Abstract
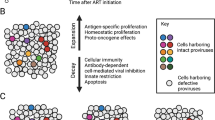
Although antiretroviral therapy (ART) suppresses viral replication to clinically undetectable levels, human immunodeficiency virus type 1 (HIV-1) persists in CD4+ T cells in a latent form that is not targeted by the immune system or by ART1,2,3,4,5. This latent reservoir is a major barrier to curing individuals of HIV-1 infection. Many individuals initiate ART during chronic infection, and in this setting, most proviruses are defective6. However, the dynamics of the accumulation and the persistence of defective proviruses during acute HIV-1 infection are largely unknown. Here we show that defective proviruses accumulate rapidly within the first few weeks of infection to make up over 93% of all proviruses, regardless of how early ART is initiated. By using an unbiased method to amplify near-full-length proviral genomes from HIV-1-infected adults treated at different stages of infection, we demonstrate that early initiation of ART limits the size of the reservoir but does not profoundly affect the proviral landscape. This analysis allows us to revise our understanding of the composition of proviral populations and estimate the true reservoir size in individuals who were treated early versus late in infection. Additionally, we demonstrate that common assays for measuring the reservoir do not correlate with reservoir size, as determined by the number of genetically intact proviruses. These findings reveal hurdles that must be overcome to successfully analyze future HIV-1 cure strategies.




Similar content being viewed by others
References
Finzi, D. et al. Identification of a reservoir for HIV-1 in patients on highly active antiretroviral therapy. Science 278, 1295–1300 (1997).
Chun, T.W. et al. Presence of an inducible HIV-1 latent reservoir during highly active antiretroviral therapy. Proc. Natl. Acad. Sci. USA 94, 13193–13197 (1997).
Wong, J.K. et al. Recovery of replication-competent HIV despite prolonged suppression of plasma viremia. Science 278, 1291–1295 (1997).
Siliciano, J.D. et al. Long-term follow-up studies confirm the stability of the latent reservoir for HIV-1 in resting CD4+ T cells. Nat. Med. 9, 727–728 (2003).
Ruelas, D.S. & Greene, W.C. An integrated overview of HIV-1 latency. Cell 155, 519–529 (2013).
Ho, Y.-C. et al. Replication-competent non-induced proviruses in the latent reservoir increase barrier to HIV-1 cure. Cell 155, 540–551 (2013).
Finzi, D. et al. Latent infection of CD4+ T cells provides a mechanism for lifelong persistence of HIV-1, even in patients on effective combination therapy. Nat. Med. 5, 512–517 (1999).
Crooks, A.M. et al. Precise quantitation of the latent HIV-1 reservoir: implications for eradication strategies. J. Infect. Dis. 212, 1361–1365 (2015).
Archin, N.M. & Margolis, D.M. Emerging strategies to deplete the HIV reservoir. Curr. Opin. Infect. Dis. 27, 29–35 (2014).
Deeks, S.G. HIV: shock and kill. Nature 487, 439–440 (2012).
Archin, N.M. et al. Administration of vorinostat disrupts HIV-1 latency in patients on antiretroviral therapy. Nature 487, 482–485 (2012).
Siliciano, J.D. & Siliciano, R.F. Enhanced culture assay for detection and quantitation of latently infected, resting CD4+ T cells carrying replication-competent virus in HIV-1-infected individuals. Methods Mol. Biol. 304, 3–15 (2005).
Laird, G.M. et al. Rapid quantification of the latent reservoir for HIV-1 using a viral outgrowth assay. PLoS Pathog. 9, e1003398 (2013).
Strain, M.C. et al. Highly precise measurement of HIV DNA by droplet digital PCR. PLoS One 8, e55943 (2013).
Rouzioux, C., Mélard, A. & Avéttand-Fénoël, V. in Human Retroviruses (eds. Vicenzi, E. & Poli, G.) 1087, 261–270 (Humana Press, 2013).
Henrich, T.J., Gallien, S., Li, J.Z., Pereyra, F. & Kuritzkes, D.R. Low-level detection and quantitation of cellular HIV-1 DNA and 2-LTR circles using droplet digital PCR. J. Virol. Methods 186, 68–72 (2012).
