Abstract
The glucose-lowering effect of sodium-glucose cotransporter 2 (SGLT2) inhibitors is reduced in patients with diabetes who have chronic kidney disease (CKD). In the present study, we examined the effect of an SGLT2 inhibitor on the salt sensitivity of blood pressure (BP), circadian rhythm of BP, and sympathetic nerve activity (SNA) in nondiabetic CKD rats. Uninephrectomized Wistar rats were treated with adenine (200 mg/kg/day) for 14 days. After stabilization with a normal-salt diet (NSD, 0.3% NaCl), a high-salt diet (HSD, 8% NaCl) was administered. Mean arterial pressure (MAP) was continuously monitored using a telemetry system. We also analyzed the low frequency (LF) of systolic arterial pressure (SAP), which reflects SNA. In adenine-induced CKD rats, HSD consumption for 5 days significantly increased the mean MAP from 106 ± 2 to 148 ± 3 mmHg. However, MAP was decreased to 96 ± 3 mmHg within 24 h after switching back to a NSD (n = 7). Treatment with an SGLT2 inhibitor, luseogliflozin (10 mg/kg/day, p.o., n = 7), significantly attenuated the HSD-induced elevation of MAP, which was associated with a reduction in LF of SAP. These data suggest that treatment with an SGLT2 inhibitor attenuates the salt sensitivity of BP, which is associated with SNA inhibition in nondiabetic CKD rats.
Similar content being viewed by others
Introduction
Diabetes mellitus is the leading cause of chronic kidney disease (CKD) and hypertension [1]. Sodium-glucose cotransporter 2 (SGLT2) inhibitors are antihyperglycemic drugs used for the treatment of diabetes [2] and have been shown to improve diabetic complications in patients with type 2 diabetes [3], including cardiovascular (CV) death [4, 5]. Furthermore, several clinical studies have shown that the administration of an SGLT2 inhibitor remarkably decreases blood pressure (BP), and meta-analyses have indicated that both systolic BP (SBP) and diastolic BP (DBP) are significantly decreased by treatment with an SGLT2 inhibitor [6, 7]. More recently, a randomized, double-blind, placebo-controlled clinical trial reported that treatment with an SGLT2 inhibitor significantly decreased 24-h ambulatory BP in patients with uncontrolled nocturnal hypertension and type 2 diabetes who were treated with antihypertensive agents [8].
Interestingly, several clinical studies have shown that the SGLT2 inhibitor-induced reduction in BP is not concomitant with a notable change or compensatory increase in heart rate (HR) [8,9,10,11,12,13,14,15]. Sano et al. [16] reported that treatment with the selective SGLT2 inhibitor luseogliflozin effectively decreased HR in patients with type 2 diabetes with high HR (≥70 beats/min before treatment). These data suggest that SGLT2 inhibitors can attenuate sympathetic nerve activity (SNA). Our previous animal studies using a telemetry system showed that treatment with an SGLT2 inhibitor significantly decreased BP and SNA in high salt-treated obese rats [9] and rats with metabolic syndrome [10]. Yoshikawa et al. [17] utilized similar methods to show that an SGLT2 inhibitor significantly lowered sympathetic vasoconstrictor activity during active periods in streptozotocin-induced diabetic rats. SGLT2 inhibitors have also been shown to significantly decrease norepinephrine production and turnover in brown adipose tissue in mice [18] and the high fat-induced elevation of norepinephrine levels in the kidneys and heart in rats [19]. Jordan et al. [20] administered an SGLT2 inhibitor for 4 days to patients with type 2 diabetes treated with metformin and showed that there was no change in muscle SNA, despite a significant reduction in BP. Although the precise mechanism responsible for SNA reduction is unclear, several studies have reported that SGLT2 inhibitors can modulate urinary sodium excretion [9, 21, 22].
