Abstracts
Wingless (Wg)/Wnt family proteins are essential for animal development and adult homeostasis. Drosophila Wg secreted from the dorsal-ventral (DV) midline in wing discs forms a concentration gradient that is shaped by diffusion rate and stability of Wg. To understand how the gradient of extracellular Wg is generated, we compared the secretion route of NRT-Wg, an artificial membrane-tethered form of Wg that is supposedly not secreted but still supports fly development, to that of wild-type Wg. We found that wild-type Wg is secreted by both conventional Golgi transport and via extracellular vesicles (EVs), and NRT-Wg can be also secreted via EVs. Furthermore, wild-type Wg secreted by Golgi transport diffused and formed Wg gradient but Wg-containing EVs did not diffuse at all. In case of Wg stability, Sol narae (Sona), a metalloprotease that cleaves Wg, contributes to generate a steep Wg gradient. Interestingly, Wg was also produced in the presumptive wing blade region, which indicates that NRT-Wg on EVs expressed in the blade allows the blade cells to proliferate and differentiate without Wg diffused from the DV midline. We propose that EV-associated Wg induces Wg signaling in autocrine and juxtaposed manners whereas Wg secreted by Golgi transport forms gradient and acts in the long-range signaling, and different organs differentially utilize these two types of Wg signaling for their own development.
Similar content being viewed by others
Introduction
Wnt family is a group of essential morphogens for animal development and homeostasis by governing multiple cellular functions such as cell proliferation, differentiation, cell survival, stem cell maintenance, and cell polarity [1,2,3,4,5]. Secreted Wnts form a concentration gradient that is shaped by diffusion and stability of Wnt [6]. This Wnt gradient is essential for embryonic muscle development, tail formation, and tissue polarity because the transcriptional induction of Wnt-responding genes is dependent on the concentration of gradient along the axis [7,8,9,10]. Fly Wg, a homolog of mammalian Wnt-1 [11,12,13,14], and the fly wing disc have been used to study Wnt gradient because Wg is heavily expressed at the DV midline of the wing pouch region, diffuses, and forms a concentration gradient [15]. Wg diffuses about ten cells and induces Wg effector genes such as distal-less (dll) and vestigial (vg) depending on the concentration of Wg [15,16,17].
NRT-Wg, an artificial membrane-tethered protein generated by the fusion of the transmembrane domain of Neurotactin and Wg, has been assumed not secreted [18]. Surprisingly, wg[KO, NRT-Wg] flies that have NRT-wg gene in place of wild-type wg gene develop normally, so authors claimed that the secretion of Wg is not necessary for Wg signaling and cells exposed to Wg in earlier development have cellular memory for the later development [19]. However, wg[KO, NRT-Wg] flies have defects in cell fate specification and proliferation, which lead to delayed organ development, malformed intestine, and sterility [19, 20]. Therefore, NRT-Wg cannot fully replace Wg for Wg signaling, and it is necessary to clarify differences between Wg and NRT-Wg.
Another factor to consider for Wg gradient is the stability of extracellular Wg. A hypothetical Wg-cleaving protease was proposed to explain the steepness of Wg gradient at the DV midline in a mathematical model [21]. A candidate for such protease is Sona belonged to a disintegrin and metalloprotease with thrombospondin motif (ADAMTS) family. Sona generates N-terminal domain (NTD) and C-terminal domain (CTD) by cleaving two sites in the linker region of Wg, and the epitope of the well-known anti-Wg antibody 4D4 is present in between the two cleavage sites [22]. Wg-NTD required for secretion of Wg is homologous to a class of lipid-interacting proteins whereas Wg-CTD is homologous to cytokine groups that activate cellular signaling [23,24,25]. Transient expression of Wg-CTD but not Wg-NTD in flies induces Wg signaling, demonstrating that Wg-CTD has Wg signaling activity. However, Wg-CTD is more specialized for cell proliferation than full-length Wg, and is less stable than full-length Wg [22]. Spatiotemporal regulation of the ratio between the two forms of Wg by Sona may be essential for fly development.
Wnts are secreted by both conventional Golgi transport and exosome-mediated secretion pathways, but it is still largely unknown how these differentially secreted Wnts contribute to the formation of Wnt gradient [26,27,28]. Exosomes, a major EV population with about 100 nm diameter, are intraluminal vesicles in the multivesicular body (MVB) that are released to extracellular space by the fusion of MVB with the plasma membrane [29, 30]. We recently reported a new population of EVs that are smaller than exosomes [31]. Both exosomes and this small EV group contain the EV marker CD63, but another EV marker CD81 is present only in exosomes. Furthermore, exosomes enhance their own secretion while the other group promotes cell proliferation [31].
We studied both diffusion and stability of Wg and NRT-Wg to understand the extracellular Wg gradient, and found a few important features. Firstly, Wg is secreted by both Golgi transport and via EVs whereas NRT-Wg is secreted only via EVs as a membrane-bound form. Secondly, Wg secreted by Golgi transport diffuses about ten cells but Wg-containing EVs diffuse only one cell distance. Thirdly, Sona contributes to form a steep Wg gradient. Lastly, Wg expressed in the blade region is important for wing growth and development.
