Abstract
Diagnostic testing for primary ciliary dyskinesia (PCD) usually includes transmission electron microscopy (TEM), nasal nitric oxide, high-speed video microscopy, and genetics. Diagnostic performance of each test should be assessed toward the development of PCD diagnostic algorithms. We systematically reviewed the literature and quantified PCD prevalence among referrals and TEM detection rate in confirmed PCD patients. Major electronic databases were searched until December 2015 using appropriate terms. Included studies described cohorts of consecutive PCD referrals in which PCD was confirmed by at least TEM and one additional test, in order to compare the index test performance with other test(s). Meta-analyses of pooled PCD prevalence and TEM detection rate across studies were performed. PCD prevalence among referrals was 32% (95% CI: 25–39%, I2 = 92%). TEM detection rate among PCD patients was 83% (95% CI: 75–90%, I2 = 90%). Exclusion of studies reporting isolated inner dynein arm defects as PCD, reduced TEM detection rate and explained an important fraction of observed heterogeneity (74%, 95% CI: 66–83%, I2 = 66%). Approximately, one third of referrals, are diagnosed with PCD. Among PCD patients, a significant percentage, at least as high as 26%, is missed by TEM, a limitation that should be accounted toward the development of an efficacious PCD diagnostic algorithm.
Similar content being viewed by others
Main
Primary ciliary dyskinesia (PCD) is caused by dysfunctional motile cilia and it is characterized by impaired mucociliary clearance which predisposes patients to recurrent respiratory infections. Patients usually suffer from lifelong rhinorrhea, chronic wet cough, progressive loss of lung function and eventually develop structural damage of the airways, and bronchiectasis (1). The main clinical manifestations that lead to consideration of PCD diagnostic testing are situs abnormalities, a history of neonatal respiratory distress syndrome, a family history of PCD, male infertility, and chronic productive cough in the absence of more common causes of chronic lung disease (2).
Confirmation of a positive PCD diagnosis remains challenging as no single diagnostic test has been shown to have 100% sensitivity and specificity, thus a combination of diagnostic tests is usually needed for a final decision (3). Specialized diagnostic testing is currently available only in few specialized centers and includes the measurement of nasal nitric oxide (nNO) (4), assessment of ciliary motility (5), and ciliary ultrastructure (6), while a few centers have also introduced genetic testing in clinical practice (7). Overall, PCD diagnostic testing is expensive and time consuming (8,9), which underlines the need to estimate the prevalence of PCD among referrals. This estimate is useful to know for cost-benefit analyses as higher prevalence of PCD among referrals corresponds to a lower proportion of non-PCD patients that are referred for PCD diagnostic testing and lower economic burden for the healthcare system and/or the patient family and vice versa. Furthermore, different centers follow various diagnostic algorithms for PCD diagnosis (10), indicating the need for the development of an evidence-based diagnostic decision tree for PCD. Such an approach requires the prior assessment of summary estimates of the diagnostic performance of each individual diagnostic test. Application of the Bayes Theorem on estimates of diagnostic performance along with information about the prior probability of disease (prevalence of PCD among referrals) will allow the calculation of positive predictive values and negative predictive values for different diagnostic tests and algorithms (11).
In the past, assessment of ciliary ultrastructure abnormalities with transmission electron microscopy (TEM) was considered to be the “gold standard” for the diagnosis of PCD (6). However, for several years now, guidelines highlight that TEM cannot be considered as a gold-standard test (2) as a substantial subset of PCD patients display normal axonemal ultrastructure and cannot be identified through TEM (12). These patients usually carry biallelic mutations in the DNAH11 gene and their ciliary motility is characterized by a flickering movement (13). Furthermore, another subset of PCD patients may also remain unidentified by TEM, as specific ultrastructural defects such as nexin link defects, are not be easily discernible by standard TEM (14). Several studies reporting TEM findings in different cohorts of PCD patients demonstrate wide variation in the percentage of PCD patients missed by TEM ranging from below 10% (15,16) to over 30% (17,18). Although some reviews and editorial papers reported that this percentage is equal to approximately 30% (3,19), this estimate is not based on a systematic review of the entire published evidence.
This study systematically reviewed the published evidence aiming to quantify the prevalence of PCD diagnosis in cohorts of suspect cases referred for PCD diagnostic testing and to estimate the diagnostic detection rate of TEM in PCD patients in whom diagnosis was confirmed with a combination of tests.