Clinical studies have indicated that the blood glucose-lowering effect induced by SGLT2 inhibitors was significantly attenuated in patients with diabetes and CKD who had a reduced glomerular filtration rate [23]. However, the effects of an SGLT2 inhibitor on BP and SNA in nondiabetic patients with CKD have not previously been investigated. Therefore, we examined the effects of an SGLT2 inhibitor on BP and SNA in nondiabetic adenine-induced CKD rats. To determine the salt sensitivity of BP, we administered a high-salt diet (HSD) to nondiabetic adenine-induced CKD rats.
Methods
Animals
All experimental procedures were carried out according to the guidelines for the care and use of animals established by Kagawa University. Wistar rats were purchased from Japan SLC Inc. (Shizuoka, Japan) and were housed in specific pathogen-free animal facilities at a controlled temperature (24 ± 2 °C) and humidity (55 ± 5%) with a 12-h light-dark cycle. Rats were given standard chow prior to telemetry implantation and had access to water ad libitum.
Protocols
Five-week-old male Wistar rats were subjected to right nephrectomy. After 1 week of acclimatization, rats were treated with vehicle (0.5% carboxymethylcellulose, p.o., n = 6) or adenine (200 mg/kg per day, p.o., n = 14) for 2 weeks. Next, a radiotelemetry device was implanted in all animals. Thereafter, rats received a normal-salt diet (NSD, 0.3% NaCl, OYC, Tokyo, Japan) for 9 days. Subsequently, the chow was switched to a HSD (8% NaCl, OYC, Tokyo, Japan), and adenine-treated rats were treated with vehicle (n = 7) or luseogliflozin (10 mg/kg per day, p.o., n = 7). After 5 days, the chow was again switched to a NSD for all animals (Supplementary Fig. 1).
Sample collection
At the end of the observation period, overnight-fasted animals were sacrificed by an overdose of sodium pentobarbital (250 mg/kg, i.p.). Blood was collected for the measurement of blood urea nitrogen (BUN) levels. Left kidney tissue was harvested, fixed in 10% formalin (pH 7.4) for 48 h, and embedded in paraffin for periodic acid-Schiff (PAS) staining.
Telemetry system
A radiotelemetry system (Data Science International, Saint Paul, Minnesota, USA) was used for the measurement of BP in conscious animals at 8 weeks of age. A telemetry catheter was inserted into the femoral artery, and animals were maintained under stress-free conditions for 7 days for recovery (Supplementary Fig. 1). All animals underwent a 24-h acclimatization period on a telemetry receiver panel. Twenty four-hour BP measurements were taken during the experimental period. Moreover, we calculated the 12-h dark-period (18∶00–06∶00) and 12-h light-period (06∶00–18∶00) mean arterial pressure (MAP) to evaluate the BP circadian rhythm. We also investigated SNA with the low frequency (LF) of SBP, locomotor activity (LA), and circadian rhythm using the telemetry system as previously described [9, 17].
Statistical analysis
Data are presented as the mean ± SEM. Statistical comparisons of the differences between values were performed using a t-test, one-way analysis (ANOVA) of variance followed by Tukey’s multiple comparison test, or two-way ANOVA. P values < 0.05 were considered statistically significant.
Results
Effects of luseogliflozin on renal injury and salt-induced BP elevation in adenine-induced CKD rats
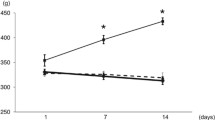
Compared with the vehicle-treated rats, adenine-induced CKD rats exhibited reduced body weight. Treatment with luseogliflozin did not affect body weight in adenine-induced CKD rats (data not shown). In adenine-induced CKD rats, severe renal histological injury was observed (Fig. 1a), and plasma BUN levels were significantly increased, while luseogliflozin did not affect histological changes or plasma BUN levels (Fig. 1b).
