Abstract
This mini-review provides a narrative analysis of the emerging therapeutic potential of Patisiran in managing Transthyretin Cardiac Amyloidosis (ATTR-CA). ATTR-CA, characterized by the deposition of misfolded transthyretin proteins in cardiac tissue, leads to progressive heart failure, significantly impacting affected individuals' quality of life and prognosis. Current treatment options for ATTR-CA are limited, necessitating the exploration of novel interventions like Patisiran. Patisiran, an RNA interference therapeutic, targets hepatic transthyretin protein production, thereby reducing amyloid deposits. While proven effective and safe in treating ATTR amyloidosis with polyneuropathy, its impact on cardiac manifestations is less studied. Positive outcomes include improved health status, enhanced quality of life, and preservation of functional capacity. Studies demonstrate sustained benefits, highlighting the potential for long-term positive effects. Reductions in cardiac amyloid burden and regression and prevention of deterioration in various cardiac parameters suggest a potential disease-modifying effect. Despite promising results, infusion-related reactions and adverse events necessitate careful consideration of long-term tolerability. Overall, Patisiran emerges as a promising intervention, offering hope for improved patient outcomes in the complex landscape of ATTR-CA management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Transthyretin cardiac amyloidosis (ATTR-CA) is a debilitating and often underdiagnosed condition characterised by the deposition of misfolded transthyretin proteins in the cardiac tissue, leading to progressive heart failure [1]. This rare yet severe form of amyloidosis significantly impacts the quality of life and prognosis of affected individuals [2]. ATTR-CA can be either hereditary (familial) or non-hereditary (wild-type), depending on the presence or absence of a genetic mutation in the TTR gene [3]. Despite recent advances in understanding the disease, effective management strategies remain limited, necessitating a thorough investigation into emerging therapeutic interventions [4]. The current treatment options for ATTR-CA are limited and mainly focus on supportive care, and symptom management [4, 5].
Patisiran is a novel RNA interference therapeutic that targets the hepatic production of the transthyretin protein, thereby sharply reducing the supply of transthyretin to the amyloid deposits, in the hope of limiting or even stopping ongoing deposition of new amyloid in the body [6]. Patisiran is effective and safe in treating ATTR amyloidosis with polyneuropathy, a subtype of ATTR that affects the peripheral nervous system [7]. However, the effects of patisiran on the cardiac manifestations of ATTR need to be better studied and understood [8]. Several clinical trials have evaluated the efficacy and tolerability of patisiran in patients with ATTR-CA, with promising results [6, 8]. However, these trials have used different methods, endpoints, and populations, making comparing and synthesising their findings difficult. Therefore, there is a need for a review of the existing literature on patisiran for ATTR-CA. This paper aims to conduct a narrative analysis of the available evidence on patisiran’s efficacy in managing ATTR-CA.
Another well studied treatment option is Tafamidis, a transthyretin stabilizer that prevents the misfolding of the transthyretin protein. This was the first FDA-approved treatment ATTR-CM, significantly reducing cardiovascular mortality and hospitalizations [9, 10].
2 Methodology
A literature search was conducted to identify relevant studies and publications on Patisiran's efficacy in managing ATTR-CA. Table 1. Databases such as PubMed, MEDLINE, Embase, and Cochrane Library were searched. The search strategy included keywords such as “patisiran”, “transthyretin cardiac amyloidosis”, “cardiac disease”, “patisiran efficacy”, “clinical trials”, “cardiac disease treatment” and related terms. Inclusion criteria include studies reporting on the use of Patisiran in managing ATTR-CA, including clinical trials and observational studies. Non-English language studies were excluded. No time limit was placed in the search. Two reviewers conducted The selection process independently, with disagreements resolved through discussion or consultation with a third reviewer. A qualitative narrative analysis approach was employed to explore and synthesize the narrative elements present in the selected studies. This involved identifying common themes and patterns.
3 Current evidence on the efficacy of Patisiran
The available studies vary in design, ranging from randomized controlled trials to prospective studies. Table 2. In most studies, the dose of Patisiran was administered intravenously every three weeks. The mean dose across the studies is 0.3 mg/kg. The mean duration of the studies spans from 12 to 24 months, providing insights into both short-term and relatively longer-term effects. The participant cohorts ranged from 16 to 360 individuals.