Methods
Search Strategy and Selection Criteria
The electronic databases PubMed, SCOPUS and Google Scholar were searched from inception until December 2015 using combinations of the keywords “Electron Microscopy” and “Ciliary Motility Disorders” as Medical Subject Headings (MeSH) or individual terms and “Primary Ciliary Dyskinesia” OR “PCD” and combinations either in the title or in the abstract. The reference lists of the retrieved studies and reviews were further searched for additional reports. The included studies were identified after two reviewers (P.K., S.I.P.) independently screened the title and abstract of the obtained electronic search results and final selection was based on full text evaluation. A third researcher (P.K.Y.) resolved any discrepancies. The guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) were followed (20).
For the prevalence of PCD among referrals estimate, studies were selected according to the following inclusion criteria: Cohorts of consecutive referrals for PCD testing and, in addition to TEM, at least one more test such as high-speed video microscopy (HSVM), nNO, or genetic testing for confirmation of PCD diagnosis.
For the detection rate of TEM among confirmed PCD patients estimate, studies were selected according to the following criteria: Cohorts of consecutive PCD patients, reporting of TEM findings and confirmation of PCD diagnosis with at least one more test such as HSVM, nNO, or genetic testing.
Confirmation of PCD diagnosis was set to rely on TEM and at least one additional test, thus potentiating the comparison of the index test against other diagnostic test(s). Studies that confirmed PCD with TEM only were excluded. Studies with overlapping patient populations were cross-checked and only the study with the largest and most recent population was selected.
Data Extraction
The year of publication, name of author, study design, country of origin, study population, number of patients referred for PCD testing, number of patients confirmed as PCD, and number of patients with a reported ultrastructural defect identified by TEM were recorded. The distribution of ultrastructural defects among the TEM-positive patients and the age range of participating patients were recorded additionally where available. The data were extracted independently by two reviewers (P.K., S.I.P.) and consensus was reached for all data. Reporting of the included studies underwent quality assessment based on the Meta-analysis Of Observational Studies in Epidemiology (MOOSE) criteria (21). Not all of the MOOSE criteria were examined, as the assessment of reporting quality involved only the specific criteria that were relevant to the purposes of this study. Quality characteristics were assessed descriptively in order to detect any low quality evidence that could influence the results.
Analysis
In order to combine the data on PCD prevalence among patients referred for PCD diagnostic testing, we performed a meta-analysis of proportions using a random effects model. Meta-analysis of proportions allows the calculation of the pooled prevalence of PCD across studies containing binomial data with the numerator defined as the number of PCD patients identified by a combination of diagnostic tests (TEM and nNO or HSVM or genetic testing) and the denominator as the total number of consecutive referrals for PCD testing (22). The random effects allow for each study to be assigned a weight which includes the within study variance and the between studies variance (23). Furthermore, when only limited numbers of studies were available, and heterogeneity was I2 > 0%, we applied the Hartung, Knapp, Sidik, and Jonkman (HKSJ) approach which uses a Student T distribution, instead of a Normal distribution for the effects’ estimates. This method applies an ad-hoc correction and yields more conservative results (24).
The same method was used for the estimation of the pooled percentage of patients that are identified by TEM with the numerator defined as the number of PCD patients identified by abnormal TEM and the denominator defined as the number of PCD patients identified by a combination of diagnostic tests (TEM and nNO or HSVM or genetic testing). Lastly, the fraction of PCD patients with different ultrastructural defects (isolated ODA, combined ODA+IDA, MTD) was calculated.
Heterogeneity was assessed with the I2 which describes the proportion of total variation in the effect estimate that results from the between-studies heterogeneity and ranges from 0 to 100% (25). Subgroup analysis was planned a priori based on factors that (i) could affect referral or diagnostic patterns, (ii) could lead to a different cohort of referrals, or (iii) could detect a different spectrum of the examined disease. Such factors were the region specific referral patterns (in series of tests results from a specific country were excluded one at a time), the number of tests used to confirm PCD (exclusion of studies that performed only two tests), the sample size (exclusion of studies with number of referrals below the median number of referrals of all included studies) and whether an isolated Inner Dynein Arm (IDA) defect was considered diagnostic for PCD (exclusion of studies that reported isolated IDA defects as diagnostic). Isolated IDA defects as a diagnostic feature for PCD remains to date controversial because IDA are usually characterized by low contrast (6,26). In addition, none of the reported 32 genes, which harbor pathogenic mutations for PCD, have been found to affect only IDA (27). All calculations were performed using STATA (Version 12, StataCorp, College Station, TX) with the command metaprop for binomial data (22).