Impact of luseogliflozin treatment on renal injury. Adenine-induced CKD rats fed a high-salt diet (HSD) exhibited (a) widespread tubular dilation in the kidneys and (b) increased levels of blood urea nitrogen (BUN). Luseogliflozin treatment did not affect these changes. Bar = 50 μm. **P < 0.01 vs. vehicle group
In vehicle-treated rats, HSD significantly increased the average 24-h MAP from 111 ± 1 to 126 ± 1 mmHg. In adenine-induced CKD rats, the HSD-induced increase in MAP was significantly enhanced (from 106 ± 2 to 148 ± 3 mmHg). In both animal groups, MAP decreased immediately after switching to a NSD. Treatment with luseogliflozin significantly attenuated the salt-induced increase in MAP (134 ± 4 mmHg) in adenine-induced CKD rats (Fig. 2).
Impact of luseogliflozin treatment on mean arterial pressure (MAP). Average 24-h MAP on a normal-salt diet (NSD, 0.3% NaCl), on a HSD (8% NaCl), and after switching back to a NSD in adenine-induced CKD rats with or without luseogliflozin treatment. *P < 0.05, **P < 0.01, ***P < 0.001 vs. vehicle group; #P < 0.05 vs. adenine group
Figures 3a–c show the average 12-h MAP during the light (inactive) and dark (active) periods, respectively. In vehicle-treated rats, a significant difference in MAP was observed between the inactive and active periods before initiating HSD treatment (114 ± 1 vs. 109 ± 0.4 mmHg, respectively, Fig. 3a). In contrast, MAP did not differ significantly between the inactive and active periods in adenine-induced CKD rats, indicating a nondipper pattern of BP. After 5 days of HSD consumption, a difference in MAP was observed between the inactive and active periods in vehicle- and adenine-treated rats. In luseogliflozin-treated adenine-induced CKD rats, a difference in MAP was also observed between the inactive and active periods, although these changes were not significantly different (Fig. 3b). After switching from a HSD to a NSD, the BP circadian rhythm patterns immediately returned to the levels at baseline (Fig. 3c). The average values of the difference in MAP between the inactive and active periods are summarized in Fig. 3d. As shown in Supplementary Figs. 2–5, the changes in SBP and DBP were similar to those in MAP.
Impact of luseogliflozin treatment on MAP circadian rhythm. Average 12-h MAP in the inactive and active periods (a) during NSD feeding, (b) after 5 days on a HSD, and (c) 2 days after switching to a NSD. *P < 0.05, **P < 0.01, ***P < 0.001 vs. the inactive period of the respective group. d Differences in 12-h MAP between the inactive and active periods in adenine-induced CKD rats with or without luseogliflozin treatment. *P < 0.05, **P < 0.01 vs. vehicle group
Effects of luseogliflozin on SNA in adenine-induced CKD rats
We assessed the effects of luseogliflozin on LF (0.25–0.75 Hz) of SBP, which reflects the level of sympathetic vasoconstrictor activity [9, 24]. Figure 4a shows the average 24-h LF of SBP. During feeding with a NSD, adenine-induced CKD rats showed significantly reduced LF of SBP compared with vehicle-treated rats (3.1 ± 0.3 vs. 4.8 ± 0.2 mmHg², respectively), which was in response to an increase in BP (Fig. 2). Feeding with a HSD did not change SNA in all animals. Among the adenine-induced CKD rats, luseogliflozin-treated CKD rats exhibited a lower SNA than vehicle-treated CKD rats on the fifth day of HSD consumption (2.1 ± 0.1 mmHg² vs. 2.6 ± 0.3 mmHg², respectively). In all groups, SNA returned to basal values within 24 h after switching to NSD. However, the luseogliflozin-induced reduction in SNA persisted until the end of the experimental period in CKD rats (2.8 ± 0.3 mmHg² vs. 4.2 ± 0.5 mmHg², adenine + luseogliflozin vs. adenine), indicating the continuing efficacy of luseogliflozin.