Results
Extracellular Wg/Wnts are present in soluble multi-protein complex, exosomes, and a new group of EVs
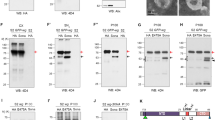
Proteins and EVs secreted to extracellular space in vivo are released to culture media in cell cultures, and can be fractionated from conditioned media [27, 32]. We recently reported a new fraction that contains EVs smaller than exosomes [31]. To examine whether Wg is also present in this new fraction, conditioned media from the tub-wg S2 cell culture were obtained, precleared and centrifuged at 100,000 × g to yield two different fractions: a pellet fraction (P100) containing exosomes and a supernatant fraction (SNΔ1). This SNΔ1 fraction was centrifuged at 200,000 × g, and a P200 pellet fraction and a supernatant fraction (SNΔ2) were obtained [31] (Fig. 1a).
a A scheme for biochemical fractions from cultured media. b Western analysis for full-length Wg in various fractions. Full-length Wg from S2 tub-wg cell culture was detected with the anti-4D4 (Wg) antibody. An exosome marker, Syntaxin 1A (Syx1A), was detected in both P100 and P200 fraction. c–e Human Wnts in various fractions. wnt1-HA, wnt3a-HA and wnt4-HA cDNA constructs were expressed in HEK 293 cells. All Wnts were detected with anti-HA antibody. Both P100 and P200 fractions were confirmed by anti-CD63 antibody. f Various fractions from culture of S2 cells expressing NRT-HA-wg and sona-HA cDNAs are analyzed. GFP-Wg was used as a positive control for cleavage by Sona [22]. Full-length NRT-HA-Wg and a cleaved fragment generated by Sona are marked by a red arrow and a black arrowhead, respectively. g–i Images of extracellular NRT-HA-Wg taken at the basal region of the wing disc from the late 3rd instar larvae. NRT-HA-Wg was transiently expressed for 18 h in en > NRT-HA-wg, GFP, Gal80ts larvae in (g). The boxed region in (g) was magnified in (i). Diagram of NRT-HA-Wg on EVs. NRT-HA-Wg has a HA tag between transmembrane domain of NRT and Wg [18] in (h). Extracellular HA+ 4D4+, HA+ 4D4-, and HA- 4D4+ structures are marked with a circle, an arrow, and an arrowhead, respectively in (i). Scale bar, 60 μm except (i), 6.5 μm.
Wg was present in all fractions including the P200 fraction (Fig. 1b). Syntaxin 1A, a fly exosome marker, and CD63, a mammalian exosome marker, were present in both P100 and P200 fractions in S2 cell culture similar to the results from mammalian cell culture (Fig. S1) [31, 33]. Human Wnt1, Wnt3a, and Wnt4 were also present in all three fractions with different proportions: Wnt1 and Wnt3a are predominantly present in P100 EVs whereas Wnt4 is mainly present in SNΔ2 fraction (Fig. 1c–e). Therefore, both Wg and Wnts are secreted by Golgi transport and via EVs.
NRT-Wg is present in both P100 and P200 EVs
NRT-HA-Wg has a transmembrane domain of NRT, so theoretically it should be secreted as a membrane-bound form on EVs [34, 35] (Fig. 1h). To test this, the fractions were prepared from conditioned media of S2 cells transfected with NRT-HA-wg cDNA (Materials and Methods). GFP-Wg as a positive control was detected in all three fractions whereas ~140 kDa NRT-HA-Wg was detected in P100 and P200 but not in SNΔ2 fraction (Fig. 1f’-f’’’). Sona cleaved GFP-Wg in P100, P200, and SNΔ2 fractions, indicating that Sona cleaves Wg regardless of secretion routes. Sona also cleaved NRT-HA-Wg in both P100 and P200 fractions, and generated a fragment corresponding to Wg-CTD (Figs. 1f-f”’ and S2) [22]. Furthermore, extracellular HA+ 4D4− (NRT-NTD-L1 like fragments) and HA− 4D4+ (CTD-L1 like fragments) puncta that are cleaved products of NRT-HA-Wg were detected in en > NRT-HA-wg, GFP, Gal80ts wing discs (Figs. 1g–i and S3). Therefore, NRT-HA-Wg tethered to EVs are secreted and cleaved by Sona.
EVs do not diffuse in wing discs
We then compared the patterns of Wg and NRT-Wg in Canton-S (CS) and wg[KO, NRT-Wg] wing discs, respectively. Both apical and basal extracellular Wg in CS discs was more broadly distributed than extracellular NRT-Wg in wg[KO, NRT-Wg] discs (Figs. 2a–d and S4), consistent with Chaudhary et al. [36]. NRT-Wg was localized mostly to the plasma membrane that may represent NRT-Wg tethered to the plasma membrane, and in some vesicular structures that may represent NRT-Wg-containing EVs.
Yellow lines demarcate the anterior and the posterior boundary in (e), (f), and (h). a–d Distribution patterns of extracellular Wg in the DV boundary of wing discs in the late 3rd instar CS and wg[KO, NRT-Wg] larvae. Images in (a) and (b) were taken by focusing at basal extracellular matrix (ECM) while those in (c) and (d) were taken by focusing at apical ECM. e, f Both HA-Wg and NRT-HA-Wg were transiently expressed in the posterior region by en-Gal4 driver. Dll was induced up to ten cells in the anterior region of en > GFP, HA-wg, Gal80ts discs in (e). NRT-HA-Wg induced Dll cell-autonomously and in juxtaposed cells in en > GFP, NRT-HA-wg, Gal80ts discs (white arrow) in (f). g No diffusion of GFP+ signals from CD63-GFP+ LacZ+ clones generated by flp-out method [67]. h Extracellular CD63-GFP visualized by anti-GFP antibody in the en > CD63-GFP, lacZ wing disc. Scale bar, (a), (b) 40 μm; (e–h) 60 μm.