3.1 Positive outcomes
The APOLLO-B trial conducted by Zubair et al. [11] showed a significant improvement in health status and quality of life (QoL) among patients receiving intravenous Patisiran. The positive impact extended across all domains of the Kansas City Cardiomyopathy Questionnaire (KCCQ), with patients reporting enhanced enjoyment of life and increased engagement in hobbies and recreational activities. This shows Patisiran's potential to alleviate cardiac symptoms and enhance overall well-being. Further affirming the positive outcomes, Mathew et al. [12] highlighted that Patisiran played a crucial role in preserving functional capacity, health status, and QoL in patients diagnosed with ATTR cardiac amyloidosis. Even after 18 months of treatment, the sustained positive effects emphasize the potential for long-term benefits, reinforcing the significance of early initiation of Patisiran therapy.
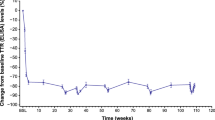
The phase 3 trial led by Mathew et al. [12] echoed these findings, demonstrating Patisiran's efficacy in preserving functional capacity, particularly evidenced by a lower decline in the 6-min walk distance at Month 12 compared to the placebo group. Despite some infusion-related reactions, the study underscores the tangible benefits of Patisiran in maintaining patients' ability to engage in daily activities. Rebecca et al. [13] provided insights from a study involving patients treated with Patisiran for 24 months, showcasing sustained benefits across clinical endpoints such as 6-min walk time (6MWT), KCCQ-OS, and N-terminal prohormone of brain natriuretic peptide (NT-proBNP). During the double-blind phase (12 months), the Patisiran group experienced a mean reduction in 6-min walk test (6MWT) distance of 8.1 m (± 5.7) from baseline compared to 25.4 m (± 5.6) in the placebo group, indicating an improvement in walking distance in the Patisiran group. This benefit was sustained even after both groups received Patisiran in the open-label extension (OLE) (another 12 months). In this OLE phase, the previously Patisiran-treated group maintained a mean 6MWT reduction of 7.8 m (± 7.0), while the group originally on placebo showed a continued decline, with a mean reduction of 26.0 m (± 6.1) from baseline. Even those transitioning from placebo to Patisiran exhibited relative stabilisation or slowing disease progression, reinforcing the drug's positive impact.
A prospective study by Lisi et al. [14] involving 31 subjects with ATTRh demonstrated Patisiran's effectiveness in stabilizing cardiac disease. In an analysis of 7 patients with cardiac amyloidosis and polyneuropathy (out of 31 patients enrolled in the study), Patirisan significantly improved global longitudinal strain (GLS) and left atrial stiffness index (LASI), indicating positive cardiac outcomes.
Marianna et al. [15] conducted a study involving 32 patients diagnosed with hereditary ATTR-CM, (16 patients treated with Patisiran and 16 untreated controls who were retrospectively matched). The study utilized cardiac magnetic resonance imaging to investigate cardiac amyloid regression. The study observed a reduction in extracellular volume (ECV) in 38% of patients treated with Patisiran compared to none in the untreated group. These findings suggest Patisiran's potential to promote cardiac amyloid regression.
Masatoshi et al. [16], in a randomised, double-blind, placebo-controlled phase 3 trial, demonstrated Patisiran's ability to prevent the deterioration of left ventricular global longitudinal strain (LV GLS) for up to 18 months. The study suggested that basal longitudinal strain may be a sensitive marker for treatment associations with cardiac manifestations in ATTRv amyloidosis. Scott et al. [17] further validated these findings in an 18-month randomised, double-blind, placebo-controlled phase 3 trial. Patisiran treatment yielded promising results. While the mean reduction in left ventricular wall thickness (− 0.9 ± 0.4 mm) was statistically significant compared to placebo, it did not reach the established clinically meaningful threshold of > 2 mm. However, a significantly greater proportion of patients in the Patisiran group (29.1%) experienced a reduction in wall thickness compared to placebo (4.0%). Patisiran also demonstrated significant reductions in global longitudinal strain (21.3% vs. 8.0% in placebo) and N-terminal prohormone of brain natriuretic peptide (NT-proBNP) levels (55% reduction). 31.6% of evaluable patients in the Patisiran group achieved a substantial decrease (≥ 30% and ≥ 300 pg/mL at month 18) in NT-proBNP, a marker of cardiac stress, compared to none in the placebo group. These findings suggest potential for Patisiran to halt or reverse disease progression, although larger mean reductions in wall thickness and global longitudinal strain are necessary to meet clinically meaningful thresholds.
