Abstract
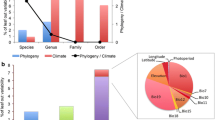
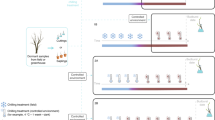
The relative roles of temperature and day length in driving spring leaf unfolding are known for few species, limiting our ability to predict phenology under climate warming1,2. Using experimental data, we assess the importance of photoperiod as a leaf-out regulator in 173 woody species from throughout the Northern Hemisphere, and we also infer the influence of winter duration, temperature seasonality, and inter-annual temperature variability. We combine results from climate- and light-controlled chambers with species’ native climate niches inferred from georeferenced occurrences and range maps. Of the 173 species, only 35% relied on spring photoperiod as a leaf-out signal. Contrary to previous suggestions, these species come from lower latitudes, whereas species from high latitudes with long winters leafed out independent of photoperiod. The strong effect of species’ geographic–climatic history on phenological strategies complicates the prediction of community-wide phenological change.


Similar content being viewed by others
Change history
14 November 2016
In the version of this Letter originally published, the last sentence should have read 'Therefore, photoperiod may be expected to constrain climate-driven shifts in spring leaf unfolding only at lower latitudes'. This error as been corrected in all versions of the Letter.
References
Koerner, C. & Basler, D. Phenology under global warming. Science 327, 1461–1462 (2010).
Richardson, A. D. et al. Climate change, phenology, and phenological control of vegetation feedbacks to the climate system. Agr. Forest Meteorol. 169, 156–173 (2013).
Saikkonen, K. et al. Climate change-driven species’ range shifts filtered by photoperiodism. Nat. Clim. Change 2, 239–242 (2012).
Keenan, T. F. et al. Net carbon uptake has increased through warming-induced changes in temperate forest phenology. Nat. Clim. Change 4, 598–604 (2014).
Menzel, A. & Fabian, P. Growing season extended in Europe. Nature 397, 659 (1999).
Buitenwerf, R., Rose, L. & Higgins, S. I. Three decades of multi-dimensional change in global leaf phenology. Nat. Clim. Change 5, 364–368 (2015).
Richardson, A. D. et al. Influence of spring and autumn phenological transitions on forest ecosystem productivity. Phil. Trans. R. Soc. B 365, 3227–3246 (2010).
Heide, O. M. Dormancy release in beech buds (Fagus sylvatica) requires both chilling and long days. Physiol. Plant. 89, 187–191 (1993).
Basler, D. & Koerner, C. Photoperiod sensitivity of bud burst in 14 temperate forest tree species. Agr. Forest Meteorol. 165, 73–81 (2012).
Zohner, C. M. & Renner, S. S. Perception of photoperiod in individual buds of mature trees regulates leaf-out. New Phytol. 208, 1023–1030 (2015).
Heide, O. M. Daylength and thermal time responses of budburst during dormancy release in some northern deciduous trees. Physiol. Plant. 88, 531–540 (1993).
Laube, J. et al. Chilling outweighs photoperiod in preventing precocious spring development. Glob. Change Biol. 20, 170–182 (2014).
Polgar, C., Gallinat, A. & Primack, R. B. Drivers of leaf-out phenology and their implications for species invasions: insights from Thoreau’s Concord. New Phytol. 202, 106–115 (2014).
Vitasse, Y., Lenz, A. & Körner, C. The interaction between freezing tolerance and phenology in temperate deciduous trees. Front. Plant Sci. 5, 541 (2014).
Fu, Y. H. et al. Declining global warming effects on the phenology of spring leaf unfolding. Nature 526, 104–107 (2015).
Wareing, P. F. Growth studies in woody species. V. Photoperiodism in dormant buds of Fagus sylvatica L. Physiol. Plant. 6, 692–706 (1953).
Falusi, M. & Calamassi, R. Bud dormancy in beech (Fagus sylvatica L.). Effect of chilling and photoperiod on dormancy release of beech seedlings. Tree Physiol. 6, 429–438 (1990).
