Abstract
Purpose
To describe the frequency, characteristics, and treatment outcome of persistent fetal vasculature (PFV) in children undergoing surgery for congenital and infantile cataract in the first 2 years of life.
Patients and methods
Observational population-based cohort study with case identification through active surveillance and standardised data collection via a national clinical network, the British Isles Congenital Cataract Interest Group (BCCIG).
Results
The IoLunder2 cohort comprises 246 children undergoing surgery for bilateral and unilateral congenital and infantile cataract in the first 2 years of life. A total of 58/246 (24%) children had PFV (%): overall, 46/95 (46%) with unilateral cataract, and 12/141 (8%) with bilateral disease. Anterior segment vascular remnants were more common in bilateral than unilateral disease (75 vs 11%, P=0.01). At 1 year after surgery, 20% of children with bilateral PFV and 24% with unilateral had achieved normal vision for age within the operated eye. The prevalence of post-operative glaucoma was 9% (of children with bilateral disease) and 4% (unilateral).
Conclusion
PFV is significantly more common than previously reported, and outcomes are comparable to that for congenital and infantile cataract overall.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Persistent fetal vasculature (PFV) comprises a spectrum of clinical features caused by failure of the normal process of regression or apoptosis of the fetal intraocular vasculature, which usually occurs during the second trimester. PFV was previously termed persistent hyperplastic primary vitreous (PHPV), due to the characteristic feature of a plaque of remnant tissue sitting on the posterior face of a cataractous lens, attached to a persistent stalk of hyaloid vasculature. However, any part of the fetal vasculature can persist, resulting in iris, pupillary, or optic nerve vascular remnants.1 Congenital pupillary fibrovascular membranes, rather than being a separate entity, are histopathologically similar to the retrolenticular manifestations of PFV.2
Cataract is a frequent secondary event in PFV due to invasion of the vascular remnants, lenticular swelling, intralenticular haemorrhage, or calcification.1, 3 PFV features have been found in a fifth of cases of unilateral congenital or infantile cataract, but are much less commonly noted in bilateral cataract.4 Cataract surgery in children with PFV is associated with a higher incidence of adverse events and worse outcomes.5, 6 In addition, bilateral cases of PFV may be associated with systemic abnormalities, which impact on a child’s global development.1, 7
The British Isles study of primary intraocular lens implantation in children under 2 years with congenital or infantile cataract (IoLunder2) is a prospective observational cohort study, undertaken through the British Isles Congenital Cataract Interest Group (BCCIG), a collaborative clinical research network of ophthalmologists involved in the management of children with congenital and infantile cataract. IoLunder2, which comprises a nationally representative group of children who have undergone cataract extraction with or without intraocular lens implantation in the United Kingdom and Ireland, aims to describe treatment outcomes and determine the predictors of outcome. We report here on the morphology of PFV and outcomes following surgery within this cohort.
Materials and methods
Children eligible for inclusion into the IoLunder2 study were those resident in the British Isles undergoing primary surgery for congenital or infantile cataract in the first 2 years of life. Active surveillance was undertaken over a 2-year study period (January 2009 to December 2010) through a national clinical network, the British Isles Congenital Cataract Interest Group (BCCIG). Following parental consent, standardised data collection using a structured proforma was undertaken through the network. The IoLunder2 study methodology has been reported in detail previously.8
A standardised classification of the presence and morphology of PFV was developed for the study. PFV was classified within one of these three mutually exclusive categories using an anatomically derived taxonomy (Supplementary e-supplement):
-
1
Isolated posterior capsular plaque/persistent posterior tunica vasculosa.
-
2
Anterior PFV only (persistent iridohyaloid/pupillary membrane/anterior tunica vasculosa).
-
3
Complex PFV (posterior plaque plus other manifestation).
Within the complex PFV category, the presence of other signs (anterior PFV, persistent hyaloid artery, centrally dragged ciliary processes, optic nerve head hypoplasia / dysplasia, macular abnormalities, and retinal detachment) were dichotomised as present or absent.
