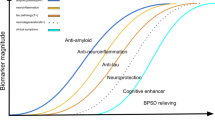
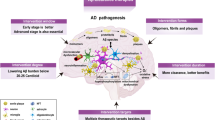
Abstract
Many drugs that target amyloid-β (Aβ) in Alzheimer disease (AD) have failed to demonstrate clinical efficacy. However, four anti-Aβ antibodies have been shown to mediate the removal of amyloid plaque from brains of patients with AD, and the FDA has recently granted accelerated approval to one of these, aducanumab, using reduction of amyloid plaque as a surrogate end point. The rationale for approval and the extent of the clinical benefit from these antibodies are under intense debate. With the aim of informing this debate, we review clinical trial data for drugs that target Aβ from the perspective of the temporal interplay between the two pathognomonic protein aggregates in AD — Aβ plaques and tau neurofibrillary tangles — and their relationship to cognitive impairment, highlighting differences in drug properties that could affect their clinical performance. On this basis, we propose that Aβ pathology drives tau pathology, that amyloid plaque would need to be reduced to a low level (~20 centiloids) to reveal significant clinical benefit and that there will be a lag between the removal of amyloid and the potential to observe a clinical benefit. We conclude that the speed of amyloid removal from the brain by a potential therapy will be important in demonstrating clinical benefit in the context of a clinical trial.




Similar content being viewed by others
References
Karran, E., Mercken, M. & De Strooper, B. The amyloid cascade hypothesis for Alzheimer’s disease: an appraisal for the development of therapeutics. Nat. Rev. Drug Discov. 10, 698–712 (2011).
Goate, A. et al. Segregation of a missense mutation in the amyloid precursor protein gene with familial Alzheimer’s disease. Nature 349, 704–706 (1991).
Sherrington, R. et al. Cloning of a gene bearing missense mutations in early-onset familial Alzheimer’s disease. Nature 375, 754–760 (1995).
Rogaev, E. I. et al. Familial Alzheimer’s disease in kindreds with missense mutations in a gene on chromosome 1 related to the Alzheimer’s disease type 3 gene. Nature 376, 775–778 (1995).
Jonsson, T. et al. A mutation in APP protects against Alzheimer’s disease and age-related cognitive decline. Nature 488, 96–99 (2012).
Morris, J. C. The Clinical Dementia Rating (CDR): current version and scoring rules. Neurology 43, 2412–2414 (1993).
Knopman, D. S., Jones, D. T. & Greicius, M. D. Failure to demonstrate efficacy of aducanumab: an analysis of the EMERGE and ENGAGE trials as reported by Biogen, December 2019. Alzheimers Dementia 17, 696–701 (2021).
US Food and Drug Administration. Drug approval package: Aduhelm (aducanumab-avwa). Office of Neurology’s summary review memorandum. FDA https://www.accessdata.fda.gov/drugsatfda_docs/nda/2021/761178Orig1s000TOC.cfm (2021).
Alexander, G. C. et al. Revisiting FDA approval of aducanumab. N. Engl. J. Med. 385, 769–771 (2021).
Dalton, R. M., Krishnan, H. S., Parker, V. S., Catanese, M. C. & Hooker, J. M. Coevolution of atomic resolution and whole-brain imaging for tau neurofibrillary tangles. ACS Chem. Neurosci. 11, 2513–2522 (2020).
Leuzy, A. et al. Tau PET imaging in neurodegenerative tauopathies-still a challenge. Mol. Psychiatry 24, 1112–1134 (2019).
Villemagne, V. L., Dore, V., Burnham, S. C., Masters, C. L. & Rowe, C. C. Imaging tau and amyloid-beta proteinopathies in Alzheimer disease and other conditions. Nat. Rev. Neurol. 14, 225–236 (2018).
Jagust, W. Imaging the evolution and pathophysiology of Alzheimer disease. Nat. Rev. Neurosci. 19, 687–700 (2018).
Thal, D. R., Rub, U., Orantes, M. & Braak, H. Phases of A beta-deposition in the human brain and its relevance for the development of AD. Neurology 58, 1791–1800 (2002).
Grothe, M. J. et al. In vivo staging of regional amyloid deposition. Neurology 89, 2031–2038 (2017).
