Abstract
Purpose
Anastomotic leakage (AL) is one of the most pernicious complications after esophagectomy for patients with esophageal or esophagogastric junction cancer (EC or EJC). The application of fibrin sealant (FS) may be advantageous for reducing the incidence of AL. This study aims to evaluate the safety and effectiveness of FS in preventing AL in patients undergoing McKeown esophagectomy.
Methods
In this multicenter, prospective, randomized controlled trial, we planned to recruit 360 patients aged 18–75 years with resectable EC or EJC and the interim analysis was performed when the number of participants reaches 180. Patients assigned to the FS group received McKeown esophagectomy with 2.5ml FS applied to the cervical anastomosis, while patients in the control group received surgery alone. The primary endpoint was the incidence of cervical AL within the first 3 months postoperatively.
Result
From February 2019 to November 2021, 180 patients were recruited, with 89 in the FS group and 91 in the control group. There was no statistically difference between the incidence of AL between the two groups [6.7% (6/89) in the FS vs. 14.3% (13/91) in the control group, P = 0.16]. Complications was comparable (P = 0.76) between the FS group (42 of 89, 47.2%) and the control group (45 of 91, 49.5%). No adverse events related to FS or deaths occurred postoperatively.
Conclusion
The application of FS intraoperatively is feasible and does not increase the risk of complications, and its effectiveness for the prevention of AL needs to be revalidated after the completion of patient enrollment.
Trial registration
This trial was registered at ClinicalTrials.gov (NCT03847857) on February 19th, 2019.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
As the seventh most common malignancy worldwide, esophageal cancer (EC) yields approximately 477,900 new cases and 375,000 deaths as estimated in 2015 [1, 2]. Surgery remains to be the most important and effective treatment for EC. Among all surgical methods, right-sided trans-thoracic approach, especially the McKeown esophagectomy, had shown superiority in radical lymphadenectomy as well as sufficient resection margin [3]. Nevertheless, compared with intrathoracic anastomosis [4], cervical anastomosis in the McKeown/three-hole esophagectomy is associated with a higher risk of anastomotic leakage (AL) that would prolonged hospital stay and increase considerable morbidity and mortality [4, 5]. According to past studies, the incidence of AL ranges from 10.6 to 26.1% [6,7,8]. Therefore, additional precautions for AL are of great importance during perioperative period, among which fibrin sealant (FS) had already shown potential convenience and efficacy.
Anecdotal reports have attempted to use FS to prevent gastrointestinal AL after surgery which successfully enhances postoperative recovery [9,10,11,12]. However, studies about the applications of FS to prevent AL after esophagectomy was still of insufficient improvement [13, 14]. Our previous phase II trial reported a significantly low cervical anastomotic leakage rate of 3.5% with intraoperative application of FS during esophagectomy [15]. Based on this, the present study was designed to further assess the role of porcine FS (Bioseal) in the prevention of cervical AL for patients with EC undergoing Mckeown esophagectomy. The study has met the criteria for interim analysis as prespecified, and here the interim results are displayed.
2 Materials and methods
2.1 Study design
The PLACE030 trial was a multicenter, prospective, randomized controlled trial, planning to recruit 360 patients in 4 years. Six centers participated in this study (Sun Yat-sen University Cancer Center, Zhongshan Hospital of Sun Yat-Sen University, Cancer Hospital of Shantou University Medical College, Shanghai Chest Hospital, People’s Hospital of Jieyang, and Sichuan Cancer Hospital).The interim analysis would be performed when the number of participants reaches 180 (Fig. 1). Enrolled patients were randomized 1:1 into two groups. Two point five ml of FS (BioSeal; Guangzhou Bioseal Biotech Co., Ltd., China) was applied to cover the cervical anastomotic site in the treatment group. The effectiveness of the application of Bioseal in reducing the chance of AL was assessed. All included patients were supposed to provide written informed consent. This trial was approved by the institutional ethical committee (B2018-011-01) and was registered at ClinicalTrials.gov (NCT03847857).