Statistical analyses were performed using STATA SE 10/11 (Stata Corp., Chicago, IL, USA). Children with unilateral cataract were analysed separately from those with bilateral disease. Findings of descriptive analysis are presented as proportions for binary or categorical data, with 95% confidence intervals, where appropriate.
Ethics and research governance approval was granted by the relevant United Kingdom/Ireland bodies, and the research adhered to the tenets of the Declaration of Helsinki.
Results
Two hundred fifty-four (83%) of eligible children were recruited into IOLunder2. Complete pre and per operative data are available for 246, and post-operative outcomes at 1 year following surgery were available for 221. PFV was present in 58 of the 246 children (23.6%, Table 1): 46 of the 99 children with unilateral cataract (46.5%, 95% confidence interval CI 36–56%), and a significantly lower proportion of children with bilateral cataract (12/147 children, 8.2%, 95% CI 0–24%). Compared with other children with unilateral congenital and infantile cataract, those with PFV presented to clinicians at a similar age. All children with unilateral congenital cataract associated with PFV had negative family histories for congenital cataract. Compared with other children with bilateral cataract, diagnosis of PFV cataract occurred at a later age, children were less likely to have a family history of cataract, and more likely to be affected by a systemic disorder or anomaly, although these differences did not reach statistical significance (Table 2).
Manifestations of PFV in unilateral and bilateral cataract
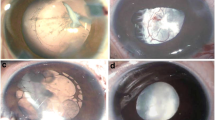
The most common finding in PFV-associated unilateral cataract was persistent posterior tunica vasculosa lentis, manifesting as either a posterior plaque or membrane (41 children, 92%) (Figure 1a). However, for half of all children with a persistent hyaloid artery (9/18), there was no other retrolental vascular manifestation (Figure 1a). Bilateral PFV was more likely to have present as something other than the classic retrolental plaque: in comparison with children with unilateral PFV, a significantly higher proportion of children with bilateral PFV-associated cataract had iris, pupil, or anterior capsular vascular remnants (75 vs 11%, χ2-test for difference between proportions or DIP P=0.01, 95% CI DIP 21–71%). Bilateral PFV also showed evidence of within-child phenotypic variation: in two of the seven cases of bilateral PFV, children had asymmetric manifestations of persistence in both eyes. In comparison with congenital and infantile cataract with no evidence of PFV, bilateral and unilateral cataract eyes with evidence of persistent vasculature were no more likely to be microphthalmic. No child with bilateral disease had stretched ciliary processes, while eight children (18%, 95% CI 7–29%) with unilateral PFV exhibited ciliary process traction (Figure 1b).
Systemic disorders in children with PFV-related cataract
Five of the 46 (11%) children with unilateral cataract associated with PFV were also affected by a non-ocular disorder or anomaly (Table 2). These systemic abnormalities comprised: congenital toxoplasmosis, trisomy 21, haemophilia, and in two children for whom a formal diagnosis is awaited, lower intestinal and genitourinary malformations, and atrioseptal congenital cardiac anomaly. The child with congenital toxoplasmosis manifested complex PFV, while the other four children had isolated posterior hyaloid stalks.
Over half of the children, 7/12, with bilateral cataract and PFV had systemic disorders (Table 3). Three children had hereditary ocular or systemic disease, but for the majority of children with a systemic disorder a formal diagnosis or genotype has yet to be determined.
Outcomes following surgery for cataract associated with persistent fetal vasculature
All children with PFV underwent primary surgery involving anterior capsulotomy, lens aspiration, primary posterior capsulotomy, and primary anterior vitrectomy. Patent hyaloid stalks underwent diathermy prior to stalk division. A total of 16/46 unilateral PFV eyes (35%), and 4/12 eyes of children with bilateral PFV (33%) underwent primary IOL implantation. One year following surgery for unilateral PFV cataract, 24% of operated eyes had achieved vision within normal range for age (95% CI 12–36) and 10% had only perception of light vision or worse (CI 1–19%). The proportion of children with bilateral PFV cataract achieving normal vision in the better eye or with both eyes open was 20% (CI 0–43%) and all children had vision better than perception of light. The subgroup of children with complex PFV achieved relatively poorer outcomes. No child with optic nerve head or retinal involvement achieved vision within the normal range for age 1 year after surgery, compared with 35% (CI 20–47%) of children with unilateral PFV not affecting the fundus.