Impact of luseogliflozin treatment on sympathetic nerve activity (SNA) and circadian rhythm. Low frequency (LF) of systolic blood pressure (SBP) reflects the level of SNA. (a) Average 24-h LF of SBP during feeding with a NSD (0.3% NaCl) or HSD (8% NaCl) and after switching back to a NSD in adenine-induced CKD rats with or without luseogliflozin treatment. *P < 0.05, **P < 0.01, ***P < 0.001 vs. vehicle group; ##P < 0.01 vs. adenine group. Average 12-h LF of SBP in the inactive and active periods (b) during feeding with a NSD, (c) after 5 days of a HSD, and (d) 2 days after switching to a NSD. *P < 0.05, **P < 0.01, ***P < 0.001 vs. the inactive period of the respective group; #P < 0.05, ###P < 0.001 vs. the active period of the vehicle group; †P < 0.05 vs. the active period of the adenine group
Figure 4b–d shows the average 12-h LF of SBP during the inactive and active periods. During feeding with a NSD, the circadian rhythm of LF of SBP (Fig. 4b) was similar to that of MAP (Fig. 3a). Specifically, a significant difference between inactive and active periods of LF of SBP (6.4 ± 0.3 mmHg² vs. 3.0 ± 0.2 mmHg², respectively) was observed in vehicle-treated rats, while adenine-induced CKD rats exhibited a nondipper pattern of LF of SBP and treatment (Fig. 4b). After 5 days of HSD feeding, both vehicle- and adenine-treated CKD rats showed reduced LF of SBP in the active period but not in the inactive period. Interestingly, the LF value of SBP in luseogliflozin-treated CKD rats in the active period was significantly lower than that of vehicle-treated CKD rats (1.9 ± 0.1 mmHg² vs. 2.6 ± 0.4 mmHg², respectively, Fig. 4c). After switching from a HSD to a NSD, the LF of SBP during both the inactive and active periods did not change in vehicle-treated rats. However, a difference in the LF of SBP between the inactive and active periods was apparent in adenine-induced CKD rats. Furthermore, the increase in LF of SBP observed in adenine-induced CKD rats during the active period was inhibited by luseogliflozin treatment (Fig. 4d).
Effects of luseogliflozin on LA in HS-treated adenine-induced CKD rats
Figure 5a shows the average 24-h LA. During feeding with a NSD, the average 24-h LA was lower in adenine-induced CKD rats than in vehicle-treated rats. In adenine-induced CKD rats, treatment with luseogliflozin tended to decrease LA, although the changes were not significantly different. After 5 days of HSD feeding, LA was markedly increased from 2.7 ± 0.1 to 4.4 ± 0.2 counts/minute in vehicle-treated rats. However, the HSD-induced increase in LA was significantly lower in vehicle- or luseogliflozin-treated adenine-induced CKD rats (2.6 ± 0.3 and 2.2 ± 0.2 counts/minute, respectively). After switching from a HSD to a NSD, LA immediately returned to the respective baseline level in all groups.
Impact of luseogliflozin treatment on locomotor activity (LA) and circadian rhythm. a Average 24-h LA during feeding with a NSD (0.3% NaCl) or a HSD (8% NaCl) and after switching back to a NSD in adenine-induced CKD rats with or without luseogliflozin treatment. *P < 0.05, **P < 0.01, ***P < 0.001 vs. vehicle group. Average 12-h LA in the inactive and active periods (b) during feeding with a NSD, (c) after 5 days of a HSD, and (d) 2 days after switching to a NSD. *P < 0.05, **P < 0.01, ***P < 0.001 vs. the inactive period of the respective group; #P < 0.05, ##P < 0.01, ###P < 0.001 vs. the active period of the vehicle group
Figure 5b–d depicts the average 12-h LA during the inactive and active periods. During feeding with a NSD, LA was significantly greater in the active period than in the inactive period (Fig. 5b). Moreover, both vehicle- and luseogliflozin-treated adenine-induced CKD rats exhibited decreased LA during the active period. After 5 days of HSD treatment, LA was significantly increased during the inactive and active periods in vehicle-treated rats but not in adenine-induced CKD rats. In these animals, luseogliflozin did not significantly affect LA (Fig. 5c). After switching from a NSD to a HSD, LA levels returned to the respective basal levels in all animals (Fig. 5d).
