Abstract
Determining sex in Cervidae species is crucial for wildlife management, conservation, breeding programs, disease surveillance, and forensic investigations. While existing genetic sexing methods have proven effective, they often involve time-consuming and resource-intensive processes. In response to the growing need for a more efficient and cost-effective technique, this study aimed to develop a novel loop-mediated isothermal amplification (LAMP) method for sex detection in various deer species. We designed LAMP primers specific to Cervidae based on sex chromosome-located genes, AmelogeninX, and SRY. Analytical specificity was confirmed using 108 samples from red deer, roe deer, and fallow deer, and species specificity was also tested in other deer and mammal species. Sex determination results were fast (40 min) and accurate (100% reliability). This technique has the advantage of allowing researchers to identify the sex of different deer species using the same method. Using the isothermal amplification properties of LAMP, this new method provides a simple, quick, and accurate tool to help researchers, wildlife managers, and forensic investigators with important sex-related questions in cervid populations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The family Cervidae comprises a diverse assemblage of majestic deer species, playing pivotal roles in both natural ecosystems and captive breeding programs. Understanding the sex distribution within these populations is essential for a comprehensive grasp of the species’ reproductive dynamics, genetic diversity, and population management (Yamauchi et al. 2000; Han et al. 2007; Barbosa et al. 2009; Yamazaki et al. 2011; Paul et al. 2019). In many population genetics and ecology studies where non-invasively collected samples are used, the lack of morphological information necessitates the use of genetic methods for sex identification (Nittu et al. 2021). Individual cases have been recorded where the sex of an animal could only be accurately determined by genetic tests due to a congenital disorder, such as hermaphroditism (Pajares et al. 2009; Kropatsch et al. 2013). Fetal sex determination using the circulation cell-free DNA (ccfDNA) of uniparous mammals with long gestation benefits the management of economically important domestic animals as well as captive wild populations (Aucamp et al. 2023). Genetic sexing can be employed to monitor and study disease outbreaks within cervid populations, particularly in cases where sex-related differences in disease susceptibility or transmission exist. Understanding sex-specific responses to diseases can aid in implementing targeted control and management strategies (Moazami-Goudarzi et al. 2021). In areas where hunting is permitted, genetic sexing can help enforce hunting regulations and quotas. By identifying the sex of harvested animals, wildlife agencies can ensure sustainable hunting practices and prevent over-harvesting of specific sexes, contributing to the long-term conservation of cervid populations (Wilson and White 1998; Marealle et al. 2010; Haines et al. 2012; Corlatti et al. 2019). As a part of forensic investigations in cases of illegal hunting or poaching, genetic sexing can be employed to identify the sex of the poached animals aiding law enforcement efforts, when the sample is a carcass or remains of a specimen, hindering morphological identification (Takahashi et al. 1998; Wilson and White 1998; Hamlin et al. 2021; Morf et al. 2021; Zenke et al. 2022).
While traditional methods of sex determination, such as cytogenetic analysis (Cao et al. 2005; Fiorillo et al. 2013) and PCR-based techniques, have provided valuable insights into cervid genetics (Pfeiffer and Brenig 2005; Han et al. 2007; Qiao et al. 2007; Kim et al. 2008; Barbosa et al. 2009; Gurgul et al. 2010; Paul et al. 2019), the need for an alternative approach that offers higher efficiency and quicker turnaround times is becoming evident. In the studies conducted on sex detection in deer species, male-specific Y-chromosome markers were identified using a traditional PCR method, with mitochondrial, autosomal, or X-chromosome markers employed as internal controls. The loop-mediated isothermal amplification (LAMP) technique, introduced in the early 2000s, has revolutionized molecular diagnostics by enabling rapid and isothermal amplification of target DNA sequences (Notomi et al. 2000). The LAMP method uses a unique set of four to six primers to amplify the target region under isothermal conditions, eliminating the need for thermocycling. This property reduces time, energy consumption, and the need for expensive devices, making the method highly suitable for resource-limited settings (Wong et al. 2018; Hassan et al. 2022). Several experiments conducted with the LAMP technique also confirm that the method is suitable for examining samples collected non-invasively, such as faeces, urine, saliva, skin tape discs, milk, and hair (Fernandes et al. 2022; Singhla et al. 2022; Taslimi et al. 2023; Sritong et al. 2024; Zuraik et al. 2024).
