulation downstream of active promoters and activation of MYCN enhanced recruitment (Extended Data Fig. 10d, e).
MYC was able to bind USP11, but was unable to promote association of USP11 with BRCA1 (Extended Data Fig. 10f). MYCN was more efficient than MYC in recruiting BRCA1 to chromatin (Extended Data Fig. 10g). MYC and MYCN differ in their response to transcriptional stress because MYC, in contrast to MYCN, is able to recruit MIZ1 to buffer excessive transcriptional activation29. We hypothesize that MYC and MYCN engage different mechanisms of coping with deregulated transcriptional elongation and that MYCN-amplified neuroblastoma tumours are characterized by the ability of MYCN to engage an USP11–BRCA1-dependent pathway that suppresses the accumulation of stalled RNAPII (Extended Data Fig. 10h).
Methods
Detailed information about cell lines, antibodies, reagents, commercial kits, primers, siRNA and shRNA sequences and used software is reported in Supplementary Table 2.
Cell culture
Neuroblastoma cell lines (IMR-32, IMR-5, SH-EP and SMS-KAN) were verified by single tandem repeat profiling and grown in RPMI 1640 (Sigma-Aldrich and Thermo Fisher Scientific). HEK293TN, Plat-E, KPC and NIH-3T3 cells were grown in DMEM (Sigma-Aldrich and Thermo Fisher Scientific). Medium was supplemented with 10% fetal calf serum (Biochrom and Sigma-Aldrich) and penicillin–streptomycin (Sigma-Aldrich). All cells were routinely tested for mycoplasma contamination. Where indicated, cells were treated with 4-OHT (Sigma-Aldrich), flavopiridol (Sigma-Aldrich), THZ1 (Hycultec) or etoposide (Sigma-Aldrich). For double-thymidine block, cells were treated for 16 h with 2 mM thymidine (Sigma-Aldrich), released for 8 h into normal medium and then blocked again (2 mM, 16 h). For release, cells were washed with PBS before medium was added.
For the clonogenic assay, 1 × 105 SH-EP MYCN–ER cells previously infected and selected as described in ‘Transfection and lentiviral infection’ were plated and treated from the following day onwards with 200 nM 4-OHT or ethanol for 6 days. Fresh medium with 4-OHT or ethanol was added every second day. Cells were fixed with 3.7% formaldehyde for 20 min. After aspirating the medium, crystal violet solution (0.1% crystal violet, 20% ethanol in ddH2O) was added for overnight staining and cells were washed the next day with ddH2O.
Transfection and lentiviral infection
Transfection with siRNA was performed using the RNAiMAX reagent according to the manufacturer’s protocol. Cells were collected 72 h after transfection. Transfection with cDNA was performed using PEI (Sigma-Aldrich). Cells were collected 48 h after transfection. For lentivirus production, HEK293TN cells were transfected using PEI. Lentivirus expressing shRNA targeting BRCA1 was produced by transfection with the pGIPZ plasmid together with the packaging plasmid psPAX.2 and the envelope plasmid pMD2.G. Lentivirus expressing shRNA targeting USP11 was produced by transfection with the pLKO plasmid. Virus-containing supernatant was collected 48 h and 72 h after transfection. SH-EP cells were infected with lentiviral supernatants in the presence of 4 μg ml−1 polybrene for 24 h. Cells were selected for 2 days with puromycin (2 μg ml−1; IMR5 0.5 μg ml−1) and afterwards plated for the experiment.
Immunoblot and immunoprecipitation
Whole-cell extracts were prepared using NP-40 buffer (50 mM Tris (pH 8.0), 150 mM NaCl, 1% NP-40) with three rounds of freeze–thaw cycles or RIPA buffer (50 mM HEPES, 140 mM NaCl, 1 mM EDTA, 1% triton X-100, 0.1% sodium deoxycholate and 0.1% SDS) containing protease and phosphatase inhibitor cocktails (Sigma-Aldrich). Lysates were cleared by centrifugation, separated on SDS or Bis-Tris gels and transferred to a PVDF membrane (Millipore).
For immunoprecipitation, cells were resuspended in lysis buffer containing 20 mM HEPES-KOH (pH 7.8), 140 mM KCl, 0.2 mM EDTA, 0.1% NP-40 supplemented with a cocktail of protease and phosphatase inhibitors. After brief sonication, samples were incubated on ice for 30 min and cleared by centrifugation. Co-immunoprecipitation was carried out in lysis buffer using 2 μg of antibodies and 1–2 mg lysate.
For immunoblots showing multiple proteins with similar molecular mass, one representative loading control is shown. As loading control, vinculin, actin, tubulin or CDK2 was used. For gel source data, see Supplementary Fig. 1.
Flow cytometry analysis
Flow cytometry (fluorescence-activated cell sorting, FACS) analysis was performed as previously described30. Subconfluent cells were labelled with 20 μM BrdU (Sigma-Aldrich) for 1 h. Cells were collected, washed with ice-cold PBS and fixed in 80% ethanol overnight at −20 °C. Cells were washed with cold PBS and incubated in 2 M HCl/0.5% triton X-100 for 30 min at room temperature. Cell pellets were neutralized by incubating with Na2B4O7. The pellet was incubated with anti-BrdU–FITC antibody (BioLegend) diluted in 100 μl 1% BSA and 0.5% Tween-20 in PBS for 30 min at room temperature in the dark. After washing with PBS, the cells were resuspended in PBS with RNase A (24 μg ml−1) and propidium iodide (54 μM) and incubated for 30 min at 37 °C.
For FACS using propidium iodide, cells were collected by trypsinization, washed with cold PBS and fixed in 80% ethanol overnight at −20 °C. After washing with PBS, the cells were resuspended in PBS with RNase A (24 μg ml−1) and propidium iodide (54 μM), and incubated for 30 min at 37 °C.
For FACS using annexin V and propidium iodide, the supernatant of the respective cultures was combined with cells collected by trypsinization and washed with cold PBS. Cell pellets were resuspended in 100 μl 1× annexin V-binding buffer (10 mM HEPES pH 7.4, 140 mM NaCl and 2.5 mM CaCl2) and 2 μl annexin V/Pacific Blue dye and incubated for 15 min at room temperature in the dark. Afterwards, 400 μl 1× binding buffer and propidium iodide (54 μM) were added and the samples were stored cold and dark until analysis.
Subsequent analysis of all FACS experiments was performed on a BD FACSCanto II flow cytometer using BD FACSDIVA Software.
DNA–RNA immunoprecipitation
DNA–RNA immunoprecipitation (DRIP) was performed as previously described31. In brief, cells were digested with 0.5% SDS and proteinase K overnight. DNA was extracted with phenol/chloroform and precipitated with ethanol. DNA was digested using a cocktail of restriction enzymes (Bsrg1, EcoR1, HindIII, SspI and XbaI) overnight at 37 °C. For RNase H-treated samples, DNA was additionally incubated with RNase H overnight. DNA was purified as described above. S9.6 antibody, which detects RNA–DNA hybrids32, was coupled to A/G-Dynabeads (Invitrogen). DNA in 1× binding buffer (10 mM NaPO4 pH 7.0, 140 mM NaCl and 0.05% triton X-100) was added to the antibody-coupled beads overnight. After extensive washing, DNA was eluted with elution buffer (50 mM Tris-HCl pH 8.0, 10 mM EDTA and 0.5% SDS) and treated for 2 h at 45 °C with proteinase K. After DNA extraction, locus-specific DRIP signals were assessed by qPCR.
