Abstract
The design and synthesis of lincomycin derivatives modified at the C-6 and C-7 positions are described. A substituent at the C-7 position is a 5-aryl-1,3,4-thiadiazol-2-yl-thio group that generates antibacterial activities against macrolide-resistant Streptococcus pneumoniae and Streptococcus pyogenes carrying an erm gene. An additional modification at the C-6 position was explored in application of information regarding pirlimycin and other related compounds. These dual modifications were accomplished by using methyl α-thiolincosaminide as a starting material. As a result of these dual modifications, the antibacterial activities were improved compared with those of compounds with a single modification at the C-7 position. The antibacterial activities of selected compounds in this report against macrolide-resistant S. pneumoniae and S. pyogenes with an erm gene were superior to those of telithromycin.
Similar content being viewed by others
Introduction
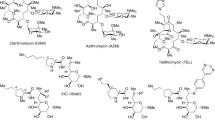
Macrolide antibiotics have had a significant role in treatment of respiratory infections. Clarithromycin1 and azithromycin2 synthesized from 14-membered erythromycin are clinically important macrolides and widely used over many years (Figure 1). However, prevalence of macrolide resistance in Gram-positive bacteria such as S. pneumoniae has been causing serious clinical problems.3, 4 Clarithromycin and azithromycin are inactive against resistant strains of S. pneumoniae, which have a mechanism of ribosome methylation by an erm gene and decreased antibacterial activities against resistant strains with a mef gene, which expresses the efflux pump. Although telithromycin (TEL)5 derived from natural erythromycin has improved antibacterial activities against resistant S. pneumoniae with an erm gene and/or a mef gene, safety problems6 of TEL made its clinical use difficult. Another promising example is a group of 16-membered azalides7, 8 that are effective against resistant S. pneumoniae with an erm gene. However, these compounds are still in the research phase and have not entered development. Novel therapeutic agents that are effective against the resistant S. pneumoniae without concerns of adverse events are required in clinical sites.
Lincomycin (LCM)9 (Figure 2) is one of secondary metabolites of Streptomyces lincolnensis and has been used as antibacterial agent mainly against Gram-positive bacteria. Chemical modification of LCM led to clindamycin (CLDM),10 which has an enhanced antibacterial activity against S. pneumoniae and an improved pharmacokinetic profile. Although LCM and CLDM are distinct from macrolide antibiotics in their chemical structures, they have cross-resistance against S. pneumoniae with an erm gene.11 The cross-resistance is consistent with the result of X-ray crystallographic studies that revealed the overlap of binding site on 50S ribosome. Thus, CLDM shows almost no antibacterial activity against these resistant pathogens as shown in Table 1. CLDM, however, is attractive because of its safety profile and effectiveness against resistant pathogens expressing a drug efflux pump. In addition, CLDM has no gastrointestinal side effect caused by modulating motilin receptor.12 Furthermore, CLDM has been reported to be effective for invasive group A streptococcal infections caused by S. pyogenes.13 A simple chemical structure of CLDM compared with that of macrolide is attractive from the viewpoint of production cost. Based on these reasons, we have been expanding chemical modifications of lincosamide, in order to generate a novel antibacterial agent that is effective against resistant S. pneumoniae and S. pyogenes with erm and mef genes.
It has been reported that modifications at the C-7 position of LCM tend to provide comparable antibacterial activity to that of LCM.14, 15 against susceptible strains. We reported chemical modifications of LCM and clarified that (7S)-7-deoxy-7-thiolincomycin derivatives16, 17, 18, 19, 20, 21, 22 exhibited moderate to strong antibacterial activities against S. pneumoniae and S. pyogenes with an erm gene. Compound 1 that has a phenyl-thiadiazol-2-yl-thio moiety to the C-7 position showed response (very weak antibacterial activities) against those resistant pathogens (Table 1). A nitro group at the 2-position and methoxy groups at the 4- and 5-positions in the benzene ring of 1 had a key role for enhancement of the antibacterial activity against S. pneumoniae with an erm gene.21, 22
The ring-size modification of a proline moiety linked to the N-6 position of lincosamides was reported by several groups23, 24, 25, 26, 27. As for the piperidine derivatives, pirlimycin23 has been used for animal drugs and VIC-10555524, 25, 26 displays enhanced in vitro and in vivo activities with superior efficacy over clindamycin in several experimental models (Figure 2). Although these compounds did not show antibacterial activities against resistant Gram-positive bacteria with an erm gene, in vitro and/or in vivo properties of (7S)-7-deoxy-7-thiolincomycin derivatives are expected to be improved by the modification of the proline moiety. In this article, we report optimization of the proline moiety of (7S)-7-deoxy-7-(5-(2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio)lincomycin derivatives, in order to generate novel LCM derivatives exhibiting stronger antibacterial activities than TEL.
Chemistry
Synthesis of important intermediates that have 4′-substituted piperidine-2′-carbonyl group instead of a natural 4′-propyl proline part is shown in Scheme 1. A racemic mixture of 2,4-cis-piperidine-carboxylic acids was prepared by a reported procedure28 from 4-alkylpyridine. The protection of an amine gave carboxylic acids (±)-4a, (±)-4b and (±)-4c. Condensation of these carboxylic acids with methyl α-thiolincosaminide29 gave 1′-N-Boc protected compounds 5a, 5b and 5c as a 1:1 diastereomeric mixture. These mixtures showed two spots on TLC, but complete separation by silica gel chromatography was difficult. After exploring a solvent for selective precipitation, we found that less polar isomer was precipitated from ethyl acetate and more polar isomer was enriched in mother liquor. Birkenmeyer et al.23 reported that more active 2′-β-4′-β-cis isomers of pirlimycin derivatives showed more polar profile on TLC. Based on the report, the more polar isomer of 5b was assigned as a 2′-β-4′-β-cis isomer. The 2′-β-4′-β-cis isomers of 5b were converted to tetra-O-trimethylsilyl intermediate, which was followed by regioselective deprotection of the 7-O-trimethylsilyl group and silica gel column chromatography, afforded 6b. The other 2′-α-4′-α-cis isomer 6b′ was prepared from less polar isomer (5b′) as well. Regarding 4′-ethyl and 4′-n-butyl derivatives, 2′-β-4′-β-cis isomers 6a and 6c were obtained in the similar manner to 6b.
Synthesis of (7S)-7-deoxy-7-(5-phenyl-1,3,4-thiadiazol-2-yl-thio)-LCM derivatives modified at the C-6 position is shown in Scheme 2. Introductions of the 5-substituted 1,3,4-thiadiazol-2-yl-thio group at the C-7 position were achieved by Mitsunobu reaction and subsequent deprotection led to target compounds 7a–7k. In order to obtain information on the relationship between stereochemistry in the piperidine moiety and antibacterial activity, 2′-α-4′-α-cis isomer 7b′ was also synthesized in the similar manner to 7a–7k from 6b′. Compound 7b was more polar than 7b′ on TLC as in the case of 6b and 6b′. A major reason for relatively low yield of these Mitsunobu reactions was explained by generation of an N-connected byproduct in the thiadiazole moiety instead of the desired S-connected derivative. 1′-Methylated compounds 8d, 8e and 8f were prepared by reductive N-methylation of the corresponding secondary amine. Synthesis of VIC-105555 and 10 were conducted based on the reported procedure24 as shown in Scheme 3. The reduction of 9 gave a diastereomeric mixture of VIC-105555 and 10. We separated these isomers and a more polar isomer was assigned as VIC-105555.
Results and discussion
Antibacterial activities of LCM derivatives that have the 5-(2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio group at the C-7 position and the 4′-alkyl piperidin-2′-carbonylamino group at the C-6 position are shown in Table 2. Regarding stereoisomers 7b and 7b′, which are 4′-propyl derivatives, antibacterial activities of 7b were clearly more potent than those of 7b′ as expected. According to the result, the assignment of stereochemistry in the piperidine moiety was also supported by antibacterial activity. Similarly, VIC-105555 showed reported antibacterial activities against S. pneumoniae and S. pyogenes without an erm gene and antibacterial activities of stereoisomer 10 was much weaker than those of VIC-105555. Compared with a natural proline analog 2, transformation to 4′-n-propylpiperidine-2′-carbonyl analog remarkably improved the antibacterial activities against resistant S. pneumoniae and S. pyogenes with an erm gene. 4′-Ethyl analog (7a) showed improved antibacterial activities against susceptible strains of S. pneumoniae and S. pyogenes compared to 2. However, its antibacterial activities against resistant S. pneumoniae with an erm gene were weaker than those of 7b. In contrast, 4′-n-butyl analog (7c) exhibited more potent antibacterial activities than 7b against all of the test organisms. Antibacterial activities of these derivatives were significantly superior to TEL against S. pyogenes with an erm gene.