The products of the LAMP reaction can be detected with the naked eye, with the help of intercalating dyes or pH indicators, by measuring turbidity, or with agarose gel electrophoresis (Mori et al. 2001; Goto et al. 2009; Tanner et al. 2015). These methods, except for electrophoresis, do not require an additional step and can already be detected in the PCR tube during the reaction (Khamlor et al. 2015; Dini et al. 2016). The LAMP method has also various application possibilities in human medicine and veterinary sciences, particularly in diagnosing infectious diseases, animal husbandry, and food testing (Zorkóczy et al. 2023).
Implementing the LAMP technique in cervid sex determination could offer numerous advantages compared to traditional PCR-based methods, including simplicity, robustness, and cost-effectiveness (Wong et al. 2018; Soroka et al. 2021; Iqbal et al. 2022; Sharma et al. 2024). In response to the pressing demand for this more streamlined sex detection method in Cervidae species, we aimed to develop a novel LAMP-based assay that targets specific sex-linked genes. Additionally, the method’s isothermal nature enables rapid amplification and detection within a single reaction, making it particularly suitable for on-site applications or studies involving large sample sizes (Nguyen et al. 2024).
Materials and methods
Sample collection and DNA extraction
Muscle samples were collected from registered shootings by hunters with a license from red deer (Cervus elaphus, n = 36), fallow deer (Dama dama, n = 36), and roe deer (Capreolus capreolus, n = 36), ensuring an equal distribution and knowledge of sexes (50%-50%), from various regions in Hungary. Genomic DNA was isolated using a FavorPrepTM Tissue Genomic DNA Extraction Mini Kit (Favorgen Biotech, Ping-Tung, Taiwan) following the provided procedural guidelines. The concentration of the extracted DNA was measured using a Qubit 2.0 Fluorometer (Life Technologies Corporation, Carlsbad, CA, USA), and the quality was tested using a 1% agarose gel stained with GelGreen™ Nucleic Acid Gel Stain (Biotium, Fremont, CA, USA). Isolated DNA from the tissue samples were stored at − 20 °C until subsequent analysis.
Design of deer-specific lamp primers
All available sequences of the sex-determining region Y (SRY) and Amelogenin X (AmelX) genes of the three targeted deer species were downloaded from the National Center for Biotechnology Information (NCBI) GenBank database https://www.ncbi.nlm.nih.gov/genbank/, accessed on 05 August 2023) (Table S1). The selected homolog sequences were aligned in the MEGA11 (Tamura et al. 2021) (Molecular Evolutionary Genetics Analysis) computing platform to identify conserved regions. The deer-specific primers for the Lamp reactions were designed based on the reference sequences KJ542362 (Cervus elaphus AmelX, 596 bp) and DQ888692 (Cervus elaphus SRY, 690 bp) using PrimerExplorerV5 (PrimerExplorerV5) (Eiken Chemical Co., Ltd., Japan; http://primerexplorer.jp/e/). Since, based on the alignment, there may be some differences in the SRY sequences within the three species, we designed loop primers with the help of the same program based on the information of the previously generated four primers in order to increase specificity and efficiency. The AmelX marker was used as an endogenous control. These two sets of primers were synthesized, and the information about the primer sets used in this study is listed in Table 1.
Using GenBank (BLAST, Basic Local Alignment Search Tool), we performed a query with the chosen conserved sequences to find further matches, with the expectation of expanding the use of the primers designed in this study to other cervid species.
Testing lamp primers in target species
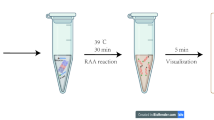
The designed primers were examined on a total of 108 control samples of three species with known sex (n = 18 per sex per species). The LAMP reactions were compiled separately for the AmelX and SRY markers: each reaction (5 μl in volume) consisted of 3 μl 10X Isothermal Mastermix (ISO-004, OptiGene, Horsham, West Sussex, UK), 0.2 μl FIP/BIP primers (40 μM stock), 0.2 μl F3/B3 primers (5 μM stock), 0.2 μl LF/LB primers (20 μM stock, used only in the SRY reaction), 0.3 μl Betaine (5 M stock, ThermoFisher Scientific, Waltham, MA, USA), 1 ng DNA template, and PCR grade-H2O to volume. PCR tubes were placed into a pre-heated thermocycler set to 65 °C for 50 min. A non-template control (NTC) was also used as the negative control in all LAMP experiments. Qualitative assessments of amplifications were conducted using two different end-point analyses: (a) 1 μL GelRed™ Nucleic Acid Gel Stain (10,000X stock, Biotium, Fremont, CA, USA) diluted with nuclease-free water 1:10 (v/v) was added to each tube and detected under blue-light with Glite 900 BW Gel Scanner; (b) samples were transferred to 2% agarose gel stained with the same gel stain used in method (a), run for 20 min and detected under blue-light (Glite 900 BW Gel Scanner).