Results
Eligible Studies
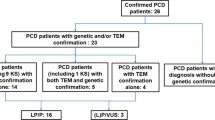
A total of 2,253 studies were retrieved through online search and 6 additional studies were identified through references’ screening. Among the retrieved studies, 2,097 were excluded based on title or abstract. Of the 161 studies that were assessed in full detail, 12 were review studies, 3 were guidelines or editorials, 56 were case–control or case–series studies, 33 did not include data on TEM results and 5 did not include data on PCD patients. A total of 36 studies were included in the qualitative synthesis. Of these, 25 studies were excluded at the last step and 11 were eventually included in the quantitative synthesis ( Figure 1 ). The 25 studies that were excluded at the last step prior to quantitative synthesis, as well as the main reason for their exclusion, are presented separately in Supplementary Table S1 online. The majority of the studies (n = 19) presented in Supplementary Table S1 online, were excluded from further analysis because PCD diagnosis was established only based on the TEM results, although in some of these studies (n = 8), additional tests were partly also performed.
PRISMA diagram. PRISMA diagram for the search strategy and selected studies.
Study Characteristics
Included studies and their descriptive characteristics are presented in Table 1 . All the studies confirmed PCD diagnosis with at least one more test in addition to TEM. The vast majority of studies used HSVM as an additional test with the exception of Leigh et al. which used genetic testing (16). Furthermore, approximately 36% of the studies also included nNO in the diagnostic work-up. Three studies were performed in the United Kingdom (15,28,29) and three studies were performed in the United States of America (16,30,31) while the remaining were performed in other European countries. The majority of the studies provided information about the number of consecutive referrals that underwent PCD diagnostic testing and were included in the meta-analysis regarding PCD prevalence among cohorts of respiratory referrals (n = 2,475) (15,18,28,29,30,31,32). The study by Stannard et al. although focused on PCD patients with a positive TEM diagnosis, it also performed HSVM and reported patients diagnosed with abnormal beating and normal ultrastructure and as a result, it was also included in the PCD prevalence meta-analysis (15). All the included studies, with the exception of Shappiro et al. (30) provided data on ultrastructural assessment for all the PCD diagnosed patients and were also included in the meta-analysis of the TEM detection rate (n = 728) (4,15,16,18,28,29). The most common ultrastructural findings in the assessed studies were isolated Outer Dynein Arm (ODA) defects and combined ODA and IDA (ODA+IDA) defects as well as tubular defects and normal ultrastructure (NU). A subset of studies also reported isolated IDA defects (15,28,33). Lastly, two of the included studies also reported a phenotype with lack of multiple cilia (acilia) in some patients (15,17).
Assessment of Reporting Quality
Quality assessment results for the included studies are presented in Figure 2 . Overall, the analyzed studies were characterized by good methodology and all of them appropriately described the diagnostic tests performed. On the other hand, there were some studies that did not report the recruitment period or did not describe in detail the patients’ characteristics. Only a few of the studies discussed potential study limitations although this was expected, as most of them were of a descriptive nature. Finally, a small number of studies (16,28,29,31,32) reported some efforts to reduce bias during the evaluation of diagnostic tests such as blinded assessment or assessment by more than one evaluator. The results of reporting quality assessments for each study are presented in detail in Supplementary Table S2 online.
Quality assessment results. Reporting quality assessment results for the included studies.
Data Synthesis
The pooled prevalence of newly diagnosed PCD patients in cohorts of consecutive referrals of suspect cases was 32% (95% CI: 25–39%, I2 = 92%) ( Figure 3 ). The ad-hoc correction using HKSJ resulted in the same pooled prevalence estimate but wider confidence intervals (32%, 95% CI: 20–44%, I2 = 92%) A series of subgroup analyses were performed by excluding each time studies from each individual country, studies that performed only two tests for PCD confirmation, and studies with low sample size. Overall, subgroup analyses did not yield significant differences from the original analysis (Supplementary Figures S1 and S2 online) but the heterogeneity in the effect estimate was explained by the exclusion of UK studies (prevalence: 41%, 95% CI: 37–45%, I2 = 0%) (Supplementary Figure S3 online).
Forest plot of the prevalence of primary ciliary dyskinesia (PCD) among cohorts of suspect patients. Forest plot of the proportion of referred patients for PCD testing that have eventually PCD confirmed.