High-throughput sequencing
ChIP and ChIP–seq analyses were performed as previously described33. For spike-in experiments (ChIP-Rx), 10% of fixed NIH-3T3 or KPC mouse cell lines were added before lysis. Cells were treated with 1% formaldehyde for 10 min at room temperature following 5 min of incubation with glycine. After cell lysis (5 mM PIPES pH 8.8, 5 mM KCl and 0.5% NP40), nuclei were resuspended in RIPA buffer (50 mM HEPES pH 7.9, 140 mM NaCl, 1% triton X-100, 0.1% deoxycholate, 0.1% SDS and 1 mM EDTA containing protease and phosphatase inhibitor cocktails) and DNA was fragmented to a size of <500 bp using a Branson sonifier. Antibodies were bound to protein A/G-Dynabeads (Invitrogen) and immunoprecipitated. After extensive washing, chromatin was eluted with 1% SDS and crosslinking was reverted overnight. Phenol/chloroform extraction was used for DNA purification. After DNA extraction occupancy of different proteins was assessed by qPCR. qPCR analyses show mean ± s.d. of technical triplicates as well as an overlay of each data point to indicate the distribution of the data.
ChIP–seq was performed as previously described34. Purified DNA was end-repaired, A-tailed, ligated to Illumina adaptors, size-selected (200 bp) and purified with a gel-extraction kit. DNA fragments were amplified by 15–18 cycles of PCR, and the library size and amount of library were specified with the Biorad Experion Automated Electrophoresis system or Fragment Analyzer (Advanced Analytical). The library was subjected to Illumina GAIIx or Illumina NextSeq 500 sequencing, according to the manufacturer’s instructions. After base-calling using the CASAVA software (GAIIx sequencing) or Illumina’s FASTQ Generation software v.1.0.0 (NextSeq 500 sequencing), high quality PF-clusters were selected for further analysis.
RNA sequencing was performed as previously described24 using an Illumina NextSeq 500. RNA was extracted using RNeasy mini columns (Qiagen) including on-column DNase I digestion. mRNA was isolated using the NEBNext Poly(A) mRNA Magnetic Isolation Module (NEB) and library preparation was performed with the NEBNext Ultra RNA Library Prep Kit for Illumina following the instruction manual. Libraries were size-selected using Agencourt AMPure XP Beads (Beckman Coulter) followed by amplification with 12 PCR cycles. Library quantification and size determination was performed with the Biorad Experion Automated Electrophoresis system or Fragment Analyzer (Advanced Analytical).
For 4-thiouridine (4sU)-labelled nascent RNA sequencing, SH-EP MYCN–ER cells were cultured at a density of 5 million cells per plate 24 h before treatment with 4-OHT. Before collection of RNA using the Qiagen miRNeasy kit, nascent RNA was labelled by adding 200 μM of 4sU (Sigma-Aldrich) in RPMI medium to the cells for 15 min under normal culture conditions. After extraction and quantification of total RNA by Nanodrop, equal amounts were labelled with biotin (Pierce) in the presence of DMF–HPDP buffer. Free biotin removal was carried out by chloroform–isoamyl alcohol extraction, after which RNA was resuspended into nuclease free water. Dynabeads MyOne Streptavidin T1 beads (Life Technologies) were used for enrichment of biotinylated RNA, which was then eluted by 100 mM DTT and cleaned by RNeasy MinElute cleanup kit. The nascent RNA concentration was then measured using a RiboGreen RNA assay kit and equal amounts were used for library preparation. Before library preparation, rRNA was depleted using a NEB rRNA depletion kit and then all eluted material was processed using the NEB Ultra Directional kit with 17 PCR cycles. The libraries were then sequenced for 75 cycles using an Illumina NextSeq 500 system.
Global run-on followed by next-generation sequencing (GRO–seq) was carried out as previously described, with a few modifications35. In brief, cells were washed twice with cold 1× PBS and collected in 1× PBS. Cells were resuspended in 10 ml swelling buffer (10 mM Tris-HCl pH 7.5, 2 mM MgCl2 and 3 mM CaCl2) and incubated for 5 min on ice. After centrifugation (400g, 10 min), cells were resuspended in 10 ml swelling buffer containing 10% glycerol. Then, 10 ml lysis buffer (10 mM Tris-HCl pH 7.5, 2 mM MgCl2, 3 mM CaCl2, 10% glycerol and 1% Igepal) was added with gently swirling and the sample was incubated for 5 min on ice. The volume of cell suspension was brought to 45 ml with lysis buffer. Resultant nuclei were washed with 10 ml freezing buffer (40% glycerol, 50 mM Tris-HCl pH 8.0, 5 mM MgCl2 and 0.1 mM EDTA). The pellet was resuspended in 100 μl freezing buffer per 1 × 107 of nuclei.
For run-on reactions, resuspended nuclei were mixed with the same amount of run-on reaction buffer (10 mM Tris-HCl pH 8.0, 5 mM MgCl2, 300 mM KCl, 1 mM DTT, 500 μM ATP, 500 μM GTP, 500 μM Br-UTP, 2 μM CTP, 200 U ml−1 Superase In and 1% N-laurylsarcosine) and incubated 7 min at 30 °C. The reaction was blocked by adding 600 μl Trifast, thoroughly vortexing and incubating for 5 min. RNA was extracted by adding 160 μl chloroform, precipitated with ethanol and resuspended in 20 μl H2O. RNA was next subjected to DNase treatment (Turbo DNA-free kit, Thermo Fisher) twice according to manufacturer’s protocol. RNA was fragmented using an Ambion RNA fragmentation kit and end-repaired by T4 PNK (NEB).
Anti-BrdU agarose beads (Santa-Cruz, 50 μl per sample) were washed twice and then blocked for 1 h with blocking buffer (1× binding buffer, 0.1% polyvinylpyrrolidone and 0.1% BSA). Beads were washed twice with binding buffer (0.25× SSPE, 0.05% Tween20, 37.5 mM NaCl and 1 mM EDTA) and mixed with RNA in 700 μl binding buffer for 1 h on a rotating wheel. After binding, beads were washed twice with binding buffer, twice with low-salt wash buffer (0.2× SSPE, 0.05% Tween20 and 1 mM EDTA), one time with high-salt wash buffer (0.2× SSPE, 137.5 mM NaCl, 0.05% Tween20 and 1 mM EDTA) and twice with TE with 0.05% Tween20. Finally, RNA was eluted four times with 100 μl elution buffer (50 mM Tris pH 7.5, 150 mM NaCl, 0.1% SDS, 20 mM DTT and 1 mM EDTA) at 37 °C. RNA was purified with TRIzol and chloroform as described above. RNA was decapped using RppH (NEB) with 10× NEB Thermopol buffer for 1 h at 37 °C. Reactions were stopped by adding EDTA and incubation at 65 °C for 5 min. Afterwards RNA was extracted as described above. Library preparation was done according to the manufacturer’s instructions using a NEBNext Multiplex Small RNA Library Prep Kit for Illumina.
shRNA screening
A total of 18,290 shRNAs of the Open Biosystems (Dharmacon; GE Lifesciences) pGIPZ shRNA library (releases 6.1–6.12) were screened in two pools (consisting of 9,589 and 8,701 individual shRNAs) and each pool in two independent biological replicates. Upon reannotation using the human transcriptome hg19 (Homo_sapiens.GRCh37.74.cDNA.all.fa), 12,931 shRNAs were found to target 9,601 genes. Of these, 1, 8, 10, 96, 429, 2,098 and 6,959 were targeted with 7, 6, 5, 4, 3, 2 or 1 individual shRNAs, respectively. Following lentiviral packaging of the plasmids, SH-EP MYCN–ER cells were transduced with the viral library at a multiplicity of infection of 0.1. Infected cells were selected with puromycin for three days and the screen was started immediately after selection was completed. Cells were collected before the treatment with ethanol or 4-OHT (condition ‘start’) and after two weeks of treatment with either ethanol (control) or 4-OHT (activation of MYCN). Cell numbers were counted regularly over the entire course of the screening experiment to verify an equal number of population doublings in both conditions. Every 2–3 days, the cells of both conditions were split and supplied with fresh 4-OHT or ethanol. The number of cells was always kept at a minimum of 1.0 × 107 cells to guarantee a sufficient minimum representation of all shRNAs. Genomic DNA was isolated using DNAzol (Invitrogen) followed by ethanol precipitation. shRNA hairpin sequences were recovered from the genomic host cell DNA in an amplification reaction using custom PCR primers that specifically bind to the pGIPZ vector sequences that flank the shRNA sequences. For each condition, 70 identical PCRs of 24 cycles and with 1 μg of genomic DNA each were carried out and corresponding products were pooled and gel-purified. The quantity and size of the purified PCR products was determined using the Experion Automated Electrophoresis System (Bio-Rad). Sample-specific DNA libraries were subjected to Illumina GAIIx single-read sequencing according to the manufacturer’s instructions. Mapping of the sample-specific reads and initial bioinformatics analyses were done as described in ‘Bioinformatics analysis and statistics’. Using Z-score statistics for the effect strengths, we identified 104 shRNAs targeting 99 genes that were strongly depleted upon growth of SH-EP MYCN–ER cells in the presence of 4-OHT, but much less depleted in the absence of 4-OHT.