Table 3 shows antibacterial activities of 4-fluoro-2-nitrophenyl analogs. The antibacterial activities of 7d, 7e and 7f indicated that the effect of a fluorine atom on antibacterial activity against S. pneumoniae and S. pyogenes was unclear. However, antibacterial activities of 7f against S. pneumoniae with an erm gene were generally more potent than those of TEL as well as 7c. On the other hand, 7d, 7e and 7f exhibited enhanced antibacterial activities against Haemophilus influenzae compared with the corresponding compounds 7a, 7b and 7c without a fluorine atom. Compounds 8d, 8e and 8f, which have a methyl group at the 1′-position showed weaker antibacterial activities against resistant S. pneumoniae and S. pyogenes with an erm gene than corresponding 1′-demethyl compounds, respectively.
Antibacterial activities of 2-nitrophenyl derivatives that have substituent(s) in the benzene ring are shown in Table 4. We previously reported22 that the introduction of a substituent such as a methylamino group and a methoxy group to the 5-position of the benzene ring of LCM analog possessing a natural 4′-propyproline moiety dramatically improved the antibacterial activities and their antibacterial activities were further enhanced by additional substitution at the 4-position of the benzene ring. According to the antibacterial activities of 7g and 7h, ring size expansion from a pyrrolidine ring found in the original LCM to the piperidine ring enhanced antibacterial activities against S. pneumoniae and S. pyogenes as in the case of the 2-nitrophenyl derivatives and 4-fluoro-2-nitrophenyl derivatives. In addition, the conversion of 4,5-dimethoxy-2-nitrophenyl derivative 3 (Figure 2) to a piperidine analog gave 7j that exhibited apparently more potent antibacterial activities against resistant S. pneumoniae and S. pyogenes with an erm gene than TEL. Although antibacterial activities of 4′-ethyl derivative (7i) against resistant S. pneumoniae with an erm gene were remarkably weakened, 4′-n-butyl derivative (7k) showed more potent antibacterial activities against resistant S. pneumoniae and S. pyogenes with an erm gene than TEL. It should be noted that antibacterial activities of 7k against resistant strains with an erm gene are almost same as those against susceptible strains. These results encouraged us to investigate the antibacterial activities of Mycoplasma pneumoniae. It is reported that macrolide resistant M. pneumoniae has been prevailing in China, Japan and other countries.30 Compounds tested showed potent antibacterial activities against resistant M. pneumonia, except for 7h (#18).
In summary, we previously reported that 1 possessing the 5-phenyl-1,3,4-thiadiazol-2-yl-thio group at the C-7 position showed weak antibacterial activities against the resistant pathogens. In the course of our continuous chemical derivatization, target antibacterial activities were significantly improved. Compound 3 that has two methoxy groups at the benzene ring exhibited potency comparable to that of TEL. In this article, an additional modification at the C-6 position led to 7k possessing the 4-n-butylpiperidine-2-carbonyl group instead of the 4-n-propylpyrrolidin-2-carbonyl group at the C-6 position. Selected compound 7k exhibited significantly potent antibacterial activities against the resistant pathogens than TEL. This series of compounds has a nitro group at the benzene ring. Although it is known that a nitro group has a risk for mutagens and carcinogens, there are drugs and drug candidates that have a nitro group.
Conclusions
A series of LCM derivatives that have the 5-(2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio moiety at the C-7 position in S-configuration and the 4-alkylpipelidine-2-carbonylamino group at the C-6 position was synthesized. Introductions of a substituent at the C-7 position were accomplished by the Mitsunobu reaction, and stereochemistry of 4-alkylpiperidine-2-carbonyl moiety was assigned by the reported information on the relationship between stereochemistry and polarity. Additional modification at the C-6 position improved antibacterial activities of (7S)-1,3,4-thiadiazol-2-yl-thio LCM derivatives. In this study, we found the 4-n-butylpiperidine-2-carbonylamino group had the most potent functionality at the C-6 position against resistant S. pneumoniae and S. pyogenes with an erm gene. In particular, compound 7k exhibited the most potent antibacterial activities among all our (7S)-1,3,4-thiadiazol-2-yl-thio-LCM derivatives and apparently more potent in vitro than TEL against the resistant strains. Summary of SAR between antibacterial activity against resistant S. pneumoniae with an erm gene and (7S)-7-(5-aryl-1,3,4-thiadiazol-2-yl-thio)-LCM derivatives is shown in Figure 3. This series of LCM derivatives is promising to overcome maclolide-resistant S. pneumoniae and S. pyogenes with an erm gene.
Experimental procedures
General
1H NMR spectra were measured with Varian Gemini-300 (Varian, Inc., Palo Alto, CA, USA) for 300 MHz, JEOL JNM-GSX 400 (JEOL Ltd, Tokyo, Japan) for 400 MHz or BRUKER Ascend 400 NMR spectrometer (BRUKER Corporation, Coventry, UK) for 400 MHz in CDCl3 or CD3OD with 0.03% tetramethylsilane as an internal standard. MS spectra were obtained on a JEOL JMS-FABmate spectrometer or JEOL JMS-700 mass spectrometer or Agilent Technologies 6530-Q-TOF LC/MS mass spectrometer (Agilent Technologies, Santa Clara, CA, USA). The optical rotations were recorded with Jasco P-2300 digital polarimeter (Jasco, Tokyo, Japan). Column chromatography was performed with silica gel 60 N (Kanto Chemical, Tokyo, Japan, spherical, neutral).
Mixture 5a of methyl 6-N-[(2′S, 4′R)-1′-N-(tert-butoxycarbonyl)-4′-ethylpiperidine-2′-carbonyl]-α-thiolincosaminide and methyl 6-N-[(2′R, 4′S)-1′-N-(tert-butoxycarbonyl)-4′-ethylpiperidine-2′-carbonyl]-α-thiolincosaminide
To a solution of (±)-4a (6.66 g, 25.9 mmol) in N,N′-dimethylformamide (45 ml) were added 1-hydroxybenzotriazole (3.50 g, 25.9 mmol), N,N′-dicyclohexylcarbodiimide (6.05 g, 29.3 mmol), methyl α-thiolincosaminide (7.37 g, 29.1 mmol) and triethylamine (10.0 ml, 71.7 mmol), and stirred at room temperature for overnight. To the mixture was added H2O and filtrated. Ethyl acetate was added to the filtrate and washed with saturated aqueous NaHCO3. The organic phase was dried over Na2SO4, filtrated and concentrated under reduced pressure. The resulting residue was purified by silica gel column chromatography (hexane/ethyl acetate=50/50 to ethyl acetate, then ethyl acetate to ethyl acetate/MeOH=90/10) to afford methyl 6-N-[1′-N-(tert-butoxycarbonyl)-4′-ethylpiperidine-2′-carbonyl]-α-thiolincosaminide (18.5 g, 84.9%, (2′S, 4′R) isomer: (2′R, 4′S) isomer=ca 50:50) as a colorless solid. To this colorless solid was added ethyl acetate, and insoluble matter was filtrated off and ethyl acetate solution was concentrated under reduced pressure to afford mixture 5a (7.31 g, 60% de ((2′S, 4′R): (2′R, 4′S)=80:20)) as a colorless solid. 1H NMR (400 MHz, CDCl3) δ 6.72 (br s, 0.8H), 6.55 (d, J=8.6 Hz, 0.2H), 5.30–5.36 (m, 1H), 4.58–4.65 (m, 0.2 H), 4.46 (br s, 0.8H), 4.07–4.26 (m, 5H), 3.95–3.98 (m, 0.8H), 3.90 (br s, 0.2H), 3.47–3.62 (m, 2H), 3.31–3.41 (m, 1H), 2.80–3.03 (m, 1H), 2.55–2.70 (m, 2H), 2.17 (s, 3H), 1.96–2.04 (m, 1H), 1.78–1.88 (m, 1H), 1.61–1.74 (m, 1H), 1.44–1.49 (m, 9H), 1.28–1.38 (m, 4H), 1.20–1.24 (m, 3H), 0.91 (t, J=7.3 Hz, 3H).