Eriksson, S. et al. Comparative analysis of measures of viral reservoirs in HIV-1 eradication studies. PLoS Pathog. 9, e1003174 (2013).
Günthard, H.F. et al. Antiretroviral treatment of adult HIV infection: 2014 recommendations of the International Antiviral Society–USA Panel. J. Am. Med. Assoc. 312, 410–425 (2014).
Archin, N.M. et al. Immediate antiviral therapy appears to restrict resting CD4+ cell HIV-1 infection without accelerating the decay of latent infection. Proc. Natl. Acad. Sci. USA 109, 9523–9528 (2012).
Jain, V. et al. Antiretroviral therapy initiated within 6 months of HIV infection is associated with lower T cell activation and smaller HIV reservoir size. J. Infect. Dis. 208, 1202–1211 (2013).
Deng, K. et al. Broad CTL response is required to clear latent HIV-1 due to dominance of escape mutations. Nature 517, 381–385 (2015).
Delviks-Frankenberry, K. et al. Mechanisms and factors that influence high-frequency retroviral recombination. Viruses 3, 1650–1680 (2011).
Jetzt, A.E. et al. High rate of recombination throughout the human immunodeficiency virus type 1 genome. J. Virol. 74, 1234–1240 (2000).
Yu, H., Jetzt, A.E., Ron, Y., Preston, B.D. & Dougherty, J.P. The nature of human immunodeficiency virus type 1 strand transfers. J. Biol. Chem. 273, 28384–28391 (1998).
Hwang, C.K., Svarovskaia, E.S. & Pathak, V.K. Dynamic copy choice: steady state between murine leukemia virus polymerase and polymerase-dependent RNase H activity determines frequency of in vivo template switching. Proc. Natl. Acad. Sci. USA 98, 12209–12214 (2001).
Yu, Q. et al. Single-strand specificity of APOBEC3G accounts for minus strand deamination of the HIV genome. Nat. Struct. Mol. Biol. 11, 435–442 (2004).
Kieffer, T.L. et al. G–>A hypermutation in protease and reverse transcriptase regions of human immunodeficiency virus type 1 residing in resting CD4+ T cells in vivo. J. Virol. 79, 1975–1980 (2005).
Pierson, T.C. et al. Molecular characterization of preintegration latency in human immunodeficiency virus type 1 infection. J. Virol. 76, 8518–8531 (2002).
Pillai, S.K. et al. Role of retroviral restriction factors in the interferon-α-mediated suppression of HIV-1 in vivo. Proc. Natl. Acad. Sci. USA 109, 3035–3040 (2012).
Harper, M.S. et al. Interferon-α subtypes in an ex vivo model of acute HIV-1 infection: expression, potency and effector mechanisms. PLoS Pathog. 11, e1005254 (2015).
Stacey, A.R. et al. Induction of a striking systemic cytokine cascade prior to peak viremia in acute human immunodeficiency virus type 1 infection, in contrast to more modest and delayed responses in acute hepatitis B and C virus infections. J. Virol. 83, 3719–3733 (2009).
Kearney, M.F. et al. Origin of rebound plasma HIV includes cells with identical proviruses that are transcriptionally active before stopping of antiretroviral therapy. J. Virol. 90, 1369–1376 (2016).
Maldarelli, F. et al. HIV latency. Specific HIV integration sites are linked to clonal expansion and persistence of infected cells. Science 345, 179–183 (2014).
Wagner, T.A. et al. HIV latency. Proliferation of cells with HIV integrated into cancer genes contributes to persistent infection. Science 345, 570–573 (2014).
Schröder, A.R.W. et al. HIV-1 integration in the human genome favors active genes and local hotspots. Cell 110, 521–529 (2002).
Cohn, L.B. et al. HIV-1 integration landscape during latent and active infection. Cell 160, 420–432 (2015).
Simonetti, F.R. et al. Clonally expanded CD4+ T cells can produce infectious HIV-1 in vivo. Proc. Natl. Acad. Sci. USA 113, 1883–1888 (2016).