3.2 Negative outcomes
Despite the promising positive outcomes observed in evaluating Patisiran's efficacy in managing transthyretin cardiac amyloidosis, considering negative outcomes provides a comprehensive understanding. Most adverse events were mild to moderate based on severity across all the studies. In the study led by Mathew et al. [18], patients receiving Patisiran experienced infusion-related reactions, arthralgia, and muscle spasms compared to the placebo group. This indicates that while the drug demonstrates positive effects in preserving functional capacity, notable adverse events are associated with its administration. Rebecca et al. [13] highlighted infusion-related adverse events in their study, albeit generally mild or moderate in severity. These events were consistent with the known safety profile of Patisiran, and no new safety concerns, including cardiac events, were identified during the study. The most common related adverse event observed was infusion-related reactions.
Acknowledging these negative outcomes and adverse events reported in the studies is crucial. While Patisiran shows promise in improving patients' well-being with transthyretin cardiac amyloidosis, physicians must weigh these potential drawbacks against the observed benefits. Careful monitoring and consideration of individual patient profiles are important in ensuring a balanced approach to the clinical management of this complex condition.
4 Discussion
The positive outcomes observed across multiple studies, including improvements in health status, quality of life, and preservation of functional capacity, signify a potential paradigm shift in managing transthyretin cardiac amyloidosis. Patisiran emerges as a promising therapeutic intervention that addresses cardiac manifestations and positively influences overall patient well-being. The sustained positive effects reported, especially in studies with extended durations, suggest the potential for long-term benefits with Patisiran therapy. This is particularly significant in chronic conditions like transthyretin cardiac amyloidosis, where maintaining or improving patients' quality of life over an extended period is a primary goal. It is also important to consider the impact of the cost of Patirisan on these patients as Patirisan has an annual cost of between $451,430 and $677,145 per patient. The cost is dependent on weight and the monthly copay in the insured may vary [19].
Findings indicating a reduction in cardiac amyloid burden, regression of cardiac amyloid, and prevention of deterioration in various cardiac parameters suggest a potential disease-modifying effect of Patisiran. This could alter the natural progression of the disease, offering hope for improved prognosis and outcomes. The positive impact on functional capacity, as seen in measures like the 6-min walk distance, along with improvements in health status and quality of life, underscores the holistic benefits of Patisiran beyond traditional clinical endpoints. These improvements can translate into meaningful enhancements in patients' daily lives and activities.
The studies consistently report infusion-related reactions, arthralgia, and muscle spasms as adverse events associated with Patisiran administration. While generally mild or moderate, these events raise concerns about the tolerability of long-term therapy. To minimize infusion-related reactions, patisiran is administered with premedication including dexamethasone, paracetamol, and H1 and H2 blockers. It is important to note that dexamethasone should be used with caution in patients with heart failure, a condition commonly observed in the study population. The diverse participant cohorts across the studies reflect the complexity of transthyretin cardiac amyloidosis. However, this diversity also introduces variability in patient characteristics, disease stages, and responses to treatment. Generalising findings to broader populations may require careful consideration of this heterogeneity. While longer-term studies provide insights into sustained benefits, some investigations have a shorter duration. Longer follow-up periods would provide a more comprehensive understanding of the durability of Patisiran's effects and its impact on disease progression over time.
Supportive treatment is essential alongside Patisiran for managing ATTR-CM. This includes cautious use of diuretics to avoid excessive volume reduction, as the myocardium in these patients is highly preload-dependent [20]. Additionally, SGLT-2 inhibitors have shown favorable effects on heart failure symptoms, hospitalizations, and overall cardiovascular mortality, making them a valuable add-on therapy to definitive management strategies [21].