Methyl 6-N-[(2′S, 4′R)-1′-N-(tert-butoxycarbonyl)-4′-ethylpiperidine-2′-carbonyl]- 2,3,4-tris-O-(trimethylsilyl)-α-thiolincosaminide (6a)
To a solution of mixture 5a (7.31 g, 14.8 mmol, 60% de ((2′S, 4′R):(2′R, 4′S)=80:20)) in pyridine (38 ml) were added trimethylchlorosilane (8.0 ml, 6.3 mmol) and hexamethyldisilazane (6.0 ml, 2.8 mmol), and stirred at room temperature for overnight. The mixture was added to saturated aqueous NaHCO3 and was extracted with ethyl acetate, washed with brine. The organic phase was dried over Na2SO4, filtrated and concentrated under reduced pressure. To the resulting residue were added methanol (45 ml) and 6 N acetic acid (2.7 ml), and stirred at room temperature for 3 h. The mixture was added to saturated aqueous NaHCO3 and concentrated under reduced pressure. The resulting mixture was extracted with ethyl acetate, dried over Na2SO4, filtrated and concentrated under reduced pressure. The resulting residue was purified by silica gel column chromatography (hexane/ethyl acetate=19/1 to 3/1) to afford 6a (5.56 g, 30% from (±)-4a) as a colorless solid. 1H NMR (400 MHz, CDCl3) δ 6.40 (d, J=9.2 Hz, 1H), 5.17 (d, J=5.5 Hz, 1H), 4.30–4.38 (m, 1H), 4.19 (dd, J=9.0, 6.3 Hz, 1H), 4.12 (dd, J=9.5, 5.5 Hz, 1H), 4.07 (d, J=8.3 Hz, 1H), 3.93–4.02 (m, 1H), 3.90–3.93 (m, 1H), 3.62 (dd, J=9.5, 2.6 Hz, 1H), 3.48–3.59 (m, 1H), 3.31–3.40 (m, 1H), 2.83–2.89 (m, 1H), 2.05 (s, 3H), 1.99–2.03 (m, 1H), 1.78–1.89 (m, 1H), 1.57–1.63 (m, 1H), 1.47 (s, 9H), 1.29–1.39 (m, 2H), 1.20–1.29 (m, 1H), 1.17 (d, J=6.4 Hz, 3H), 0.90 (t, J=7.3 Hz, 3H), 0.19 (s, 9H), 0.13–0.16 (m, 18H); MS (ESI) m/z 709 (M+H)+.
Methyl 6-N-[(2′R, 4′S)-1′-N-(tert-butoxycarbonyl)-4′-(n-propyl)piperidine-2′-carbonyl] -α-thiolincosaminide (5b′) and mixture 5b of methyl 6-N-[(2′S, 4′R)-1′-N-(tert-butoxycarbonyl)-4′-(n-propyl)piperidine-2′-carbonyl]-α-thiolincosaminide and methyl 6-N-[(2′R, 4′S)-1′-N-(tert-butoxycarbonyl)-4′-(n-propyl)piperidine-2′-carbonyl]-α-thiolincosaminide
Compound (±)-4b (11.7 g, 43.1 mmol), 1-hydroxybenzotriazole (7.55 g, 55.8 mmol), N,N′-dicyclohexylcarbodiimide (11.0 g, 53.3 mmol) and methyl α-thiolincosaminide (14.2 g, 56.1 mmol) in N,N′-dimethylformamide (100 ml) were treated for 12 h according to the similar procedure as described for the preparation of mixture 5a to afford methyl 6-N-[1′-N-(tert-butoxycarbonyl)-4′-(n-propyl)piperidine-2′-carbonyl]-α-thiolincosaminide (18.5 g, 84.9%, (2′S, 4′R) isomer: (2′R, 4′S) isomer=ca. 50:50) as a colorless solid. To this colorless solid was added ethyl acetate and insoluble matter was collected by filtration to afford 5b′ (3.2 g, 15%) as a colorless solid and ethyl acetate solution was concentrated under reduced pressure to afford mixture 5b (13.5 g, 62%, 20% de ((2′S, 4′R): (2′R, 4′S)=60:40)) as a colorless solid. 5b′: 1H NMR (400 MHz, CDCl3) δ 6.55 (d, J=8.6 Hz, 1H), 5.34 (d, J=5.5 Hz, 1H), 4.62 (br s, 1H), 4.11–4.27 (m, 4H), 3.99 (d, J=9.4 Hz, 1H), 3.90 (br s, 1H), 3.48–3.57 (m, 2H), 3.32–3.42 (m, 1H), 2.85–2.94 (m, 2H), 2.64 (br s, 1H), 2.16 (s, 3H), 1.94–2.03 (m, 1H), 1.77–1.87 (m, 1H), 1.62–1.69 (m, 1H), 1.50–1.59 (m, 1H), 1.47 (s, 9H), 1.23–1.38 (m, 5H), 1.22 (d, J=6.4 Hz, 3H), 0.89 (t, J=6.9 Hz, 3H); MS (ESI) m/z 507 (M+H)+. 5b: 1H NMR (400 MHz, CDCl3) δ 6.74 (br s, 0.6H), 6.54 (d, J=8.7 Hz, 0.4H), 5.30–5.35 (m, 1H), 4.60 (br s, 0.4H), 4.45 (br s, 0.6H), 3.88–4.27 (m, 6H), 3.47–3.62 (m, 2H), 3.28–3.42 (m, 1H), 2.61–3.06 (m, 3H) 2.16 (m, 3H), 1.92–2.02 (m, 1H), 1.74–1.86 (m, 1H), 1.61–1.67 (m, 1H), 1.49–1.54 (m, 1 H), 1.42–1.49 (m, 9H), 1.25–1.37 (m, 5H), 1.19–1.25 (m, 3H), 0.86–0.91 (m, 3H).
Methyl 6-N-[(2′S, 4′R)-1′-N-(tert-butoxycarbonyl)-4′-(n-propyl)piperidine-2′-carbonyl]-2,3,4-tris-O-(trimethylsilyl)-α-thiolincosaminide (6b)
Mixture 5b (13.5 g, 26.6 mmol, 20% de ((2′S, 4′R): (2′R, 4′S)=60:40), trimethylchlorosilane (17.0 ml, 133 mmol) and hexamethyldisilazane (27.9 ml, 133 mmol) in pyridine (50 ml) were treated for 40 min according to the similar procedure as described for the preparation of 6a and the crude tetrakis-O-trimethylsilyl intermediate and 6 N acetic acid (5.8 ml) in MeOH (138 ml) were treated for 2.5 h according to the similar procedure as described for the preparation of 6a to afford 6b (9.28 g, 48% in 2 steps from diastereomeric mixture 5b) as a colorless solid. 1H NMR (400 MHz, CDCl3) δ 6.36 (d, J=9.4 Hz, 1H), 5.17 (d, J=5.5 Hz, 1H), 4.30–4.37 (m, 1H), 4.09–4.19 (m, 2H), 4.06 (d, J=8.3 Hz, 1H), 3.95–4.03 (m, 1H), 3.98–3.92 (m, 1H), 3.61 (dd, J=9.5, 2.6 Hz, 1H), 3.44–3.55 (m, 1H), 3.3–3.41 (m, 1H), 2.92 (d, J=6.5 Hz, 1H), 2.06 (s, 3H), 2.00–2.05 (m, 1H), 1.78–1.90 (m, 1H), 1.49–1.64 (m, 2H), 1.42–1.49 (m, 9H), 1.20–1.37 (m, 5H), 1.17 (d, J=6.4 Hz, 3H), 0.85–0.92 (m, 3H), 0.19–0.20 (m, 9H), 0.13–0.15 (m, 18H); MS (ESI) m/z 723 (M+H)+.
Methyl 6-N-[(2′R, 4′S)-1′-N-(tert-butoxycarbonyl)-4′-(n-propyl)piperidine-2′-carbonyl]-2,3,4-tris-O-(trimethylsilyl)-α-thiolincosaminide (6b′)
Compound 5b′ (1.00 g, 1.97 mmol), trimethylchlorosilane (1.26 ml, 9.87 mmol) and hexamethyldisilazane (2.07 ml, 9.87 mmol) in pyridine (4.0 ml) were treated for 30 min according to the similar procedure as described for the preparation of 6a and the crude tetrakis-O-trimethylsilyl intermediate was treated with 6 N acetic acid (0.42 ml) in MeOH (10 ml) for 1 h according to the similar procedure as described for the preparation of 6a to afford 6b′ (926 mg, 65% in 2 steps) as a colorless solid. 1H NMR (400 MHz, CDCl3) δ 6.56 (d, J=8.9 Hz, 1H), 5.21 (d, J=5.5 Hz, 1H), 4.24–4.31 (m, 1H), 4.11–4.20 (m, 2H), 4.08 (d, J=8.1 Hz, 1H), 4.02–4.04 (m, 1H), 3.63 (dd, J=9.5, 2.6 Hz, 1H), 3.32–3.51 (m, 3H), 2.04 (s, 3H), 1.86–1.93 (m, 1H), 1.70–1.85 (m, 2H), 1.44–1.49 (m, 10H), 1.21–1.38 (m, 5H), 1.15 (d, J=6.5 Hz, 3H), 0.89 (t, J=6.9 Hz, 3H), 0.19 (s, 9H), 0.15 (s, 9H), 0.14 (s, 9H); MS (ESI) m/z 723 (M+H)+.