2.2 Participants and randomization
Patients aged 18 to 75 who were diagnosed with histologically confirmed squamous cell carcinoma or adenocarcinoma of the thoracic esophagus or esophagogastric junction staged as T1-4aN0-3M0 (according to the 8th AJCC edition), would be included. Other inclusion criteria included: normal liver and kidney functions, WHO performance status (PS) of 0–1, and expected survival for more than 6-month. The exclusion criteria are as follows: (1) surgically contraindicated because of cardiac, respiratory, hepatic, renal diseases, or other uncontrollable status; (2) history of gastric surgery that precludes the replacement of the esophagus with gastric graft; (3) prior definitive chemoradiotherapy; (4) history of or concomitant hemorrhagic diseases; (5) history of diabetes over 10 years with poorly controlled blood glucose level; (6) concomitant peripheral neuropathy; (7) known hypersensitivity to the porcine fibrin sealant products; and (8) pregnancy or lactation status. Enrolled patients would be randomized in a 1:1 ratio to receive Bioseal application or surgery alone, using computer-generated random numbers provided by the Sun Yat-sen University Cancer Center Clinical Trial Center.
2.3 Treatment and procedures
For diagnosis and accurate staging, the following workup is performed: chest and abdomen contrasted computed tomography, esophagogastroduodenoscopy (EGD) with endoscopic ultrasonography, electrocardiogram, lung spirometry, and cervical ultrasonography for every patient. And bronchoscopy with endobronchial ultrasonography, positron emission tomography-computed tomography, and emission computed tomography, if necessary.
Consultant surgeons in each center performed the operations, with a McKeown esophagectomy and two-field lymphadenectomy through thoracotomy or a minimally invasive approach (MIE). Total lymphadenectomy includes the resection of bilateral recurrent laryngeal nerve nodes, peri-esophageal nodes, subcarinal nodes, pulmonary ligament nodes, diaphragmatic nodes, pericardiac nodes, lesser curvature nodes, left gastric nodes, common hepatic nodes, splenic nodes, and celiac nodes. An end-to-side cervical esophagogastric anastomosis was constructed via a circular stapler, wrapped by interrupted sutures for reinforcement. The gastric remanent will be closed using a linear stapler and wrapped by seromuscular-layer interrupted sutures. For patients in the treatment arm, 2.5ml of Bioseal was irrigated over the cervical anastomotic site before would closure. Afterward, a nasogastric tube, cervical drainage tube, and jejunum feeding tube were placed.
All patients were transferred to the intensive care unit for the first postoperative day and readmitted to the general ward on the second day when appropriate. Oral feeding was commenced after excluding the presence of AL on the seventh postoperative day.
2.4 Outcome
The incidence of cervical AL within the first 3 months postoperatively is the primary endpoint. Cervical AL is defined as a full-thickness gastrointestinal defect involving any of the following including the esophagus, anastomosis or gastric conduit. AL was diagnosed by visualization of anastomotic dehiscence or fistula through EGD, barium leakage by esophageal barium X-ray, digestive juice leakage from the cervical wound or chest tube, and leakage of methylene blue dye from the cervical wound or chest tube after oral administration. If the latter two circumstances occur, EGD or esophageal barium study was mandatory to confirm cervical AL.
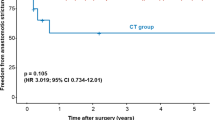
Secondary endpoints include incidence of postoperative anastomotic stricture (diagnosed based on dysphagia and the diameter of the anastomotic orifice ≤ 10 mm in esophageal barium study), postoperative morbidity and mortality, cervical wound infection rate, postoperative time to first oral feeding, 2-year overall survival, and disease-free survival.
2.5 Follow-up
The patients would be followed up in outpatient clinics once every three months in the first year, then once every six months thereafter until the study ends or death. Physical examination, routine blood tests, tumor markers, chest x-ray, esophageal barium x-ray, and cervical and abdominal ultrasonography will be performed at every visit. Contrast-enhanced cervico-thoraco-abdominal computed tomography and EGD will be performed once per year. Patients’ information will be sent to the central office for formal analysis.