To correlate the diffusion range of Wg and its activity, we transiently overexpressed HA-Wg and NRT-HA-Wg, and checked the expression range of Dll. As reported [18, 37,38,39], transiently overexpressed HA-Wg and GFP-Wg induced Dll in about ten cells from the source but NRT-HA-Wg induced Dll only at the source (Figs. 2e, f and S5). Consistent with this, extracellular HA+ or 4D4+ structures representing NRT-HA-Wg and its cleaved products also did not diffuse in both basal and apical regions (Figs. 1g and S6). These results raised two possibilities: EVs do not diffuse in general or only NRT-Wg-containing EVs do not diffuse. To distinguish these two, we examined the distribution pattern of the EV marker, CD63-GFP [33]. In fact, GFP signals were restricted to the CD63-GFP+ clone, and extracellular GFP signals were not detected in the anterior region of en > CD63-GFP, lacZ wing discs (Fig. 2g, h). Thus, EVs do not diffuse in wing discs.
The Wg-NTD domain may be essential for the long-range diffusion of Wg
If Wg tethered to EVs do not diffuse, Wg secreted by Golgi transport may be solely responsible for the formation of Wg gradient in wing discs. To prove this point and examine which domain of Wg is required for diffusion, we constructed two transgenes that encode Wg-NTD and Wg-CTD fragments, each containing the Wg signal peptide at the amino-terminus [22]. Both Wg-NTD and Wg-CTD fragments were present in all biochemical fractions, indicating that they are secreted by both Golgi transport and via EVs in S2 cells (Fig. 3a, b).
Yellow lines mark the AP boundary of wing discs in (d) and (e). a, b CX, P100, P200, and SNΔ2 fractions from S2 cells expressing GFP-wg-NTD cDNA in (a) and wg-mycCTD cDNA in (b). c–e Extracellular GFP signal representing GFP-Wg-NTD at the basal ECM and LacZ signal representing the en region are compared in upper (c) and extracellular 4D4 signal representing Wg-MycCTD and GFP signal representing the en region are compared in lower (c). The plotted intensity of extracellular GFP and 4D4 signal in (c) were measured in the boxed regions in (d’) and (e’), respectively. Extracellular GFP signals representing GFP-Wg-NTD protein diffused (arrow in d’’). Extracellular 4D4 signals representing Wg-CTD in (e) did not diffuse (arrow in e’’). Scale bar, 100 μm.
Based on the secretion of both Wg-NTD and Wg-CTD fragments by Golgi transport, we expected that both fragments may diffuse in wing discs. However, Wg-NTD diffused in en > GFP-wg-NTD wing discs whereas Wg-CTD did not diffuse at all in en > wg-myc-CTD wing discs (Figs. 3c–e and S7). This result is consistent with our previous report that the level of Dll increases only inside of the Wg-CTD-expressing clones [22]. In sum, the NTD domain may be essential for the diffusion of Wg.
Sona contributes to form a steep gradient of extracellular Wg
Wg gradient is also affected by stability of Wg, which is regulated by proteolytic enzymes. Sona is one of the strong candidates because it generates unstable Wg-CTD by cleaving stable full-length Wg [22]. We visualized the patterns of extracellular Wg at the basal region of wing discs in CS and two sona mutants (n = 7, each) (Fig. 4a, c, e). The intensity of extracellular Wg signals visualized by 4D4 was measured and plotted to generate Wg gradient curves. Several differences were detected between control and sona mutants (Fig. 4b, d, f). The peaks at the DV midline were higher and the areas under the gradient curves were larger in wild-type than sona mutants, which may be due to the promotion of Wg secretion by Sona [40]. Most importantly, the slopes of Wg gradient in sona mutants were shallower (a = 31.528 in sona13; a = 22.202 in sona18) than wild-type (a = 87.225). A similar phenomenon was observed in wg > sona RNAi wing discs (Fig. S8). This shallow gradient in sona mutants may be due to increase in the half-life of full-length Wg by lack of Wg-cleaving activity of Sona, which allows more diffusion of full-length Wg.
Distribution patterns of extracellular Wg in the DV boundary of late 3rd instar wing discs in CS (a), sona13 (c) and sona18 (e) larvae. Extracellular Wg at basal ECM was detected with anti-4D4 antibody. The signal intensities of extracellular Wg in the boxed regions encompassing the dorsal and ventral wing pouch in (a), (c) and (e) (n = 7, each) are co-plotted in (b), (d) and (f). The average intensity of background signal was 2.09 × 102, approximately one tenth or less than the lowest intensity of all samples. The trend line was drawn by averaging all seven curves and the slope was calculated by regression analysis with the values obtained from the dorsal edge to the DV midline. Scale bar, 60 μm.
Wg is expressed in the presumptive wing blade region
We have shown that NRT-HA-Wg is tethered to EVs and cannot diffuse (Fig. 2). Then, the question still remains how NRT-Wg can support fly development in wg[KO, NRT-Wg] flies. Although the long-term cellular memory by prior exposure to Wg may be an attractive explanation [19], we considered another possibility: Wg expressed in the presumptive wing blade region (‘blade’ hereafter) is important for the normal wing development of wg[KO, NRT-Wg] flies.
It has been well documented that wg is expressed in both peripodial epithelium and disc proper in early larval wing discs [41,42,43,44], which are confirmed in sona > GFP/wg-lacZ flies using LacZ as a marker for wg transcription (Fig. 5a–c). The wg-lacZ was heavily expressed in the anterior ventral domain and at the DV midline in wing discs of early third instar larvae (Fig. 5a, b). After the mid-third instar stage, expression of wg became more prominent at the DV midline and the presumptive hinge region but became weaker in the blade (Fig. 5c–e). Interestingly, other wg reporters, wg-Gal4 and wg[KO, Gal4] [19, 45], were also expressed in the blade with similar patterns to the wg-lacZ reporter at a much higher level (Fig. 5e–g). Besides, a specific region near the anterior-posterior (AP) boundary did not express wg in all wg reporters (asterisks in Fig. 5a–g). In case of sona, it was expressed as a patchy pattern in the entire pouch during larval stages (Fig. 5a–c, h). Since Sona promotes Wg secretion as well as cleaves Wg, dynamic changes in the transcriptional patterns of wg and sona may be essential for optimizing the amount of full-length Wg and Wg-CTD in any given location of the wing disc.