5 Future directions
Continuous and long-term safety monitoring is pivotal to Patisiran's future trajectory. The observed adverse events highlight the necessity of extended surveillance to identify potential late-onset effects. This monitoring is essential for assessing the safety profile over an extended duration and informing clinicians about the sustained tolerability of the treatment. The long-term safety data will play a pivotal role in guiding clinical decision-making, ensuring that healthcare practitioners can make informed choices about the risks and benefits of Patisiran therapy. Moreover, exploring the underlying mechanisms of Patisiran's effects on transthyretin cardiac amyloidosis is vital. A better understanding of the molecular and cellular pathways impacted by Patisiran could uncover novel therapeutic targets. This exploration is essential for refining the current treatment strategies and potentially identifying additional points of intervention. By elucidating the mechanisms driving Patisiran's efficacy, studies can discover new avenues for drug development and combination therapies, enhancing ATTR-CA management.
The progression of research should aim to identify factors that influence individual responses to Patisiran. The move toward personalized treatment approaches is a promising avenue for tailoring interventions based on patient-specific characteristics. By recognizing and understanding predictive factors, clinicians can optimize treatment plans, ensuring patients receive the most effective and well-tolerated therapies. This approach aligns with the broader trend in medicine towards precision medicine, where treatments are tailored to each patient's unique genetic, molecular, and clinical profiles.
Furthermore, future clinical trials should adopt broader inclusion criteria to enhance the generalizability of findings and ensure that Patisiran's efficacy is understood across diverse patient populations. This involves considering age, comorbidities, and disease stages when designing studies. A more inclusive approach to clinical trials will generate a richer dataset, allowing for a more comprehensive understanding of Patisiran's performance in real-world scenarios. This is particularly important for ensuring that the benefits observed in specific patient groups apply to a broader spectrum of individuals.
Vutrisiran is emerging as a promising treatment option for transthyretin amyloidosis with cardiomyopathy (ATTR-CM). In the HELIOS-A trial, which primarily focused on ATTRv-PN (Hereditary transthyretin amyloidosis with polyneuropathy), a cardiac outcomes analysis demonstrated a reduction in NT-proBNP levels and improved echocardiographic parameters at 18 months. The HELIOS-B trial is set to further investigate vutrisiran's effects on ATTR-CM over a 36-month period in a randomized, double-blind, placebo-controlled study. Vutrisiran is administered as a 25 mg subcutaneous injection every 3 months, compared to Patisiran, which is given at 0.3 mg/kg every 3 weeks. Unlike Patisiran, which requires premedication to limit infusion-related reactions, vutrisiran does not require premedication. This could be a significant advantage, particularly for patients with heart failure, where dexamethasone has to be used with caution [22].
6 Limitations and strengths of study
Relying exclusively on English-language studies introduces language bias, as valuable studies published in other languages may be overlooked. Moreover, The variability in study designs, participant cohorts, and treatment durations across the reviewed studies introduces heterogeneity. This impacts the generalizability of findings. However, this review goes beyond presenting findings and engages in critical discussions regarding the implications of the results. Additionally, including future directions emphasizes the forward-looking nature of the review, contributing to its relevance for clinicians.
7 Conclusion
This mini-review analyses the current evidence regarding Patisiran's efficacy in managing ATTR-CA. ATTR-CA, a debilitating condition with significant impacts on patients' quality of life and prognosis, has been a challenging area for therapeutic interventions. Patisiran, a novel RNA interference therapeutic targeting hepatic transthyretin production, shows promising outcomes in the reviewed studies. The positive outcomes observed across various trials, including improvements in health status, quality of life, and preservation of functional capacity, underscore Patisiran's potential as a transformative intervention. These positive effects were sustained over different durations, indicating the prospect of long-term benefits. Noteworthy improvements in clinical endpoints such as the 6-min walk distance, KCCQ scores, and N-terminal prohormone of brain natriuretic peptide (NT-proBNP) levels suggest a multifaceted impact on the disease. Despite these promising results, it is crucial to acknowledge the limitations and negative outcomes associated with Patisiran. Infusion-related reactions, arthralgia, and muscle spasms were reported in some studies, highlighting the importance of careful monitoring and individualised treatment approaches. The diversity in participant cohorts and the variable duration of studies necessitate cautious generalisation of findings.