2.6 Statistical analysis
All patients are included in the intention-to-treat population. Sample sizes are calculated according to a projected cervical anastomotic leakage rate of 15% and 5% for patients assigned to the treatment arm and the control arm, respectively. We assumed an overall two-sided significance level of 5% and a statistical power of 80%. The randomization ratio between the experimental and control arms is 1:1. With 4 years’ accrual, 2 years’ follow-up, and one planned interim analysis, the calculated sample size is 324 patients. The final number of patients is 360 with 180 per arm assuming a 10% drop-out. An interim analysis was prespecified after 180 patients (50%) have been enrolled. According to the O’Brien-Fleming algorithm, the two-sided significance level will be 0.005 in the interim analysis and 0.048 in the final analysis. The postoperative cervical AL rate, anastomotic stricture rate, morbidity, and mortality rate are calculated by percentage and compared by Pearson’s chi-squared test. All statistical analyses were performed using SPSS 22.0 (IBM Corporation, USA).
3 Results
3.1 Baseline characteristics
From February 2019 to November 2021, 180 patients from 6 Chinese centers were randomly allocated to the FS group (n = 89) or control group (n = 91). Baseline characteristics are summarized in Table 1. Most patients enrolled were male gender (80.6%), with tumors located in the thoracic region (96.7%) and had squamous cell carcinoma (96.7%). As for preoperative treatment, 42 (47.2%) of the 89 patients had received neoadjuvant therapy in the FS group, comparing with 47 (51.6%) of the 91 patients in the control group (P = 0.948) Table 2.
3.2 Surgery
All 180 patients underwent McKeown esophagectomy and two-field lymphadenectomy through MIE, including robot-assisted MIE [3 of 89 (3.4%) in the FS group vs. 9 of 91 (9.9%) in the control group, P = 0.08]. Two patients (1.1%) in the FS group received R1 resection due to microscopically positive resection margins. The mean number of dissected lymph nodes was 28(IQR: 21–35)in the FS group compared to 28 (21–34)in the control group (P = 0.96). The average operating time was similar in two groups [250 9IQR: 200–2800minutes in the FS group and 250 (IQR: 205–280) minutes in the control group, P = 0.98]. As for estimated blood loss volume, FS group [77 (IQR: 50–100) ml] tend to have a smaller average value than the control group [100 (IQR: 50–100) ml, P = 0.046].
3.3 Histopathological results
For those patients who received surgery alone, most patients were stage II-III [70.1% (65 of 92)], while patients with pathological stage I [53.4% (47 of 88)]accounted for the largest proportion in those who received neoadjuvant therapy (Table 2). The pathological complete response (pCR) was achieved in 14 (34.1%) of the 41 patients in the FS group, compared with a pCR rate of 31.9% (15 of 47) in the control group (P = 0.82).
3.4 Anastomotic leakage
No significant difference in the incidence of AL was observed between two groups, with 6.7% (6 of 89) in the FS group and 14.3% (13 of 91) in the control group, respectively (P = 0.16). Among all patients who experienced AL, one patient in each group was diagnosed with a gastric stump fistula and underwent a second operation to reconstruct the gastric stump. The other 17 patients were all successfully managed after conservative treatment, including fasting, cervical dressing, and endoscopic treatment.
3.5 Other morbidities
In total, 87 of the 180 (48.3%) patients suffered from postoperative complications and incidence of complications was comparable (P = 0.76) between the FS group (42 of 89, 47.2%) and the control group (45 of 91, 49.5%). Besides cervical AL, the most common complications were pneumonia [FS group: 9.0% (8 of 89) versus control group: 15.4% (14 of 91), P = 0.28] and anastomotic stricture [14.6% (13 of 89) versus 8.8% (8 of 91), P = 0.42]. One patient (1.1%) in the FS group experienced intrathoracic gastric conduit fistula and eventually recovered through endoscopic treatment 93 days after the surgery. One patient (1.1%) of the control group experienced surgical site infection. 12 (6.7%) cases of recurrent laryngeal nerve injury were observed, including 5 (5.6%) in the FS group and 7 (7.7%) in the control group (P = 0.80). There was no significant difference in other complications between two groups, including arrhythmia, heart failure, respiratory failure, pneumothorax, ARDS, atelectasis, and pyothorax (Table 3). No FS-related adverse events were observed, and no deaths occurred within 90 days after surgery either.