The detection rate of TEM in PCD diagnosed patients was 83% (95% CI: 75–90%, I2 = 90%) ( Figure 4 ). The detection rate of TEM in PCD after the ad-hoc HKSJ correction was also 83% (95% CI: 74–92%, I2 = 90%). Subgroup analyses were also performed for this estimate by excluding each time, studies that performed only two tests for PCD confirmation, studies with low sample size and studies which reported isolated IDA defects. The subgroup analyses, with the exception of one, did not demonstrate significant differences compared to the original analysis (Supplementary Figures S4 and S5 online). The subgroup analysis that included studies that did not report isolated IDA defects resulted in a marked reduction in the detection rate of TEM and explained an important fraction of the observed heterogeneity (detection rate: 74%, 95% CI: 66–83%, I2 = 66%) ( Figure 5 ). Similarly as before, the HKSJ ad-hoc correction resulted in wider confidence intervals (detection rate: 74%, 95% CI: 61–87%, I2 = 66%).
Forest plot of the detection rate of transmission electron microscopy (TEM) across all studies. Forest plot of the detection rate of TEM in cohorts of patients that have primary ciliary dyskinesia confirmed with a combination of diagnostic tests (all included studies).
Forest plot of the detection rate of transmission electron microscopy (TEM) across studies excluding IDA. Forest Plot of the detection rate of TEM across the included studies that did not report an isolated IDA defect.
Among PCD patients, an isolated ODA defect was identified in 28% (95% CI: 19–36%, I2 = 80%) (Supplementary Figure S6 online) while a combined ODA+IDA defect was identified in 26% (95% CI: 14–39%, I2 = 95%) (Supplementary Figure S7 online). HKSJ ad-hoc correction resulted in an estimate of 28% (95% CI: 18–38%) for the fraction of patients with isolated ODA and in an estimate of 26% (95% CI: 9–43%) for the patients with a combined ODA+IDA defect. The fraction of patients identified with a tubular defect was 10% (95% CI: 3–18%, I2 = 93%, HKSJ ad-hoc correction: 10%, 95% CI: 0–25%) (Supplementary Figure S8 online).
Discussion
In this systematic review and meta-analysis, we found the pooled prevalence of PCD diagnosis in cohorts of consecutive referrals of suspect cases for PCD diagnostic testing to be 32%. This finding suggests that one third of the patients suspected for PCD, are indeed affected by the disease. The high heterogeneity observed in the estimate can be at least partly explained by the inhomogeneity in diagnostic protocols and referral patterns between the different studies. Some studies used different number or combination of diagnostic tests for eliciting PCD diagnosis. The study by Shappiro et al. used TEM along with genetics and nNO for confirmation of PCD diagnosis (30) while other studies used TEM and HSVM (15,17,31,33,34) or TEM, HSVM, and nNO (18,28,29,32). Different referral patterns between the included studies may have also influenced the heterogeneity in the final estimate. Although the referral of patients for PCD diagnostic testing should be based on combinations of classical PCD features (3), to date, the decision to refer patients does not result from a suspect manifestations scoring system but rather from the clinicians’ awareness of PCD and personal experience with the disease. The lack of such a scoring system is reflected in differences in the referral patterns across different countries or different centers (18,35,36). The recent publication by Leigh et al. regarding the association of specific clinical features with the likelihood of PCD (37) and the development of PICADAR clinical scoring tool (38) constitute the first steps toward the introduction of a universal clinical scoring system and referral algorithm for PCD in the primary care clinical setting. The performance of future referral algorithms can be compared with the current estimate of the prevalence of PCD among referrals reported here, which is essentially compromised by the variability in the referral patterns of the different centers.
Furthermore, among consecutive diagnosed PCD patients that underwent ultrastructural assessment, we calculated the detection rate of TEM to be 83%. In all the included studies, positive PCD diagnosis was based on a combination of at least two or three diagnostic tests and ciliary ultrastructural assessment using TEM was part of the diagnostic work-up. The estimated detection rate means that approximately 17% of PCD patients do not exhibit ciliary abnormalities on TEM analysis. This analysis also displayed significant heterogeneity across studies. In subgroup analysis, after exclusion of studies which reported isolated IDA defects as an abnormal diagnostic TEM finding, we found the detection rate estimate to be 74% and the heterogeneity to be markedly reduced. The resulting pooled estimate suggests that 26% of PCD patients do not exhibit abnormal ultrastructure and this estimate is much closer to the empirically quoted estimate of 30% (19). Among the confirmed PCD patients, 28% were characterized by an isolated ODA defect, 26% were characterized by a combined ODA+IDA defect and 10% by tubular defects. However, among the remaining two categories of ultrastructural defects reported in the individual studies, Central Pair (CP) and Radial Spoke (RS) defects, it is possible that some of the patients also have microtubular disorganization (MTD) as some genetic mutations result in both or either CP and MTD defects through the disruption of radial spokes (39,40). As a result, the estimated 10% fraction for tubular defects could be an underestimation, probably affected by the underlying categorization of certain TEM defects such as CP, RS, and MTD within the included studies.