Marker sequencing
Sanger-sequencing was applied to determine whether the targeted AmelX and SRY regions were amplified in the three deer species. Both markers per species were amplified with locus specific F3/B3 primers in singleplex PCR reactions. The PCR reactions (25 μl in volume) consisted of 5 μL DreamTaq™ Green PCR Master Mix (ThermoFisher Scientific, Waltham, MA, USA), 0.5 μM F3, and 0.5 μM B3 primer, 1 ng DNA template, and PCR grade-H2O to volume. PCR was carried out in an AppliedBiosystems 2720 Thermal Cycler with the following conditions: an initial 95ºC for 10 s. followed by 36 cycles of 20 s at 94ºC, annealing of 20 s at 54ºC, and 20 s at 72ºC. Qualitative assessments of singleplex amplifications were conducted using 2% agarose gel stained with GelRed™ Nucleic Acid Gel Stain (Biotium, Fremont, CA, USA).
Amplification products were purified using GenEluteTM PCR Clean-Up Kit (Sigma–Aldrich, St. Louis, MO, USA). Both DNA strands were sequenced using the BigDye® Terminator v.1.1 Cycle Sequencing Kit (Thermo Fisher Scientific, Waltham, MA, USA) following the manufacturer’s recommendations. PCR amplicons were sequenced by an ABI Prism 3130XL Genetic Analyzer (Applied Biosystems, Waltham, MA, USA), according to the manufacturer’s guidelines. Sequence analyses were performed using Sequencing Analysis Software 5.1 (Applied Biosystems, Waltham, MA, USA), and alignment using Sequencher™ 5.4.6 software (Gene Codes Corp, Ann Arbor, MI, USA).
Analytical specificity and sensitivity of the LAMP deer sex assay
For assessing the specificity and applicability of the two primer sets and the developed assays, isolated DNA from spotted deer (Axis axis, n = 8), sambar deer (Rusa unicolor, n = 6), and barking deer (Muntiacus muntjak, n = 2) was included, in addition to red deer, roe deer, and fallow deer. These samples had previously been verified for sex using the traditional PCR method (Zenke et al. 2022). Additionally, DNA samples from seven non-deer species (sheep, human, dog, cat, horse, cattle, and swine; both males and females) were examined as part of the study. AmelX and SRY Lamp reactions, with these samples of DNA, had conditions identical to those described above for amplification of primary target species.
To assess the optimal reaction time, the reaction tubes separately assembled of 6–6 samples for the two markers were placed in a thermocycler at 65 °C for 35, 40, 45, and 50 min, respectively. Sensitivity evaluations of the two developed Lamp assays were performed using serially diluted genomic DNA with concentrations 1.0, 0.5, 0.25, 0.125, and 0.062 ng/μl. The amount of the DNA was estimated using a Qubit™ Fluorometer. Loop-mediated amplification of 1 μl of diluted sample was performed using the conditions described above for LAMP reactions.
Results
Primer design
By aligning the available target sequences in the three investigated species, it was possible to identify a SRY gene section that shows 100% similarity in red deer and fallow deer, and 93–100% similarity in roe deer compared to the former two species. While only one LAMP primer set was generated for the SRY gene, five primer sets were created for the AmelX gene. The location of the chosen primers designed for this research on both genes’ sections is shown in Fig. 1. Since differences in the generated primer binding sequences are possible in at least one position in fallow deer and up to at least six positions in roe deer, loop primers (LF and LB) for SRY gene section were also designed (Fig. 1). Based on the results of the in silico analysis of the same gene sections of different deer species, our LAMP primers designed for conserved gene sections show a high degree of sequence homology with other species as well (Table S2).
Alignment and identification of gene segments in the SRY (A) and Amelogenin X (B) sequences in red deer (Cervus elaphus), fallow deer (Dama dama), and roe deer (Capreolus capreolus) by MEGA program. The grey arrow indicates a possible different nucleotide position from the designed primer sequence for fallow deer and the yellow arrows for roe deer sequences
As part of primer testing, traditional monoplex PCR amplification of both markers in the three deer species examined revealed the appropriate length of the specific gene segments in the corresponding sexes. The products were verified by sequencing, and the obtained base sequences were compared with sequences available in GenBank. As a result, 100% homology was achieved with sequences corresponding to the respective species and sex. The AmelX and SRY gene segments detected in the three species were deposited into the NCBI GenBank with the identification codes OR515036-OR515041.
