Abstract
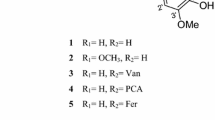
Cyperus rotundus L. (Suada, Sueda, family: Cyperaceae) is vastly spread in several world’s subtropical and tropical regions. It had variable traditional uses and bioactivities. A new flavonol derivative: cyperaflavoside (myricetin 3,3′,5′-trimethyl ether 7-O-β-d-glucopyranoside) and five flavonoids: vitexin, orientin, cinaroside, quercetin 3-O-β-d-glucopyranoside, and myrcetin 3-O-β-d-glucopyranoside were separated from the methanolic extract of C. rotundus aerial parts. Their structures were verified based on UV, IR, NMR (1D and 2D), HRESIMS, and comparison with literature. All metabolites were assessed for their 5-lipoxygenase inhibitory potential. All compounds possessed 5-lipoxygenase inhibitory potentials with IC50s 5.1, 4.5, 5.9, 4.0, 3.7, and 2.3 μM, respectively, in comparison to indomethacin (IC50 0.98 μM). These results supported the traditional uses of C. rotundus in treating inflammation and its related symptoms.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Agrawal, P.K., 1992. NMR spectroscopy in the structural elucidation of oligosaccharides and glycosides. Phytochemistry 31, 3307–3330.
Ahmad, M., Mahayrookh, Mehjabeen, Rehman, A.B., Jahan, N., 2012. Analgesic, antimicrobial and cytotoxic effect of Cyperus rotundus ethanol extract. Pakistan J. Pharmacol. 29, 7–13.
Al-Attas, A.A.M., El-Shaer, N.S., Mohamed, G.A., Ibrahim, S.R.M., Esmat, A., 2015. New antiinflammatory sesquiterpenes from the rhizomes of Costus speciosus. J. Ethnopharmacol. 176, 365–374.
Al-Musayeib, N.M., Mohamed, G.A., Ibrahim, S.R.M., Ross, S.A., 2013. Lupeol-3-O-decanoate, a new triterpene ester from Cadaba farinosa Forsk. growing in Saudi Arabia Med. Chem. Res. 22, 5297–5302.
Bawden, K., Quant, J., Raman, A., 2002. An alpha-amylase assay for the guided fractionation of anti-diabetic plants. Fitoterapia 2, 167.
Boulos, L., 1983. Medicinal Plants of North Africa. Reference Publications, Algonac, pp. 82.
Boulos, L., El-Hadidi, M.N., 1984. The Weed Flora of Egypt. The American University in Cairo Press, Cairo, pp. 58.
Braca, A., Bilia, A.R., Mendez, J., Morelli, I., 2001. Myricetin glycosides from Licania densiflora. Fitoterapia 72, 182–185.
Collenette, S., 1999. Wild flowers of Saudi Arabia King of Saudi Arabia: National Commission for Wild life Conservation and Development (NCWCD) and Sheila Collenette. King Fahd National Library, Kingdom of Saudi Arabia, pp. 286.
Daswani, P.G., Brijesh, S., Tetali, P., Birdi, T.J., 2011. Studies on the activity of Cyperus rotundus Linn. tubers against infectious diarrhea. Indian J. Pharmacol. 43, 340–344.
García-Lafuente, A., Guillamón, E., Villares, A., Rostagno, M.A., Martínez, J.A., 2009. Flavonoids as anti-inflammatory agents: implications in cancer and cardiovascular disease. Inflamm. Res. 58, 537–552.
Gautam, R., Jachak, S.M., 2009. Recent developments in antiinflammatory natural products. Med. Res. Rev. 29, 767–820.
Harborne, J.B., 1994. The Flavonoids Advances in Research Since 1986. Chapman and Hall, London.
Ibrahim, S.R.M., Mohamed, G.A., Al-Musayeib, N.M., 2012. New constituents from the rhizomes of Egyptian Iris germanica L. Molecules 17, 2587–2598.
Jeong, S., Miyamoto, T., Inagaki, M., Kim, Y., Higuchi, R., 2000. Rotundines AC, three novel sesquiterpene alkaloids from Cyperus rotundus. J. Nat. Prod. 63, 673–675.
Jin, J.H., Lee, D.U., Kim, Y.S., Kim, H.P., 2011. Anti-allergic activity of sesquiterpenes from the rhizomes of Cyperus rotundus. Arch. Pharm. Res. 34, 223–228.