In situ PLA
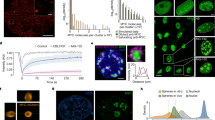
PLA was performed using the Duolink In situ Kit (Sigma-Aldrich) according to the manufacturer’s protocol. For counterstaining of nuclei and actin, Hoechst 33342 (Sigma-Aldrich) and Alexa Fluor 568–phalloidin (Thermo Fisher Scientific), respectively, were used. Images were taken with a confocal microscope (Nikon Ti-Eclipse) with 60× magnification and on an Operetta High-Content Imaging System with 40× magnification. For quantification, not less than 300 cells were analysed using ImageJ (W. Rasband, NIH). Images from Operetta were analysed using Harmony High Content Imaging and Analysis Software. P values were calculated using a two-tailed Wilcoxon rank-sum test.
DNA fibre assay
DNA fibre assays to analyse replication fork progression and origin firing were essentially carried out as previously described36. Cells were first incubated with 4-OHT for 6 h and afterwards with 5-chloro-2-deoxyuridine (CldU, 25 μM) for 20 min, followed by 5-iodo-2-deoxyuridine (IdU, 25 μM; both from Sigma-Aldrich) for 1 h. DNA fibres were spread on glass slides. After acid treatment, CldU- and IdU-labelled tracts were detected by 1 h incubation at 20 °C with rat anti-BrdU antibody (dilution 1:400 detects BrdU and CldU; AbD Serotec) and mouse anti-BrdU antibody (1:150, detects BrdU and IdU; Becton Dickinson). Slides were fixed in 4% paraformaldehyde in PBS and incubated for 2 h at 20 °C with an Alexa Fluor 555-conjugated goat anti-rat antibody (dilution 1:150) or Alexa Fluor 488-conjugated goat anti-mouse antibody (dilution 1:150; both from Molecular Probes/Thermofisher). Fibre images were acquired by fluorescence microscopy using the Axio Scope A1 running with the microscope software ZEN (both from Zeiss) for image acquisition and processing. For analysis of fibre images, the imaging software ImageJ was used. Statistical analysis of replication fork progression was performed using the two-tailed, unpaired t-test with additional Welch’s correction in GraphPad Prism v.5.0, because of unequal variances between samples.
Immunofluorescence staining
Cells were infected and selected by puromycin treatment as previously described. After selection, cells were plated in a 96-well plate and incubated with ethanol or 4-OHT from the following day onwards for 24 h (200 nM). The last two hours, etoposide was added as positive control (25 μM). Cells were fixed with 3.7% paraformaldehyde in PBS. After removing paraformaldehyde and washing, cells were permeabilized with 0.2% triton X-100 in PBS and blocked with 3% BSA in PBS. Samples were stained with primary antibodies against γH2A.X (1:400) and 53BP1 (1:500) in 3% BSA in PBS overnight at 4 °C and after washing incubated with secondary antibody (1:400) for 1 h at room temperature. Images were taken with an Operetta High-Content Imaging System with 20× magnification. Images were analysed using Harmony High Content Imaging and Analysis Software.
Bioinformatics analysis and statistics
Base-calling was performed using Illumina’s FASTQ Generation software v.1.0.0 and sequencing quality was tested using the FastQC script.
For ChIP–seq, reads were mapped to hg19 using Bowtie137 with default parameters, and samples were normalized to the number of mapped reads in the smallest sample. Peak-calling for BRCA1, MYCN and total RNAPII with MACS1438 was performed with the corresponding input sample as control and with variable settings for duplicates (--keep-dup; (antibody/biological condition/value): BRCA1 ± 4-OHT: 5; BRCA1 ± 4-OHT, ±flavopiridol, ±etoposide: 3; MYCN ± 4-OHT: 5; DCP1A ± 4-OHT, ±BRCA1 shRNA: 1; MYCN pRRL–MYCN: 1; BRCA1 pWZL–MYCN/pWZL–empty: 5; total RNAPII ethanol: 5) and P value (--pvalue; (antibody/biological condition/value): BRCA1 ± 4-OHT: 1 × 10−6; BRCA1 ± 4-OHT, ±flavopiridol, ±etoposide: 1 × 10−7; MYCN ± 4-OHT: 1 × 10−11; DCP1A ± 4-OHT, ±BRCA1 shRNA: 1 × 10−6; MYCN pRRL–MYCN: 1 × 10−9; BRCA1 pWZL–MYCN/pWZL–empty: 1 × 10−9; total-RNAPII ethanol: 1 × 10−14). Bedgraph files were generated using the genomecov function from BEDtools39 and the Integrated Genome Browser40 was used to visualize these density files. Annotation of peaks to the next RefSeq gene was done with BEDtools closestBed and a promoter was defined as a region ±1 kb relative to the transcription start site (TSS). Read density graphs were obtained using the computeMatrix function from DeepTools41 at a resolution of 10 bp. For de novo motif analysis the DREME algorithm implemented in the MEME suite42 was used with default settings and similarities between identified motifs and known motifs were examined with TOMTOM and three databases (‘JASPAR Core vertebrates 2014’, ‘Jolma 2013’ and ‘Uniprobe mouse’). Relative motif frequencies of E-boxes were counted with an in-house script and curves were smoothed with a moving window of 50 bp. Travelling ratios for RNAPII ChIP–seq were calculated by counting reads using the BEDtools ‘coverage’ function (parameter: –F 0.51) around the TSS (−30 to +300 bp), within gene bodies (+300 bp to TES) and at the TES (TES to +1 kb) of Ensembl genes. A pseudocount per kb was added, gene body counts were normalized to the length of the gene and, finally, TSS counts were divided by gene body counts. Metagene window plots were generated with NGStools43 of the indicated gene group. MYCN-activated and -repressed genes are defined by positive and negative log2(change in expression), respectively, of genes with Benjamini–Hochberg q-values (±4-OHT) less than 0.05, with an expression filter that rejects genes with less than 2 CPM.
Classification of genes with strong, weak or no G/C skew was done with skewR31 using the human hg19 genome, all expressed genes and the parameters: -z 60 –s hg19.fa -m model/GC_SKEW_1mil.hmm -g UCSC_Main_on_Human_refGene_hg19_TSS_toTES.bed -b UCSC_hg19_CpGislands.bed. Gene expression changes in SH-EP MYCN–ER cells after activation of MYCN of the respective class of genes was plotted as kernel density with a bandwidth of 0.05. P values were calculated with a two-tailed Wilcoxon one-sample signed-rank test with μ = 0.