The average days for staying in the intensive care unit were significantly shorter in the FS group than in the control group [FS group: 2(IQR:1–2)days versus control group: 4 (IQR:1–4) days, P = 0.02], while the hospitalization stay was comparable[FS group: 16 (IQR:9–17) days versus control group: 17 (IQR:9–20) days, P = 0.64]. As for oral feeding initiating time postoperatively, no significant difference was observed between two groups. [FS group: 14 (IQR:8–13) days vs. control group: 15 (IQR:8–14) days, P = 0.43].
4 Discussion
To the best of our knowledge, this is the first prospective, phase III, randomized controlled study focusing on the efficacy and safety of FS in the prevention of AL after McKeown esophagectomy. In this interim analysis, 180 patients were enrolled, with 89 in the FS group and 91 in the control group. The incidence of AL in the FS group (6.7%) was numerically lower than that in the control group(14.3%), but the difference was not statistically significant (P = 0.16) and will be tested after the completion of patient enrollment. The incidence of other complications was comparable between two groups.
Prophylactic application of FS in esophageal surgery to prevent AL has been reported in previous studies. Upadhyaya et al. [13] carried out a prospective clinical trial, in which 45 infants were enrolled and underwent esophagectomy for congenital esophageal atresia with tracheoesophageal fistula. In the study arm, end-to-end esophagogastric anastomosis was performed with the application of FS. The AL rate was 9.1% in the study group, significantly lower than 43% in the control group. Additionally, a lower risk of anastomotic stenosis and perioperative mortality was also observed in the study arm. Saldana-Cortes et al. [14] conducted a study that focused on puerile esophagectomy for caustic injury in 38 patients. In their study, colon interposition was used as the reconstruction conduit. The result showed that the incidence of cervical AL was 28.5% in the patients who received FS, lower than that 50% in the control group. Both studies conclude that the application of FS can reduce the risk of AL and is feasible in the clinical practice. Another pilot study was reported by Haverkamp et al [16], evaluating the feasibility of fibrin-coated collagen patches (Tachosil) on cervical esophageal anastomosis in 11 adult patients who successfully received esophagogastrostomy with the application of Tachosil. As the result showed, 18.2% (2 of the 11) patients were diagnosed with AL. The study’s conclusion confirmed the technical feasibility of applying Tachosil to esophageal anastomosis. In 2021, Yan Huang et al. [17] carried out a single-center retrospective study that consisted of 227 patients with esophageal or esophagogastric junction cancer undergoing McKeown esophagectomy. As a consequence, the cervical AL rate was lower in the FS group (FS group: 4.7% [4 of 82] vs. control group: 19.9% [28 of 141], P = 0.01). Multivariate logistic regression was performed in the study, identifying that the intraoperative application of FS was an independent protective factor for the reduction of AL (odds ratio: 0.17, 95% confidence interval: 0.06–0.52). Additionally, the study also indicated that rates of most postoperative complications were comparable between two groups. However, as the fact that these studies are either retrospective studies or prospective studies with insufficient sample size, their significance of them needs to be further confirmed. Thus, more evidence is needed for the role of FS in preventing AL after esophagectomy. Unlike all previous studies, this is the first prospective, phase III, randomized controlled study, with a large predicted sample size of 360. The result of interim analysis demonstrated the safety of FS, with no statistical differences observed between two groups when the incidence of postoperative complications was compared.