Yellow asterisks mark specific regions near the AP boundary where neither wg nor sona is expressed. Yellow arrows indicate absence of wg transcription in all wg enhancers examined. a, b Transcriptional pattern of wg and sona in sona > GFP, wg-lacZ wing discs of the late 2nd instar larvae. a is magnified five times in (b). c, d The transcriptional pattern of wg and sona in mid 3rd instar wing discs. c is magnified nine times in (d). e–g LacZ pattern of three wg reporters in late 3rd instar wing discs: wg-lacZ (e), wg-Gal4 > lacZ (f) and wg[KO; Gal4]>lacZ (g). h GFP pattern in sona > GFP late 3rd instar wing discs. i, j G-trace analysis for wg-Gal4 and wg[KO; Gal4] drivers in late 3rd instar wing discs. RFP signals represent the real-time expression of the driver, and GFP signals represent cell lineage that had expressed the driver during earlier time of development. Scale bar, 100 μm except (b), 50 μm.
To check whether the pattern of these wg reporters represents the transcription pattern of wg, a lineage analysis was performed using G-trace system with wg-Gal4 drivers [46]. In this cell-autonomous FLP-out marking system, cells expressing a given Gal4 at a sampling time are marked with RFP representing ‘real-time’ expression, while cells that have expressed the given Gal4 during the earlier developmental time are marked with GFP representing ‘lineage’. The ‘real-time' RFP patterns induced by these wg-Gal4 drivers were similar to the LacZ patterns induced by wg-Gal4 and wg[KO, Gal4] drivers (Fig. 5i”, j”). Besides, a region near AP boundary devoid of wg expression was also detected (asterisks in Fig. 5i”, j”). The ‘lineage’ GFP signals were present in almost all disc proper cells plus anterior peripodial epithelium, consistent with the wide expression of Wg in early larval wing discs (Fig. 5i’, j’). The patterns of these wg reporters were also fairly similar to those of Wg protein in the blade (Fig. S9). In sum, Wg expressed in the blade may be essential for the wing development of not only wg[KO, NRT-Wg] but also normal flies.
Patterns of vgQE and Delta in the wing blade correlate with that of wg
Pattern of wg in the blade suggests that the blade cells may express other genes similar or opposite pattern to wg, and we identified two such genes, vestigial (vg) and Delta (Dl). It has been shown that the expression of vg in the blade is carried out by the vgQE-enhancer [17, 47, 48]. Interestingly, GFP representing wg transcription and LacZ representing vg transcription in the blade of wg-Gal4/UAS-GFP; vgQE-lacZ wing discs were mutually exclusive (Fig. 6a, b). For instance, the region near the AP boundary had a low level of GFP but a high level of LacZ (asterisks in Fig. 6a’, a”). Similar results were obtained in wg[KO, Gal4]/UAS-GFP; vgQE-lacZ wing discs (Fig. S10).
The specific region near the AP boundary is marked with yellow asterisks. a, b GFP and LacZ patterns in wg > GFP; vgQE-lacZ wing disc (a) and the black and white images (a’, a”). Intensities of red and green signals along the white-line in (a) are plotted in (b). c, d As a control, Wg-lacZ and Dl patterns in wg-lacZ/en-Gal4; UAS-GFP wing discs are shown (c). Overexpression of Dl induced the LacZ expression in the posterior region of wg-lacZ/en-Gal4; UAS-GFP, UAS-Dl wing discs (d). e, f LacZ and Dl patterns in wg > lacZ wing discs (e). Intensities of red and green signals on the white-line in (e) are plotted in (f). Scale bar, (a), (e) 60 μm; (c), (d) 100 μm.
Dl is a Notch ligand acting upstream of wg, and regulates wg transcription in regions for future wing margin and veins [49]. In fact, the level of wg-lacZ was increased by overexpression of Dl in the blade (Fig. 6c, d). Dl was also expressed at a higher level along the future vein regions in a similar pattern to GFP in wg > GFP wing discs, suggesting that Dl in the blade turns on wg transcription (Fig. 6e, f). Thus, expression of wg, vg, and Dl in similar or mutually exclusive pattern in the blade may be crucial to specify veins and interveins as well as to modulate the rate of cell proliferation in different parts of the blade.
Discussion
It had been widely accepted for decades that secreted Wg diffuses in the extracellular space, forms a concentration gradient, and induces gene expression in a concentration-dependent manner. Here, we report that Wg tethered to EVs does not diffuse and induces only a short-range Wg signaling in autocrine and juxtacrine manners (Fig. 7a). In contrast, soluble Wg in a multi-protein complex secreted by conventional Golgi transport diffuses, forms a gradient, and induces a long-range signaling (Fig. 7b). Furthermore, Sona contributes to the formation of steep Wg gradient in wing discs because it cleaves stable Wg and generates unstable Wg-CTD.
a A model to explain the behavior of Wg secreted by EVs. Wg-containing EVs are channeled into two pathways. (1) When Sona is absent, Wg-containing EVs bind to the juxtaposed cells and induce Wg signaling. (2) When Sona is present, Wg is cleaved to Wg-CTD, Wg-CTD then binds to the juxtaposed cells and induces Wg signaling. b A model to explain the behavior of Wg secreted by conventional Golgi transport in two different pathways. (1) When Sona is absent, WgFL in multi-protein complex diffuses about 10 cells, forms Wg gradient and induces the long-range signaling. (2) When Sona is present, WgFL in the multi-protein complex is cleaved by Sona, Wg-CTD binds to cells nearby and induces Wg signaling. The distance that WgFL travels depends on the level of Sona.