Discussion
The blood glucose-lowering effect induced by SGLT2 inhibitors is directly proportional to the glomerular filtration rate [23]. The present study examined the effect of an SGLT2 inhibitor on the salt sensitivity of BP and SNA in nondiabetic CKD rats. Experiments were performed in nondiabetic adenine-induced CKD rats to avoid the influence of changes in blood sugar levels. Our results showed that administration of the SGLT2 inhibitor luseogliflozin significantly attenuated the HS-induced elevation of BP in adenine-induced CKD rats and was associated with a reduction in SNA. These data suggest that treatment with an SGLT2 inhibitor decreases the salt sensitivity of BP by attenuating SNA in nondiabetic CKD rats, independent of blood glucose changes.
Impaired renal function predominantly contributes to the pathogenesis of salt sensitivity in BP [25]. In patients with CKD whose glomerular ultrafiltration capability is reduced, BP becomes salt sensitive [25, 26]. In the present study, we confirmed that HSD-induced increases in BP were significantly augmented in adenine-induced CKD rats. Interestingly, treatment with luseogliflozin significantly attenuated the development of HSD-induced hypertension in nondiabetic adenine-induced CKD rats. Since blood sugar levels were not influenced by luseogliflozin, BP reduction appears to be induced via a blood glucose-independent mechanism. These data are consistent with those from previous studies showing that treatment with empagliflozin prevented salt-induced increases in BP without changing blood sugar levels in obese rats with normal kidney function [27]. As indicated in previous studies [27,28,29], SGLT2 inhibitors induce natriuresis, which may mediate the SGLT2 inhibitor-induced reduction in BP in hypertensive subjects. However, a limitation of the present study is the failure to identify luseogliflozin-induced natriuresis, given the difficulties in monitoring the accurate urinary excretion rate of sodium in adenine-treated uninephrectomized rats fed an 8% NaCl diet because of severe polyuria.
It has also been reported that SNA plays an important role in the development of salt-dependent hypertension [30, 31]. In the present study, treatment with luseogliflozin significantly attenuated the HSD-induced BP elevation in CKD rats, which was associated with a reduction in SNA. These data are consistent with previous reports showing that SGLT2 inhibitors decrease SNA in salt-treated obese rats [9] and metabolic syndrome rats [10]. The precise mechanism responsible for the SGLT2 inhibitor-induced reduction in SNA is not clear. However, our data indicate that this reduction is not accompanied by reductions in blood sugar levels and LA (Fig. 5). We therefore speculate that the SGLT2 inhibitor-induced reduction in SNA is associated with the effect of the inhibitor on natriuresis, as reported by other investigators [32, 33]. However, further studies are required to clarify the precise molecular mechanism for this effect.
In agreement with previous studies in patients with CKD [34], the adenine-induced CKD rats in the present study showed a nondipper pattern in BP circadian rhythm. However, short-term feeding with a HSD induced increased BP elevation in the active period compared with the inactive period, resulting in a significant difference in BP between the two periods. These data are consistent with those from previous studies in which BP elevation induced by short-term treatment with a HSD was significantly greater in the active period than in the inactive period in Dahl salt-sensitive hypertensive rats [35]. It remains unclear why short-term HSD elicits a greater BP elevation in the active period, although the effect may be attributable to differences in drinking water volume and urinary volume between the active and inactive periods. It is also possible that a HSD alters eating habits and rhythms in rodents, which can have a marked influence on BP circadian rhythms [36].