The results of this analysis suggest that PCD diagnosis cannot rely only on TEM examination. As a test of characterization of morphologic features, TEM holds substantial subjectivity and may be influenced by the overall quality of the obtained sample (41). The other routinely used PCD diagnostic tests, the measurement of nNO and HSVM, have both been reported to perform better than TEM. More specifically, a recent meta-analysis of nNO measurements in PCD patients has demonstrated a pooled sensitivity of 93 to 95% depending on vellum closure status during testing (42) while a number of studies have reported high sensitivity values for HSVM ranging from 89 (43) to 100% (29). Although our findings suggest that PCD diagnosis should not rely only on TEM, performance of this test may still be beneficial since determination of an ultrastructural defect or confirmation of its absence may guide genetic testing wherever this is available. This is particularly important for centers that lack access to whole-exome or whole-genome sequencing and rely on genotyping of specific PCD genes. In this setting, prior identification of ultrastructural defects allows prioritization of which genes to be sequenced based on known genotype-TEM findings correlations (44). In addition, recent studies have suggested associations between the ultrastructural phenotype and severity of clinical features and disease progression (17,45), thus highlighting the potential of TEM analysis to facilitate the identification of clinically significant phenotypic subgroups among PCD patients. Furthermore, a recent study by Knowles et al. has highlighted the milder clinical phenotype in PCD individuals with bi-allelic mutations in RSPH1 and mainly CP defects in a small subset of cross-sections (46).
IDA imaging with TEM is more difficult compared to ODA due to the low contrast of IDA coupled with the frequent presence of nonspecific biological or technical artifacts (6,26). Furthermore, as ODA and IDA are multiprotein complexes of different axonemal dynein polypeptides which include heavy, intermediate, and light chain polypeptides, the composition and variability of which, may affect the visualization of these structures under TEM. In more detail, it has been shown that IDA composition is more diverse compared to ODA (47) as well as that the periodicity of IDA is higher compared to the periodicity of ODA along the axoneme (48). As a result specific tools have been proposed in order to allow for the clearer visualization of IDA (and ODA) in electron micrographs such as averaged TEM pictures (49) and Markham rotation (50) although none has received widespread application. Overall, the presence of an isolated IDA defect in PCD remains controversial as, up to now, none of the reported 32 genes which harbor pathogenic mutations for PCD, has been found to affect only IDA (27). Several genes affect both ODA and IDA (27), while CCDC39 and CCDC40, which have been recently shown to cause loss of IDA, cause as well disruption of the axonemal organization (51) and more severe disease (45). As a result, the analysis which excluded studies reporting isolated IDA defects provides a more reliable estimate of the TEM detection rate.
This is to our knowledge, the first study that summarizes the evidence from cohorts of consecutive referrals and informs about the prevalence of PCD among these cohorts and the detection rate of TEM among these patients. This meta-analysis benefited from including data from a large number of suspect cases referrals and PCD cases from many centers. However, as most of the included studies were retrospective, the possibility of selection or misclassification bias cannot be ruled out. Furthermore, it is acknowledged that, although the number of studies reporting TEM findings in PCD patients is quite extensive, the studies that were finally included in this meta-analysis are only eleven. However, the goal of this systematic review was to estimate the detection rate of TEM in PCD confirmed cases, thus the included studies should have had both criteria, consecutive referrals and the PCD diagnosis confirmed by a combination of diagnostic tests and not only by TEM. This design may have led to a smaller number of included studies but it enabled the calculation of a more reliable estimate for the detection rate of TEM in PCD. In 8 of the 10 included cohorts, it was clearly stated that all PCD patients were diagnosed by at least one more diagnostic test in addition to TEM. In the remainder two cohorts (Leigh et al. (16) and Shoemark et al. (28)), a fraction of their patients were not diagnosed by another diagnostic test (in addition to TEM). This represents a limitation of the analysis probably leading to a slight overestimation of the diagnostic performance of TEM. The TEM detection rate in the sensitivity analysis which excluded cohorts reporting isolated IDA defects as a diagnostic finding is not affected, as these two studies (16,28) were not included in the analysis.