The secretion and diffusion of Wg tethered to EVs
EVs including exosomes have recently drawn a lot of attention because of their signaling activity and medical application [50, 51]. We found that Wg/Wnts are present in both P100 and P200 EV fractions (Fig. 1). These P100 and P200 fractions promote exosome production and cell proliferation, respectively [31]. In addition to Wnt activity as mitogens [17, 18, 52], Wnt3a and Wnt5b promote exosome production in primary cultured microglia and melanoma cells, respectively [53, 54]. Therefore, Wnts on EVs may be one of the key proteins that promote cell proliferation and exosome production in a juxtacrine manner.
Wnt1 and Wnt3a were abundantly present especially in the P100 fraction whereas Wnt4 was abundantly present in the SNΔ2 fraction (Fig. 1). Therefore, Wnt1 and Wnt3 may be preferentially secreted as tethered forms to EVs and act in a juxtacrine manner. Consistent with this speculation, Wnt3a expressed from Paneth cells induces a short-range Wnt signaling in the intestinal stem cell niche [55] and the majority of Wnt3a is secreted as a membrane-associated form in MDCK cell culture [56]. These Wnt/Wg secreted by multiple secretion routes may play distinct roles in different tissues. Since glycosyl groups in Wnts determine by which pathway Wnts are secreted [56], it will be interesting to find the potential link between glycosyl groups and the secretion route of Wg in various fly tissues. Moreover, NRT-Wg signaling to the Frizzled 2 (Fz2) rather than the Fz1 receptor plays an important role in cell viability [36]. Thus, each Fz receptor may exhibit differential binding affinity for Wg in multi-protein complex, Wg on EVs, or Wg-CTD.
Wing vs. other organ defects in wg[KO, NRT-Wg] flies
wg[KO, NRT-Wg] flies that lack Wg secreted by Golgi transport have normal wing margin bristles, so NRT-Wg on EVs seems to be fully responsible for the differentiation of wing margin bristles. However, the size of their wings is about 10–12% smaller than wild-type wings [19], suggesting that the diffusion of Wg secreted by Golgi transport is required for wing growth to about 10–12%, and Wg on EVs expressed in the blade is mainly responsible for wing growth of wg[KO, NRT-Wg] flies. Based on these data on wg[KO, NRT-Wg] flies, one can speculate that Wg on EVs plays more significant roles for wing growth and development than Wg secreted by Golgi transport in wild-type flies.
Although wings of wg[KO, NRT-Wg] flies are almost normal, these flies show defects in cell proliferation and fate specification during gut development [20]. wg[KO, NRT-Wg] flies also show severe defects in proximo-distal patterning of Malphigian/ureter tubules and neuronal specification in the visual system [57, 58]. Thus, Wg secreted by Golgi transport and formation of Wg gradient seem to play more significant roles than Wg on EVs in these aforementioned organs compared to wings.
Wg-cleaving proteases in the formation of Wg gradient
The clear difference between the slope of Wg gradient in sona and wild-type discs demonstrates that Sona affects Wg gradient. The peak level of extracellular Wg at the DV midline of sona wing discs was lower than that in wild-type, which is consistent with the involvement of Sona in Wg secretion (Fig. 4) [22]. To interpret the slope of Wg gradient, it is necessary to consider that Wg signals detected by the 4D4 antibody are mostly from full-length Wg and Wg-NTD but little from unstable Wg-CTD [22]. The steep Wg gradient in wild-type wing discs indicates that only a small fraction of full-length Wg is diffused far from the DV midline probably because Sona efficiently cleaves full-length Wg. By the same token, the shallow Wg gradient in sona wing discs indicates that full-length Wg is increased in its level by lack of Sona activity, and diffuses farther.
Tiki is another Wnt-cleaving metalloprotease identified in Xenopus, and its homologs are found in vertebrates but not in flies [59, 60]. Tiki cuts the N-terminal residues of several Wnts, which minimizes receptor-binding capacity and reduces the solubility of extracellular Wnt [61, 62]. Whether Tiki affects Wnt gradient has not yet been reported. It is possible that other Wg-cleaving proteases besides Sona may exist and affect Wg signaling in flies because cleaved products of Wg are repeatedly detected in the absence of Sona in S2 cell culture [22].
Distinct expression patterns of Wg and Sona in the wing blade
It has been widely accepted that Wg secreted from the cells in the DV midline of the wing discs is responsible for the growth and differentiation of the entire wing. We and others have shown that Wg is expressed not only in the DV midline but also in the blade region. Wg is heavily expressed in the ventral region of wing discs during the early larval stage [41,42,43,44] (Fig. 5). We showed that wg is also expressed in the blade during the late third instar stage. Furthermore, wg is heavily expressed in all veins of adult wings [63], which is consistent with our finding that pattern of Wg is similar to that of Dl in the blade region of the wing disc in the late 3rd larval stage (Fig. 6).