To correlate BRCA1 occupancy in promoter-proximal regions before and after activation of MYCN–ER with gene expression, 7,812 expressed genes (log2(CPM) > 1.28) with a BRCA1 peak in the promoter (±1 kb relative to the TSS) were grouped into 15 equally sized bins and the mean of BRCA1 occupancy in a region of ±100 bp around the peak summit was plotted, including ±95% confidence interval.
For ChIP-Rx–seq, reads were mapped independently to the human hg19 and mouse mm10 (spike-in) genome and a spike-in normalization factor was calculated by dividing the number of mapped reads of the spike-in of the smallest sample by the number of mapped reads of the spike-in for each sample. For each sample, this factor was multiplied by the number of reads that map to the human genome and all BAM files for subsequent analysis were adjusted to this read count. Peak-calling of the MYCN ChIP-Rx sample ‘+4-OHT, −BRCA1 shRNA’ was done with MACS14 (keep-dup: 1, P value: 1 × 10−10) with separate peak-calling for the corresponding input sample using the same parameters as the treated sample. Overlapping peaks in the input were then subtracted from the MYCN ChIP-Rx sample resulting in 692 peaks in promoters of RefSeq genes (±1 kb relative to TSS). Read density plots around these TSS were then drawn with a resolution of 10 bp using the ‘computeMatrix’ and ‘plotProfile’ function from DeepTools.
For GRO–seq, reads of all biological conditions (±4-OHT, ±BRCA1 shRNA) were pooled and the 3′ adaptor was trimmed using Cutadapt44 keeping at least 30 bp. Untrimmed and trimmed reads passing the filter were then mapped to hg19 using TopHat245 and Bowtie246, including spliced reads. TSS with a called RNAPII peak (ethanol condition) with a maximum distance of 250 bp to the TSS were used as view point for a heat map drawn with ‘computeMatrix’ and ‘plotHeatmap’ from DeepTools at a resolution of 1 bp. The resulting heat map is sorted based on the distance of the TSS to the RNAPII peak and additionally contains read coverage from mRNA-sequencing (sample: ‘ethanol’) and ChIP–seq (samples: ‘total RNAPII (N20) ethanol’ and corresponding input).
Raw data from DRIPc–seq47 were downloaded from GEO (GSE70189, sample GSM1720613), strand-specifically mapped to hg19 with Bowtie1, and the read density around TSSs of genes with either up- or downstream MYCN peaks was calculated with DeepTools. Bidirectional genes and genes with multiple annotated TSSs were removed before the density matrix was calculated and the mean profile was drawn. To identify the number of BRCA1/MYCN-bound genes with R-loops, coordinates of R-loops from DRIP–seq experiments from NT2 (two replicates), K562, E14 and NIH-3T3 cells were obtained from GEO (GSE70189) and converted to hg19 if necessary. A consensus R-loops dataset was generated as defined by (1) overlap by at least 1 bp in all samples, (2) overlap by at least 1 bp in an RNase A-treated sample and (3) not overlapping with an RNase H-treated sample. The resulting R-loops were then annotated to the next TSS, MYCN and BRCA1 peak and filtered as indicated. P values were calculated with a permutation test (n = 10,000).
Coordinates of polyA sites were downloaded from UCSC (UHR; two replicates)48, extended in both directions by 15 bp and overlapping polyA sites from both replicates were extracted. For each annotated RefSeq TSS, the first downstream intronic polyA site in a region of TSS to +5 kb that is not in a window of ±1 kb around a TES was used to analyse RNAPII density.
For mRNA sequencing, reads were mapped to hg19 using TopHat245 and Bowtie246 and samples were normalized to the number of mapped reads in the smallest sample. Reads per gene were counted using the ‘summarizeOverlaps’ function from the R package ‘GenomicAlignments’ using the ‘union’ mode and Ensembl genes. Non- and weakly expressed genes were removed (mean count over all samples < 1 (BRCA1 shRNA experiment) or < 1.28 (MYCN–ER ± 4-OHT; GSE78957)). Differentially expressed genes were called with edgeR and P values were adjusted for multiple testing using the Benjamini–Hochberg procedure. Gene set enrichment analyses49 were done with the ‘Hallmark’, ‘C2’ and ‘C7’ databases from MSigDB50, 1,000 permutations and default settings. For bin plots, genes were sorted on the basis of the indicated criterion, grouped into equally sized bins and the mean or median was calculated. P values for the difference in slopes were calculated using a linear model and analysis of variance (ANOVA). To calculate P values comparing the median of different groups in box plots, a two-tailed Wilcoxon signed-rank test (for paired data) or a two-tailed Wilcoxon rank-sum test (unpaired data) was used.
To correlate MYCN-dependent gene expression changes with gene expression in neuroblastoma tumour samples, normalized expression values from GSE6256451 were downloaded from GEO, row-wise median-centred and the top 400 differentially expressed genes between stage 4 and stage 1/2/3/4s were defined with: (meanstage4 − meanstage1,2,3,4s)/(s.d.stage4 + s.d.stage1,2,3,4s). For each tumour sample, the Pearson’s correlation coefficient between the median-centred expression and the gene expression changes between scramble shRNA 4-OHT versus scramble shRNA ethanol was calculated. Kaplan–Meier survival curves stratified by BRCA1 expression from the same dataset were calculated using the R2: Genomics Analysis and Visualization Platform (http://r2.amc.nl) in ‘scanning’ mode.
Methylation data from human neuroblastoma tumour samples and corresponding meta information were taken from GSE76269 and the R2 platform was used for visualization of the methylation status of probes within the BRCA1 genomic locus. The ratio of methylation is defined as signal (methylated)/signal (methylated + unmethylated).
For 4sU-seq, fastq generation, quality check, read mapping, and normalization of mapped reads and differential expression analyses, the analysis was carried out in the same way as for mRNA sequencing, with the difference being that the reads falling in exons were not considered. In total, 18,213 genes were grouped into 39 equally sized bins.
For analysing the shRNA screen, fastq files were mapped to a custom reference database that contains the guide-stem sequences of all screened shRNAs. Mapping was done using Bowtie v.0.12.8, not allowing any mismatches. Mapped reads per sample were counted and combined in one count matrix. Average values of size-normalized counts for corresponding conditions from replicate experiments were used to calculate relevant fold changes to determine enrichment or depletion of individual shRNAs between conditions. A Z-score statistic was applied to all calculated log2(fold change in abundance) (indicating the number of standard deviations an effect is above or below the mean of the overall population of shRNAs), to compare the results from the two separately screened shRNA pools and to determine cut-off values for a combined hit selection. shRNAs with Z-scores of Z < −3 for the change in abundance between the ethanol-treated and the 4-OHT-treated conditions were defined as ‘synthetic lethal hits’ as long as the Z-score for the change in abundance between the starting condition and the ethanol-treated control condition was Z > −3.
In box plots, the central line reflects the median and the borders of the boxes show the interquartile range of the plotted data. The whiskers extend to 1.5× the interquartile range and outliers are shown as dots.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this paper.
References
Rickman, D. S., Schulte, J. H. & Eilers, M. The expanding world of N-MYC-driven tumors. Cancer Discov. 8, 150–163 (2018).
Nie, Z. et al. c-Myc is a universal amplifier of expressed genes in lymphocytes and embryonic stem cells. Cell 151, 68–79 (2012).
Rahl, P. B. et al. c-Myc regulates transcriptional pause release. Cell 141, 432–445 (2010).
Pao, G. M. et al. Role of BRCA1 in brain development. Proc. Natl Acad. Sci. USA 111, E1240–E1248 (2014).
Kortlever, R. M. et al. Myc cooperates with Ras by programming inflammation and immune suppression. Cell 171, 1301–1315 (2017).
de Pretis, S. et al. Integrative analysis of RNA polymerase II and transcriptional dynamics upon MYC activation. Genome Res. 27, 1658–1664 (2017).
Lorenzin, F. et al. Different promoter affinities account for specificity in MYC-dependent gene regulation. eLife 5, e15161 (2016).