No child with congenital cataract-related PFV was diagnosed with glaucoma pre-operatively, and during the first post-operative year secondary glaucoma was diagnosed in 4% of children with unilateral (n=2, 95% CI 0–10%) and 9% with bilateral PFV cataract (n=1, 95% CI=0–15%). Of the two unilateral cases that developed secondary glaucoma, one had a posterior plaque only, the other a plaque and stalk. The bilateral cases had posterior plaque only. The frequency of post-operative glaucoma was not higher for eyes with anterior manifestations of PFV in either bilateral or unilateral cataract.
Discussion
We have found that the proportion of children who undergo surgery for congenital cataract associated with PFV is significantly higher than previously reported, even allowing for the widening of the definition of PFHV from that of PHPV. Forty-two of the 95 children with unilateral cataract (44%) within this present cohort would previously have been diagnosed with PHPV, which remains a significantly higher proportion. This is unlikely to reflect a ‘true’ increase in the incidence of PFV, as such a rise would require a dramatic biological influence. Instead, our findings suggest that there is improved clinical recognition of the signs of PFV. Furthermore, features of PFV may only be visible during removal of the cataractous lens, and children with PFV may present earlier in life. Thus a study that recruits children undergoing surgery in early life may report a higher frequency of affected children. An Austrian study that prospectively recruited 31 children aged 0–15 years old with unilateral cataract reported that all had some feature of persistent vasculature.7 Two large-scale population-based studies have described PFV as only affecting a relatively low proportion of children with cataract. Rahi et al4 reported that 22% of children aged 0–15 years diagnosed with unilateral congenital or infantile cataract in a national study were affected by PFV, and investigators in Toronto reported the frequency to be 11% in children aged under 16 years at surgery for unilateral cataract.9 These study populations described children diagnosed at any age with congenital and infantile cataract. This difference from the IoLunder2 study may partly explain the observed differences in frequency of PFV, as children with PFV manifestations may be diagnosed earlier than those without. The converse was, however, true for the IoLunder2 cohort where PFV was associated with later diagnosis in bilateral cataract.
In total, 36% of children with bilateral cataract, and 20% of those with unilateral cataract within the IoLunder2 cohort achieved normal vision for age within the operated eye 1 year following surgery.8 For the majority of children with congenital cataract associated with PFV, there is no evidence of worse outcome when compared with reported outcomes for the wider group of children within the IoLunder2 cohort. Other investigators have theorised that the ocular maldevelopment secondary to failure of vascular regression results in worse functional and structural outcomes,1, 5 with worse vision for affected children and higher rates of post-operative glaucoma, and previous studies have reported a higher rate of per- and post-operative complications in eyes affected by PFV,6 but within IoLunder2 the only association with poor outcome was for those with optic nerve head or retinal anomalies in associated with PFV. Congenital and infantile cataract per se are reflections of significant insult to lens development, and it may be that there is no ‘summative’ negative impact from the additional presence of persistent vasculature. The early outcomes from IoLunder2 should be broadly predictive of later outcomes,10 although more cases of post-operative glaucoma are likely to emerge.11
Stretched ciliary processes were once considered pathognomic for PFV,3 but are now known to be present in other vitreoretinopathies such as retinopathy of prematurity or vitreoretinal dysplasia.1 Our findings suggest that children with bilateral PFV are less likely to have ciliary traction, but also more likely to have anterior segment manifestations, and more likely to have a non-ophthalmic disorder, but the significance of this is unclear. There may be different pathogenetic pathways involved for children with bilateral and unilateral disease. Emerging evidence suggests that in the majority of children affected by bilateral congenital and infantile cataract where there is no obvious associated ‘syndrome’, it is possible to identify a plausible pathogenic single-gene mutation.12 The aetiology of PFV is poorly understood, but animal models have identified a role for the failure of both the macrophage-orchestrated extrinsic apoptotic pathway13 and the tumour suppressor gene intrinsic apoptotic pathway14 in the pathogenesis of the persistence of the nutrient fetal vasculature. One gene complex implicated in the failure of ocular fetal vasculature apoptosis via the intrinsic pathway is frizzled/wnt,15 which codes for a transmembrane protein. This protein is also implicated in the allelic congenital human retinal vascular development disorders of Norrie disease and familial exudative vitreoretinopathy, with the latter being described clinically as a ‘hereditary form of retinopathy of prematurity (ROP)’. It has also been suggested that PFV is a result of failure of apoptosis due to reduced exposure to VEGF. This pathway has been supported by the changing patterns of VEGF receptors on mammalian fetal intraocular vasculature,16 and is a pathway of current clinical interest due to the use of VEGF inhibitors as treatment for ROP.