Specificity and sensitivity of LAMP reactions
The LAMP reaction for the AmelX marker gave positive results in red, roe, and fallow deer species, males and females from all 108 samples. The reactions aimed at detecting the SRY marker resulted in positive outcomes for all male samples (n = 54) and negative results for female samples (n = 54) of all three species. The optimization of different reaction times showed that SRY primers fully discriminated females from males when LAMP reactions were performed for 40 min (Fig. S1). Visualization of the amplified products was possible by adding an intercalating dye (GelRed™) directly to the reaction tubes at the end of the reaction and then detecting them with blue light screening; the same results were obtained with agarose gel electrophoresis (Fig. 2). A patent application for the method has been initiated, with the application base number P2300426.
Possibilities of detecting the amplification of AmelX and SRY markers using the LAMP method from one female and one male sample per species. A: on agarose gel, B and C: mixed with intercalating dye under blue light illumination with two different displays (♀: female sample, ♂: male sample, + : true positive, - : true negative, RC = reagent control)
The analytical sensitivity of the 40-minute LAMP test for red deer was determined at 0.25 ng DNA, while it was 0.5 ng DNA for fallow deer and roe deer for both the AmelX and SRY reactions (Fig. S2). The designed AmelX primers show low species specificity, since visible LAMP products were detectable for mouflon, human, dog, cat, horse, cattle, and swine DNA (male and female). However, SRY primers show higher specificity to deer DNA since an obvious LAMP product was only detectable using male mouflon DNA (Fig. S3).
Regarding the DNA test of the other three deer species, spotted, sambar, and barking deer, all LAMP reactions for the AmelX marker gave positive results if the amount of used DNA reached 1 ng. Positive SRY results were obtained with the Lamp reaction in animals that proved to be male with the traditional PCR test (Fig. S4). These products were also verified by sequencing. AmelX obtained from spotted, sambar, and barking deer were deposited into the NCBI GenBank with the identification codes PP358594-PP358596. SRY gene segments detected in spotted and sambar deer were deposited into the NCBI GenBank with the identification codes PP358597-PP358598.
Discussion
LAMP techniques have been developed so far to genetically test the sex of many wild bird and fish species (Hsu and Tsai 2011; Chan et al. 2012; Centeno-Cuadros et al. 2017; Elnomrosy et al. 2022), as well as various economically important domestic animal species (Hirayama et al. 2006, 2013; Khamlor et al. 2015; Kim et al. 2015; Dini et al. 2016). Sex identification in Cervidae is among the most critical parameters for conservation efforts and legal hunting. However, due to the lack of morphological information during the collection of non-invasive samples and in most forensic cases, a genetic method is needed to perform such identification. Methods developed with a universal primer system designed for conserved DNA segments allow for sex testing from diverse biological samples of several different deer species (Zenke et al. 2022). As far as the authors know, this is the first application of LAMP in identifying the sex of Cervidae and clearly shows the potential application of LAMP in molecular ecology and conservation efforts, as well as in the forensic field. The LAMP primers designed for the AmelX gene as a reaction control were successfully utilized in all investigated deer species (most commonly found in Europe and India), and positive results were obtained in all cases during the examination of the female and male samples. However, the LAMP primers designed for the SRY gene, though appearing less than ideal based on the in silico tests, proved efficient in the in vitro, even on DNA samples from roe deer and other deer species from India, where several base deviations can occur in the primer binding regions. This is probably also due to the loop primers, which help the reaction work more efficiently by providing increased starting points for DNA synthesis, resulting in shorter amplification time and higher specificity. The primers designed in this study can be uniformly employed for the three antlered wild species found in Europe (red deer and European roe deer) and other parts of the world (fallow deer). Based on our in silico and in vitro testing, our developed method can also be applied to cervids in other continents in the future after in vitro testing of certain species and populations. The developed robust and rapid LAMP assays showed the advantage of simple detection (evaluated by visual inspection), rapid reaction time (40 min), isothermal conditions (less equipment required), and high efficiency. Therefore, the method is more economical and practical than conventional PCR. The LAMP assay can be easily performed in the field and is a valuable tool for detecting sex ratios in wild populations. The sensitivity and specificity of the assay were established using a diverse set of cervid samples, encompassing various species and populations.
Remarkably, LAMP does not necessarily require expensive reagents (such as fluorescently labeled primers) or electrophoresis for amplified product detection. The examination of the results of the reactions with an intercalating dye proved to be sufficiently sensitive and reliable, which is supported by the identical results of detection on agarose gel. However, the subsequent addition of the intercalating dye at the end of the reaction is considered an extra step, and the re-opening of the reaction tubes includes the possibility of potential contamination (Quoc et al. 2018; Kumar et al. 2021). Nevertheless, the method did not cause problems if done with sufficient care.