Mattsson, N., Palmqvist, S., Stomrud, E., Vogel, J. & Hansson, O. Staging beta-amyloid pathology with amyloid positron emission tomography. JAMA Neurol. 76, 1319–1329 (2019).
Jansen, W. J. et al. Prevalence of cerebral amyloid pathology in persons without dementia: a meta-analysis. JAMA 313, 1924–1938 (2015).
Braak, H. & Braak, E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 82, 239–259 (1991).
Crary, J. F. et al. Primary age-related tauopathy (PART): a common pathology associated with human aging. Acta Neuropathol. 128, 755–766 (2014).
Lowe, V. J. et al. Cross-sectional associations of tau-PET signal with cognition in cognitively unimpaired adults. Neurology 93, e29–e39 (2019).
Pontecorvo, M. J. et al. Relationships between flortaucipir PET tau binding and amyloid burden, clinical diagnosis, age and cognition. Brain 140, 748–763 (2017).
De Strooper, B. & Karran, E. The cellular phase of Alzheimer’s disease. Cell 164, 603–615 (2016).
Villemagne, V. L. et al. Amyloid β deposition, neurodegeneration, and cognitive decline in sporadic Alzheimer’s disease: a prospective cohort study. Lancet Neurol. 12, 357–367 (2013).
Mattsson-Carlgren, N. et al. The implications of different approaches to define AT(N) in Alzheimer disease. Neurology 94, e2233–e2244 (2020).
Pontecorvo, M. J. et al. A multicentre longitudinal study of flortaucipir (18F) in normal ageing, mild cognitive impairment and Alzheimer’s disease dementia. Brain 142, 1723–1735 (2019).
Knopman, D. S. et al. Association of initial beta-amyloid levels with subsequent flortaucipir positron emission tomography changes in persons without cognitive impairment. JAMA Neurol. 78, 217–228 (2021).
Sanchez, J. S. et al. The cortical origin and initial spread of medial temporal tauopathy in Alzheimer’s disease assessed with positron emission tomography. Sci. Transl. Med. 13, eabc0655 (2021).
Hong, W. et al. Diffusible, highly bioactive oligomers represent a critical minority of soluble Abeta in Alzheimer’s disease brain. Acta Neuropathol. 136, 19–40 (2018).
Walsh, D. M. & Selkoe, D. J. Amyloid β-protein and beyond: the path forward in Alzheimer’s disease. Curr. Opin. Neurobiol. 61, 116–124 (2020).
Benilova, I., Karran, E. & De Strooper, B. The toxic Abeta oligomer and Alzheimer’s disease: an emperor in need of clothes. Nat. Neurosci. 15, 349–357 (2012).
Karran, E. & Hardy, J. A critique of the drug discovery and phase 3 clinical programs targeting the amyloid hypothesis for Alzheimer disease. Ann. Neurol. 76, 185–205 (2014).
Doody, R. S. et al. A phase 3 trial of semagacestat for treatment of Alzheimer’s disease. N. Engl. J. Med. 369, 341–350 (2013).
Bateman, R. J. et al. A gamma-secretase inhibitor decreases amyloid-beta production in the central nervous system. Ann. Neurol. 66, 48–54 (2009).
De Strooper, B. Lessons from a failed gamma-secretase Alzheimer trial. Cell 159, 721–726 (2014).
Bray, S. J. Notch signalling in context. Nat. Rev. Mol. Cell Biol. 17, 722–735 (2016).
Doody, R. S. et al. Peripheral and central effects of gamma-secretase inhibition by semagacestat in Alzheimer’s disease. Alzheimers Res. Ther. 7, 36 (2015).
Egan, M. F. et al. Randomized trial of verubecestat for prodromal Alzheimer’s disease. N. Engl. J. Med. 380, 1408–1420 (2019).
Egan, M. F. et al. Randomized trial of verubecestat for mild-to-moderate Alzheimer’s disease. N. Engl. J. Med. 378, 1691–1703 (2018).
Hampel, H. et al. The β-secretase BACE1 in Alzheimer’s disease. Biol. Psychiatry 89, 745–756 (2021).
Racke, M. M. et al. Exacerbation of cerebral amyloid angiopathy-associated microhemorrhage in amyloid precursor protein transgenic mice by immunotherapy is dependent on antibody recognition of deposited forms of amyloid beta. J. Neurosci. 25, 629–636 (2005).