In conclusion, our data indicate that treatment with an SGLT2 inhibitor decreases the salt sensitivity of BP, which is associated with the attenuation of SNA in CKD rats. BP control is one of the most important aspects of cardiovascular disease management in patients with CKD [37, 38]. In this regard, Wanner et al. [39] showed that although treatment with an SGLT2 inhibitor elicits fewer glucose-lowering effects in patients with type 2 diabetes and CKD than in type 2 diabetic patients without CKD, its cardioprotective effects are not influenced by reduced renal function. The effect of an SGLT2 inhibitor in nondiabetic CKD patients is currently being investigated in a large clinical study (https://www.astrazeneca.com/media-centre/press-releases/2016/astrazeneca-announces-two-new-phase-IIIb-trials-for-Forxiga-in-chronic-kidney-disease-and-chronic-heart-failure-120920161.html#). Although the primary endpoint of this study is renal outcome, the effects of the SGLT inhibitor on BP are expected to be clarified in this patient population.
References
Yamazaki D, Hitomi H, Nishiyama A. Hypertension with diabetes mellitus complications. Hypertension Res. 2018;41:147–56.
List JF, Whaley JM. Glucose dynamics and mechanistic implications of SGLT2inhibitors in animals and humans. Kidney Int Suppl. 2011. https://doi.org/10.1038/ki.2010.512.
Fitchett DH. Empagliflozin and cardio-renal outcomes in patients with type 2 diabetes and cardiovascular disease - implications for clinical practice. Eur Endocrinol. 2018;14:40–9.
Abdul-Ghani M, Del Prato S, Chilton R, DeFronzo RA. SGLT2 inhibitors and cardiovascular risk: lessons learned from the EMPA-REG OUTCOME Study. Diabetes care 2016;39:717–25.
Fitchett D, Zinman B, Wanner C, Lachin JM, Hantel S, Salsali A, et al. Heart failure outcomes with empagliflozin in patients with type 2 diabetes at high cardiovascular risk: results of the EMPA-REG OUTCOME(R) trial. Eur Heart J. 2016;37:1526–34.
Baker WL, Smyth LR, Riche DM, Bourret EM, Chamberlin KW, White WB. Effects of sodium-glucose co-transporter 2 inhibitors on blood pressure: a systematic review and meta-analysis. J Am Soc Hypertens. 2014;8:262–75.e269.
Vasilakou D, Karagiannis T, Athanasiadou E, Mainou M, Liakos A, Bekiari E, et al. Sodium-glucose cotransporter 2 inhibitors for type 2 diabetes: a systematic review and meta-analysis. Ann Intern Med. 2013;159:262–74.
Kario K, Okada K, Kato M, Nishizawa M, Yoshida T, Asano T, et al. 24-hour blood pressure-lowering effect of an SGLT-2 inhibitor in patients with diabetes and uncontrolled nocturnal hypertension: results from the randomized, placebo-controlled SACRA Study. Circulation. 2018. https://doi.org/10.1161/circulationaha.118.037076.
Wan N, Rahman A, Hitomi H, Nishiyama A. The effects of sodium-glucose cotransporter 2 inhibitors on sympathetic nervous activity. Front Endocrinol. 2018;9:421.
Rahman A, Fujisawa Y, Nakano D, Hitomi H, Nishiyama A. Effect of a selective SGLT2 inhibitor, luseogliflozin, on circadian rhythm of sympathetic nervous function and locomotor activities in metabolic syndrome rats. Clin Exp Pharmacol Physiol. 2017;44:522–5.
Maegawa H, Tobe K, Tabuchi H, Nakamura I. Baseline characteristics and interim (3-month) efficacy and safety data from STELLA-LONG TERM, a long-term post-marketing surveillance study of ipragliflozin in Japanese patients with type 2 diabetes in real-world clinical practice. Expert Opin Pharmacother. 2016;17:1985–94.
Chilton R, Tikkanen I, Cannon CP, Crowe S, Woerle HJ, Broedl UC, et al. Effects of empagliflozin on blood pressure and markers of arterial stiffness and vascular resistance in patients with type 2 diabetes. Diabetes, Obes Metab. 2015;17:1180–93.
Rosenstock J, Jelaska A, Zeller C, Kim G, Broedl UC, Woerle HJ. Impact of empagliflozin added on to basal insulin in type 2 diabetes inadequately controlled on basal insulin: a 78-week randomized, double-blind, placebo-controlled trial. Diabetes, Obes Metab. 2015;17:936–48.