In conclusion, we developed a LAMP-based technique for identifying the sex of several deer species. The development of a quick LAMP-based sex detection approach for Cervidae species is expected to make a substantial contribution to wildlife management, conservation efforts, and breeding programs. This method is considered a powerful and dependable alternative to PCR. By offering a simple and reliable tool, researchers and wildlife managers can swiftly and efficiently address sex-related inquiries within cervid populations, thereby making a valuable contribution to the overall comprehension and conservation of these iconic and ecologically consequential organisms.
Data availability
The data will be available with the corresponding author upon request.
References
Aucamp J, van der Zwan H, Geldenhuys Z, Abera A, Louw R, van der Sluis R (2023) Diagnostic applications and limitations for the use of cell-free fetal DNA (cffDNA) in animal husbandry and wildlife management. Res Vet Sci 158:106–116. https://doi.org/10.1016/j.rvsc.2023.03.013
Barbosa AM, Fernández-García JL, Carranza J (2009) A new marker for rapid sex identification of red deer (Cervus elaphus). Hystrix Ital J Mammal 20(2):169–172
Cao X, Jiang H, Zhang X (2005) Polymorphic karyotypes and sex chromosomes in the tufted deer (Elaphodus cephalophus): cytogenetic studies and analyses of sex chromosome-linked genes. Cytogenet Genome Res 109(4):512–518. https://doi.org/10.1159/000084212
Centeno-Cuadros A, Abbasi I, Nathan R (2017) Sex determination in the wild: a field application of loop-mediated isothermal amplification successfully determines sex across three raptor species. Mol Ecol Resour 17(2):153–160. https://doi.org/10.1111/1755-0998.12540
Chan K-W, Liu P-C, Yang W-C, Kuo J, Chang C-T, Wang C-Y (2012) A novel loop-mediated isothermal amplification approach for sex identification of Columbidae birds. Theriogenology 78(6):1329–1338. https://doi.org/10.1016/j.theriogenology.2012.05.034
Corlatti L, Sanz-Aguilar A, Tavecchia G, Gugiatti A, Pedrotti L (2019) Unravelling the sex-and age-specific impact of poaching mortality with multievent modeling. Front Zool 16:1–8. https://doi.org/10.1186/s12983-019-0321-1
Dini P, Van Poucke M, Herrera C, Peelman L, Daels P (2016) Preimplantation gender determination on equine embryos using LAMP. J Equine Vet Sci 41:75–75. https://doi.org/10.1016/j.jevs.2016.04.070
Elnomrosy SM, Hagag NM, AbdAllah MI, Kolenda R, Zacharski M (2022) Application of loop-mediated isothermal amplification (LAMP) in sex identification of parrots bred in Egypt. Biology 11(4):565. https://doi.org/10.3390/biology11040565
Fernandes LP, Rocha MN, Duarte CG, Minozzo JC, do, Monte-Neto RL, Felicori LF (2022) Validation of a colorimetric LAMP to detect Loxosceles experimental envenomation. Toxicon 216:50–56. https://doi.org/10.1016/j.toxicon.2022.06.017
Fiorillo BF, Sarria-Perea JA, Abril VV, Duarte JMB (2013) Cytogenetic description of the amazonian brown brocket Mazama nemorivaga (Artiodactyla, Cervidae). Comp Cytogenet 7(1):25. https://doi.org/10.3897/compcytogen.v7i1.4314
Goto M, Honda E, Ogura A, Nomoto A, Hanaki K-I (2009) Colorimetric detection of loop-mediated isothermal amplification reaction by using hydroxy naphthol blue. Biotechniques 46(3):167–172. https://doi.org/10.2144/000113072
Gurgul A, Radko A, Słota E (2010) Characteristics of X-and Y-chromosome specific regions of the amelogenin gene and a PCR-based method for sex identification in red deer (Cervus elaphus). Mol Biol Rep 37:2915–2918. https://doi.org/10.1007/s11033-009-9852-4
Haines AM, Elledge D, Wilsing LK, Grabe M, Barske MD, Burke N, Webb SL (2012) Spatially explicit analysis of poaching activity as a conservation management tool. Wildl Soc Bull 36(4):685–692. https://doi.org/10.1002/wsb.194
Hamlin BC, Meredith EP, Rodzen J, Strand JM (2021) OdoPlex: an STR multiplex panel optimized and validated for forensic identification and sex determination of north American mule deer (Odocoileus hemionus) and white-tailed deer (Odocoileus virginianus). Forensic Sci Int Anim Environ 1:100026. https://doi.org/10.1016/j.fsiae.2021.100026