As a swine-derived agent, Bioseal is manufactured from porcine-derived thrombin and fibrinogen, with the function to generate a stable cross-linked fibrin clot by stimulating the common pathway of the coagulation cascade. A gel-like material will be formed after the application of the fibrin clot on the surface of surgical wounds. Thereafter, the gel-like material will promote the healing of wounds by repairing tissue defects as well as forming a fibrin matrix where fibroblasts and capillary endothelial cell proliferation happen, which is favorable for the formation of granulation tissue. Furthermore, during the early phase of postoperative period, FS can promote the maturation process of granulation tissue, which is critical for anastomotic healing [18, 19]. Additionally, FS could provide a watertight esophageal anastomosis by additional sealing, which will result in lower occurrence of AL after the surgery owing to the avoidance of preceding microleakage [19].
This current study has its limitations. First, most enrolled tumors were squamous cell carcinoma located at the thoracic esophagus, hence the applicability in adenocarcinoma warrants further study. Second, the healing process of intrathoracic anastomosis may have a different physiologic pattern than the cervical one, thus the conclusion from this study may not be generalizable to intrathoracic anastomsis such as an Ivor-Lewis procedure.
In conclusion, our interim analysis suggested that the application of Bioseal intraoperatively is feasible and does not increase the risk of complications.
Availability of data and materials
The data of this study are available on resonable request. The data are not publicly available due to privacy or ethical restrictions.
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018:GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018. https://doi.org/10.3322/caac.21609.
Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J. Cancer statistics in China, 2015. CA Cancer J Clin. 2016. https://doi.org/10.3322/caac.21338.
Chinese Society of Esophageal Cancer, China Anti-cancer Association. Chinese expert consensus on thoracic lymph node dissection in the radical resection of esophageal cancer (2017 edition). Chin J Dig Surg. 2017;16(11):1087–90.
Jones CE, Watson TJ. Anastomotic leakage following esophagectomy. Thorac Surg Clin. 2015. https://doi.org/10.1016/j.thorsurg.2015.07.004.
Turrentine FE, Denlinger CE, Simpson VB, Garwood RA, Guerlain S, Agrawal A, Friel CM, LaPar DJ, Stukenborg GJ, Jones RS. Morbidity, mortality, cost, and survival estimates of gastrointestinal anastomotic leaks. J Am Coll Surg. 2015. https://doi.org/10.1016/j.jamcollsurg.2014.11.002.
Kassis ES, Kosinski AS, Ross P Jr, Koppes KE, Donahue JM, Daniel VC. Predictors of anastomotic leak after esophagectomy: an analysis of the society of thoracic surgeons general thoracic database. Ann Thorac Surg. 2013. https://doi.org/10.1016/j.athoracsur.2013.07.119.
Takeuchi H, Miyata H, Gotoh M, Kitagawa Y, Baba H, Kimura W, Tomita N, Nakagoe T, Shimada M, Sugihara K, Mori M. A risk model for esophagectomy using data of 5354 patients included in a japanese nationwide web-based database. Ann Surg. 2014. https://doi.org/10.1097/SLA.0000000000000644.
van Hagen P, Hulshof MC, van Lanschot JJ, Steyerberg EW, van Berge Henegouwen MI, Wijnhoven BP, Richel DJ, Nieuwenhuijzen GA, Hospers GA, Bonenkamp JJ, Cuesta MA, Blaisse RJ, Busch OR, ten Kate FJ, Creemers GJ, Punt CJ, Plukker JT, Verheul HM, Spillenaar Bilgen EJ, van Dekken H, van der Sangen MJ, Rozema T, Biermann K, Beukema JC, Piet AH, van Rij CM, Reinders JG, Tilanus HW, van der Gaast A. ; CROSS Group. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med 2012; DOI: https://doi.org/10.1056/NEJMoa1112088.
Hu H, Zhang L, Lu J, et al. The value of biomedical fibrin sealant applied in esophagectomy under thoracoscopy. China Prac Med. 2009;4(15):35–6.
Hirst NA, Tiernan JP, Millner PA, Jayne DG. Systematic review of methods to predict and detect anastomotic leakage in colorectal surgery. Colorectal disease: the official journal of the Association of Coloproctology of Great Britain and Ireland. 2014. https://doi.org/10.1111/codi.12411.