Sjostrom CD, Johansson P, Ptaszynska A, List J, Johnsson E. Dapagliflozin lowers blood pressure in hypertensive and non-hypertensive patients with type 2 diabetes. Diabetes Vasc Dis Res. 2015;12:352–8.
Leiter LA, Yoon KH, Arias P, Langslet G, Xie J, Balis DA, et al. Canagliflozin provides durable glycemic improvements and body weight reduction over 104 weeks versus glimepiride in patients with type 2 diabetes on metformin: a randomized, double-blind, phase 3 study. Diabetes Care. 2015;38:355–64.
Sano M, Chen S, Imazeki H. Changes in heart rate in patients with type 2 diabetes mellitus after treatment with luseogliflozin: subanalysis of placebo-controlled, double-blind clinical trials. J Diabetes Investig. 2018;9:638-41.
Yoshikawa T, Kishi T, Shinohara K, Takesue K, Shibata R, Sonoda N, et al. Arterial pressure lability is improved by sodium-glucose cotransporter 2 inhibitor in streptozotocin-induced diabetic rats. Hypertension Res. 2017;40:646–51.
Chiba Y, Yamada T, Tsukita S, Takahashi K, Munakata Y, Shirai Y, et al. Dapagliflozin, a sodium-glucose co-transporter 2 inhibitor, acutely reduces energy expenditure in BAT via neural signals in mice. PloS ONE. 2016;11:e0150756.
Matthews VB, Elliot RH, Rudnicka C, Hricova J, Herat L, Schlaich MP. Role of the sympathetic nervous system in regulation of the sodium glucose cotransporter 2. J Hypertens. 2017;35:2059–68.
Jordan J, Tank J, Heusser K, Heise T, Wanner C, Heer M, et al. The effect of empagliflozin on muscle sympathetic nerve activity in patients with type II diabetes mellitus. J Am Soc Hypertens. 2017;11:604–12.
Heise T, Jordan J, Wanner C, Heer M, Macha S, Mattheus M, et al. Pharmacodynamic effects of single and multiple doses of empagliflozin in patients with type 2 diabetes. Clin Therapeutics 2016;38:2265–76.
Guyenet PG. Putative mechanism of salt-dependent neurogenic hypertension: cell-autonomous activation of organum vasculosum laminae terminalis neurons by hypernatremia. Hypertension. 2017;69:20–2.
Kelly MS, Lewis J, Huntsberry AM, Dea L, Portillo I. Efficacy and renal outcomes of SGLT2 inhibitors in patients with type 2 diabetes and chronic kidney disease. Postgrad Med. 2019;131:31–42.
Oliveira-Sales EB, Toward MA, Campos RR, Paton JF. Revealing the role of the autonomic nervous system in the development and maintenance of Goldblatt hypertension in rats. Auton Neurosci. 2014;183:23–9.
Kimura G, Frem GJ, Brenner BM. Renal mechanisms of salt sensitivity in hypertension. Curr Opin Nephrol Hypertens. 1994;3:1–12.
Kimura G. Glomerular function reserve and sodium sensitivity. Clin Exp Nephrol. 2005;9:102–13.
Takeshige Y, Fujisawa Y, Rahman A, Kittikulsuth W, Nakano D, Mori H, et al. A sodium-glucose co-transporter 2 inhibitor empagliflozin prevents abnormality of circadian rhythm of blood pressure in salt-treated obese rats. Hypertens. 2016;39:415–22.
Rahman A, Kittikulsuth W, Fujisawa Y, Sufiun A, Rafiq K, Hitomi H, et al. Effects of diuretics on sodium-dependent glucose cotransporter 2 inhibitor-induced changes in blood pressure in obese rats suffering from the metabolic syndrome. J Hypertens. 2016;34:893–906.