Muhar, M. et al. SLAM-seq defines direct gene-regulatory functions of the BRD4–MYC axis. Science 360, 800–805 (2018).
Ginno, P. A., Lim, Y. W., Lott, P. L., Korf, I. & Chédin, F. GC skew at the 5′ and 3′ ends of human genes links R-loop formation to epigenetic regulation and transcription termination. Genome Res. 23, 1590–1600 (2013).
Quinet, A., Lemaçon, D. & Vindigni, A. Replication fork reversal: players and guardians. Mol. Cell 68, 830–833 (2017).
Gorthi, A. et al. EWS–FLI1 increases transcription to cause R-loops and block BRCA1 repair in Ewing sarcoma. Nature 555, 387–391 (2018).
Scully, R. et al. BRCA1 is a component of the RNA polymerase II holoenzyme. Proc. Natl Acad. Sci. USA 94, 5605–5610 (1997).
Zhang, X. et al. Attenuation of RNA polymerase II pausing mitigates BRCA1-associated R-loop accumulation and tumorigenesis. Nat. Commun. 8, 15908 (2017).
Hatchi, E. et al. BRCA1 recruitment to transcriptional pause sites is required for R-loop-driven DNA damage repair. Mol. Cell 57, 636–647 (2015).
Chen, L. et al. R-ChIP using inactive RNase H reveals dynamic coupling of R-loops with transcriptional pausing at gene promoters. Mol. Cell 68, 745–757 (2017).
Brannan, K. et al. mRNA decapping factors and the exonuclease Xrn2 function in widespread premature termination of RNA polymerase II transcription. Mol. Cell 46, 311–324 (2012).
Almada, A. E., Wu, X., Kriz, A. J., Burge, C. B. & Sharp, P. A. Promoter directionality is controlled by U1 snRNP and polyadenylation signals. Nature 499, 360–363 (2013).
Hernández, G. et al. Decapping protein EDC4 regulates DNA repair and phenocopies BRCA1. Nat. Commun. 9, 967 (2018).
Chang, C. T., Bercovich, N., Loh, B., Jonas, S. & Izaurralde, E. The activation of the decapping enzyme DCP2 by DCP1 occurs on the EDC4 scaffold and involves a conserved loop in DCP1. Nucleic Acids Res. 42, 5217–5233 (2014).
Büchel, G. et al. Association with Aurora-A controls N-MYC-dependent promoter escape and pause release of RNA polymerase II during the cell cycle. Cell Rep. 21, 3483–3497 (2017).
Orthwein, A. et al. A mechanism for the suppression of homologous recombination in G1 cells. Nature 528, 422–426 (2015).
Maertens, G. N., El Messaoudi-Aubert, S., Elderkin, S., Hiom, K. & Peters, G. Ubiquitin-specific proteases 7 and 11 modulate Polycomb regulation of the INK4a tumour suppressor. EMBO J. 29, 2553–2565 (2010).
Tavana, O. et al. HAUSP deubiquitinates and stabilizes N-Myc in neuroblastoma. Nat. Med. 22, 1180–1186 (2016).
Jaenicke, L. A. et al. Ubiquitin-dependent turnover of MYC antagonizes MYC/PAF1C complex accumulation to drive transcriptional elongation. Mol. Cell 61, 54–67 (2016).
Farrell, A. S. & Sears, R. C. MYC degradation. Cold Spring Harb. Perspect. Med. 4, a014365 (2014).
Li, J. et al. EYA1’s conformation specificity in dephosphorylating phosphothreonine in Myc and its activity on Myc stabilization in breast cancer. Mol. Cell. Biol. 37, e00499-16 (2016).
Hu, Y. et al. PARP1-driven poly-ADP-ribosylation regulates BRCA1 function in homologous recombination-mediated DNA repair. Cancer Discov. 4, 1430–1447 (2014).
Kouzine, F. et al. Transcription-dependent dynamic supercoiling is a short-range genomic force. Nat. Struct. Mol. Biol. 20, 396–403 (2013).
Vo, B. T. et al. The interaction of Myc with Miz1 defines medulloblastoma subgroup identity. Cancer Cell 29, 5–16 (2016).
Schülein-Völk, C. et al. Dual regulation of Fbw7 function and oncogenic transformation by Usp28. Cell Rep. 9, 1099–1109 (2014).
Ginno, P. A., Lott, P. L., Christensen, H. C., Korf, I. & Chédin, F. R-loop formation is a distinctive characteristic of unmethylated human CpG island promoters. Mol. Cell 45, 814–825 (2012).
Boguslawski, S. J. et al. Characterization of monoclonal antibody to DNA·RNA and its application to immunodetection of hybrids. J. Immunol. Methods 89, 123–130 (1986).
Walz, S. et al. Activation and repression by oncogenic MYC shape tumour-specific gene expression profiles. Nature 511, 483–487 (2014).
Chen, X. et al. Integration of external signaling pathways with the core transcriptional network in embryonic stem cells. Cell 133, 1106–1117 (2008).
Gardini, A. Global run-on sequencing (GRO–seq). Methods Mol. Biol. 1468, 111–120 (2017).
Klusmann, I. et al. p53 activity results in DNA replication fork processivity. Cell Rep. 17, 1845–1857 (2016).
Langmead, B., Trapnell, C., Pop, M. & Salzberg, S. L. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol. 10, R25 (2009).
Zhang, Y. et al. Model-based analysis of ChIP–seq (MACS). Genome Biol. 9, R137 (2008).
Quinlan, A. R. & Hall, I. M. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010).
Freese, N. H., Norris, D. C. & Loraine, A. E. Integrated genome browser: visual analytics platform for genomics. Bioinformatics 32, 2089–2095 (2016).
Ramírez, F., Dündar, F., Diehl, S., Grüning, B. A. & Manke, T. deepTools: a flexible platform for exploring deep-sequencing data. Nucleic Acids Res. 42, W187–W191 (2014).
Bailey, T. L. et al. MEME SUITE: tools for motif discovery and searching. Nucleic Acids Res. 37, W202–W208 (2009).
Shen, L., Shao, N., Liu, X. & Nestler, E. ngs.plot: quick mining and visualization of next-generation sequencing data by integrating genomic databases. BMC Genomics 15, 284 (2014).
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J 17, 10–12 (2011).
Kim, D. et al. TopHat2: accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol. 14, R36 (2013).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Sanz, L. A. et al. Prevalent, dynamic, and conserved R-Loop structures associate with specific epigenomic signatures in mammals. Mol. Cell 63, 167–178 (2016).
Derti, A. et al. A quantitative atlas of polyadenylation in five mammals. Genome Res. 22, 1173–1183 (2012).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl Acad. Sci. USA 102, 15545–15550 (2005).
Liberzon, A. et al. The Molecular Signatures Database (MSigDB) hallmark gene set collection. Cell Syst. 1, 417–425 (2015).
Su, Z. et al. An investigation of biomarkers derived from legacy microarray data for their utility in the RNA-seq era. Genome Biol. 15, 523 (2014).
Kim, Y. H. et al. Combined microarray analysis of small cell lung cancer reveals altered apoptotic balance and distinct expression signatures of MYC family gene amplification. Oncogene 25, 130–138 (2006).
Acknowledgements
This work was supported by grants from the European Research council (AuroMYC), the German Cancer Aid (111300), the Federal Ministry of Education and Research (SYSMED) and the German Research Foundation (WO 2108/1-1). We thank J. Dirks and M. Brockmann for initial experiments on USP11.