Simple diffusion of Wg from the DV midline to the entire blade may not be adequate to finely control the activity of Wg signaling in the local environment. In line with this, wg and sona are expressed in patchy patterns in the blade (Fig. 5), which may allow modulation of the amount of full-length Wg and Wg-CTD at any given location. Such fine-tuning of full-length Wg vs. Wg-CTD may be essential for coordinating rates of cell proliferation and patterning [22]. We propose that Wg tethered to EVs is secreted from cells at the DV midline to initiate neuronal differentiation for the formation of wing margin bristles by the short-range signaling, whereas Wg secreted by Golgi transport diffuses to form Wg gradient and support the growth of cells about ten cells from the DV midline. Most importantly, the low amount of Wg secreted from the cells in the blade via EVs play important roles in growth and patterning of structures such as veins and interveins. It will be interesting to find out whether these findings on fly Wg are relevant to Wnts for the development and growth of other animals.
Materials and methods
Drosophila strains, transgenic lines, and DNA constructs
UAS-wg-mycCTD, UAS-GFP-wg-NTD, sona13, and sona18 flies are described elsewhere [22, 40]. UAS-CD63-GFP [33], UAS-GFP-wg [64], wg[KO; Gal4], wg[KO, NRT-Wg] [65], UAS-NRT-HA-Wg [18], wg-Gal4 [45], and vgQE-lacZ [66] were kindly provided from other labs. All other fly lines were obtained from the Bloomington stock center. Wnt cDNAs were obtained from J. W. Kim. pUAST-CD63-GFP was constructed with CD63-GFP cDNA in CD63-pEGFP C2 (Addgene). The pUAST-NRT-HA-wg was constructed by recombining the pUAST vector with NRT cDNA (WG35) and HA(flu)-wg cDNA (WG143). These WG35 and WG143 constructs were kindly provided by G. Struhl.
Cell lines, cell culture, and exosome preparation
Drosophila S2 tub-wg and S2 cell lines were obtained from DGRC. Drosophila S2 cell were grown in M3 media (Sigma-Aldrich) supplemented with 10% IMS (Sigma-Aldrich) at 25 °C. Stable cell lines were grown with hygromycin in 10% IMS M3 media, and S2 tub-wg cells were cultured in 10% FBS M3 media. Transfections were carried out with Cellfectin (Invitrogen) according to the manufacturerʼs instructions. For the preparation of EVs (P100, P200), 7–40 ml of conditioned media obtained from cultures (1.25 × 106 cells/ml) were used as described [31]. Human HEK293 cells obtained from KCBL were grown in RPMI 1640 media (Gibco) supplemented with 10% FBS (Gibco) at 37 °C with 5% CO2.
Immunocytochemistry and Western analysis
Fly larvae were cultured at 25 °C unless stated otherwise. Wing discs from the late third instar larvae were used for intracellular staining and extracellular staining [15]. For immunocytochemistry, we used 1:100; 4D4 (DSHB, mouse), 1:200; HA (Roche, rat), 1:200; GFP (Abcam, rabbit), 1:100; Dl (DSHB, mouse), 1:100; Dll (Santa Cruz, goat). For extracellular staining, we used ten times more antibodies than for intracellular staining. Fluorescent images were captured using a Zeiss LSM laser scanning confocal microscope and processed with Adobe Photoshop. Plot analysis and intensity measurements were performed on raw data processed with Zen 2012 and ImageJ under identical conditions. Western analysis was carried out as described [40]. For western analysis, we used 1:5,000; HA (Santa Cruz, rabbit), 1:1,000; HA (Roche, rat), 1:1,000; Wg (DSHB, mouse), 1:1,000; CD63 (SBI, rabbit), 1:1,000; CD63 (Santa Cruz, mouse), 1:1,000; GFP (Abcam, rabbit), and 1:1,000; Syx1A (DSHB, mouse).
References
Teo JL, Kahn M. The Wnt signaling pathway in cellular proliferation and differentiation: a tale of two coactivators. Adv Drug Deliv Rev. 2010;62:1149–55.
Steinhart Z, Angers S. Wnt signaling in development and tissue homeostasis. Development. 2018;145:dev146589.
Masckauchan TN, Shawber CJ, Funahashi Y, Li CM, Kitajewski J. Wnt/beta-catenin signaling induces proliferation, survival and interleukin-8 in human endothelial cells. Angiogenesis. 2005;8:43–51.
Humphries AC, Mlodzik M. From instruction to output: Wnt/PCP signaling in development and cancer. Curr Opin Cell Biol. 2018;51:110–6.
Nusse R, Fuerer C, Ching W, Harnish K, Logan C, Zeng A, et al. Wnt signaling and stem cell control. Cold Spring Harb Symp Quant Biol. 2008;73:59–66.
Logan CY, Nusse R. The Wnt signaling pathway in development and disease. Annu Rev Cell Dev Biol. 2004;20:781–810.
Kiecker C, Niehrs C. A morphogen gradient of Wnt/beta-catenin signalling regulates anteroposterior neural patterning in Xenopus. Development. 2001;128:4189–201.
Yamaguchi TP. Heads or tails: Wnts and anterior-posterior patterning. Curr Biol. 2001;11:R713–724.
Petersen CP, Reddien PW. Wnt signaling and the polarity of the primary body axis. Cell. 2009;139:1056–68.
Simons M, Mlodzik M. Planar cell polarity signaling: from fly development to human disease. Annu Rev Genet. 2008;42:517–40.
Sharma RP. Wingless - a new mutant in D. melanogaster. Drosoph Inf Serv. 1973;50:134.
Nusse R, Varmus HE. Many tumors induced by the mouse mammary tumor virus contain a provirus integrated in the same region of the host genome. Cell. 1982;31:99–109.
Clevers H. Wnt/beta-catenin signaling in development and disease. Cell. 2006;127:469–80.