Mixture 5c of methyl 6-N-[(2′S, 4′R)-1′-N-(tert-butoxycarbonyl)-4′-(n-butyl)piperidine-2′-carbonyl]-α-thiolincosaminide and methyl 6-N-[(2′R, 4′S)-1′-N-(tert-butoxycarbonyl)-4′-(n-butyl)piperidine-2′-carbonyl]-α-thiolincosaminide
Compound (±)-4c (12.6 g, 44.2 mmol), 1-hydroxybenzotriazole (7.77 g, 57.5 mmol), N,N′-dicyclohexylcarbodiimide (11.0 g, 53.3 mmol) and methyl α-thiolincosaminide (14.6 g, 57.5 mmol) in N,N′-dimethylformamide (120 ml) were treated for 20 h according to the similar procedure as described for the preparation of mixture 5a to afford methyl 6-N-[1′-N-(tert-butoxycarbonyl)-4′-(n-butyl)piperidine-2′-carbonyl]-α-thiolincosaminide (20.0 g, 87%, (2′S, 4′R) isomer: (2′R, 4′S) isomer=ca 50:50) as a colorless solid. To this colorless solid (14.5 g) was added ethyl acetate and insoluble matter was filtrated off and ethyl acetate solution was concentrated under reduced pressure to afford mixture 5c (8.15 g, 80% de ((2′S, 4′R): (2′R, 4′S)=90:10)) as a colorless solid. 1H NMR (400 MHz, CDCl3) δ 6.85 (br s, 0.9H), 6.58 (d, J=8.7 Hz, 0.1H), 5.30–5.34 (m, 1H), 4.64 (br s, 0.1H), 4.46 (br s, 0.9H), 3.93–4.25 (m, 6H), 3.51–3.63 (m, 2H), 3.28–3.38 (m, 1H), 2.80–2.98 (m, 2H), 2.14–2.16 (m, 3H), 1.93–2.02 (m, 1H), 1.68–1.86 (m, 3H), 1.54 (br s, 1H), 1.44–1.47 (m, 9H), 1.19–1.35 (m, 7H), 1.19–1.25 (m, 3H), 0.86–0.91 (m, 3H).
Methyl 6-N-[(2′S, 4′R)-1′-N-(tert-butoxycarbonyl)-4′-(n-butyl)piperidine-2′-carbonyl]-2,3,4-tris-O-(trimethylsilyl)-α-thiolincosaminide (6c)
Mixture 5c (8.15 g, 15.7 mmol, 80% de ((2′S, 4′R): (2′R, 4′S)=90:10), trimethylchlorosilane (100 ml, 78.3 mmol) and hexamethyldisilazane (16.4 ml, 78.3 mmol) in pyridine (30 ml) were treated for 20 min according to the similar procedure as described for the preparation of 6a and the crude tetrakis-O-trimethylsilyl intermediate and 6 N acetic acid (3.4 ml) in MeOH (88 ml) were treated for 40 min according to the similar procedure as described for the preparation of 6a to afford 6c (8.20 g, 71% in 2 steps from diastereomeric mixture 5c) as a colorless solid. 1H NMR (400 MHz, CDCl3) δ 6.34 (d, J=9.3 Hz, 1H), 5.17 (d, J=5.5 Hz, 1H), 4.30–4.36 (m, 1H), 4.09–4.15 (m, 2H), 4.05 (d, J=8.3 Hz, 1H), 3.95–4.03 (m, 1H), 3.88–3.90 (m, 1H), 3.61 (dd, J=9.5, 2.6 Hz, 1H), 3.43–3.44 (m, 1H), 3.32–3.44 (m, 1H), 2.96 (d, J=6.5 Hz, 1H), 2.05 (s, 3H), 2.00–2.04 (m, 1H), 1.79–1.89 (m, 1H), 1.49–1.87 (m, 1H), 1.46 (s, 9H), 1.19–1.33 (m, 7H), 1.16 (d, J=6.4 Hz, 3H), 0.85–0.92 (m, 3H), 0.17–0.19 (m, 9H), 0.12–0.15 (m, 18H); MS (ESI) m/z 737 (M+H)+.
Methyl (7S)-7-deoxy-6-N-[(2′S, 4′R)-4′-ethylpiperidine-2′-carbonyl]-7-[5-(2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio]-α-thiolincosaminide (7a)
To a solution of 6a (270 mg, 0.381 mmol) in tetrahydrofuran (5 ml) at 0 °C were added triphenylphosphine (200 mg, 0.762 mmol) and diethlazodicarboxylate (0.150 ml, 0.823 mmol), and stirred at 0 °C for 10 min, and 5-(2-nitrophenyl)-1,3,4-thiadiazole-2-thiol (200 mg, 0.836 mmol) was added and stirred at room temperature for 3 h. The mixture was concentrated under reduced pressure and added methanol (10 ml), 5 N hydrochloric acid (0.1 ml) and stirred at room temperature for 1 h. The mixture was concentrated under reduced pressure. To the resulting residue was added trifluoroacetic acid (3.0 ml) and stirred at room temperature for 30 min. The mixture was concentrated under reduced pressure and the resulting residue was purified by preparative TLC (CHCl3/CH3OH/28% aq NH4OH=20/1/0.1) to afford 7a (52.5 mg, 22%) as colorless solid. [α]D25 +100° (c 1.3, CH3OH); 1H NMR (400 MHz, CD3OD) δ 8.10 (d, J=7.6 Hz, 1H), 7.76–7.86 (m, 3H), 5.28 (d, J=5.6 Hz, 1H), 4.67 (dd, J=10.1, 2.5 Hz, 1H), 4.46 (qd, J=6.8, 2.5 Hz, 1H), 4.38–4.43 (m, 1H), 4.07–4.12 (m, 1H), 3.87–3.89 (m, 1H), 3.56 (dd, J=10.4, 3.3 Hz, 1H), 3.26–3.31 (m, 1H), 3.13–3.20 (m, 1H), 2.56–2.67 (m, 1H), 1.99 (s, 3H), 1.95–1.98 (m, 1H), 1.68–1.75 (m, 1H), 1.56 (d, J=6.8 Hz, 3H), 1.36–1.50 (m, 1H), 1.25–1.34 (m, 2H), 0.98–1.11 (m, 2H), 0.89–0.95 (m, 3H); MS (FAB) m/z 614 (M+H)+; HRMS (ESI) m/z calcd for C25H36N5O7S3 614.1771, found 614.1768 (M+H)+.
Methyl (7S)-7-deoxy-7-[5-(2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio]-6-N-[(2′S, 4′R)-4′-(n-propyl)piperidine-2′-carbonyl]-α-thiolincosaminide (7b)
Reaction of 6b (200 mg, 0.277 mmol) with 5-(2-nitrophenyl)-1,3,4-thiadiazole-2-thiol (114 mg, 0.476 mmol) afforded 7b as a colorless solid in 24% yield by the similar procedure to 7a. [α]D26 +83° (c 0.17, CH3OH); 1H NMR (400 MHz, CD3OD) δ 8.09–8.13 (m, 1H), 7.77–7.86 (m, 3H), 5.29 (d, J=5.6 Hz, 1H), 4.63–4.69 (m, 1H), 4.41–4.47 (m, 1H), 4.43 (d, J=10.2 Hz, 1H), 4.06–4.12 (m, 1H), 3.87–3.91 (m, 1H), 3.56 (dd, J=10.2, 3.4 Hz, 1H), 3.25–3.31 (m, 1H), 3.19–3.25 (m, 1H), 3.08–3.18 (m, 1H), 2.58–2.68 (m, 1H), 1.99 (s, 3H), 1.65–1.72 (m, 1H), 1.56 (d, J=7.1 Hz, 3H), 1.24–1.34 (m, 5H), 0.98–1.11 (m, 2H), 0.85–0.95 (m, 3H); MS (FAB) m/z 628 (M+H)+; HRMS (ESI) m/z calcd for C26H38N5O7S3 628.1928, found 628.1926 (M+H)+.