Doody, R. S. et al. Phase 3 trials of solanezumab for mild-to-moderate Alzheimer’s disease. N. Engl. J. Med. 370, 311–321 (2014).
Honig, L. S. et al. Trial of solanezumab for mild dementia due to Alzheimer’s disease. N. Engl. J. Med. 378, 321–330 (2018).
Siemers, E. R. et al. Phase 3 solanezumab trials: secondary outcomes in mild Alzheimer’s disease patients. Alzheimers Dement. 12, 110–120 (2016).
Bateman, R. J. et al. The DIAN-TU next generation Alzheimer’s prevention trial: adaptive design and disease progression model. Alzheimers Dement. 13, 8–19 (2017).
Wang, G. et al. A novel cognitive disease progression model for clinical trials in autosomal-dominant Alzheimer’s disease. Stat. Med. 37, 3047–3055 (2018).
Salloway, S. et al. A trial of gantenerumab or solanezumab in dominantly inherited Alzheimer’s disease. Nat. Med. 27, 1187–1196 (2021).
Ultsch, M. et al. Structure of crenezumab complex with Abeta shows loss of beta-hairpin. Sci. Rep. 6, 39374 (2016).
Farlow, M. et al. Safety and biomarker effects of solanezumab in patients with Alzheimer’s disease. Alzheimers Dement. 8, 261–271 (2012).
Yoshida, K. et al. Pharmacokinetics and pharmacodynamic effect of crenezumab on plasma and cerebrospinal fluid beta-amyloid in patients with mild-to-moderate Alzheimer’s disease. Alzheimers Res. Ther. 12, 16 (2020).
Meilandt, W. J. et al. Characterization of the selective in vitro and in vivo binding properties of crenezumab to oligomeric Aβ. Alzheimers Res. Ther. 11, 97 (2019).
Salloway, S. et al. Amyloid positron emission tomography and cerebrospinal fluid results from a crenezumab anti-amyloid-beta antibody double-blind, placebo-controlled, randomized phase II study in mild-to-moderate Alzheimer’s disease (BLAZE). Alzheimers Res. Ther. 10, 96 (2018).
Yang, T. et al. Target engagement in an alzheimer trial: crenezumab lowers amyloid β oligomers in cerebrospinal fluid. Ann. Neurol. 86, 215–224 (2019).
Brothers, H. M., Gosztyla, M. L. & Robinson, S. R. The physiological roles of amyloid-β peptide hint at new ways to treat Alzheimer’s disease. Front. Aging Neurosci. 10, 118 (2018).
Bard, F. et al. Peripherally administered antibodies against amyloid beta-peptide enter the central nervous system and reduce pathology in a mouse model of Alzheimer disease. Nat. Med. 6, 916–919 (2000).
Demattos, R. B. et al. A plaque-specific antibody clears existing beta-amyloid plaques in Alzheimer’s disease mice. Neuron 76, 908–920 (2012).
Sperling, R. A. et al. Amyloid-related imaging abnormalities in amyloid-modifying therapeutic trials: recommendations from the Alzheimer’s Association Research Roundtable Workgroup. Alzheimers Dement. 7, 367–385 (2011).
Rosen, W. G., Mohs, R. & Davis, K. L. A new rating scale for Alzheimer’s disease. Am. J. Psychiatry 141, 1356–1364 (1984).
Gélinas, I., Gauthier, L., Mcintyre, M. & Gauthier, S. Development of a functional measure for persons with Alzheimer’s disease: the disability assessment for dementia. Am. J. Occup. Ther. 53, 471–481 (1999).
Salloway, S. et al. Two phase 3 trials of bapineuzumab in mild-to-moderate Alzheimer’s disease. N. Engl. J. Med. 370, 322–333 (2014).
Liu, E. et al. Amyloid-β 11C-PiB-PET imaging results from 2 randomized bapineuzumab phase 3 AD trials. Neurology 85, 692–700 (2015).
Bohrmann, B. et al. Gantenerumab: a novel human anti-Abeta antibody demonstrates sustained cerebral amyloid-beta binding and elicits cell-mediated removal of human amyloid-beta. J. Alzheimers Dis. 28, 49–69 (2012).
Ostrowitzki, S. et al. Mechanism of amyloid removal in patients with Alzheimer disease treated with gantenerumab. Arch. Neurol. 69, 198–207 (2012).