Mann MC. The occurrence and hereditary behavior of two new dominant mutations in an inbred strain of Drosophila melanogaster. Genetics. 1923;8:27–36.
Strigini M, Cohen SM. Wingless gradient formation in the Drosophila wing. Curr Biol. 2000;10:293–300.
Delanoue R, Legent K, Godefroy N, Flagiello D, Dutriaux A, Vaudin P, et al. The Drosophila wing differentiation factor Vestigial-Scalloped is required for cell proliferation and cell survival at the dorso-ventral boundary of the wing imaginal disc. Cell Death Differ. 2004;11:110–22.
Neumann CJ, Cohen SM. A hierarchy of cross-regulation involving Notch, wingless, vestigial and cut organizes the dorsal/ventral axis of the Drosophila wing. Development. 1996;122:3477–85.
Zecca M, Basler K, Struhl G. Direct and long-range action of a wingless morphogen gradient. Cell. 1996;87:833–44.
Alexandre C, Baena-Lopez A, Vincent JP. Patterning and growth control by membrane-tethered Wingless. Nature. 2014;505:180–5.
Tian A, Duwadi D, Benchabane H, Ahmed Y. Essential long-range action of Wingless/Wnt in adult intestinal compartmentalization. PLoS Genet. 2019;15:e1008111.
Wartlick O, Kicheva A, Gonzalez-Gaitan M. Morphogen gradient formation. Cold Spring Harb Perspect Biol. 2009;1:a001255.
Won JH, Kim GW, Kim JY, Cho DG, Kwon B, Bae YK, et al. ADAMTS Sol narae cleaves extracellular Wingless to generate a novel active form that regulates cell proliferation in Drosophila. Cell Death Dis. 2019;10:564.
Janda CY, Waghray D, Levin AM, Thomas C, Garcia KC. Structural basis of Wnt recognition by Frizzled. Science. 2012;337:59–64.
Bazan JF, Janda CY, Garcia KC. Structural architecture and functional evolution of Wnts. Dev Cell. 2012;23:227–32.
von Maltzahn J, Zinoviev R, Chang NC, Bentzinger CF, Rudnicki MA. A truncated Wnt7a retains full biological activity in skeletal muscle. Nat Commun. 2013;4:2869.
Port F, Basler K. Wnt Trafficking: new Insights into Wnt Maturation, Secretion and Spreading. Traffic. 2010;11:1265–71.
Gross JC, Chaudhary V, Bartscherer K, Boutros M. Active Wnt proteins are secreted on exosomes. Nat Cell Biol. 2012;14:1036–45.
Gross JC, Boutros M. Secretion and extracellular space travel of Wnt proteins. Curr Opin Genet Dev. 2013;23:385–90.
Thery C, Zitvogel L, Amigorena S. Exosomes: composition, biogenesis and function. Nat Rev Immunol. 2002;2:569–79.
Thery C, Ostrowski M, Segura E. Membrane vesicles as conveyors of immune responses. Nat Rev Immunol. 2009;9:581–93.
Lee SS, Won JH, Lim GJ, Han J, Lee JY, Cho KO, et al. A novel population of extracellular vesicles smaller than exosomes promotes cell proliferation. Cell Commun Signal. 2019;17:95.
Beckett K, Monier S, Palmer L, Alexandre C, Green H, Bonneil E, et al. Drosophila S2 cells secrete wingless on exosome-like vesicles but the wingless gradient forms independently of exosomes. Traffic. 2013;14:82–96.
Panakova D, Sprong H, Marois E, Thiele C, Eaton S. Lipoprotein particles are required for Hedgehog and Wingless signalling. Nature. 2005;435:58–65.
Barthalay Y, Hipeau-Jacquotte R, de la Escalera S, Jimenez F, Piovant M. Drosophila neurotactin mediates heterophilic cell adhesion. EMBO J. 1990;9:3603–9.
de la Escalera S, Bockamp EO, Moya F, Piovant M, Jimenez F. Characterization and gene cloning of neurotactin, a Drosophila transmembrane protein related to cholinesterases. EMBO J. 1990;9:3593–601.
Chaudhary V, Hingole S, Frei J, Port F, Strutt D, Boutros M. Robust Wnt signaling is maintained by a Wg protein gradient and Fz2 receptor activity in the developing Drosophila wing. Development. 2019;146:dev174789.
Katanaev VL, Solis GP, Hausmann G, Buestorf S, Katanayeva N, Schrock Y, et al. Reggie-1/flotillin-2 promotes secretion of the long-range signalling forms of Wingless and Hedgehog in Drosophila. EMBO J. 2008;27:509–21.
Neumann CJ, Cohen SM. Long-range action of Wingless organizes the dorsal-ventral axis of the Drosophila wing. Development. 1997;124:871–80.
Lecuit T, Cohen SM. Proximal-distal axis formation in the Drosophila leg. Nature. 1997;388:139–45.
Kim GW, Won JH, Lee OK, Lee SS, Han JH, Tsogtbaatar O, et al. Sol narae (Sona) is a Drosophila ADAMTS involved in Wg signaling. Sci Rep. 2016;6:31863.
Couso JP, Bate M, Martinez-Arias A. A wingless-dependent polar coordinate system in Drosophila imaginal discs. Science. 1993;259:484–9.
Ng M, Diaz-Benjumea FJ, Vincent JP, Wu J, Cohen SM. Specification of the wing by localized expression of wingless protein. Nature. 1996;381:316–8.
Blair SS. A role for the segment polarity gene shaggy-zeste white 3 in the specification of regional identity in the developing wing of Drosophila. Dev Biol. 1994;162:229–44.