Caffarra, A. & Donnelly, A. The ecological significance of phenology in four different tree species: effects of light and temperature on bud burst. Int. J. Biometeorol. 55, 711–721 (2011).
Vitasse, Y. & Basler, D. What role for photoperiod in the bud burst phenology of European beech. Eur. J. Forest Res. 132, 1–8 (2013).
Ghelardini, L., Santini, A., Black-Samuelsson, S., Myking, T. & Falusi, M. Bud dormancy in elm (Ulmus spp.) clones—a case study of photoperiod and temperature responses. Tree Physiol. 30, 264–274 (2010).
Way, D. A. & Montgomery, R. A. Photoperiod constraints on tree phenology, performance and migration in a warming world. Plant Cell Environ. 38, 1725–1736 (2015).
Lechowicz, M. J. Why do temperate deciduous trees leaf out at different times? Adaptation and ecology of forest communities. Am. Nat. 124, 821–842 (1984).
Chuine, I. & Beaubien, E. Phenology is a major determinant of temperate tree range. Ecol. Lett. 4, 500–510 (2001).
Chuine, I. Why does phenology drive species distribution? Phil. Trans. R. Soc. B 365, 3149–3160 (2010).
Zohner, C. M. & Renner, S. S. Common garden comparison of the leaf-out phenology of woody species from different native climates, combined with herbarium records forecasts long-term change. Ecol. Lett. 17, 1016–1025 (2014).
Wang, T. et al. The influence of local spring temperature variance on temperature sensitivity of spring phenology. Glob. Change Biol. 20, 1473–1480 (2014).
Vitasse, Y., Porte, A. J., Kremer, A., Michalet, R. & Delzon, S. Responses of canopy duration to temperature changes in four temperate tree species: relative contributions of spring and autumn leaf phenology. Oecologia 161, 187–198 (2009).
Lenz, A., Hoch, G., Vitasse, Y. & Körner, C. Convergence of leaf-out towards minimum risk of freezing damage in temperate trees. Funct. Ecol. 30, 1480–1490 (2016).
Lenoir, J. & Svenning, J.-C. Climate-related range shifts—a global multidimensional synthesis and new research directions. Ecography 38, 15–28 (2015).
Tiffney, B. H. & Manchester, S. R. The influence of physical environment on phytogeographic continuity and phylogeographic hypotheses in the Northern Hemisphere Tertiary. Int. J. Plant Sci. 162, 3–17 (2001).
Vitasse, Y. & Basler, D. Is the use of cuttings a good proxy to explore phenological responses of temperate forests in warming and photoperiod experiments? Tree Physiol. 34, 174–183 (2014).
Siegel, S. & Castellan, N. J. Non-Parametric Statistics for the Behavioral Sciences 213–214 (MacGraw Hill, 1988).
Dismo v1.1-1 (Hijmans, R. J., Phillips, S., Leathwick, J. & Elith, J., 2011); http://cran.r-project.org/web/packages/dismo/index.html
WORLDCLIM v1.3 (Hijmans, R. J., Cameron, S. E., Parra, J. L., Jones P. G. & Jarvis, A., 2004); http://datadryad.org/handle/10255/dryad.12700
Harris, I., Jones, P. D., Osborn, T. J. & Lister, D. H. Updated high-resolution grids of monthly climatic observations - the CRU TS3.10 Dataset. Int. J. Climatol. 34, 623–642 (2014).
Peel, M. C., Finlayson, B. L. & McMahon, T. A. Updated world map of the Koeppen–Geiger climate classification. Hydrol. Earth Syst. Sci. 11, 1633–1644 (2007).
Breiman, L. Random forest. Mach. Learn. 45, 15–32 (2001).
Cutler, D. R. et al. Random forests for classification in ecology. Ecology 88, 2783–2792 (2007).
de Villemereuil, P., Wells, J. A., Edwards, R. D. & Blomberg, S. P. Bayesian models for comparative analysis integrating phylogenetic uncertainty. BMC Evol. Biol. 12, 102 (2012).
Pagel, M. Inferring the historical patterns of biological evolution. Nature 401, 877–884 (1999).