Nguyen NT, Nguyen CT, Stevens CM, Steward E, Paya M. The efficacy of fibrin sealant in prevention of anastomotic leak after laparoscopic gastric bypass. J Surg Res. 2004. https://doi.org/10.1016/j.jss.2004.05.005.
Martin I, Au K. Does fibrin glue sealant decrease the rate of anastomotic leak after a pancreaticoduodenectomy? Results of a prospective randomized trial. HPB: the official journal of the International Hepato Pancreato Biliary Association. 2013. https://doi.org/10.1111/hpb.12018.
Upadhyaya VD, Gopal SC, Gangopadhyaya AN, et al. Role of fibrin glue as a sealant to esophageal anastomosis in cases of congenital esophageal atresia with tracheoesophageal fistula. World J Surg. 2007. https://doi.org/10.1007/s00268-007-9244-7.
Saldana-Cortes JA, Larios-Arceo F, Prieto-Diaz-Chavez E, et al. Role of fibrin glue in the prevention of cervical leakage and strictures after esophageal reconstruction of caustic injury. World J Surg. 2009. https://doi.org/10.1007/s00268-009-9949-x.
Yao-Bin L, Jian-Hua F, Yan H, et al. Fibrin sealant for esophageal anastomosis: a phase II study. World J Gastrointest Oncol. 2020. https://doi.org/10.4251/wjgo.v12.i6.651.
Haverkamp L, Ruurda JP, van Hillegersberg R. Technical feasibility of Tachosil application on esophageal anastomoses. Gastroent Res. 2015. https://doi.org/10.1155/2015/534080.
Yan H, Yihuai H, Yaobin L, et al. Evaluation of Fibrin Sealant in Prevention of Cervical Anastomotic Leakage after McKeown Esophagectomy: a Single- Center, Retrospective Study. Ann Surg Oncol. 2021. https://doi.org/10.1245/s10434-021-09877-0.
Vakalopoulos KA, Daams F, Wu Z, Timmermans L, Jeekel JJ, Kleinrensink GJ, van der Ham A, Lange JF. Tissue adhesives in gastrointestinal anastomosis: a systematic review. J Surg Res. 2013. https://doi.org/10.1016/j.jss.2012.12.043.
Holmer C, Praechter C, Mecklenburg L, Heimesaat M, Rieger H, Pohlen U. Anastomotic stability and wound healing of colorectal anastomoses sealed and sutured with a collagen fleece in a rat peritonitis model. Asian J Surg. 2014. https://doi.org/10.1016/j.asjsur.2013.07.008.
Acknowledgements
We thank all the patients and their families who participated in this study. The study was supported in part by the National Natural Science Foundation of China (grant numbers 81402003 and 81972614).
Clinical trial registration statement
This study is registered at https://clinicaltrials.gov (NCT03847857)
Funding
The study was funded by the National Natural Science Foundation of China (grant numbers 81402003 and 81972614). All authors have completed the Unified Competing Interest form and declare no conflict of interest.
Author information
Authors and Affiliations
Contributions
Hong Yang conceived of the study and contributed to project design; Hong Yang, Zerui Zhao, Weizhao Huang, Geng Wang, Teng Mao, Wenqiang Lv and Lin Peng recruited patients for the study; Zhichao Li and Jiyang Chen performed data collection; Hong Yang, Zerui Zhao, Zhichao Li and Jiyang Chen analyzed and interpreted the data; Zerui Zhao and Zhichao Li drafted the manuscript; Hong Yang and Zerui Zhao contributed to editing and revising the article; authors read and approved the final version.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by the Ethics Committee of Sun Yat-sen University Cancer Center (B2018-011-01).
Informed consent was obtained from all individual participants included in the study.
Competing interests
All authors have completed the Unified Competing Interest form and declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhao, Z., Li, Z., Huang, W. et al. Fibrin sealant for the prevention of anastomotic leakage after esophagectomy for esophageal or esophagogastric junction cancer: interim report of a prospective, phase III, randomized controlled study. Holist Integ Oncol 2, 27 (2023). https://doi.org/10.1007/s44178-023-00050-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44178-023-00050-z