Phillips RG, Whittle JR. Wingless expression mediates determination of peripheral nervous system elements in late stages of Drosophila wing disc development. Development. 1993;118:427–38.
Giraldez AJ, Copley RR, Cohen SM. HSPG modification by the secreted enzyme Notum shapes the Wingless morphogen gradient. Dev Cell. 2002;2:667–76.
Evans CJ, Olson JM, Ngo KT, Kim E, Lee NE, Kuoy E, et al. G-TRACE: rapid Gal4-based cell lineage analysis in Drosophila. Nat Methods. 2009;6:603–5.
Maves L, Schubiger G. A molecular basis for transdetermination in Drosophila imaginal discs: interactions between wingless and decapentaplegic signaling. Development. 1998;125:115–24.
Klein T, Arias AM. Different spatial and temporal interactions between Notch, wingless, and vestigial specify proximal and distal pattern elements of the wing in Drosophila. Dev Biol. 1998;194:196–212.
Kooh PJ, Fehon RG, Muskavitch MA. Implications of dynamic patterns of Delta and Notch expression for cellular interactions during Drosophila development. Development. 1993;117:493–507.
Zhang Y, Liu Y, Liu H, Tang WH. Exosomes: biogenesis, biologic function and clinical potential. Cell Biosci. 2019;9:19.
Zhang ZG, Buller B, Chopp M. Exosomes - beyond stem cells for restorative therapy in stroke and neurological injury. Nat Rev Neurol. 2019;15:193–203.
Baena-Lopez LA, Franch-Marro X, Vincent JP. Wingless promotes proliferative growth in a gradient-independent manner. Sci Signal. 2009;2:ra60.
Ekstrom EJ, Bergenfelz C, von Bulow V, Serifler F, Carlemalm E, Jonsson G, et al. WNT5A induces release of exosomes containing pro-angiogenic and immunosuppressive factors from malignant melanoma cells. Mol Cancer. 2014;13:88.
Hooper C, Sainz-Fuertes R, Lynham S, Hye A, Killick R, Warley A, et al. Wnt3a induces exosome secretion from primary cultured rat microglia. BMC Neurosci. 2012;13:144.
Farin HF, Jordens I, Mosa MH, Basak O, Korving J, Tauriello DV, et al. Visualization of a short-range Wnt gradient in the intestinal stem-cell niche. Nature. 2016;530:340–3.
Yamamoto H, Awada C, Hanaki H, Sakane H, Tsujimoto I, Takahashi Y, et al. The apical and basolateral secretion of Wnt11 and Wnt3a in polarized epithelial cells is regulated by different mechanisms. J Cell Sci. 2013;126:2931–43.
Beaven R, Denholm B. Release and spread of Wingless is required to pattern the proximo-distal axis of Drosophila renal tubules. Elife. 2018;7:e35373.
Apitz H, Salecker I. Spatio-temporal relays control layer identity of direction-selective neuron subtypes in Drosophila. Nat Commun. 2018;9:2295.
Vincent JP, Magee T. Tiki casts a spell on Wnt. Cell. 2012;149:1426–7.
Sanchez-Pulido L, Ponting CP. Tiki, at the head of a new superfamily of enzymes. Bioinformatics. 2013;29:2371–4.
Zhang X, Abreu JG, Yokota C, MacDonald BT, Singh S, Coburn KL, et al. Tiki1 is required for head formation via Wnt cleavage-oxidation and inactivation. Cell. 2012;149:1565–77.
Zhang X, MacDonald BT, Gao H, Shamashkin M, Coyle AJ, Martinez RV, et al. Characterization of Tiki, a New Family of Wnt-specific Metalloproteases. J Biol Chem. 2016;291:2435–43.
Klein T. Wing disc development in the fly: the early stages. Curr Opin Genet Dev. 2001;11:470–5.
Pfeiffer S, Ricardo S, Manneville JB, Alexandre C, Vincent JP. Producing cells retain and recycle Wingless in Drosophila embryos. Curr Biol. 2002;12:957–62.
Baena-Lopez LA, Alexandre C, Mitchell A, Pasakarnis L, Vincent JP. Accelerated homologous recombination and subsequent genome modification in Drosophila. Development. 2013;140:4818–25.
Prasad M, Bajpai R, Shashidhara LS. Regulation of Wingless and Vestigial expression in wing and haltere discs of Drosophila. Development. 2003;130:1537–47.
Pignoni F, Zipursky SL. Induction of Drosophila eye development by decapentaplegic. Development. 1997;124:271–8.
Acknowledgements
We thank J. Jung and J.Y. Kim for their critical reading of this paper. We are in debt to J. P. Vincent, K. Basler, S. Eaton, S. M. Cohen, L. S. Shashidhara, R. Holmgren for fly lines, Gary Struhl, J.W. Kim, K. Basler, and J. P. Vincent for DNA constructs, and two reviewers who helped to improve this report. We thank Bloomington Stock Center, Drosophila Genetic Resource Center, Korea Drosophila Resource Center, and Developmental Studies Hybridoma Bank for fly strains and antibodies. This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2017R1A2B4009254, 2019R1H1A2039726, and 2019R1A6A1A10073887).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by E. Baehrecke
Rights and permissions
About this article
Cite this article
Won, JH., Cho, KO. Wg secreted by conventional Golgi transport diffuses and forms Wg gradient whereas Wg tethered to extracellular vesicles do not diffuse. Cell Death Differ 28, 1013–1025 (2021). https://doi.org/10.1038/s41418-020-00632-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41418-020-00632-8
- Springer Nature Limited