Primary prevention of unilateral cataract is an important goal as visual outcomes for unilateral remains poor. Understanding the aetiology and pathogenesis of PFV may contribute to this aim, and may increase the understanding of the vascularisation of the fetal retina and possibly of the entire fetal cerebral system. PFV is thus a worthy candidate for future genetic research, but will require meticulous ‘phenotyping’, that is, detailed description of morphology with pre-operative clinical imaging, ahead of investigation into genetic markers of the disorder.
Prompt diagnosis affording prompt treatment remains the most important management intervention for achieving good visual outcomes in congenital and infantile cataract. The early age at diagnosis for the majority of children with PFV-related congenital cataract can be considered as an indicator of the success of the UK’s ocular anomaly screening programme. Children undergo a red reflex test within 72 h of birth, and a second examination with their general practitioner (family doctor) as part of the 6–8 week infant physical examination screening. However, this cohort consists only of children who have undergone cataract surgery. There may be a small number of children who did not undergo cataract surgery as late diagnosis had resulted in amblyopia sufficiently dense to be unlikely to respond successfully to treatment. Six children (50%) with bilateral PFV cataract were diagnosed after 8 weeks of age. It is unclear whether these represent cases of ‘late’ diagnosis, or whether cases were diagnosed at a later age following progression of PFV-related lens opacity.
The IoLunder2 study is able to provide population level data on the prevalence and manifestations of PFV within children undergoing surgery for congenital and infantile cataract in the first 2 years of life, better informing clinicians on the characteristics and outcomes of this complex, and uncommon disorder. Ongoing follow up of this cohort will provide natural history and outcomes for children who have undergone surgery for PFV-related cataract.

References
Goldberg MF . Persistent fetal vasculature (PFV): an integrated interpretation of signs and symptoms associated with persistent hyperplastic primary vitreous (PHPV). LIV Edward Jackson Memorial Lecture. Am J Ophthalmol 1997; 124 (5): 587.
Lambert SR, Buckley E, Lenhart P, Zhang Q, Grossniklaus H . Congenital fibrovascular pupillary membranes: clinical and histopathologic findings. Ophthalmology 2012; 119 (3): 634–641.
Reese AB . Persistent hyperplastic primary vitreous. Am J Ophthalmol 1955; 40 (3): 317–331.
Rahi JS, Dezateux C . Congenital and infantile cataract in the United Kingdom: underlying or associated factors. British Congenital Cataract Interest Group. Invest Ophthalmol Vis Sci 2000; 41 (8): 2108–2114.
Sisk RA, Berrocal AM, Feuer WJ, Murray TG . Visual and anatomic outcomes with or without surgery in persistent fetal vasculature. Ophthalmology 2010; 117 (11): 2178–2183.
Morrison DG, Wilson ME, Trivedi RH, Lambert SR, Lynn MJ . Infant Aphakia Treatment Study: effects of persistent fetal vasculature on outcome at 1 year of age. J AAPOS 2011; 15 (5): 427–431.