Methyl (7S)-7-deoxy-7-[5-(2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio]- 6-N-[(2′R, 4′S)-4′-(n-propyl)piperidine-2-carbonyl]-α-thiolincosaminide (7b′)
Reaction of 6b′ (200 mg, 0.277) mmol) with 5-(2-nitrophenyl)-1,3,4-thiadiazole-2-thiol (104 mg, 0.435 mmol) afforded 7b′ as a light yellow solid in 25% yield by the similar procedure to 7a. [α]D26 +110° (c 0.21, CH3OH); 1H NMR (400 MHz, CD3OD) δ 8.08–8.12 (m, 1H), 7.75–7.85 (m, 3H), 5.27 (d, J=5.9 Hz, 1 H), 4.66 (dd, J=10.1, 2.5 Hz, 1H), 4.39–4.49 (m, 2H), 4.07–4.13 (m, 1H), 3.83–3.85 (m, 1H), 3.56 (dd, J=10.2, 3.3 Hz, 1H), 3.24–3.29 (m, 1H), 3.09–3.15 (m, 1H), 2.58–2.66 (m, 1H), 2.01 (s, 3H), 1.94–1.99 (m, 1H), 1.65–1.73 (m, 1H), 1.58 (d, J=6.9 Hz, 3H), 1.18–1.42 (m, 5H), 0.99–1.12 (m, 2H), 0.90 (t, J=7.2 Hz, 3H); MS (FAB) m/z 628 (M+H)+; HRMS (ESI) m/z calcd for C26H38N5O7S3 628.1928, found 628.1921 (M+H)+.
Methyl (7S)-6-N-[(2′S, 4′R)-4′-(n-butyl)piperidine-2′-carbonyl]-7-deoxy-7-[5-(2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio]-α-thiolincosaminide (7c)
Reaction of 6c (500 mg, 0.678 mmol) with 5-(2-nitrophenyl)-1,3,4-thiadiazole-2-thiol (211 mg, 0.882 mmol) afforded 7c as a colorless solid in 21% yield by the similar procedure to 7a. [α]D25 +93° (c 0.48, CH3OH); 1H NMR (400 MHz, CD3OD) δ 8.08–8.12 (m, 1H), 7.76–7.87 (m, 3H), 5.29 (d, J=5.6 Hz, 1H), 4.68 (dd, J=10.1, 2.3 Hz, 1H), 4.46 (dd, J=6.8, 2.4 Hz, 1H), 4.42 (d, J=10.2 Hz, 1H), 4.11 (dd, J=10.2, 5.6 Hz, 1H), 3.88–3.91 (m, 1H), 3.59–3.63 (m, 1H), 3.57 (dd, J=10.2, 3.2 Hz, 1H), 3.38 (dd, J=11.9, 2.9 Hz, 1H), 3.15–3.22 (m, 1H), 2.63–2.72 (m, 1H), 1.99 (s, 3H), 1.68–1.76 (m, 1H), 1.52–1.58 (m, 4H), 1.24–1.34 (m, 6H), 1.02–1.17 (m, 3H), 0.84–0.92 (m, 3H); MS (FAB) m/z 642 (M+H)+; HRMS (ESI) m/z calcd for C27H40N5O7S3 642.2084, found 642.2092 (M+H)+.
Methyl (7S)-7-deoxy-6-N-[(2′S, 4′R)-4′-ethylpiperidine-2′-carbonyl]-7-[5-(4-fluoro-2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio]-α-thiolincosaminide (7d)
Reaction of 6a (180 mg, 0.253 mmol) with 5-(4-fluoro-2-nitrophenyl)-1,3,4-thiadiazole-2-thiol (80.0 mg, 311 mmol) afforded 7d as a light yellow solid in 39% yield by the similar procedure to 7a. [α]D25 +89° (c 0.53, CH3OH); 1H NMR (400 MHz, CD3OD) δ 7.98 (dd, J=8.3, 2.5 Hz, 1H), 7.85 (dd, J=8.7, 5.3 Hz, 1H), 7.64 (ddd, J=8.7, 7.7, 2.5 Hz, 1H), 5.27 (d, J=5.6 Hz, 1H), 4.67 (dd, J=10.0, 2.4 Hz, 1H), 4.46 (qd, J=6.8, 2.4 Hz, 1H), 4.41 (dd, J=10.1, 0.9 Hz, 1H), 4.10 (dd, J=10.3, 5.9 Hz, 1H), 3.88 (dd, J=3.2, 0.9 Hz, 1H), 3.55 (dd, J=10.3, 3.2 Hz, 1H), 3.26–3.30 (m, 1H), 3.14–3.20 (m, 1H), 2.64 (td, J=12.8, 2.8 Hz, 1H), 1.99 (s, 3H), 1.95–1.98 (m, 1H), 1.68–1.75 (m, 1H), 1.56 (d, J=6.8 Hz, 3H), 1.37–1.50 (m, 1H), 1.26–1.35 (m, 2H), 1.00–1.11 (m, 2H), 0.92 (t, J=7.5 Hz, 3H); MS (FAB) m/z 632 (M+H)+; HRMS (ESI) m/z calcd for C25H35FN5O7S3 632.1677, found 632.1674 (M+H)+.
Methyl (7S)-7-deoxy-6-N-[(2′S, 4′R)-4-ethyl-1′-N-methylpiperidine-2′-carbonyl]-7-[5-(4-fluoro-2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio]-α-thiolincosaminide (8d)
To a solution of compound 7d (52 mg, 0.082 mmol) in MeOH (3 ml) at 0 oC were added 36% aqueous formaldehyde (100 μl, 1.2 mmol), acetic acid (50 μl, 0.79 mmol) and NaBH(OAc)3 (120 mg, 0.57 mmol) and stirred at room temperature for 4 h. The mixture was concentrated under reduced pressure. Ethyl acetate was added to the residue and washed with saturated aqueous NaHCO3. The organic phase was dried over MgSO4, filtrated and concentrated under reduced pressure. The resulting residue was purified by preparative TLC (CHCl3/MeOH/28% aq NH4OH=10/1/0.1) to afford 8d (41 mg, 77%) as a light yellow solid. [α]D25 +80° (c 0.92, CH3OH); 1H NMR (400 MHz, CD3OD) δ 7.98 (dd, J=8.3, 2.6 Hz, 1H), 7.85 (dd, J=8.7, 5.3 Hz, 1H), 7.61–7.67 (m, 1H), 5.28 (d, J=5.6 Hz, 1H), 4.69 (dd, J=10.1, 2.6 Hz, 1H), 4.48 (qd, J=6.9, 2.6 Hz, 1H), 4.43 (dd, J=10.1, 0.8 Hz, 1H), 4.12 (dd, J=10.2, 5.6 Hz, 1H), 3.83–3.87 (m, 1H), 3.57 (dd, J=10.2, 2.9 Hz, 1H), 2.93–3.99 (m, 1H), 2.58–2.64 (m, 1H), 2.25 (s, 3H), 2.06–2.15 (m, 1H), 1.98–2.01 (m, 3H), 1.85–1.91 (m, 1H), 1.69–1.76 (m, 1H), 1.58 (d, J=6.9 Hz, 3H), 1.23–1.34 (m, 5H), 0.87–0.94 (m, 3H); MS (FAB) m/z 646 (M+H)+; HRMS (ESI) m/z calcd for C27H39FN5O7S3 646.1834, found 646.1833 (M+H)+.
Methyl (7S)-7-deoxy-7-[5-(4-fluoro-2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio]-6-N-[(2′S, 4′R)-4′-(n-propyl)piperidine-2′-carbonyl]-α-thiolincosaminide (7e)
Reaction of 6b (2.80 g, 3.87 mmol) with 5-(4-fluoro-2-nitrophenyl)-1,3,4-thiadiazole-2-thiol (1.19 mg, 4.59 mmol) afforded 7e as a light yellow solid in 39% yield by the similar procedure to 7a except for using toluene as a solvent of Mitunobu reaction. [α]D26 +93° (c 0.20, CH3OH); 1H NMR (400 MHz, CD3OD) δ 7.99 (dd, J=8.3, 2,7 Hz, 1H), 7.85 (dd, J=8.6, 5.2 Hz, 1H), 7.60–7.69 (m, 1H), 5.27 (d, J=5.6 Hz, 1H), 4.66 (dd, J=10.1, 2.6 Hz, 1H), 4.43–4.50 (m, 1H), 4.41 (d, J=10.1 Hz, 1H), 4.10 (dd, J=10.3, 5.6 Hz, 1H), 3.85–3.90 (m, 1H), 3.55 (dd, J=10.3, 3.3 Hz, 1H), 3.12–3.19 (m, 1H), 2.57–2.67 (m, 1H), 1.99 (s, 3H), 1.92–1.97 (m, 1H), 1.65–1.74 (m, 1H), 1.56 (d, J=6.8 Hz, 3H), 1.19–1.42 (m, 5H), 0.99–1.12 (m, 2H), 0.90 (t, J=7.2 Hz, 3H); MS (FAB) m/z 646 (M+H)+; HRMS (ESI) m/z calcd for C26H37FN5O7S3 646.1834, found 646.1836 (M+H)+.