Bosque, A., Famiglietti, M., Weyrich, A.S., Goulston, C. & Planelles, V. Homeostatic proliferation fails to efficiently reactivate HIV-1 latently infected central memory CD4+ T cells. PLoS Pathog. 7, e1002288 (2011).
Massanella, M., Gianella, S., Lada, S.M., Richman, D.D. & Strain, M. Quantification of total and 2-LTR (long terminal repeat) HIV DNA, HIV RNA and herpesvirus DNA in PBMCs. Bio Protoc. 5, e1492 (2015).
Lorenzo-Redondo, R. et al. Persistent HIV-1 replication maintains the tissue reservoir during therapy. Nature 530, 51–56 (2016).
Rose, P.P. & Korber, B.T. Detecting hypermutations in viral sequences with an emphasis on G–> A hypermutation. Bioinformatics 16, 400–401 (2000).
Kim, M. et al. A primary CD4+ T cell model of HIV-1 latency established after activation through the T cell receptor and subsequent return to quiescence. Nat. Protoc. 9, 2755–2770 (2014).
Rosenbloom, D.I.S. et al. Designing and interpreting limiting dilution assays: general principles and applications to the latent reservoir for human immunodeficiency virus-1. Open Forum Infect. Dis. 2, ofv123 (2015).
Acknowledgements
We thank the study participants who have made this research possible. We also thank G. Laird for critical advice and discussion, L. Alston, H. McHugh and D. Xu for assistance with study participants, V. Walker-Sperling for providing the Ba-L virus, C. Pohlmeyer for providing a sample from a viremic subject, and all of the members of the Siliciano laboratory for valuable discussion and advice. This work was supported by the Genomics and Sequencing Core at the UCSD Center for AIDS Research (P30AI036214 (D.D.R.)), the Pendleton Charitable Trust (D.D.R.), the VA San Diego Healthcare System (D.D.R.), the Veterans Medical Research Foundation (D.D.R.), the Martin Delaney CARE and DARE Collaboratories (US National Institutes of Health (NIH) grants AI096113 (M.C.S., D.D.R. and R.F.S.) and 1U19AI096109 (S.G.D. and R.F.S.)), an ARCHE Collaborative Research Grant from the Foundation for AIDS Research (amFAR 108165-50-RGRL (R.F.S.)), the Johns Hopkins Center for AIDS Research grant P30AI094189 (R.F.S.), the NIH grants 43222 (R.F.S.) and R21AI113147-02 (J.D.S.), the Howard Hughes Medical Institute (R.F.S.), and the Bill and Melinda Gates Foundation (R.F.S.).
Author information
Authors and Affiliations
Contributions
K.M.B., R.A.P., S.G.D. and R.F.S. designed the experiments; K.M.B., A.J.M., R.A.P., M.G.S., A.A.C., J.L., M.C.S. and S.M.L. performed the experiments; K.M.B., A.J.M., S.B.L., Y.-C.H., D.D.R., J.D.S. and R.F.S. analyzed the data; R.H. and A.A.C. managed study participant recruitment; S.G.D. provided patient samples; and K.M.B. and R.F.S. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Tables 1–2 (PDF 1253 kb)
Rights and permissions
About this article
Cite this article
Bruner, K., Murray, A., Pollack, R. et al. Defective proviruses rapidly accumulate during acute HIV-1 infection. Nat Med 22, 1043–1049 (2016). https://doi.org/10.1038/nm.4156
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.4156
- Springer Nature America, Inc.
This article is cited by
-
The model student: GPT-4 performance on graduate biomedical science exams
Scientific Reports (2024)
-
Bispecific antibodies promote natural killer cell-mediated elimination of HIV-1 reservoir cells
Nature Immunology (2024)
-
HIV-1 subtype A1, D, and recombinant proviral genome landscapes during long-term suppressive therapy
Nature Communications (2024)
-
Phenotypic characterization of single CD4+ T cells harboring genetically intact and inducible HIV genomes
Nature Communications (2023)
-
Estimating the contribution of CD4 T cell subset proliferation and differentiation to HIV persistence
Nature Communications (2023)