Han S-H, Cho I-C, Lee S-S, Tandang L, Lee H, Oh H-S, Kim BS, Oh M-Y (2007) Identification of species and sex of Korean roe deer (Capreolus pygargus Tianschanicus) using SRY and CYTB genes. Integr Biosci 11(2):165–168. https://doi.org/10.1080/17386357.2007.9647331
Hassan MM, Grist LF, Poirier AC, La Ragione RM (2022) JMM profile: loop-mediated isothermal amplification (LAMP): for the rapid detection of nucleic acid targets in resource-limited settings. J Med Microbiol 71(5):001522. https://doi.org/10.1099/jmm.0.001522
Hirayama H, Kageyama S, Takahashi Y, Moriyasu S, Sawai K, Onoe S, Watanabe K, Kojiya S, Notomi T, Minamihashi A (2006) Rapid sexing of water buffalo (Bubalus bubalis) embryos using loop-mediated isothermal amplification. Theriogenology 66(5):1249–1256. https://doi.org/10.1016/j.theriogenology.2006.03.036
Hirayama H, Kageyama S, Moriyasu S, Sawai K, Minamihashi A (2013) Embryo sexing and sex chromosomal chimerism analysis by loop-mediated isothermal amplification in cattle and water buffaloes. J Reprod Dev 59(4):321–326. https://doi.org/10.1262/jrd.2013-028
Hsu R-J, Tsai H-J (2011) Performing the labeled microRNA pull-down (LAMP) assay system: an experimental approach for high-throughput identification of microRNA-target mRNAs. Methods Mol Biol Clifton NJ 764:241–247. https://doi.org/10.1007/978-1-61779-188-8_16
Iqbal BN, Arunasalam S, Divarathna MV, Jabeer A, Sirisena P, Senaratne T, Muthugala R, Noordeen F (2022) Diagnostic utility and validation of a newly developed real time loop mediated isothermal amplification method for the detection of SARS CoV-2 infection. J Clin Virol Plus 2(3):100081. https://doi.org/10.1016/j.jcvp.2022.100081
Khamlor T, Pongpiachan P, Parnpai R, Punyawai K, Sangsritavong S, Chokesajjawatee N (2015) Bovine embryo sex determination by multiplex loop-mediated isothermal amplification. Theriogenology 83(5):891–896. https://doi.org/10.1016/j.theriogenology.2014.11.025
Kim BJ, Lee Y-S, An J, Park H-C, Okumura H, Lee H, Min M-S (2008) Species and sex identification of the Korean goral (Nemorhaedus Caudatus) by molecular analysis of non-invasive samples. Mol Cells 26(3):314–318. https://doi.org/10.1016/S1016-8478(23)14001-5
Kim SW, Choi JS, Sharma N, Ko YG, Do YJ, Byun M, Seong HH, Park SB, Jeong DK (2015) A novel approach for determination of chicken sexing at an early stage of development by using loop-mediated isothermal amplification method. Turk J Vet Anim Sci 39(5):583–588. https://doi.org/10.3906/vet-1410-84
Kropatsch R, Dekomien G, Akkad DA, Gerding WM, Petrasch-Parwez E, Young ND, Altmüller J, Nürnberg P, Gasser RB, Epplen JT (2013) SOX9 duplication linked to intersex in deer. PLoS ONE 8(9):e73734. https://doi.org/10.1371/journal.pone.0073734
Kumar TS, Radhika K, Rajan JJS, Makesh M, Alavandi S, Vijayan K (2021) Closed-tube field-deployable loop-mediated isothermal amplification (LAMP) assay based on spore wall protein (SWP) for the visual detection of Enterocytozoon hepatopenaei (EHP). J Invertebr Pathol 183:107624. https://doi.org/10.1016/j.jip.2021.107624
Marealle WN, Fossøy F, Holmern T, Stokke BG, Røskaft E (2010) Does illegal hunting skew Serengeti wildlife sex ratios? Wildl Biol 16(4):419–429. https://doi.org/10.2981/10-035
Moazami-Goudarzi K, Andréoletti O, Vilotte J-L, Béringue V (2021) Review on PRNP genetics and susceptibility to chronic wasting disease of Cervidae. Vet Res 52:1–15. https://doi.org/10.1186/s13567-021-00993-z
Morf NV, Kopps AM, Nater A, Lendvay B, Vasiljevic N, Webster LM, Fautley RG, Ogden R, Kratzer A (2021) STRoe deer: a validated forensic STR profiling system for the European roe deer (Capreolus capreolus). Forensic Sci Int Anim Environ 1:100023. https://doi.org/10.1016/j.fsiae.2021.100023
Mori Y, Nagamine K, Tomita N, Notomi T (2001) Detection of loop-mediated isothermal amplification reaction by turbidity derived from magnesium pyrophosphate formation. Biochem Biophys Res Commun 289(1):150–154. https://doi.org/10.1006/bbrc.2001.5921