Methyl (7S)-7-deoxy-7-[5-(4-fluoro-2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio]-6-N-[(2′S, 4′R)-1′-N-methyl-4′-(n-propyl)piperidine-2′-carbonyl]-α-thiolincosaminide (8e)
The title compound was synthesized from 7e (55 mg, 0.086 mmol) as a light yellow solid in 37% yield by the similar procedure to 8d. [α]D26 +88° (c 0.17, CH3OH); 1H NMR (400 MHz, CD3OD) δ 7.99 (dd, J=5.2 Hz, 1H), 7.85 (dd, J=8.9, 5.2 Hz, 1H), 7.61–7.67 (m, 1H), 5.28 (d, J=5.6 Hz, 1H), 4.69 (dd, J=10.2, 2.4 Hz, 1H), 4.45–4.52 (m, 1H), 4.43 (d, J=10.2 Hz, 1H), 4.12 (dd, J=10.2, 5.7 Hz, 1H), 3.84–3.86 (m, 1H), 3.57 (dd, J=10.2, 3.2 Hz, 1H), 2.92–3.00 (m, 1H), 2.57–2.65 (m, 1H), 2.25 (s, 3H), 2.10–2.18 (m, 1H), 2.00 (s, 3H), 1.83–1.90 (m, 1H), 1.68–1.74 (m, 1H), 1.57 (d, J=7.6 Hz, 3H), 1.20–1.40 (m, 7H), 0.86–0.92 (m, 3H); MS (FAB) m/z 660 (M+H)+; HRMS (ESI) m/z calcd for C27H39N5O7S3 660.1990, found 660.1995 (M+H)+.
Methyl (7S)-6-N-[(2′S, 4′R)-4′-(n-butyl)piperidine-2′-carbonyl]-7-deoxy-7-[5-(4-fluoro-2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio]-α-thiolincosaminide (7f)
Reaction of 6c (500 mg, 0.678 mmol) with 5-(4-fluoro-2-nitrophenyl)-1,3,4-thiadiazole-2-thiol (224 mg, 0.882 mmol) afforded 7f as a light yellow solid in 30% yield by the similar procedure to 7a. [α]D26 +100° (c 0.27, CH3OH); 1H NMR (400 MHz, CD3OD) δ 7.98 (dd, J=8.2, 2,6 Hz, 1H), 7.85 (dd, J=8.6, 5.2 Hz, 1H), 7.60–7.68 (m, 1H), 5.28 (d, J=5.6 Hz, 1H), 4.67 (dd, J=10.2, 2.4 Hz, 1H), 4.43–4.51 (m, 1H), 4.41 (d, J=10.2 Hz, 1H), 4.10 (dd, J=10.2, 5.6 Hz, 1H), 3.87–3.91 (m, 1H), 3.56 (dd, J=10.2, 3.2 Hz, 1H), 3.32–3.36 (m, 1H), 3.14–3.21 (m, 1H), 2.61–2.71 (m, 1H), 2.00–2.04 (m, 1H), 1.99 (s, 3H), 1.68–1.75 (m, 1H), 1.56 (d, J=7.1 Hz, 3H), 1.23–1.34 (m, 6H), 0.99–1.14 (m, 2H), 0.86–0.91 (m, 3H); MS (FAB) m/z 660 (M+H)+; HRMS (ESI) m/z calcd for C27H39FN5O7S3 660.1990, found 660.1994 (M+H)+.
Methyl (7S)-6-N-[(2′S, 4′R)-4′-(n-butyl)-1′-N-methylpiperidine-2′-carbonyl]-7-deoxy-7-[5-(4-fluoro-2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio]-α-thiolincosaminide (8f)
The title compound was synthesized from 7f (72 mg, 0.11 mmol) as a light yellow solid in 85% yield by the similar procedure to 8d. [α]D26 +94° (c 0.22, CH3OH); 1H NMR (400 MHz, CD3OD) δ 7.98 (dd, J=8.2, 2,6 Hz, 1H), 7.85 (dd, J=8.6, 5.2 Hz, 1H), 7.60–7.68 (m, 1H), 5.28 (d, J=5.6 Hz, 1H), 4.70 (dd, J=10.1, 2.7 Hz, 1H), 4.45–4.53 (m, 1H), 4.43 (d, J=10.1 Hz, 1H), 4.12 (dd, J=10.2, 5.6 Hz, 1H), 3.82–3.87 (m, 1H), 3.57 (dd, J=10.2, 3.3 Hz, 1H), 2.93–3.01 (m, 1H), 2.62–2.68 (m, 1H), 2.27 (s, 3H), 2.09–2.18 (m, 1H), 2.00 (s, 3H), 1.83–1.92 (m, 1H), 1.67–1.76 (m, 1H), 1.57 (d, J=7.1 Hz, 3H), 1.21–1.42 (m, 9H), 0.85–0.93 (m, 3H); MS (FAB) m/z 674 (M+H)+; HRMS (ESI) m/z calcd for C28H41FN5O7S3 674.2147, found 674.2160 (M+H)+.
Methyl (7S)-7-deoxy-7-[5-(5-methylamino-2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio]-6-N-[(2′S, 4′R)-4′-(n-propyl)piperidine-2′-carbonyl]-α-thiolincosaminide (7g)
Reaction of 6b (200 mg, 0.277 mmol) with 5-(5-methylamino-2-nitrophenyl)-1,3,4-thiadiazole-2-thiol (96.6 mg, 0.360 mmol) afforded 7 g as a yellow solid in 26% yield by the similar procedure to 7a. [α]D24 +91° (c 0.41, CH3OH); 1H NMR (300 MHz, CDCl3) δ 8.16 (d, J=8.7 Hz, 1H), 6.62–6.72 (m, 2H), 5.33 (d, J=5.7 Hz, 1H), 4.46–4.53 (m, 1H), 4.31 (d, J=9.9 Hz, 1H), 4.11–4.25 (m, 2H), 3.87–3.91 (m, 1H), 3.56–3.68 (m, 1H), 3.37–3.46 (m, 1H), 3.16–3.24 (m, 1H), 2.91 (s, 3H), 2.68–2.90 (m, 2H), 2.61–2.68 (m, 1H), 2.19 (s, 3H), 2.00–2.09 (m, 1H), 1.64–1.72 (m, 1H), 1.51 (d, J=6.9 Hz, 3H), 1.16–1.36 (m, 4H), 0.98–1.12 (m, 2H), 0.79–0.89 (m, 3H); MS (FAB) m/z 657 (M+H)+; HRMS (ESI) m/z calcd for C27H41N6O7S3 657.2193, found 657.2193 (M+H)+.
Methyl (7S)-7-deoxy-7-[5-(5-methoxy-2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio]-6-N-[(2′S, 4′R)-4′-(n-propyl)piperidine-2′-carbonyl]-α-thiolincosaminide (7h)
Reaction of 6b (200 mg, 0.277 mmol) with 5-(5-methoxy-2-nitrophenyl)-1,3,4-thiadiazole-2-thiol (97.0 mg, 0.360 mmol) afforded 7h as a light yellow solid in 24% yield by the similar procedure to 7a. [α]D24 +79° (c 0.12, CH3OH); 1H NMR (300 MHz, CDCl3) δ 8.11–8.20 (m, 1H), 7.07–7.16 (m, 2H), 6.62–6.72 (m, 2H), 5.36 (d, J=5.7 Hz, 1H), 4.47–4.53 (m, 1H), 4.09–4.35 (m, 3H), 3.94 (s, 3H), 3.77–3.89 (m, 1H), 3.55–3.70 (m, 1H), 3.36–3.51 (m, 1H), 3.16–3.29 (m, 1H), 2.65–2.79 (m, 2H), 2.15 (s, 3H), 1.98–2.09 (m, 1H), 1.64–1.73 (m, 1H), 1.52 (d, J=6.9 Hz, 3H), 1.17–1.36 (m, 4H), 0.98–1.13 (m, 2H), 0.77–0.89 (m, 3H); MS (FAB) m/z 658 (M+H)+; HRMS (ESI) m/z calcd for C27H40N5O8S3 658.2034, found 658.2038 (M+H)+.