In this analysis, for the vast majority of PCD patients who had normal ultrastructure and were missed by TEM, the genetic defect is not specified. This represents an important limiting factor as we do not know if all the responsible genetic defects known to date to cause PCD and normal cilia structure were represented in this subgroup. In a recent review of different PCD populations, the frequency of genetic defects, which are known to cause PCD but not detectable ultrastructure changes by TEM, was found to be approximately 30% (52). This percentage is slightly higher but close to the 26% reported here. Additional studies in large cohorts of PCD patients, reporting diagnostic tests results and responsible genetic defects are needed to inform about the precise diagnostic accuracy of TEM.
In summary, among cohorts of consecutive referrals of suspect cases for PCD testing, approximately one third are eventually confirmed as PCD patients. Among PCD cases that underwent TEM studies, a significant percentage, at least as high as 26%, were not identified by TEM. This limitation of TEM should be taken into account during the development of a universal and efficacious diagnostic algorithm for PCD.
Author Contributions
P.K. performed the search and extraction of information from the papers, organized the material, performed the statistical analysis, and prepared the first draft of the manuscript. P.K.Y. conceived the hypothesis of the manuscript, contributed to the interpretation of the findings and contributed intellectually towards the final version of the manuscript. N.M. and J.S.E contributed to the interpretation of the findings and critically revised the final version of the manuscript. K.K. provided advice during study design, contributed to the interpretation of findings and contributed towards the final version of the manuscript. S.I.P. coordinated the whole work, provided advice on the methodology of the search and selection of the papers, contributed to the interpretation of the findings, critically revised the manuscript and contributed towards the final version of the manuscript.
Statement of Financial Support
This study was supported by EU 7th Framework Program EC-GA No. 305404 BESTCILIA.
Disclosure
There are no potential or perceived conflicts of interest to disclose.
References
Boon M, Jorissen M, Proesmans M, De Boeck K. Primary ciliary dyskinesia, an orphan disease. Eur J Pediatr 2013;172:151–62.
Barbato A, Frischer T, Kuehni CE, et al. Primary ciliary dyskinesia: a consensus statement on diagnostic and treatment approaches in children. Eur Respir J 2009;34:1264–76.
Lucas JS, Burgess A, Mitchison HM, Moya E, Williamson M, Hogg C ; National PCD Service, UK. Diagnosis and management of primary ciliary dyskinesia. Arch Dis Child 2014;99:850–6.
Boon M, Meyts I, Proesmans M, Vermeulen FL, Jorissen M, De Boeck K. Diagnostic accuracy of nitric oxide measurements to detect primary ciliary dyskinesia. Eur J Clin Invest 2014;44:477–85.
Santamaria F, de Santi MM, Grillo G, Sarnelli P, Caterino M, Greco L. Ciliary motility at light microscopy: a screening technique for ciliary defects. Acta Paediatr 1999;88:853–7.
Leigh MW, O’Callaghan C, Knowles MR. The challenges of diagnosing primary ciliary dyskinesia. Proc Am Thorac Soc 2011;8:434–7.
Kim RH, A Hall D, Cutz E, et al. The role of molecular genetic analysis in the diagnosis of primary ciliary dyskinesia. Ann Am Thorac Soc 2014;11:351–9.
Lucas JS, Paff T, Goggin P, Haarman E. Diagnostic methods in primary ciliary dyskinesia. Paediatr Respir Rev 2016;18:8–17.
Hogg C. Primary ciliary dyskinesia: when to suspect the diagnosis and how to confirm it. Paediatr Respir Rev 2009;10:44–50.
Lucas JS, Leigh MW. Diagnosis of primary ciliary dyskinesia: searching for a gold standard. Eur Respir J 2014;44:1418–22.
Sox HC, Blatt MA, Higgins MC, Marton KI. Medical decision making. Philadelphia, PA: ACP Press 2007
Horani A, Brody SL, Ferkol TW. Picking up speed: advances in the genetics of primary ciliary dyskinesia. Pediatr Res 2014;75:158–64.
Schwabe GC, Hoffmann K, Loges NT, et al. Primary ciliary dyskinesia associated with normal axoneme ultrastructure is caused by DNAH11 mutations. Hum Mutat 2008;29:289–98.
Olbrich H, Cremers C, Loges NT, et al. Loss-of-function GAS8 mutations cause primary ciliary dyskinesia and disrupt the nexin-dynein regulatory complex. Am J Hum Genet 2015;97:546–54.
Stannard WA, Chilvers MA, Rutman AR, Williams CD, O’Callaghan C. Diagnostic testing of patients suspected of primary ciliary dyskinesia. Am J Respir Crit Care Med 2010;181:307–14.