Khedr, A.I.M., Ibrahim, S.R.M., Mohamed, G.A., Ahmed, H.E.A., Ahmad, A.S., Ramadan, M.A., Abd El-Baky, A.E., Yamada, K., Ross, S.A., 2016. New ursane triterpenoids from Ficus pandurata and their binding affinity for human cannabinoid and opioid receptors. Arch. Pharm. Res. 39, 897–911.
Kim, H.P., Mani, I., Iversen, L., Ziboh, V.A., 1998. Effects of naturally-occurring flavonoids and biflavonoids on epidermal cyclooxygenase and Iipoxygenase from guinea-pigs. Prostaglandins Leukot. Essent. Fatty Acids 58, 17–24.
Kim, H.P., Son, K.H., Chang, H.W., Kang, S.S., 2004. Antiinflammatory plant flavonoids and cellular action mechanisms. J. Pharmacol. Sci. 96, 229–245.
Kim, S.J., Ryu, B., Kim, H.Y., Yang, Y.I., Ham, J., Choi, J.H., et al., 2013. Sesquiterpenes from the rhizomes of Cyperus rotundus and their potential to inhibit LPS-induced nitric oxide production. Bull. Korean Chem. Soc. 34, 2207–2210.
Krishna, S., Renu, S., 2013. Isolation and identification of flavonoids from Cyperus rotundus Linn. in vivo and in vitro. J. Drug Deliv. Ther. 3, 109–113.
Lago, J.H.G., Toledo-Arruda, A.C., Mernak, M., Barrosa, K.H., Martins, M.A., Tibério, I.F.L.C., et al., 2014. Structure-activity association of flavonoids in lung diseases. Molecules 19, 3570–3595.
Lättig, J., Bohl, M., Fischer, P., Tischer, S., Tietbohl, C., Menschikowski, M., et al., 2007. Mechanism of inhibition of human secretory phospholipase A2 by flavonoids: rationale for lead design. J. Comput. Aided Mol. Des. 21, 473–483.
Lawal, O.A., Oyedeji, A.O., 2009. Chemical composition of the essential oils of Cyperus rotundus L. from South Africa. Molecules 14, 2909–2917.
Leitäo, S.G., Monache, F.D., 1998. 2″-O-Caffeoylorientin from Vitex polygama. Phytochemistry 49, 2167–2169.
Mabry, T.J., Markham, K.R., Thomas, M.B., 1970. The Systematic Identification of Flavonoids. Springer Verlag, New York, Heidelberg, Berlin.
Malikov, V.M., Yuldashev, M.P., 2002. Phenolic compounds of plants of the Scutellaria L. genus. Distribution, structure, and properties. Chem. Nat. Compd. 38, 358–406.
Mashima, R., Okuyama, T., 2015. The role of lipoxygenases in pathophysiology; new insights and future perspectives. Redox Biol. 6, 297–310.
Mayur, P., Pawan, P., Ashwin, S., Pravesh, S., 2011. Evaluation of anticonvulsant activity of roots and rhizomes of Cyperus rotundus Linn. in mice. Int. Res. J. Pharm. 2, 37–41.
Meena, A.K., Yadav, A.K., Niranjan, U.S., Singh, B., Nagariya, A.K., Verma, M., 2010. Review on Cyperus rotundus-a potential herb. Int. J. Pharm. Clin. Res. 2, 20–22.
Mohamed, G.A., 2016. Tagenols A and B: new lipoxygenase inhibitor flavonols from Tagetes minuta. Phytochem. Lett. 16, 141–145.
Mohamed, G.A., 2015. Iridoids and other constituents from Cyperus rotundus L. rhizomes. Bull. Facu. Pharm. Cairo Univ. 53, 5–9.
Mohamed, G.A., Ibrahim, S.R.M., Al-Musayeib, N.M., Ross, S.A., 2014. New anti-inflammatory flavonoids from Cadaba glandulosa Forssk. Arch. Pharm. Res. 37, 459–466.
Mohamed, G.A., Ibrahim, S.R.M., Elkhayat, E.S., Ross, S.A., Sayed, H.M., El-Moghazy, S.A.M., El-Shanawany, M.A., 2015. Blepharisides A and B, new flavonol glycosides from Blepharis ciliaris growing in Saudi Arabia. Phytochem. Lett. 11, 177–182.
Mohamed, G.A., Ibrahim, S.R.M., Ross, S.A., 2013. New ceramides and isoflavone from the Egyptian Iris germanica L. rhizomes. Phytochem. Lett. 6, 340–344.
Murakami, A., Ohigashi, H., 2007. Targeting NOX, iNOS and COX-2 in inflammatory cells: chemoprevention using food phytochemicals. Int. J. Cancer 121, 2357–2363.