Ostrowitzki, S. et al. A phase III randomized trial of gantenerumab in prodromal Alzheimer’s disease. Alzheimers Res. Ther. 9, 95 (2017).
Nilsberth, C. et al. The ‘Arctic’ APP mutation (E693G) causes Alzheimer’s disease by enhanced Aβ protofibril formation. Nat. Neurosci. 4, 887–893 (2001).
Logovinsky, V. et al. Safety and tolerability of BAN2401–a clinical study in Alzheimer’s disease with a protofibril selective Abeta antibody. Alzheimers Res. Ther. 8, 14 (2016).
Satlin, A. et al. Design of a Bayesian adaptive phase 2 proof-of-concept trial for BAN2401, a putative disease-modifying monoclonal antibody for the treatment of Alzheimer’s disease. Alzheimers Dement. 2, 1–12 (2016).
Wang, J. et al. ADCOMS: a composite clinical outcome for prodromal Alzheimer’s disease trials. J. Neurol. Neurosurg. Psychiatry 87, 993–999 (2016).
Qian, J., Betensky, R. A., Hyman, B. T. & Serrano-Pozo, A. Association of APOE genotype with heterogeneity of cognitive decline rate in Alzheimer disease. Neurology 96, e2414–e2428 (2021).
Suzuki, K. et al. Effect of apolipoprotein E epsilon4 allele on the progression of cognitive decline in the early stage of Alzheimer’s disease. Alzheimers Dement. 6, e12007 (2020).
Swanson, C. J. et al. A randomized, double-blind, phase 2b proof-of-concept clinical trial in early Alzheimer’s disease with lecanemab, an anti-Abeta protofibril antibody. Alzheimers Res. Ther. 13, 80 (2021).
Reyderman, L. et al. BAN2401 and ARIA-E in early Alzheimer’s disease: pharmacokinetic / pharmacodynamic time-to-event analysis from the phase 2 study in early Alzheimer’s disease. J. Prev. Alzheimer’s Dis. 7, S17 (2020).
Aisen, P. et al. AHEAD 3-45 study design: a global study to evaluate the efficacy and safety of treatment with BAN2401 for 216 weeks in preclinical Alzheimer’s disease with intermediate amyloid (A3 trial) and elevated amyloid (A45 trial). Alzheimers Dement. 16 (Suppl. 9), e044511 (2020).
Lord, A. et al. An amyloid-beta protofibril-selective antibody prevents amyloid formation in a mouse model of Alzheimer’s disease. Neurobiol. Dis. 36, 425–434 (2009).
Arndt, J. W. et al. Structural and kinetic basis for the selectivity of aducanumab for aggregated forms of amyloid-beta. Sci. Rep. 8, 6412 (2018).
Sevigny, J. et al. The antibody aducanumab reduces Abeta plaques in Alzheimer’s disease. Nature 537, 50–56 (2016).
Budd Haeberlein, S. et al. Evaluation of aducanumab efficacy in early Alzheimer’s disease. Presented at 15th International Conference on Alzheimer’s and Parkinson’s Diseases and related neurological disorders (2021).
Saido, T. C. et al. Dominant and differential deposition of distinct beta-amyloid peptide species, A beta N3(pE), in senile plaques. Neuron 14, 457–466 (1995).
Kuo, Y.-M., Emmerling, M. R., Woods, A. S., Cotter, R. J. & Roher, A. E. Isolation, chemical characterization, and quantitation of Aβ 3-pyroglutamyl peptide from neuritic plaques and vascular amyloid deposits. Biochem Biophys Res. Commun. 237, 188–191 (1997).
Lowe, S. L. et al. Donanemab (LY3002813) dose-escalation study in Alzheimer’s disease. Alzheimers Dementia 7, e12112 (2021).
Lobo, E. D., Hansen, R. J. & Balthasar, J. P. Antibody pharmacokinetics and pharmacodynamics. J. Pharm. Sci. 93, 2645–2668 (2004).
Irizarry, M. C. et al. O4-08-06: safety, pharmacokinetics (PK), and florbetapir F-18 positron emission tomography (PET) after multiple dose administration of LY3002813, a β-amyloid plaque-specific antibody, in Alzheimer’s disease (AD). Alzheimers Dement. 12, P352 (2016).