Data availability
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.
Abbreviations
- ATTR-CA:
-
Transthyretin cardiac amyloidosis
- KCCQ:
-
Kansas City Cardiomyopathy Questionnaire
- NT-proBNP:
-
N-terminal prohormone of brain natriuretic peptide
- RNAi:
-
RNA interference
- CRISPR/Cas9:
-
Clusters of Regularly Interspaced Short Palindromic Repeats/CRISPR-associated protein 9
- LNP:
-
Lipid nanoparticle
- TTR:
-
Transthyretin
- ATTRv:
-
Hereditary transthyretin-mediated amyloidosis
- QoL:
-
Quality of life
References
Yamamoto H, Yokochi T. Transthyretin cardiac amyloidosis: an update on diagnosis and treatment. ESC Heart Fail. 2019;6(6):1128–39. https://doi.org/10.1002/ehf2.12518.
Smiley DA, Rodriguez CM, Maurer MS. Transthyretin cardiac amyloidosis: an evolution in diagnosis and management of an “old” disease. Cardiol Clin. 2022;40(4):541–58. https://doi.org/10.1016/j.ccl.2022.06.008.
Fenoglio R, Baldovino S, Barreca A, Bottasso E, Sciascia S, Sbaiz L, Papotti M, Roccatello D. Renal involvement in transthyretin amyloidosis: the double presentation of transthyretin amyloidosis deposition disease. Nephron. 2022;146(5):481–8. https://doi.org/10.1159/000522370.
Spencer-Bonilla G, Alexander KM, Witteles RM. Advances in the diagnosis and management of transthyretin amyloid cardiomyopathy. Curr Treat Options Cardio Med. 2020;22:45. https://doi.org/10.1007/s11936-020-00844-8.
Plumadore E, Lombardo L, Cabral KP. Pharmacotherapy review: emerging treatment modalities in transthyretin cardiac amyloidosis. Am J Health-Syst Pharm. 2022;79(2):52–62. https://doi.org/10.1093/ajhp/zxab356.
Kristen AV, Ajroud-Driss S, Conceição I, Gorevic P, Kyriakides T, Obici L. Patisiran, an RNAi therapeutic for the treatment of hereditary transthyretin-mediated amyloidosis. Neurodegener Dis Manag. 2019;9(1):5–23. https://doi.org/10.2217/nmt-2018-0033.
Titze-de-Almeida SS, Brandão PRP, Faber I, Titze-de-Almeida R. Leading RNA interference therapeutics part 1: silencing hereditary transthyretin amyloidosis, with a focus on Patisiran. Mol Diagn Ther. 2020;24(1):49–59. https://doi.org/10.1007/s40291-019-00434-w.
Yang J. Patisiran for the treatment of hereditary transthyretin-mediated amyloidosis. Expert Rev Clin Pharmacol. 2019;12(2):95–9. https://doi.org/10.1080/17512433.2019.1567326.
Maurer MS, Schwartz JH, Gundapaneni B, Elliott PM, Merlini G, Waddington-Cruz M, Kristen AV, Grogan M, Witteles R, Damy T, Drachman BM. Tafamidis treatment for patients with transthyretin amyloid cardiomyopathy. N Engl J Med. 2018;379(11):1007–16.
Tschöpe C, Elsanhoury A. Treatment of transthyretin amyloid cardiomyopathy: the current options, the future, and the challenges. J Clin Med. 2022;11(8):2148.
Shah Z, Obici L, Kale P, Mitter SS, Eldhagen P, Hashimoto T, et al. Impact of Patisiran on health status and quality of life in patients with transthyretin cardiac amyloidosis. J Card Fail. 2024;30(1):134. https://doi.org/10.1016/j.cardfail.2023.10.043.
Maurer MS, Berk JL, Hanna MA, Gillmore JD, et al. Primary results from APOLLO-B open-label extension study of Patisiran in patients with transthyretin cardiac amyloidosis. J Card Fail. 2024;30(1):130–1. https://doi.org/10.1016/j.cardfail.2023.10.035.