Methyl (7S)-7-deoxy-6-N-[(2′S, 4′R)-4′-ethylpiperidine-2′-carbonyl]-7-[5-(4,5-dimethoxy-2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio]-α-thiolincosaminide (7i)
Reaction of 6a (158 mg, 0.223 mmol) with 5-(4,5-dimethoxy-2-nitrophenyl)-1,3,4-thiadiazole-2-thiol (80.0 mg, 0.267 mmol) afforded 7i as a light yellow solid in 11% yield by the similar procedure to 7a. [α]D25 +89° (c 0.46, CH3OH); 1H NMR (400 MHz, CD3OD) δ 7.75 (s, 1H), 7.21 (s, 1H), 5.28 (d, J=5.6 Hz, 1H), 4.67 (dd, J=10.0, 2.4 Hz, 1H), 4.40–4.48 (m, 2H), 4.10 (dd, J=10.3, 5.6 Hz, 1H), 3.98 (s, 3H), 3.96 (s, 3H), 3.88–3.91 (m, 1H), 3.56 (dd, J=10.3, 3.4 Hz, 1H), 3.14–3.20 (m, 1H), 2.64 (td, J=12.9, 2.7 Hz, 1H), 2.01 (s, 3H), 1.98 (br s, 1H), 1.69–1.75 (m, 1H), 1.55 (d, J=6.8 Hz, 3H), 1.39–1.49 (m, 1H), 1.24–1.34 (m, 2H), 1.00–1.12 (m, 2H), 0.92 (t, J=7.3 Hz, 3H); MS (FAB) m/z 674 (M+H)+; HRMS (ESI) m/z calcd for C27H40N5O9S3 674.1983, found 674.1988 (M+H)+.
Methyl (7S)-7-deoxy-7-[5-(4,5-dimethoxy-2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio]-6-N-[(2′S, 4′R)-4′-(n-propyl)piperidine-2′-carbonyl]-α-thiolincosaminide (7j)
Reaction of 6b (200 mg, 0.277 mmol) with 5-(4,5-dimethoxy-2-nitrophenyl)-1,3,4-thiadiazole-2-thiol (99.4 mg, 0.332 mmol) afforded 7j as a light yellow solid in 10% yield by the similar procedure to 7a. [α]D26 +97° (c 0.30, CH3OH); 1H NMR (400 MHz, CD3OD) δ 7.77 (s, 1H), 7.22 (s, 1H), 5.28 (d, J=5.6 Hz, 1H), 4.67 (dd, J=10.2, 2.4 Hz, 1H), 4.39–4.48 (m, 2H), 4.10 (dd, J=10.2, 5.6 Hz, 1H), 3.99 (s, 3H), 3.96 (s, 3H), 3.87–3.90 (m, 1H), 3.56 (dd, J=10.2, 3.0 Hz, 1H), 3.32–3.38 (m, 1H), 3.15–3.20 (m, 1H), 2.63–2.71 (m, 1H), 2.02 (s, 3H), 1.97–2.01 (m, 1H), 1.68–1.75 (m, 1H), 1.55 (d, J=7.1 Hz, 3H), 1.31–1.40 (m, 2H), 1.20–1.28 (m, 2H), 1.02–1.15 (m, 3H), 0.90 (t, J=7.2 Hz, 3H); MS (FAB) m/z 688 (M+H)+; HRMS (ESI) m/z calcd for C28H42N5O9S3 688.2139, found 688.2153 (M+H)+.
Methyl (7S)-6-N-[(2′S, 4′R)-4′-(n-butyl)piperidine-2′-carbonyl]-7-deoxy-7-[5-(4,5-dimethoxy-2-nitrophenyl)-1,3,4-thiadiazol-2-yl-thio]-α-thiolincosaminide (7k)
Reaction of 6c (200 mg, 0.271 mmol) with 5-(4,5-dimethoxy-2-nitrophenyl)-1,3,4-thiadiazole-2-thiol (106 mg, 0.353 mmol) afforded 7k as a light yellow solid in 19% yield by the similar procedure to 7a. [α]D26 +96° (c 0.49, CH3OH); 1H NMR (400 MHz, CD3OD) δ 7.75 (s, 1H), 7.21 (s, 1H), 5.28 (d, J=5.7 Hz, 1H), 4.63–4.69 (m, 1H), 4.38–4.47 (m, 2H), 4.10 (dd, J=10.1, 5.7 Hz, 1H), 3.98 (s, 3H), 3.96 (s, 3H), 3.87–3.90 (m, 1H), 3.54–3.58 (m, 1H), 3.27–3.29 (m, 1H), 3.12–3.18 (m, 1H), 2.58–2.67 (m, 1H), 2.01 (s, 3H), 1.92–2.00 (m, 1H), 1.65–1.74 (m, 1H), 1.55 (d, J=6.8 Hz, 3H), 1.46–1.52 (m, 1H), 1.20–1.34 (m, 6H), 0.99–1.11 (m, 2H), 0.86–0.93 (m, 3H); MS (FAB) m/z 702 (M+H)+; HRMS (ESI) m/z calcd for C29H44N5O9S3 702.2296, found 702.2294 (M+H)+.
Methyl 7-deoxy-7-methyl-6-N-[(2′S, 4′R)-4′-(n-propyl)piperidine-2′-carbonyl]-α-thiolincosaminide (VIC-105555) and methyl 7-deoxy-7-methyl-6-N-[(2′R, 4′S)-4′-(n-propyl)piperidine-2′-carbonyl]-α-thiolincosaminide (10)
To a solution of compound 9 (1.36 g, 3.41 mmol) in MeOH-H2O (5:2, 100 ml) were added conc. hydrochloric acid (410 μl, 4.90 mmol), platinum (IV) oxide (1.16 g, 5.12 mmom) and stirred under a hydrogen pressure of 3.9 MPa at room temperature for 24 h. The mixture was filtered and concentrated under reduced pressure. The resulting residue was added saturated aqueous NaHCO3 and extracted with ethyl acetate. The organic phase was dried over Na2SO4, filtered and concentrated under reduced pressure. The resulting residue was purified by silica gel column chromatography (CHCl3/MeOH/28% aq NH4OH=95/5/0.5 to 85/15/1.5), to afford the title compounds as colorless solids (VIC-105555; more polar, 630 mg, 46%: 10; less polar, 653 mg, 47%). VIC-105555: [α]D26 +170° (c 0.91, CH3OH); 1H NMR (400 MHz, CD3OD) δ 5.24 (d, J=5.6 Hz, 1H), 4.16 (d, J=10.0, 3.1 Hz, 1H), 4.08 (dd, J=10.3, 5.6 Hz, 1H), 4.04 (d, J=10.0 Hz, 1H), 3.79–3.81 (m, 1H), 3.51 (dd, J=10.3, 3.2 Hz, 1H), 3.24–3.29 (m, 1H), 3.10–3.16 (m, 1H), 2.62 (td, J=12.8, 2.8 Hz, 1H), 2.12–2.20 (m, 1H), 2.11 (s, 3H), 1.89–1.96 (m, 1H), 1.66–1.73 (m, 1H), 1.46–1.57 (m, 1H), 1.32–1.41 (m, 2H), 1.21–1.28 (m, 2H), 0.98–1.10 (m, 2H), 0.89–0.96 (m, 9H); MS (FAB) m/z 405 (M+H)+; HRMS (ESI) m/z calcd for C19H37N2O5S 405.2418, found 405.2422 (M+H)+. 10: [α]D26 +200° (c 1.2, CH3OH); 1H NMR (400 MHz, CD3OD) δ 5.25 (d, J=5.7 Hz, 1H), 4.14 (dd, J=9.9, 3.6 Hz, 1H), 4.09 (dd, J=10.2, 5.7 Hz, 1H), 4.06 (d, J=9.9 Hz, 1H), 3.76–3.79 (m, 1H), 3.52 (dd, J=10.2, 3.5 Hz, 1H), 3.24 (dd, J=11.6, 2.8 Hz, 1H), 3.08–3.14 (m, 1H), 2.61 (td, J=12.7, 2.7 Hz, 1H), 2.12–2.19 (m, 1H), 2.11 (s, 3H), 1.93–1.99 (m, 1H), 1.65–1.72 (m, 1H), 1.42–1.55 (m, 1H), 1.31–1.42 (m, 2H), 1.20–1.29 (m, 2H), 0.96–1.11 (m, 2H), 0.88–0.96 (m, 9H); MS (FAB) m/z 405 (M+H)+; HRMS (ESI) m/z calcd for C19H37N2O5S 405.2418, found 404.2423 (M+H)+.