Nguyen TK, Jun H, Louis JM, Mazigo E, Lee W-J, Youm HC, Shin J, Lungu DK, Kanyemba C, Ahmed MA, others (2024) Enhancing malaria detection in resource-limited areas: a high-performance colorimetric LAMP assay for Plasmodium falciparum screening. Plos One 19(2):e0298087. https://doi.org/10.1371/journal.pone.0298087
Nittu G, Bhavana PM, Shameer TT, Ramakrishnan B, Archana R, Kaushal KK, Khedkar GD, Mohan G, Jyothi M, Sanil R (2021) Simple nested allele-specific approach with penultimate mismatch for precise species and sex identification of tiger and leopard. Mol Bio Rep 48:1667–1676. https://doi.org/10.1007/s11033-021-06139-w
Notomi T, Okayama H, Masubuchi H, Yonekawa T, Watanabe K, Amino N, Hase T (2000) Loop-mediated isothermal amplification of DNA. Nucleic Acids Res 28(12):e63–e63. https://doi.org/10.1093/nar/28.12.e63
Pajares G, Balseiro A, Pérez-Pardal L, Gamarra JA, Monteagudo LV, Goyache F, Royo LJ (2009) Sry-negative XX true hermaphroditism in a roe deer. Anim Reprod Sci 112(1–2):190–197. https://doi.org/10.1016/j.anireprosci.2008.04.018
Paul S, Ghosh T, Pandav B, Mohan D, Habib B, Nigam P, Mondol S (2019) Rapid molecular assays for species and sex identification of swamp deer and other coexisting cervids in human-dominated landscapes of the Terai region and upper Gangetic Plains, northern India: implications in understanding species distribution and population parameters. J Genet 98:1–8. https://doi.org/10.1007/s12041-019-1094-1
Pfeiffer I, Brenig B (2005) X-and Y-chromosome specific variants of the amelogenin gene allow sex determination in sheep (Ovis aries) and European red deer (Cervus elaphus). BMC Genet 6:1–4. https://doi.org/10.1186/1471-2156-6-16
PrimerExplorerV5 http://primerexplorer.jp/e/
Qiao Y, Zou F, Wei K, Yue B (2007) A rapid sex-identification test for the forest musk deer (Moschus berezovskii) based on the ZFX/ZFY gene. Zool Sci 24(5):493–495. https://doi.org/10.2108/zsj.24.493
Quoc NB, Phuong NDN, Chau NNB, others (2018) Closed tube loop-mediated isothermal amplification assay for rapid detection of hepatitis B virus in human blood. Heliyon 4(3):e00561. https://doi.org/10.1016/j.heliyon.2018.e00561
Sharma A, Azam M, Verma P, Talwar V, Roy S, Veeraraghavan B, Singh R, Gaind R (2024) Application of LAMP assay for detection of carbapenem-resistant Acinetobacter calcoaceticus-Acinetobacter baumannii complex in ICU admitted sepsis patients: a rapid and cost-effective diagnostic tool. Diagn Microbiol Infect Dis 110(1):116398. https://doi.org/10.1016/j.diagmicrobio.2024.116398
Singhla T, Pikulkaew S, Boonyayatra S (2022) Performance of loop-mediated isothermal amplification technique in milk samples for the diagnosis of bovine tuberculosis in dairy cattle using a Bayesian approach. Pathogens 11(5):573. https://doi.org/10.3390/pathogens11050573
Soroka M, Wasowicz B, Rymaszewska A (2021) Loop-mediated isothermal amplification (LAMP): the better sibling of PCR? Cells 10(8):1931. https://doi.org/10.3390/cells10081931
Sritong N, Ngo WW, Ejendal KF, Linnes JC (2024) Development of an integrated sample amplification control for salivary point-of-care pathogen testing. Anal Chim Acta 1287:342072. https://doi.org/10.1016/j.aca.2023.342072
Takahashi M, Masuda R, Uno H, Yokoyama M, Suzuki M, Yoshida MC, Ohtaishi N (1998) Sexing of carcass remains of the Sika deer (Cervus nippon) using PCR amplification of the sry gene. J Vet Med Sci 60(6):713–716. https://doi.org/10.1292/jvms.60.713
Tamura K, Stecher G, Kumar S (2021) MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol 38(7):3022–3027
Tanner NA, Zhang Y, Evans TC Jr (2015) Visual detection of isothermal nucleic acid amplification using pH-sensitive dyes. Biotechniques 58(2):59–68. https://doi.org/10.2144/000114253
Taslimi Y, Habibzadeh S, Goyonlo VM, Akbarzadeh A, Azarpour Z, Gharibzadeh S, Shokouhy M, Persson J, Harandi AM, Mizbani A, others (2023) Tape-disc-loop-mediated isothermal amplification (TD-LAMP) method as noninvasive approach for diagnosis of cutaneous leishmaniasis caused by L. Tropica. Heliyon 9(11):e21397. https://doi.org/10.1016/j.heliyon.2023.e21397