Hung RR, Berk JL, Drachman B, Fontana M et al. APOLLO-B, a study of Patisiran in patients with transthyretin cardiac amyloidosis: primary long-term results from the open-label extension period. AHA. 2023. https://capella.alnylam.com/wp-content/uploads/2023/11/APOLLO-B-A-STUDY-OF-PATISIRAN-IN-PATIENTS-WITH-TRANSTHYRETIN-CARDIAC-AMYLOIDOSIS-PRIMARY-LONG-TERM-RESULTS-FROM-THE-OPEN-LABEL-EXTENSION-PERIOD.pdf
Di Lisi D, Brighina F, Di Stefano V, Portelli MC, Ortello A, Comparato F, Cannizzo N. Effect of patisiran on myocardial deformation parameters in patients with hereditary transthyretin amyloidosis. Eur Heart J Cardiovasc Imaging. 2023;24(Supplement_1):jead119.396. https://doi.org/10.1093/ehjci/jead119.396.
Fontana M, Martinez-Naharro A, Chacko L, Rowczenio D, et al. Reduction in CMR derived extracellular volume with Patisiran indicates cardiac amyloid regression. J Am Coll Cardiol Img. 2021;14(1):189–99.
Minamisawa M, Claggett B, Adams D, Kristen AV, Merlini G, et al. Association of Patisiran, an RNA interference therapeutic, with regional left ventricular myocardial strain in hereditary transthyretin amyloidosis: the APOLLO study. JAMA Cardiol. 2019;4(5):466–72. https://doi.org/10.1001/jamacardio.2019.0849.
Solomon SD, Adams D, Kristen A, Grogan M, González-Duarte A. Effects of Patisiran, an RNA interference therapeutic, on cardiac parameters in patients with hereditary transthyretin-mediated amyloidosis. Circulation. 2019;139(4):431–43.
Maurer MS, Kale P, Fontana M, Berk JL, Grogan M, Gustafsson F, et al. Patisiran treatment in patients with transthyretin cardiac amyloidosis. N Engl J Med. 2023;389(17):1553–65. https://doi.org/10.1056/NEJMoa2300757.
Williams TM, Harvey R, Fischer MA, Neelankavil J. It's time for effective and affordable therapies for cardiac amyloidosis: lessons from Patisiran. J Cardiothorac Vasc Anesth. 2024;38(7):1457–59. https://doi.org/10.1053/j.jvca.2024.02.035.
Puig-Carrion GD, Reyentovich A, Katz SD. Diagnosis and treatment of heart failure in hereditary transthyretin amyloidosis. Clin Auton Res. 2019;29:45–53.
Binder C, Duca F. Diagnosis and supportive therapeutic management of cardiac light chain amyloidosis—a cardiologist’s perspective. MEMO. 2021;14(1):89–97.
Planté-Bordeneuve V, Perrain V. Vutrisiran: a new drug in the treatment landscape of hereditary transthyretin amyloid polyneuropathy. Expert Opin Drug Discov. 2024;19(4):393–402.
Acknowledgements
None.
Funding
No funding was received for this study.
Author information
Authors and Affiliations
Contributions
GO conceptualised the study; GO, EK, IJO, CE, OO, DO, SGK, AAE, DA, AMO, KBA, OYA, EOU, IAY, OO, and NA were involved in the literature review; EK & NA extracted the data from the reviewed studies; GO, EK, IJO, CE, OO, DO, SGK, AAE, DA, AMO, KBA, OYA, EOU, IAY, OO, AV, YAA and NA wrote the final and first drafts. GO, EK, IJO, CE, OO, DO, SGK, AAE, DA, AMO, KBA, OYA, EOU, IAY, OO, AV, YAA and NA read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Olatunji, G., Kokori, E., Ogieuhi, I.J. et al. A mini review of Patisiran's efficacy in the management of transthyretin cardiac amyloidosis. Discov Med 1, 47 (2024). https://doi.org/10.1007/s44337-024-00066-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44337-024-00066-y