Mullner-Eidenbock A, Amon M, Moser E, Klebermass N . Persistent fetal vasculature and minimal fetal vascular remnants* 1: a frequent cause of unilateral congenital cataracts. Ophthalmology 2004; 111 (5): 906–913.
Solebo AL, Russell-Eggitt I, Cumberland PM, Rahi JS . Risks and outcomes associated with primary intraocular lens implantation in children under 2 years of age: the IoLunder2 cohort study. Br J Ophthalmol 2015; 99 (11): 1471–1476.
Lim Z, Rubab S, Chan YH, Levin AV . Pediatric cataract: the Toronto Experience -Etiology. Am J Ophthalmol 2010; 149 (6): 887–892.
Lambert SR, Lynn MJ, Hartmann EE, DuBois L, Drews-Botsch C, Freedman SF et al. Comparison of contact lens and intraocular lens correction of monocular aphakia during infancy: a Randomized Clinical Trial of HOTV Optotype Acuity at age 4.5 years and clinical findings at age 5 years. JAMA Ophthalmol 2014; 132: 676–682.
Chak M, Rahi JS . Incidence of and factors associated with glaucoma after surgery for congenital cataract: findings from the British Congenital Cataract Study. Ophthalmology 2008; 115 (6): 1013–1018.
Gillespie RL, O'Sullivan J, Ashworth J, Bhaskar S, Williams S, Biswas S et al. Personalized diagnosis and management of congenital cataract by next-generation sequencing. Ophthalmology 2014; 121 (11): 2124–2137.
Lang RA, Bishop JM . Macrophages are required for cell death and tissue remodeling in the developing mouse eye. Cell 1993; 74 (3): 453–462.
Hahn P, Lindsten T, Tolentino M, Thompson CB, Bennett J, Dunaief JL . Persistent fetal ocular vasculature in mice deficient in bax and bak. Arch Ophthalmol 2005; 123 (6): 797.
Lobov IB, Rao S, Carroll TJ, Vallance JE, Ito M, Ondr JK et al. WNT7b mediates macrophage-induced programmed cell death in patterning of the vasculature. Nature 2005; 437 (7057):417–421.
Gogat K, Le Gat L, Van Den Berghe L, Marchant D, Kobetz A, Gadin S et al. VEGF and KDR gene expression during human embryonic and fetal eye development. Invest Ophthalmol Vis Sci 2004; 45 (1): 7.
Acknowledgements
This work was undertaken at UCL Institute of Child Health/Great Ormond Street Hospital for Children, which received a proportion of funding from the Department of Health’s NIHR Biomedical Research Centres funding scheme. Lola Solebo is funded, and Jugnoo Rahi is supported in part by the National Institute for Health Research (NIHR) Biomedical Research Centre based at Moorfields Eye Hospital NHS Foundation Trust and UCL Institute of Ophthalmology. Lola Solebo was supported by a personal fellowship awarded by the Ulverscroft Vision Research Group. The views expressed are those of the author(s) and not necessarily those of the funders, the NHS, the NIHR, or the Department of Health.
Disclaimer
The funding organisations had no role in the design or conduct of this research.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Eye website
Supplementary information
Rights and permissions
About this article
Cite this article
Solebo, A., Russell-Eggitt, I., Cumberland, P. et al. Congenital cataract associated with persistent fetal vasculature: findings from IoLunder2. Eye 30, 1204–1209 (2016). https://doi.org/10.1038/eye.2016.159
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/eye.2016.159
- Springer Nature Limited
This article is cited by
-
Surgical outcomes of cataract surgery in anterior and combined persistent fetal vasculature using a novel surgical technique: a single center, prospective study
Graefe's Archive for Clinical and Experimental Ophthalmology (2021)
-
Cataract management in children: a review of the literature and current practice across five large UK centres
Eye (2020)
-
Intraocular lens implantation in combination with lensectomy and vitrectomy for persistent fetal vasculature
Graefe's Archive for Clinical and Experimental Ophthalmology (2020)