Leigh MW, Hazucha MJ, Chawla KK, et al. Standardizing nasal nitric oxide measurement as a test for primary ciliary dyskinesia. Ann Am Thorac Soc 2013;10:574–81.
Boon M, Smits A, Cuppens H, et al. Primary ciliary dyskinesia: critical evaluation of clinical symptoms and diagnosis in patients with normal and abnormal ultrastructure. Orphanet J Rare Dis 2014;9:11.
Yiallouros PK, Kouis P, Middleton N, et al. Clinical features of primary ciliary dyskinesia in Cyprus with emphasis on lobectomized patients. Respir Med 2015;109:347–56.
Werner C, Onnebrink JG, Omran H. Diagnosis and management of primary ciliary dyskinesia. Cilia 2015;4:2.
Moher D, Liberati A, Tetzlaff J, Altman DG ; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 2009;151:264–9, W64.
Stroup DF, Berlin JA, Morton SC, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 2000;283:2008–12.
Nyaga VN, Arbyn M, Aerts M. Metaprop: a Stata command to perform meta-analysis of binomial data. Arch Public Health 2014;72:39.
Borenstein M, Hedges L, Rothstein H. Meta-analysis: fixed effect vs. random effects, 2007. https://www.meta-analysis.com/downloads/Meta-analysis%20fixed%20effect%20vs%20random%20effects.pdf.
IntHout J, Ioannidis JP, Borm GF. The Hartung-Knapp-Sidik-Jonkman method for random effects meta-analysis is straightforward and considerably outperforms the standard DerSimonian-Laird method. BMC Med Res Methodol 2014;14:25.
Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med 2002;21:1539–58.
O’Callaghan C, Rutman A, Williams GM, Hirst RA. Inner dynein arm defects causing primary ciliary dyskinesia: repeat testing required. Eur Respir J 2011;38:603–7.
Kurkowiak M, Ziętkiewicz E, Witt M. Recent advances in primary ciliary dyskinesia genetics. J Med Genet 2015;52:1–9.
Shoemark A, Dixon M, Corrin B, Dewar A. Twenty-year review of quantitative transmission electron microscopy for the diagnosis of primary ciliary dyskinesia. J Clin Pathol 2012;65:267–71.
Jackson CL, Behan L, Collins SA, et al. Accuracy of diagnostic testing in primary ciliary dyskinesia. Eur Respir J 2016;47:837–48.
Shapiro AJ, Davis S, Olivier K, et al. Clinical symptoms associated with primary ciliary dyskinesia–results of a multi-centered study. Am J Respir Crit Care Med 2010;181:A6728
Olm MA, Kögler JE Jr, Macchione M, Shoemark A, Saldiva PH, Rodrigues JC. Primary ciliary dyskinesia: evaluation using cilia beat frequency assessment via spectral analysis of digital microscopy images. J Appl Physiol (1985) 2011;111:295–302.
Pifferi M, Bush A, Caramella D, et al. Agenesis of paranasal sinuses and nasal nitric oxide in primary ciliary dyskinesia. Eur Respir J 2011;37:566–71.
Djakow J, Svobodová T, Hrach K, Uhlík J, Cinek O, Pohunek P. Effectiveness of sequencing selected exons of DNAH5 and DNAI1 in diagnosis of primary ciliary dyskinesia. Pediatr Pulmonol 2012;47:864–75.
Nauta F, Pals G, Daniels H, Nagelkerke A, Haarman E. Diagnosis of primary ciliary dyskinesia in a Dutch cohort of 63 pediatric patients: an overview. Eur Respir J 2011;38(Suppl 55):511
O’Callaghan C, Chilvers M, Hogg C, Bush A, Lucas J. Diagnosing primary ciliary dyskinesia. Thorax 2007;62:656–7.
Kuehni CE, Frischer T, Strippoli MP, et al.; ERS Task Force on Primary Ciliary Dyskinesia in Children. Factors influencing age at diagnosis of primary ciliary dyskinesia in European children. Eur Respir J 2010;36:1248–58.
Leigh MW, Ferkol TW, Davis SD, et al. Clinical features and associated likelihood of primary ciliary dyskinesia in children and adolescents. Ann Am Thorac Soc 2016;13:1305–13.
Behan L, Dimitrov BD, Kuehni CE, et al. PICADAR: a diagnostic predictive tool for primary ciliary dyskinesia. Eur Respir J 2016;47:1103–12.