Ansary TM, Fujisawa Y, Rahman A. Responses of renal hemodynamics and tubular functions to acute sodium-glucose cotransporter 2 inhibitor administration in non-diabetic anesthetized rats. 2017;7:9555.
Fujita M, Fujita T. The role of CNS in the effects of salt on blood pressure. Curr Hypertens Rep. 2016;18:10.
Fujita T. Mechanism of salt-sensitive hypertension: focus on adrenal and sympathetic nervous systems. J Am Soc Nephrol. 2014;25:1148–55.
Khawaja Z, Wilcox CS. Role of the kidneys in resistant hypertension. Int J Hypertens. 2011;2011:143471.
Simmonds SS, Lay J, Stocker SD. Dietary salt intake exaggerates sympathetic reflexes and increases blood pressure variability in normotensive rats. Hypertension. 2014;64:583–9.
Nguy L, Johansson ME, Grimberg E, Lundgren J, Teerlink T, Carlstrom M, et al. Rats with adenine-induced chronic renal failure develop low-renin, salt-sensitive hypertension and increased aortic stiffness. Am J Physiol Regulatory, Integr Comp Physiol. 2013;304:R744–52.
Sufiun A, Rafiq K, Fujisawa Y, Rahman A, Mori H, Nakano D, et al. Effect of dipeptidyl peptidase-4 inhibition on circadian blood pressure during the development of salt-dependent hypertension in rats. Hypertension Res. 2015;38:237–43.
van den Buuse M. Circadian rhythms of blood pressure and heart rate in conscious rats: effects of light cycle shift and timed feeding. Physiol Behav. 1999;68:9–15.
Chang TI, Owens DK, Chertow GM. Lowering blood pressure to lower the risk of cardiovascular events in CKD. Am J Kidney Dis. 2014;63:900–2.
McMahon EJ, Bauer JD, Hawley CM, Isbel NM, Stowasser M, Johnson DW, et al. The effect of lowering salt intake on ambulatory blood pressure to reduce cardiovascular risk in chronic kidney disease (LowSALT CKD study): protocol of a randomized trial. BMC Nephrol 2012;13:137.
Wanner C, Lachin JM, Inzucchi SE, Fitchett D, Mattheus M, George J, et al. Empagliflozin and clinical outcomes in patients with type 2 diabetes mellitus, established cardiovascular disease, and chronic kidney disease. Circulation 2018;137:119–29.
Acknowledgements
We thank Clare Cox, PhD, from Edanz Group (www.edanzediting.com/ac) for editing a draft of this manuscript.
Funding
This study was partly a collaboration with Taisho Co., Ltd. (through A.N.). This work was also supported by the Japan Society for the Promotion of Science Grants-in-Aid for Scientific Research (KAKENHI) and the Salt Sciences Foundation (to AN). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
This study was performed at Kagawa University. All authors were involved in the acquisition, analysis, or interpretation of data. AN was involved in the conception and design of the study. NW wrote the manuscript, and AR and AN revised it critically for important intellectual content. All authors approved the final version of the manuscript and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All people designated as authors qualify for authorship.
Corresponding author
Ethics declarations
Conflict of interest
AN has received honoraria for educational meetings conducted on behalf of Taisho Co., Ltd. The authors declare that there are no other conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Wan, N., Fujisawa, Y., Kobara, H. et al. Effects of an SGLT2 inhibitor on the salt sensitivity of blood pressure and sympathetic nerve activity in a nondiabetic rat model of chronic kidney disease. Hypertens Res 43, 492–499 (2020). https://doi.org/10.1038/s41440-020-0410-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-020-0410-8
- Springer Nature Singapore Pte Ltd.
Keyword
This article is cited by
-
Associations of SGLT2 genetic polymorphisms with salt sensitivity, blood pressure changes and hypertension incidence in Chinese adults
Hypertension Research (2023)
-
Annual reports on hypertension research 2020
Hypertension Research (2022)
-
Thermoreceptor TRPV1 regulates body weight and blood pressure in the absence of thermogenin
Hypertension Research (2022)