Wessels, A. M. et al. A combined measure of cognition and function for clinical trials: the integrated Alzheimer’s disease rating scale (iADRS). J. Prev. Alzheimers Dis. 2, 227–241 (2015).
Mintun, M. A. et al. Donanemab in early Alzheimer’s disease. N. Engl. J. Med. 384, 1691–1704 (2021).
Clark, C. M. et al. Cerebral PET with florbetapir compared with neuropathology at autopsy for detection of neuritic amyloid-β plaques: a prospective cohort study. Lancet Neurol. 11, 669–678 (2012).
La Joie, R. et al. Multisite study of the relationships between antemortem [(11)C]PIB-PET Centiloid values and postmortem measures of Alzheimer’s disease neuropathology. Alzheimers Dement. 15, 205–216 (2019).
Dore, V. et al. Comparison of (18)F-florbetaben quantification results using the standard centiloid, MR-based, and MR-less CapAIBL((R)) approaches: validation against histopathology. Alzheimers Dement. 15, 807–816 (2019).
Amadoru, S. et al. Comparison of amyloid PET measured in centiloid units with neuropathological findings in Alzheimer’s disease. Alzheimers Res. Ther. 12, 22 (2020).
Jack, C. R. et al. Predicting future rates of tau accumulation on PET. Brain 143, 3136–3150 (2020).
Klein, G. et al. Gantenerumab reduces amyloid-beta plaques in patients with prodromal to moderate Alzheimer’s disease: a PET substudy interim analysis. Alzheimers Res. Ther. 11, 101 (2019).
Boche, D., Denham, N., Holmes, C. & Nicoll, J. A. Neuropathology after active Abeta42 immunotherapy: implications for Alzheimer’s disease pathogenesis. Acta Neuropathol. 120, 369–384 (2010).
Scherpelz, K. P. et al. Atomic-level differences between brain parenchymal- and cerebrovascular-seeded Abeta fibrils. Sci. Rep. 11, 247 (2021).
Maeda, J. et al. Longitudinal, quantitative assessment of amyloid, neuroinflammation, and anti-amyloid treatment in a living mouse model of Alzheimer’s disease enabled by positron emission tomography. J. Neurosci. 27, 10957–10968 (2007).
Higuchi, M. Visualization of brain amyloid and microglial activation in mouse models of Alzheimer’s disease. Curr. Alzheimer Res. 6, 137–143 (2009).
Insel, P. S. et al. Determining clinically meaningful decline in preclinical Alzheimer disease. Neurology 93, e322–e333 (2019).
Donohue, M. C. et al. The preclinical Alzheimer cognitive composite: measuring amyloid-related decline. JAMA Neurol. 71, 961–970 (2014).
Zetterberg, H. & Blennow, K. Moving fluid biomarkers for Alzheimer’s disease from research tools to routine clinical diagnostics. Mol. Neurodegener. 16, 10 (2021).
Alexander, G. C., Emerson, S. & Kesselheim, A. S. Evaluation of aducanumab for Alzheimer disease: scientific evidence and regulatory review involving efficacy, safety, and futility. JAMA 325, 1717–1718 (2021).
Alexander, G. C. & Karlawish, J. The problem of aducanumab for the treatment of Alzheimer disease. Ann. Intern. Med. 174, 1303–1304 (2021).
Cummings, J. et al. Aducanumab produced a clinically meaningful benefit in association with amyloid lowering. Alzheimers Res. Ther. 13, 98 (2021).
Doody, R. Editorial: Consequences of the FDA decision on aducanumab for patient care and research. J. Prev. Alzheimers Dis. 8, 393–395 (2021).
Dunn, B., Stein, P. & Cavazzoni, P. Approval of aducanumab for Alzheimer disease-the FDA’s perspective. JAMA Intern. Med. 181, 1276–1278 (2021).
Dunn, B., Stein, P., Temple, R. & Cavazzoni, P. An appropriate use of accelerated approval- aducanumab for Alzheimer’s disease. N. Engl. J. Med. 385, 856–857 (2021).
Petersen, R. C. Aducanumab: what about the patient? Ann. Neurol. 90, 334–335 (2021).
Rabinovici, G. D. Controversy and progress in Alzheimer’s disease - FDA approval of aducanumab. N. Engl. J. Med. 385, 771–774 (2021).