Reviewer information
Nature thanks Bruno Amati, Ashok Venkitaraman and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
S.H., J. Kalb, G.B. and A.C. performed most experiments, D.S. and C.K. performed PLA assays, S.R. performed replication assays, C.S.-V. performed immunofluorescence experiments. G.B. performed DNA–RNA immunoprecipitation and global run-on sequencing analyses, A.B. performed 4-thiouridine-sequencing analyses, J.X. and C.P.A. performed shRNA screening, S.W. analysed ChIP–seq and RNA-sequencing data, A.B., M.E. and C.P.A. analysed additional high-throughput data, J. Koster analysed methylation status in neuroblastomas, M.D., E.W., J.M., R.V., S.H. and M.E. devised and supervised experiments, and S.H. and M.E. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Characterization of SH-EP MYCN–ER cells.
a, Top, heat map showing the 400 most-differentially expressed genes between 498 low- and high-grade neuroblastomas (GSE62564). MYCN amplification status of the tumours and survival of the patients is indicated by the horizontal bars on top and the right panel illustrates gene expression changes after MYCN–ER activation (3 h 4-OHT), in SH-EP cells, of the same genes. Bottom, correlation between relative gene expression in tumours and changes in response to MYCN–ER activation. FC, fold change; NB, neuroblastoma. b, Box plots showing expression of 294 MYCN–ER-activated genes (SH-EP MYCN–ER, false-discovery rate (FDR) < 0.01 and log2((fold change in expression of 4-OHT compared to ethanol) > 0 in n = 3 biological replicates) in neuroblastomas of the indicated tumour stage with or without MYCN amplification (GSE62564). The number of tumour samples is indicated at the bottom and P values were calculated using a two-tailed Wilcoxon rank-sum test. c, Expression of selected gene sets from gene set enrichment analysis in SH-EP cells after MYCN–ER activation (3 h 4-OHT, n = 3) and in 65 MYCN-amplified versus 116 non-amplified stage 4 neuroblastomas (GSE62564). P values were calculated using a Kolmogorov–Smirnov test with 1,000 permutations and corrected for multiple testing using Benjamini–Hochberg procedure (FDR)49. NES, normalized enrichment score. d, Box plot illustrating MYCN binding to promoters (−30 to +300 bp relative to TSS) of 914 MYCN-activated and 615 repressed genes (n = 3). As control, a randomly selected group of 1,000 non-regulated expressed genes was chosen. e, Box plot illustrating mRNA levels in gene groups described above (activated, repressed and non-regulated; n = 3). f, Box plot showing the exon/intron ratio for 27,369 genes in 4sU-seq data (n = 6) compared to RNA-sequencing data (n = 6). b, d–f, In the box plots, the central line reflects the median and the borders of the boxes show the interquartile range of the plotted data. The whiskers extend to 1.5× the interquartile range and outliers are shown as dots.
Extended Data Fig. 2 Effects of MYCN on RNAPII function.
a, Metagene plots of total RNAPII (top) and RNAPII(pSer2) (bottom) in SH-EP MYCN–ER cells after 4-OHT treatment (3 h) for 14,488 expressed genes (n = 4). Data are mean ± s.e.m. b, Empirical cumulative distribution function of RNAPII travelling ratio after MYCN–ER activation (4-OHT) of 14,488 expressed genes (n = 4). c, Two-dimensional kernel density plot of total RNAPII occupancy at the TSS of 14,945 expressed genes in SH-EP MYCN–ER cells treated with ethanol (top) or after activation of MYCN (bottom). Samples are normalized either to sequencing depth or using a mouse spike-in (n = 1). r, Pearson’s correlation coefficient. d, Metagene plots of total RNAPII (top) and RNAPII(pSer2) (bottom) in SH-EP MYCN–ER cells after 4-OHT treatment (3 h; n = 4) of 1,000 non-regulated genes. Data are mean ± s.e.m.
Extended Data Fig. 3 Characterization of BRCA1 function in neuroblastoma.
a, Schematic overview of the shRNA screen in SH-EP MYCN–ER cells. Samples were analysed in duplicates after 14 days of cell culture. b, Waterfall plot (left) visualizing the depletion of the six screened shRNAs targeting BRCA1 in SH-EP MYCN–ER cells with activated MYCN (Z-score for the log2(fold change in expression of 4-OHT compared to start)). From all 12,931 individual shRNA, only the 1,000 shRNAs that were most strongly depleted in the screen following 4-OHT treatment are shown. Z-scores for the depletion of all individual shRNAs were calculated on the basis of the population mean and s.d. of the log2(fold change in expression) of all screened shRNAs (n = 2). Box plot (right) comparing depletion of all shRNAs targeting BRCA1 in SH-EP MYCN–ER cells upon activation of MYCN. The median and the lower and upper quartiles are shown, of the log2(fold change in expression of 4-OHT compared to start and ethanol compared to start) for n = 6 independent shRNAs targeting BRCA1 mRNA. Whiskers extend to 1.5× interquartile range above and below the upper and lower quartiles, respectively. c, Immunoblot (left) of BRCA1 in SH-EP MYCN–ER and in MYCN-amplified IMR5 and SMS-KAN cells showing the knockdown of BRCA1 by two shRNAs. Note the high BRCA1 levels in MYCN-amplified neuroblastoma cell lines (IMR5 and SMS-KAN) relative to the non-MYCN-amplified cell line (SH-EP). The arrow points to the BRCA1 band, asterisks denote unspecific bands. Vinculin was used as loading control. For all gel source data, see Supplementary Fig. 1. qPCR (right) of BRCA1 mRNA levels in BRCA1-depleted SH-EP MYCN–ER cells. Data are mean of technical triplicates (n = 3). d, Clonogenic assay in SH-EP MYCN–ER cells after shRNA-mediated knockdown of BRCA1 and induction of MYCN for six days. Colonies were stained with crystal violet (n = 3). e, Expression of a gene set of the 99 genes, identified in the shRNA screen, in patients with primary neuroblastoma. Each patient is ranked using a defined gene set of MYCN-amplified tumours52. MYCN amplification status of all 498 patients is indicated on the right. f, BRCA1 gene expression in 498 neuroblastoma samples (GSE62564). Tumours are sorted on the basis of BRCA1 expression. g, Survival of 498 patients with neuroblastoma (GSE62564) stratified by BRCA1 expression. Data were obtained from GEO and the R2 platform was used for grouping tumour samples by scanning mode based on BRCA1 expression. The q value reflects a Bonferroni-corrected P value (log-rank test). h, BRCA1 genomic region around the TSS with average methylation status in high- versus low-risk neuroblastoma.
Extended Data Fig. 4 Cell-cycle progression and DNA replication in SH-EP MYCN–ER cells.
a, Quantification (top) and representative FACS profiles (bottom) documenting cell-cycle distribution of BrdU/propidium iodide (PI)-stained control and BRCA1-deficient cells treated with BRCA1 shRNA 2 (shBRCA1#2) after 6 h of 4-OHT treatment. Propidium iodide staining was used for quantification. Data are mean + s.d. of biological triplicates. b, Same experimental set-up as in a, but after 48 h 4-OHT treatment. c, Percentage of apoptotic cells in control and BRCA1-deficient cells treated with shBRCA1#2 measured in a propidium iodide/annexin V FACS experiment after treatment with 4-OHT for 48 h. Data are mean + s.d. of biological triplicates. P values were calculated using an unpaired, two-tailed t-test. d, Top, fork progression rates during both labels based on the track length under the indicated conditions in control and BRCA1-deficient cells for BRCA1 shRNA 1 (shBRCA1#1). The number of analysed DNA fibres is as follows: scramble shRNA (shSCR): ethanol, n = 130, 4-OHT n = 131; shBRCA1#1: ethanol, n = 161, 4-OHT, n = 139. P values were calculated using a two-tailed, unpaired t-test with additional Welch’s correction. One representative experiment is shown (n = 3). Bottom, schematic of DNA fibre experiment labelled with CldU (red) and IdU (blue). e, Same experimental set-up as in d with shBRCA1#2. The number of DNA fibres is as follows: shSCR: ethanol, n = 95, 4-OHT, n = 69; shBRCA1#2: ethanol, n = 109, 4-OHT, n = 90. P values were calculated using a two-tailed, unpaired t-test with additional Welch’s correction. One representative experiment is shown (n = 3). d, e, Fork progressions are displayed as box plots with the central line reflecting the median; the borders of the boxes show the lower and upper quartile of the plotted data, with 10th–90th-percentile whiskers and outliers are shown as dots. f, Number of γH2A.X (left) and 53BP1 (right) foci per well/number of cells per well, indicating DNA damage in control and BRCA1-deficient cells treated with shBRCA1#2 after 24 h of 4-OHT treatment. Etoposide (Etop; 25 μM) was used as a positive control and added for the final 2 h. Data are mean + s.d. of biological triplicates. P values were calculated using an unpaired, two-tailed t-test. One representative experiment is shown (n = 3).