In vitro antibacterial activity
MIC was determined by the agar dilution method. Test strains were subjected to seed culture using sensitivity test broth (Nissui Pharmaceutical, Tokyo, Japan) cultured on blood agar plate for S. pneumoniae, S. pyogenes and H. influenzae. A 5 μl portion of cell suspension of the test strains having about 106 CFU ml−1 was inoculated into sensitivity disk agar (Nissui Pharmaceutical) supplemented with 5% horse blood and incubated at 37 °C for 20 h. Then, MIC was defined as the lowest drug concentration that prevented visible growth.

Synthesis of key intermediates having a 4-substituted piperidine 2-carbonyl group. Conditions: (a) DCC, HOBt, DMF, rt; (b) TMSCl, HMDS, pyridine, rt; (c) 6N AcOH, MeOH, rt.

Synthesis of (7S)-7-deoxy-7-(5-phenyl-1,3,4-thiadiazol-2-yl-thio)-LCM derivatives having a 4-substituted piperidine 2-carbonyl group. Conditions: (a) ArSH, DEAD, PPh3, THF or toluene, 0 °C to rt; (b) TFA, DCM, −20 °C to rt; (c) 36% HCHO, NaBH(OAc)3, AcOH, MeOH or EtOH, rt.

Synthesis of VIC-105555 and its 2′-α-4′-α-cis isomer 10. Condition: H2, PtO2, conc. HCl, MeOH-H2O, rt.
References
Morimoto, S., Takahashi, Y., Watanabe, Y. & Ōmura, S. Chemical modification of erythromycins. I. Synthesis and antibacterial activity of 6-O-methylerythromycins A. J. Antibiot. 37, 187–189 (1984).
Slobodan, D. et al. Erythromycin series. Part 13. Synthesis and structure elucidation of 10-dihydro-10-deoxo-11-methyl-11-azaerythromycin A. J. Chem. Res. Synop. 1988, 152–153 (1988).
Ajito, K., Miura, T., Furuuchi, T. & Tamura, A. Sixteen-membered macrolides: chemical modifications and future applications. Heterocycles 89, 281–352 (2014).
Sato, T. et al. In vitro antibacterial activity of modithromycin, a novel 6,11-bridged bicyclolide, against respiratory pathogens, including macrolide-resistant Gram-positive cocci. Antimicrob. Agents Chemother. 55, 1588–1593 (2011).
Denis, A. et al. Synthesis and antibacterial activity of HMR 3647 a new ketolide highly potent against erythromycin-resistant and susceptible pathogens. Bioorg. Med. Chem. Lett. 9, 3075–3080 (1999).
Clay, K. D. et al. Severe hepatotoxicity of telithromycin: three case reports and literature review. Ann. Intern. Med. 144, 415–420 (2006).
Miura, T. et al. Novel azalides derived from sixteen-membered macrolides. I. Isolation of the mobile dialdehyde and its one-pot macrocyclization with an amine. J. Antibiot. 60, 407–435 (2007).
Miura, T. et al. Novel azalides derived from 16-membered macrolides. III. Azalides modified at the C-15 and 4” positions: improved antibacterial activities. Bioorg. Med. Chem. 18, 2735–2747 (2010).
Mason, D. J., Dietz, A. & Deboer, C. Lincomycin, a new antibiotic I. Discovery and biological properties. Antimicrob. Agents Chemother. 1962, 554–559 (1962).
Birkenmeyer, R. D. & Kagan, F. Lincomycin. XI. Synthesis and structure of clindamycin. A potent antibacterial agent. J. Med. Chem. 13, 616–619 (1970).
Weisblum, B. Erythromycin resistance by ribosome modification. Antimicrob. Agents Chemother. 39, 577–585 (1995).
Tsuzuki, K. et al. Motilides, macrolides with gastrointestinal motor stimulating activity. I. O-substituted and tertiary N-substituted derivatives of 8,9-anhydroerythromycin A 6,9-hemiacetal. Chem. Pharm. Bull. 37, 2687–2700 (1989).
Shah, P. J., Vakil, N. & Kabakov, A. Role of intravenous immune globulin in streptococcal toxic shock syndrome and Clostridium difficile infection. Am. J. Health Syst. Pharm. 72, 1013–1019 (2015).
Sztaricskai, F. et al. Semisynthetic modification of antibiotic lincomycin. J. Antibiot. 49, 941–943 (1996).
Goffic, L. F. Structure activity relationships in lincosamide and streptogramin antibiotics. J. Antimicrob. Chemother. 16 (Suppl A), 13–21 (1985).
Umemura, E. et al. Synthesis of novel lincomycin derivatives and their in vitro antibacterial activities. J. Antibiot. 66, 195–198 (2013).
Wakiyama, Y. et al. Synthesis and structure–activity relationships of novel lincomycin derivatives. Part 1. Newly generated antibacterial activities against Gram-positive bacteria with erm gene by C-7 modification. J. Antibiot. 69, 368–380 (2016).
Wakiyama, Y. et al. Synthesis and structure-activity relationships of novel lincomycin derivatives. Part 2. Synthesis of 7(S)-7-deoxy-7-(4-morpholinocarbonylphenylthio)lincomycin and its 3-dimensional analysis with rRNA. J. Antibiot. 69, 428–439 (2016).
Wakiyama, Y. et al. Synthesis and structure-activity relationships of novel lincomycin derivatives part 3: discovery of the 4-(pyrimidin-5-yl)phenyl group in synthesis of 7(S)-thiolincomycin analogs. J. Antibiot. 70, 52–64 (2017).
Kumura, K. et al. Synthesis and antibacterial activity of novel lincomycin derivatives. I. Enhancement of antibacterial activities by introduction of substituted azetidines. J. Antibiot. 69, 440–445 (2016).
Kumura, K. et al. Synthesis and antibacterial activity of novel lincomycin derivatives. II. Synthesis and antibacterial activity of novel lincomycin derivatives. II. Exploring (7S -7-(5-aryl-1,3,4-thiadiazol-2-yl-thio)-7-deoxylincomycin derivatives. J. Antibiot. 70, 655–663 (2017).
Kumura, K. et al. Synthesis and antibacterial activity of novel lincomycin derivatives. III. Optimization of a phenyl thiadiazole moiety. J. Antibiot. doi:10.1038/ja.2017.59 (2017).
Birkenmeyer, R. D., Kroll, S. J., Lewis, C., Stern, K. F. & Zurenko, G. E. Synthesis and antimicrobial activity of clindamycin analogues: pirlimycin, a potent antibacterial agent. J. Med. Chem. 27, 216–223 (1984).
Lewis, J. G. et al The lincomycin derivatives possessing antibacterial activity. WO/2004/016632 A2, 26 February (2004).
Lewis, J. G. et al Novel Antimicrobial 7-Methyl Lincosamides: Pipecolamide Analogs. 43rd Interscience Conference on Antimicrobial Agents and Chemotherapy. Poster F-1389 (2004).
Lopez, S. L. et al Characterization of the Spectrum of In Vitro Activity of VIC-105555, a New Lincosamide. 44th Interscience Conference on Antimicrobial Agents and Chemotherapy. Poster F-2038 (2004).
O’Dowd, H. et al. Novel antibacterial azetidine lincosamides. Bioorg. Med. Chem. Lett 18, 2645–2648 (2008).
Shuman, R. T., Ornstein, P. L., Paschal, J. W. & Gesellchen, P. D. An improved synthesis of homoproline and derivatives. J. Org. Chem. 55, 738–741 (1990).
Schroeder, W., Bannister, B. & Hoeksema, H. Lincomycin. III. The structure and stereochemistry of the carbohydrate moiety. J. Am. Chem. Soc. 89, 2448–2453 (1967).
Eldere, J. V. et al. Overview of antimicrobial options for Mycoplasma pneumoniae pneumonia: focus on macrolide resistance. Clin. Respir. J. 11, 419–429 (2017).
Acknowledgements
We thank Dr E Shitara, Mr. A Tamura and Dr T Okutomi for valuable scientific discussion. We are grateful to Professor Emeritus Dr M Konno for supervision through our in-house drug discovery program in lincomycin field. We are also grateful to Ms. T Miyara, Ms. S Miki, Ms. K Kaneda, Dr T Murata and Mr. S Sato for contribution toward analytical chemistry; Mr. Y Takayama for biological studies; and Ms. M Takagi for manuscript. We also thank Ms. M Ishii for direction in intellectual properties.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Kumura, K., Wakiyama, Y., Ueda, K. et al. Synthesis and antibacterial activity of novel lincomycin derivatives. IV. Optimization of an N-6 substituent. J Antibiot 70, 1112–1121 (2017). https://doi.org/10.1038/ja.2017.143
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ja.2017.143
- Springer Japan KK