Salloway, S. & Cummings, J. Aducanumab, amyloid lowering, and slowing of Alzheimer disease. Neurology 97, 543–544 (2021).
Steinbrook, R. The accelerated approval of aducanumab for treatment of patients with Alzheimer disease. JAMA Intern. Med. 181, 1281 (2021).
Cummings, J. et al. Aducanumab: appropriate use recommendations. J. Prev. Alzheimers Dis. 8, 398–410 (2021).
Ossenkoppele, R. et al. Prevalence of amyloid PET positivity in dementia syndromes: a meta-analysis. JAMA 313, 1939–1949 (2015).
Niewoehner, J. et al. Increased brain penetration and potency of a therapeutic antibody using a monovalent molecular shuttle. Neuron 81, 49–60 (2014).
Weber, F. et al. Brain shuttle antibody for Alzheimer’s disease with attenuated peripheral effector function due to an inverted binding mode. Cell Rep. 22, 149–162 (2018).
Andrews, J. S. et al. Disease severity and minimal clinically important differences in clinical outcome assessments for Alzheimer’s disease clinical trials. Alzheimers Dement. 5, 354–363 (2019).
Liu, K. Y., Schneider, L. S. & Howard, R. The need to show minimum clinically important differences in Alzheimer’s disease trials. Lancet Psychiatry 8, 1013–1016 (2021).
Klunk, W. E. et al. The Centiloid project: standardizing quantitative amyloid plaque estimation by PET. Alzheimers Dement. 11, 1–15.e4 (2015).
Miles, L. A., Crespi, G. A., Doughty, L. & Parker, M. W. Bapineuzumab captures the N-terminus of the Alzheimer’s disease amyloid-beta peptide in a helical conformation. Sci. Rep. 3, 1302 (2013).
Englund, H. et al. Sensitive ELISA detection of amyloid-beta protofibrils in biological samples. J. Neurochem. 103, 334–345 (2007).
Hardy, J. The relationship between amyloid and tau. J. Mol. Neurosci. 20, 203–206 (2003).
Lantos, P. L. et al. Familial Alzheimer’s disease with the amyloid precursor protein position 717 mutation and sporadic Alzheimer’s disease have the same cytoskeletal pathology. Neurosci. Lett. 137, 221–224 (1992).
Hanger, D. P., Mann, D. M., Neary, D. & Anderton, B. H. Tau pathology in a case of familial Alzheimer’s disease with a valine to glycine mutation at position 717 in the amyloid precursor protein. Neurosci. Lett. 145, 178–180 (1992).
Hutton, M. et al. Association of missense and 5′-splice-site mutations in tau with the inherited dementia FTDP-17. Nature 393, 702–705 (1998).
Goedert, M., Crowther, R. A. & Spillantini, M. G. Tau mutations cause frontotemporal dementias. Neuron 21, 955–958 (1998).
Braak, H., Thal, D. R., Ghebremedhin, E. & Del Tredici, K. Stages of the pathologic process in Alzheimer disease: age categories from 1 to 100 years. J. Neuropathol. Exp. Neurol. 70, 960–969 (2011).
Arnsten, A. F. T., Datta, D., Tredici, K. D. & Braak, H. Hypothesis: Tau pathology is an initiating factor in sporadic Alzheimer’s disease. Alzheimers Dement. 17, 115–124 (2021).
Jack, C. R. Jr. et al. Hypothetical model of dynamic biomarkers of the Alzheimer’s pathological cascade. Lancet Neurol. 9, 119–128 (2010).
Palmqvist, S. et al. Cerebrospinal fluid and plasma biomarker trajectories with increasing amyloid deposition in Alzheimer’s disease. EMBO Mol. Med. 11, e11170 (2019).
Hansson, O. Biomarkers for neurodegenerative diseases. Nat. Med. 27, 954–963 (2021).
Cho, H. et al. Progressive tau accumulation in Alzheimer disease: 2-year follow-up study. J. Nucl. Med. 60, 1611–1621 (2019).
Jack, C. R. Jr. et al. Longitudinal tau PET in ageing and Alzheimer’s disease. Brain 141, 1517–1528 (2018).
Aschenbrenner, A. J., Gordon, B. A., Benzinger, T. L. S., Morris, J. C. & Hassenstab, J. J. Influence of tau PET, amyloid PET, and hippocampal volume on cognition in Alzheimer disease. Neurology 91, e859–e866 (2018).