Extended Data Fig. 5 Control experiments for BRCA1 recruitment.
a, Venn diagram documenting genome-wide overlap between BRCA1 and MYCN peaks. b, Relative E-box (CACGTG) frequency around BRCA1 peaks in promoter regions (±1 kb relative to the TSS). The curve is smoothed using a sliding window of 50 bp. c, Enriched DNA motifs in 12,161 BRCA1 peaks located in the promoter (±1 kb relative to the TSS) identified by de novo motif search. A region of ±50 bp around the BRCA1 peak summit was analysed and similarity to known motifs was assigned with TOMTOM and the JASPAR vertebrate motif database. E values are calculated with Fisher’s exact test corrected for the number of input sequences, and q values for the comparison to known motifs are FDR-corrected P values calculated with a null model based on sampling motif columns from all of the columns in the set of target motifs42. The top three motifs from DREME analysis are shown (n = 3). d, ChIP of BRCA1 from SH-EP MYCN–ER cells transfected either with a control siRNA (siCTR) or siRNA targeting BRCA1. Selected promoters have both a robust MYCN and an overlapping BRCA1 peak. IgG was used as control. Where indicated, 4-OHT or ethanol was added for 5 h. Data are mean + s.d. of technical triplicates (n = 1). e, ChIP of BRCA1 from SH-EP MYCN–ER cells that stably express either a control shRNA or shRNA targeting BRCA1. IgG was used as control. The experiment was carried out as in d. Data are mean + s.d. of technical triplicates (n = 1). f, ChIP of BRCA1 from SH-EP MYCN–ER cells using two different BRCA1 antibodies. 4-OHT was added for 5 h. Data are mean + s.d. of the enrichment over IgG (of technical triplicates) (n = 1). g, Browser track of the LDHA locus of a BRCA1 and MYCN ChIP–seq experiment documenting BRCA1 binding to the LDHA promoter in SH-EP MYCN–ER cells after MYCN activation (5 h), and in SH-EP cells that express ectopic MYCN or empty vector as control. For ectopically expressing cells, the MYCN browser track is shown. h, Heat map showing occupancy of BRCA1 and MYCN in SH-EP MYCN–ER cells and SH-EP cells that express ectopic MYCN or empty vector as a control. Plot is centred to TSS. i, ChIP of BRCA1 from control SH-EP and from SH-EP MYCN–ER cells treated with either 4-OHT or ethanol (5 h). Data are mean + s.d. of technical triplicates of one representative experiment (n = 2).
Extended Data Fig. 6 BRCA1 recruitment to paused RNAPII.
a, Left, ChIP of BRCA1 at the indicated loci upon treatment of SH-EP MYCN–ER cells with 4-OHT (5 h) and flavopiridol (100 nM, 3 h), where indicated. Data are mean + s.d. of technical triplicates of one representative experiment (n = 2). Right, immunoblot of cells treated as described. Asterisk denotes an unspecific band. Vinculin was used as loading control (n = 1). b, Density plots of BRCA1 occupancy in 6,887 intergenic regions after treatment with either 4-OHT or ethanol (5 h) in DMSO-treated cells (left) and flavopiridol-treated cells (100 nM, 3 h) (right). Data are mean ± s.e.m. (n = 1). c, Left, ChIP of BRCA1 in SH-EP MYCN–ER cells treated with CDK7 inhibitor THZ1 (200 nM, 4 h) or DMSO as control together with 4-OHT or ethanol. Data are mean + s.d. of technical triplicates. Right, immunoblots for RNAPII(pSer5), RNAPII(pSer2) and total RNAPII after treatment as described above. Actin was used as loading control (n = 1). d, Left, ChIP of BRCA1 at the indicated loci in MYCN-amplified IMR-5 neuroblastoma cells that express a doxycycline (DOX)-inducible shRNA targeting MYCN. Where indicated, cells were treated with flavopiridol (200 nM, 4 h) or doxycycline (1 μg ml−1, 48 h). Data are mean + s.d. of technical triplicates of one representative experiment (n = 2). Right, immunoblot of cells treated as described above. Asterisks denote unspecific bands. CDK2 was used as loading control (n = 2).
Extended Data Fig. 7 Effect of BRCA1 on MYCN-dependent RNAPII function.
a, Metagene plots of total RNAPII after 4-OHT treatment (3 h) in control (top) and BRCA1-depleted cells treated with shBRCA1#1 (bottom) on 14,488 expressed genes. Data are mean ± s.e.m. b, Immunoblot of total RNAPII (RBP1) and phosphorylated forms after knockdown of BRCA1 and activation of MYCN. One representative experiment is shown (n = 2). The arrow points to the BRCA1 band, asterisks denote unspecific bands. c, ChIP of NELF-E in control and BRCA1-depleted SH-EP MYCN–ER cells after 4-OHT treatment (4 h). Data are mean + s.d. of technical triplicates of one representative experiment (n = 3 using two different shRNAs). d, Empirical cumulative distribution function of RNAPII travelling ratio after MYCN–ER activation (3 h) in control (top) and BRCA1-depleted (bottom) cells treated with shBRCA1#2 of 14,488 expressed genes. e, Metagene plots (of RNAPII(pSer2) in control (top) and BRCA1-depleted (bottom) cells treated with shBRCA1#2 upon treatment as described above for 14,488 expressed genes. Data are mean ± s.e.m. f, Gene set enrichment analysis upon activation of MYCN (5 h) in BRCA1-depleted and control conditions (n = 3). Significantly enriched gene sets are highlighted in black (FDR q < 0.25), MYC-activated gene sets are marked in red and MYC-repressed gene sets are marked in blue. FDR was calculated using a Kolmogorov–Smirnov test with 1,000 permutations using a Benjamini–Hochberg correction for multiple testing. g, Two-dimensional kernel density plot correlating gene expression changes after MYCN–ER activation (5 h) in BRCA1-depleted and control cells. Results are shown for two shRNAs of 19,429 expressed genes. r, Pearson’s correlation coefficient.
Extended Data Fig. 8 BRCA1 promotes R-loop resolution and mRNA decapping.
a, DRIP using the S9.6 antibody, indicating R-loops at known loci within the ACTB gene. Digestion with RNase H1 was used as control for specificity of the antibody and IgG as control for unspecific chromatin binding. Data are mean + s.d. of technical triplicates of one representative experiment (n = 2). b, DRIP documenting binding of the S9.6 R-loop antibody to the indicated loci upon depletion of BRCA1 and activation of MYCN for 4 h. Data are mean + s.d. of technical triplicates of one representative experiment (n = 4). The panel shows the non-normalized data of Fig. 3d. c, ChIP of senataxin at the indicated loci in SH-EP MYCN–ER cells after 4-OHT treatment (3 h) in control or BRCA1-depleted cells. Data are mean + s.d. of technical triplicates of one representative experiment (n = 2 with 2 different antibodies). d, RNAPII density around the first downstream polyA site in SH-EP MYCN–ER cells after BRCA1 depletion using shBRCA1#2 and MYCN activation for 3 h: 1,713 genes are shown. Data are mean ± s.e.m. One representative experiment is shown (n = 3). e, Heat map of 8,077 TSSs of genes with an RNAPII peak downstream of the start site. Reads originate from GRO–seq, mRNA sequencing and total RNAPII ChIP–seq samples and genes are sorted on the basis of the distance from the TSS to the RNAPII peak.