Nagulendran, K., Velavan, S., Mahesh, R., Begum, V.H., 2007. In vitro antioxidant activity and total polyphenolic content of Cyperus rotundus rhizomes. E-J. Chem. 4, 440–449.
Nguyen, T.Y., To, D.C., Tran, M.H., Lee, J.S., Lee, J.H., Kim, J.A., Woo, M.H., Min, B.S., 2015. Anti-inflammatory flavonoids isolated from Passiflora foetida. Nat. Prod. Commun. 10, 929–931.
Nie, D., Honn, K.V., 2002. Cyclooxygenase, lipoxygenase and tumour angiogenis. Cell Mol. Life Sci. 59, 707–799.
Noreen, Y., Ringbom, T., Perera, P., Danielson, H., Bohlin, L., 1998. Development of a radiochemical cyclooxygenase-1 and -2 in vitro assay for identification of natural products as inhibitors of prostaglandin biosynthesis. J. Nat. Prod. 61, 2–7.
Radmark, O., Werz, O., Steinhilber, D., Samuelsson, B., 2007. 5-Lipoxygenase: regulation of expression and enzyme activity. Trends Biochem. Sci. 32, 332–341.
Rathee, P., Chaudhary, H., Rathee, S., Ratheem, D., Kumar, V., Kohli, K., 2009. Mechanism of action of flavonoids as anti-inflammatory agents: a review. Inflamm. Allergy Drug Targets 8, 229–235.
Sacca, R., Cuff, C.A., Ruddle, N.H., 1997. Mediators of inflammation. Curr. Opin. Immunol. 9, 851–857.
Sayed, H.M., Mohamed, M.H., Farag, S.F., Mohamed, G.A., 2001. Phytochemical and biological investigations of Cyperus rotundus L. Bull. Facu. Pharm. Cairo Univ. 39, 195–203.
Sayed, H.M., Mohamed, M.H., Farag, S.F., Mohamed, G.A., Omobuwajo, O.R.M., Proksch, P., 2008. Fructose-amino acid conjugate and other constituents from Cyperus rotundus L. Nat. Prod. Res. 22, 1487–1497.
Sayed, H.M., Mohamed, M.H., Farag, S.F., Mohamed, G.A., Proksch, P., 2007. A new steroid glycoside and furochromones from Cyperus rotundus L. Nat. Prod. Res. 21, 343–350.
Singh, N., Pandey, B.R., Verma, P., Bhalla, M., Gilca, M., 2012. Phytopharmacotherapeutics of Cyperus rotundus Linn. (Motha): an overview. Indian J. Nat. Prod. Res. 3, 467–476.
Singh, P.N., Singh, S.B., 1980. A new saponin from mature tubers of Cyperus rotundus. Phytochemistry 19, 2056.
Steinhilber, D., 1999. 5-Lipoxygenase: atarget for anti-inflammatory drugs revisited. Curr. Med. Chem. 6, 71–85.
Xu, Y., Zhang, H., Yu, C., Lu, Y., Chang, Y., Zou, Z., 2008. Norcyperone, a novel skeleton norsesquiterpene from Cyperus rotundus L. Molecules 13, 2474–2481.
Yawer, M.A., Ahmed, E., Malik, A., Ashraf, M., Rasool, M.A., Afza, N., 2007. New lipoxygenase-inhibiting constituents from Calligonum polygonoids. Chem. Bio-divers. 4, 1578–1585.
Yuldashev, M.P., Karimov, A., 2001. Flavonoids of Scutellaria ocellata and S. nepetoides. Chem. Nat. Compd. 37, 431–433.
Zhou, Z., Zhang, H., 2013. Phenolic and iridoid glycosides from the rhizomes of Cyperus rotundus L. Med. Chem. Res. 22, 4830–4835.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors’ contributions
SRMI: manuscript preparation and submission, data acquisition, analysis, and interpretation of NMR data. GAM: plant collection, concept and design of the study, and supervision of the study. RAA and KZA: shared in writing and revising the manuscript. AAE and MFZ: interpretation of biological data and sharing in writing the manuscript. All authors read and approved the final manuscript.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ibrahim, S.R.M., Mohamed, G.A., Alshali, K.Z. et al. Lipoxygenase inhibitors flavonoids from Cyperus rotundus aerial parts. Rev. Bras. Farmacogn. 28, 320–324 (2018). https://doi.org/10.1016/j.bjp.2018.04.002
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2018.04.002