Hanseeuw, B. J. et al. Association of amyloid and tau with cognition in preclinical alzheimer disease: a longitudinal study. JAMA Neurol. 76, 915–924 (2019).
La Joie, R. et al. Prospective longitudinal atrophy in Alzheimer’s disease correlates with the intensity and topography of baseline tau-PET. Sci. Transl. Med. 12, eaau5732 (2020).
Ossenkoppele, R. et al. Discriminative accuracy of [18F]flortaucipir positron emission tomography for Alzheimer disease vs other neurodegenerative disorders. JAMA 320, 1151–1162 (2018).
Busche, M. A. & Hyman, B. T. Synergy between amyloid-beta and tau in Alzheimer’s disease. Nat. Neurosci. 23, 1183–1193 (2020).
Driscoll, I. et al. Correspondence between in vivo (11)C-PiB-PET amyloid imaging and postmortem, region-matched assessment of plaques. Acta Neuropathol. 124, 823–831 (2012).
Curtis, C. et al. Phase 3 trial of flutemetamol labeled with radioactive fluorine 18 imaging and neuritic plaque density. JAMA Neurol. 72, 287–294 (2015).
Sabri, O. et al. Florbetaben PET imaging to detect amyloid beta plaques in Alzheimer’s disease: phase 3 study. Alzheimers Dement. 11, 964–974 (2015).
Clark, C. M. et al. Cerebral PET with florbetapir compared with neuropathology at autopsy for detection of neuritic amyloid-beta plaques: a prospective cohort study. Lancet Neurol. 11, 669–678 (2012).
Navitsky, M. et al. Standardization of amyloid quantitation with florbetapir standardized uptake value ratios to the Centiloid scale. Alzheimers Dement. 14, 1565–1571 (2018).
Jack, C. R. Jr. et al. Defining imaging biomarker cut points for brain aging and Alzheimer’s disease. Alzheimers Dement. 13, 205–216 (2017).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
E.K. is in an employee and stockholder of AbbVie. AbbVie has an anti-Aβ monoclonal antibody entering phase I clinical studies. B.D.S. is or has been a consultant for Eli Lilly, Biogen, Janssen Pharmaceutica, Eisai, AbbVie and other companies. B.D.S is also a scientific founder of Augustine Therapeutics and a scientific founder and stockholder of Muna Therapeutics.
Peer review
Peer review information
Nature Reviews Drug Discovery thanks David Knopman and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Biogen plans regulatory filing for aducanumab in Alzheimer’s disease based on new analysis of larger dataset from phase 3 studies: https://investors.biogen.com/news-releases/news-release-details/biogen-plans-regulatory-filing-aducanumab-alzheimers-disease
Eisai and Biogen announce detailed results of phase II clinical study of BAN2401 in early Alzheimer’s disease at Alzheimer’s Association International Conference (AAIC) 2018: https://eisai.mediaroom.com/2018-07-25-Eisai-And-Biogen-Announce-Detailed-Results-Of-Phase-II-Clinical-Study-Of-BAN2401-In-Early-Alzheimers-Disease-At-Alzheimers-Association-International-Conference-AAIC-2018
Rights and permissions
About this article
Cite this article
Karran, E., De Strooper, B. The amyloid hypothesis in Alzheimer disease: new insights from new therapeutics. Nat Rev Drug Discov 21, 306–318 (2022). https://doi.org/10.1038/s41573-022-00391-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41573-022-00391-w
- Springer Nature Limited
This article is cited by
-
Misfolded protein oligomers: mechanisms of formation, cytotoxic effects, and pharmacological approaches against protein misfolding diseases
Molecular Neurodegeneration (2024)
-
Therapeutic potential of APP antisense oligonucleotides for Alzheimer’s disease and down syndrome-related Alzheimer’s disease
Molecular Neurodegeneration (2024)
-
Histone acetylation in an Alzheimer’s disease cell model promotes homeostatic amyloid-reducing pathways
Acta Neuropathologica Communications (2024)
-
A single-domain antibody detects and neutralises toxic Aβ42 oligomers in the Alzheimer’s disease CSF
Alzheimer's Research & Therapy (2024)
-
Phenylalanine as an effective stabilizer and aggregation inhibitor of Bacillus amyloliquefaciens alpha-amylase
AMB Express (2024)