Extended Data Fig. 9 Characterization of BRCA1 recruitment and of the interaction of USP11 with MYCN.
a, ChIP of BRCA1 in SH-EP MYCN–ER cells synchronized by a double thymidine block. Cells were released and collected during the indicated cell-cycle phase after 4 h of 4-OHT treatment. IgG was used as control. Data are mean + s.d. of technical triplicates (n = 1). b, Bar plot summarizing the PLAs of MYCN and BRCA1 in G1, S and G2 phases in SH-EP MYCN–ER cells synchronized as described in a and upon 3 h 4-OHT treatment. Data are mean + s.d. of biological triplicates. For each cell-cycle phase, between 74 and 276 cells were counted. P values were calculated using an unpaired, two-tailed t-test (n = 1). c, Representative pictures of the PLA from b showing proximity of MYCN and BRCA1 in G1, S and G2 phases (green dots). Nuclei were stained with Hoechst, cytoskeleton was stained using phalloidin indicated in violet (n = 1). d, Representative FACS profiles of propidium-iodide-stained SH-EP MYCN–ER cells used for the experiment in a (n = 1). e, Immunoblot of anti-MYCN immunoprecipitates from IMR-5 MYCN-amplified neuroblastoma cells. The input corresponds to 0.75% of the amount used for the precipitation. One representative experiment is shown (n = 2). f, Immunoblot of indicated proteins after knockdown of USP11 in SMS-KAN cells and treatment with MG-132 (10 μM, 6 h), where indicated. Vinculin was used as loading control (n = 1). g, Extracted ion chromatogram of MYCN phosphorylation status from SH-EP cells that express ectopic MYCN. Values in parentheses indicate m/z ratio for each peak and the normalized target level (NL) is given for each chromatogram (n = 1). h, Immunoblot of anti-USP11 immunoprecipitates from SH-EP cells that stably express wild-type MYCN (wt), Thr58Ala (TA) or Ser62Ala (SA) mutants of MYCN. Asterisk denotes an unspecific band. One representative experiment is shown (n = 3). i, Immunoblot documenting phosphorylation status of the indicated alleles of MYCN. Actin was used as loading control. One representative experiment is shown (n = 2). j, Left, quantification of a PLA that illustrates complex formation of BRCA1 with MYCN in SH-EP cells that express ectopic MYCN or empty vector as control. Between 749 and 1,854 cells were counted for each condition. P values were calculated with a two-tailed Wilcoxon rank-sum test. In the box plot, the central line reflects the median and the borders of the boxes show the interquartile range of the plotted data. The whiskers extend to 1.5× the interquartile range, and outliers are shown as dots. Right, quantification of the corresponding immunofluorescence (IF) signals of the indicated antibodies (n = 1).
Extended Data Fig. 10 Role of DNA damage and of MYC in recruitment of BRCA1.
a, ChIP of BRCA1 in SH-EP MYCN–ER cells that were pre-treated with ATR inhibitor VE-821 (1 μM, 2 h), 4-OHT or ethanol was added for 4 h. Data are mean + s.d. of technical triplicates of one representative experiment (n = 2). b, Immunoblot of CHK1(pSer345) after treatment with ATR inhibitor VE-821, as described in a. Cells were treated with HU (5 mM) for 4 h as positive control. Asterisk denotes an unspecific band. Vinculin was used as loading control. One representative experiment is shown (n = 2). c, ChIP of BRCA1 in SH-EP MYCN–ER cells at the indicated loci after treatment (9 h) with olaparib (10 μM), talazoparib (1 μM) or DMSO as control. Data are mean + s.d. of technical triplicates of one representative experiment (n = 3). d, Immunoblot of ATM(pSer1981) after treatment with etoposide (5 μM, 3 h). Actin was used as loading control (n = 1). e, Density plots of BRCA1 occupancy after treatment with either 4-OHT or ethanol (5 h) in DMSO (left) and etoposide-treated (right) SH-EP MYCN–ER cells. f, Immunoblots of anti-USP11 immunoprecipitates from either control SH-EP cells or cells transiently transfected with MYC. CDK2 was used as loading control, asterisks denote unspecific bands. The input corresponds to 2% of the amount used for the precipitation. One representative experiment is shown (n = 3). g, Left, ChIP of BRCA1 at the indicated loci in SH-EP MYCN–ER or SH-EP MYC–ER cells treated with 4-OHT (5 h) or ethanol. Data are mean + s.d. of technical triplicates of one representative experiment (n = 2). Right, immunoblot performed with the ER antibody to detect MYCN–ER and MYC–ER in SH-EP, SH-EP MYCN–ER or SH-EP MYC–ER cells treated with 4-OHT or ethanol. CDK2 was used as loading control (n = 2). h, Model illustrating our findings. We propose that BRCA1 is recruited jointly by MYCN and Ser5-phosphorylated RNAPII. BRCA1 recruitment is strongly enhanced by blockade of CDK9, which blocks pause release, and by increasing torsional stress using etoposide, which suggests that it is predominantly a stress response to stalling of RNAPII. BRCA1, in turn, is required to prevent an MYCN-dependent accumulation of stalling RNAPII and R-loop formation via a mRNA-decapping complex. The critical signal in MYCN that enables recruitment of BRCA1 is the dephosphorylation of Thr58, a residue that—when phosphorylated—is recognized by FBXW7 and promotes turnover of MYCN. Dephosphorylation of Thr58 allows binding of USP11, which stabilizes MYCN and BRCA1 on chromatin, which suggests that a kinetic competition between MYCN turnover and dephosphorylation/deubiquitination controls the fate of RNAPII at promoters.
Supplementary information
Supplementary Figure 1
This figure contains the uncropped images of all immunoblots. We include pictures of the molecular weight standards that were used to cut each gel before incubation with the primary antibody. Because of their similar molecular weight, RNAPII/BRCA1 and MYCN/Tubulin are not blotted from the same membrane. Where indicated, loading control or inputs were reloaded. Molecular weights are indicated if a weight marker lights up with a secondary antibody.
Supplementary Table 1
Hits identified in the shRNA screen. This table summarizes the hits from the shRNA screen. Shown in green are shRNAs that were determined as “synthetic lethal” and defined as screening hits (104 shRNAs). For each gene targeted by a “synthetic lethal” shRNA (100 genes), all other recovered shRNAs targeting the same gene are shown in grey.
Supplementary Table 2
A list of all reagents and software used in the study.
Source data
Rights and permissions
About this article
Cite this article
Herold, S., Kalb, J., Büchel, G. et al. Recruitment of BRCA1 limits MYCN-driven accumulation of stalled RNA polymerase. Nature 567, 545–549 (2019). https://doi.org/10.1038/s41586-019-1030-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1030-9
- Springer Nature Limited
This article is cited by
-
Network pharmacology and experimental validation to study the potential mechanism of Tongguanteng injection in regulating apoptosis in osteosarcoma
BMC Complementary Medicine and Therapies (2024)
-
CDK12 controls transcription at damaged genes and prevents MYC-induced transcription-replication conflicts
Nature Communications (2024)
-
Recent advances in anti-tumor therapeutic approaches for the universally active transcriptional factor c-MYC
Proceedings of the Indian National Science Academy (2024)
-
Looping out of control: R-loops in transcription-replication conflict
Chromosoma (2024)
-
Sources, resolution and physiological relevance of R-loops and RNA–DNA hybrids
Nature Reviews Molecular Cell Biology (2022)