Revell, L. J. Phytools: an R package for phylogenetic comparative biology (and other things). Methods Ecol. Evol. 3, 217–223 (2012).
Panchen, Z. A. et al. Leaf out times of temperate woody plants are related to phylogeny, deciduousness, growth habit and wood anatomy. New Phytol. 203, 1208–1219 (2014).
Webb, C. O. & Donoghue, M. J. PHYLOMATIC: tree assembly for applied phylogenetics. Mol. Ecol. Notes 5, 181–183 (2005).
Bremer, B. et al. An update of the angiosperm phylogeny group classification for the orders and families of flowering plants: APG III. Bot. J. Linnean Soc. 161, 105–121 (2009).
Bell, C., Soltis, D. E. & Soltis, P. S. The age and diversification of the angiosperms re-revisited. Am. J. Bot. 97, 1296–1303 (2010).
Smith, S. A., Beaulieu, J. M. & Donoghue, M. J. An uncorrelated relaxed-clock analysis suggests an earlier origin for flowering plants. Proc. Natl Acad. Sci. USA 107, 5897–5902 (2010).
Stahl, U., Reu, B. & Wirth, C. Predicting species’ range limits from functional traits for the tree flora of North America. Proc. Natl Acad. Sci. USA 111, 13739–13744 (2014).
Gelman, A. & Hill, J. Data Analysis Using Regression and Multilevel/Hierarchical Models (Cambridge Univ. Press, 2007).
Grafen, A. The phylogenetic regression. Phil. Trans. R. Soc. B 326, 119–157 (1989).
Paradis, E., Claude, J. & Strimmer, K. APE: analyses of phylogenetics and evolution in R language. Bioinformatics 20, 289–290 (2004).
Plummer, M. JAGS: a program for analysis of Bayesian graphical models using Gibbs sampling. In Proc. 3rd Int. Workshop on Distributed Statistical Computing (eds Hornik, K., Leisch, F. & Zeileis, A.) (DSC, 2003).
R2jags v 0.04-03 (Su, Y.-S. & Yajima, M., 2014); http://CRAN.R-project.org/package=R2jags
Gelman, A. & Rubin, D. B. Inference from iterative simulation using multiple sequences. Stat. Sci. 7, 457–472 (1992).
R v3.3.1 (R Core Team, 2015); http://www.R-project.org
Acknowledgements
We thank V. Sebald and M. Wenn for help with conducting the twig-cutting experiments. B.M.B. acknowledges funding by Aarhus University and the Aarhus University Research Foundation under the AU IDEAS programme (Centre for Biocultural History) and J.-C.S. support from by the European Research Council (ERC-2012-StG-310886-HISTFUNC). The study was part of the KLIMAGRAD project sponsored by the ‘Bayerisches Staatsministerium für Umwelt und Gesundheit’.
Author information
Authors and Affiliations
Contributions
C.M.Z. and S.S.R. designed the study. C.M.Z. conducted the experiments and leaf-out observations. C.M.Z. and B.M.B. performed the analyses. C.M.Z. and S.S.R. led the writing with inputs from the other authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1406 kb)
Rights and permissions
About this article
Cite this article
Zohner, C., Benito, B., Svenning, JC. et al. Day length unlikely to constrain climate-driven shifts in leaf-out times of northern woody plants. Nature Clim Change 6, 1120–1123 (2016). https://doi.org/10.1038/nclimate3138
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nclimate3138
- Springer Nature Limited
This article is cited by
-
How consistently do species leaf-out or flower in the same order? Understanding the factors that shape this characteristic of plant communities
International Journal of Biometeorology (2023)
-
Accurate phenology analyses require bud traits and energy budgets
Nature Plants (2022)
-
Climate warming shifts the time interval between flowering and leaf unfolding depending on the warming period
Science China Life Sciences (2022)
-
Climate warming–driven phenological shifts are species-specific in woody plants: evidence from twig experiment in Kashmir Himalaya
International Journal of Biometeorology (2022)
-
Winter temperatures predominate in spring phenological responses to warming
Nature Climate Change (2020)