Wilson P, White B (1998) Sex identification of elk (Cervus elaphus canadensis), moose (Alces alces), and white-tailed deer (Odocoileus virginianus) using the polymerase chain reaction. J Forensic Sci 43(3):477–482. https://doi.org/10.1520/JFS16172J
Wong Y-P, Othman S, Lau Y-L, Radu S, Chee H-Y (2018) Loop-mediated isothermal amplification (LAMP): a versatile technique for detection of micro-organisms. J Appl Microbiol 124(3):626–643. https://doi.org/10.1111/jam.13647
Yamauchi K, Hamasaki S, Miyazaki K, Kikusui T, Takeuchi Y, Mori Y (2000) Sex determination based on fecal DNA analysis of the amelogenin gene in sika deer (Cervus nippon). J Vet Med Sci 62(6):669–671. https://doi.org/10.1292/jvms.62.669
Yamazaki S, Motoi Y, Nagai K, Ishinazaka T, Asano M, Suzuki M (2011) Sex determination of sika deer (Cervus nippon yesoensis) using nested PCR from feces collected in the field. J Vet Med Sci 73(12):1611–1616. https://doi.org/10.1292/jvms.11-0235
Zenke P, Zorkóczy OK, Lehotzky P, Ózsvári L, Pádár Z (2022) Molecular sexing and species detection of antlered European hunting game for forensic purposes. Animals 12(3):246. https://doi.org/10.3390/ani12030246
Zorkóczy OK, Bujtor Z, Szives A, Ózsvári L, Wagenhoffer Z, Zenke P (2023) A LAMP-(hurok által közvetített izotermikus sokszorosítás) technika fő alkalmazási lehetőségei az állatorvostudományban. Magy Állatorvosok Lapja 145(10):611–624. https://doi.org/10.56385/magyallorv.2023.10.611-624
Zuraik AA, Daboul Y, Awama MA, Yazigi H, Kayasseh MA, Georges M (2024) Rapid detection of FadA in Fusobacterium nucleatum using the quantitative LAMP colorimetric phenol red method in stool samples from colorectal cancer patients. Sci Rep 14(1):13739. https://doi.org/10.1038/s41598-024-62846-x
Acknowledgements
We would like to thank all the licensed hunters, who provided deer samples from Hungary. We thank Zsófia Bujtor for her contribution to some lab work. In addition, we are grateful to the two anonymous reviewers for their comments, which significantly improved the manuscript.
Funding
This work was supported by the National Research, Development and Innovation Fund of Hungary [grant numbers 2019 − 1.2.1-Egyetemi-Öko-2019-00010, financed under the 2019 − 1.2.1-Egyetemi-Öko funding scheme].
Open access funding provided by University of Veterinary Medicine.
Author information
Authors and Affiliations
Contributions
Petra Zenke and Pál Lehotzky contributed to the study conceptualization, design, and organization. Material collection and preparation were performed by Adrienn Gyurcsó, László Ózsvári, and Raveendranathanpillai Sanil. Conceptualization of methods and data analyses were performed by Petra Zenke and Orsolya K. Zorkóczy. Figures were created by Petra Zenke. The first draft was written by Petra Zenke and Orsolya K. Zorkóczy, and comments from all authors. Petra Zenke led the extensive revisions of the text and figures. All authors have read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Institutional review board statement
None of the tests described in the manuscript titled ‘Development of a Loop-Mediated Isothermal Amplification Technique for Sex Detection in Cervidae Species’ required the approval of the Ethics Committee or Institutional Review Board. There were not any experimental animals involved in the study. The samples required for the tests were obtained from animals shot in compliance with the current national and international laws in force.
Conflict of interest
The authors declare that they have no interests that could have appeared to influence the work reported in this paper.
Additional information
Handling editor: Heiko G. Rödel.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zorkóczy, O.K., Gyurcsó, A., Ózsvári, L. et al. Development of a loop-mediated isothermal amplification technique for sex detection in Cervidae species. Mamm Biol (2024). https://doi.org/10.1007/s42991-024-00457-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42991-024-00457-9