Onoufriadis A, Shoemark A, Schmidts M, et al.; UK10K. Targeted NGS gene panel identifies mutations in RSPH1 causing primary ciliary dyskinesia and a common mechanism for ciliary central pair agenesis due to radial spoke defects. Hum Mol Genet 2014;23:3362–74.
Castleman VH, Romio L, Chodhari R, et al. Mutations in radial spoke head protein genes RSPH9 and RSPH4A cause primary ciliary dyskinesia with central-microtubular-pair abnormalities. Am J Hum Genet 2009;84:197–209.
Olin JT, Burns K, Carson JL, et al.; Genetic Disorders of Mucociliary Clearance Consortium. Diagnostic yield of nasal scrape biopsies in primary ciliary dyskinesia: a multicenter experience. Pediatr Pulmonol 2011;46:483–8.
Kouis P, Papatheodorou SI, Yiallouros PK. Diagnostic accuracy of nasal nitric oxide for establishing diagnosis of primary ciliary dyskinesia: a meta-analysis. BMC Pulm Med 2015;15:153.
Boon M, Jorissen M, Jaspers M, Cuppens H, De Boeck K. Is the sensitivity of primary ciliary dyskinesia detection by ciliary function analysis 100%? Eur Respir J 2013;42:1159–61.
Horani A, Ferkol TW, Dutcher SK, Brody SL. Genetics and biology of primary ciliary dyskinesia. Paediatr Respir Rev 2016;18:18–24.
Davis SD, Ferkol TW, Rosenfeld M, et al. Clinical features of childhood primary ciliary dyskinesia by genotype and ultrastructural phenotype. Am J Respir Crit Care Med 2015;191:316–24.
Knowles MR, Ostrowski LE, Leigh MW, et al. Mutations in RSPH1 cause primary ciliary dyskinesia with a unique clinical and ciliary phenotype. Am J Respir Crit Care Med 2014;189:707–17.
Ibañez-Tallon I, Heintz N, Omran H. To beat or not to beat: roles of cilia in development and disease. Hum Mol Genet 2003;12 Spec No 1:R27–35.
Bui KH, Sakakibara H, Movassagh T, Oiwa K, Ishikawa T. Asymmetry of inner dynein arms and inter-doublet links in Chlamydomonas flagella. J Cell Biol 2009;186:437–46.
Funkhouser WK 3rd, Niethammer M, Carson JL, et al. A new tool improves diagnostic test performance for transmission em evaluation of axonemal dynein arms. Ultrastruct Pathol 2014;38:248–55.
Escudier E, Couprie M, Duriez B, et al. Computer-assisted analysis helps detect inner dynein arm abnormalities. Am J Respir Crit Care Med 2002;166:1257–62.
Antony D, Becker-Heck A, Zariwala MA, et al.; Uk10k. Mutations in CCDC39 and CCDC40 are the major cause of primary ciliary dyskinesia with axonemal disorganization and absent inner dynein arms. Hum Mutat 2013;34:462–72.
Zariwala MA, Knowles MR, Leigh MW. Primary ciliary dyskinesia. In: Pagon RA, Adam MP, Ardinger HH, et al., eds. GeneReviews(R). Seattle, WA: University of Washington, 2015.
Acknowledgements
The sponsors had no role or involvement in study design; in the collection, analysis and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Table S1
(DOCX 20 kb)
Supplementary Table S2
(DOCX 13 kb)
Supplementary Figures
(PDF 762 kb)
Rights and permissions
About this article
Cite this article
Kouis, P., Yiallouros, P., Middleton, N. et al. Prevalence of primary ciliary dyskinesia in consecutive referrals of suspect cases and the transmission electron microscopy detection rate: a systematic review and meta-analysis. Pediatr Res 81, 398–405 (2017). https://doi.org/10.1038/pr.2016.263
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pr.2016.263
- Springer Nature America, Inc.
This article is cited by
-
Recognizing clinical features of primary ciliary dyskinesia in the perinatal period
Journal of Perinatology (2024)
-
Clinical and genetic features of primary ciliary dyskinesia in a cohort of consecutive clinically suspect children in western China
BMC Pediatrics (2022)
-
Motile ciliopathies
Nature Reviews Disease Primers (2020)
-
Cost-effectiveness analysis of three algorithms for diagnosing primary ciliary dyskinesia: a simulation study
Orphanet Journal of Rare Diseases (2019)
-
Primary ciliary dyskinesia in Japan: systematic review and meta-analysis
BMC Pulmonary Medicine (2019)