Abstract
After near extirpation by nineteenth century whaling, New Zealand’s southern right whales (Eubalaena australis) are recovering strongly, calving almost exclusively at the subantarctic Auckland Islands. Right whales are capital breeders; body condition is an important driver of their breeding success. Here we use unmanned aerial vehicles to characterise variation in individual size and shape, and to quantify the size structure of the subset of the population we sampled. Of 108 whales photographically identified we gained a comprehensive set of measurements from 63 individuals, as well as length measurements for 29 calves and six non-calf whales for which the full suite of measurements were not obtainable. Lactating females (n = 32) ranged in length from 11.84 to 15.22 m, apparent non-breeding adults (n = 9) were between 11.96 and 14.92 m, while subadults (n = 28) were between 8.82 and 11.72 m long. Calves were between 5.15 and 7.53 m. Principal component analysis of the measurement data showed that widths (particularly at the positions of 30–80% along total body length) were most influential in PC1 (40.3% variance explained). Measurements of structural features (i.e. head and flukes) related more closely to PC2 (18.2% variance explained) and PC3 (14.8% variance explained). We, therefore, interpret PC2 and PC3 as representing structural size, while PC1 represents body condition. Subadults and non-breeding adults showed more variation in body condition than lactating females, highlighting the need for this demographic to maintain their body condition within a tighter range to meet the high nutritional demands of raising calves.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Of fundamental importance to conservation biology is understanding key fitness-related traits that define the health of a population. As populations thrive or decline based on the health of individuals, assessing such traits at the individual level can shed light on the health of the population as a whole. The physiological state of an animal is related to its evolutionary fitness; size and body condition influence survival and reproductive success through Darwinian selection (Darwin 1859). Obviously, body condition is important for individual survival and reproduction as both rely on stored energy reserves (Gaillard et al. 2000; Clutton-Brock and Sheldon 2010). Body condition is likely to be particularly important in defining reproductive success in capital breeders, since the burden of reproduction is often supported solely from stored energy reserves (Festa-Bianchet et al. 1998; Bonnet et al. 2002; Miller et al. 2011). Indeed, from ultrasound measurements of blubber in live right whales (Eubalaena glacialis and E. australis), Miller et al. (2011) showed large reductions in blubber thickness from pre-pregnancy to late lactation, indicating the substantial metabolic burden of reproduction. They concluded that a critical level of body fat is necessary for successful reproduction.
Most baleen whales are capital breeders; their energy stores are primarily found in subcutaneous and visceral adipose and muscle tissues (Lockyer 1986, 1987; Vikingsson 1995; Niæss et al. 1998; Christiansen et al. 2013) and can be examined to assess whale bioenergetics (e.g., Aguilar and Borrell 1990; Vikingsson 1995; Niæss et al. 1998; Miller et al. 2011). Investigations into these specific areas of endogenous energy stores often require direct interaction with the animals, and in some cases physical samples are taken from carcasses (e.g., Lockyer et al. 1985; Vikingsson 1990; Beck et al. 1993; Niæss et al. 1998). A less intrusive and more holistic approach, incorporating several key areas of lipid storage, involves measuring two components of animal size; first a relatively stable or slow-growing structural component (e.g., skeletal), followed by measurements of width, girth, or volume, that represent the stored energy reserves that are likely to change in response to fluctuations in the environment, reproductive state and overall health (Lockyer 1986, 1987, 2007; Vikingsson 1990; Hayes and Shonkwiler 2001; Perryman and Lynn 2002; Stevenson and Woods 2006; Miller et al. 2011, 2012; Christiansen et al. 2016a, 2018, 2020). These broad metrics and their relationships with prey abundance, reproductive success and seasonal fasting have been explored in several whale species, including gray (Eschrichtius robustus, Perryman and Lynn 2002; Bradford et al. 2012; Lemos et al. 2020), minke (Balaenoptera acutorostrata, Ichii et al. 1998; Niæss et al. 1998; Christiansen et al. 2014), blue (B. musculus, Durban et al. 2016), humpback (Megaptera novaengliae, Christiansen et al. 2016a) and right whales (Eubalaena sp., Miller et al. 2012; Christiansen et al. 2018), illustrating the utility of such an all-encompassing approach.
Southern right whales (SRWs; Eubalaena australis) were once found extensively throughout New Zealand waters but were hunted to near extinction by unconstrained whaling during the 1800s. This reduced the population to a total of around 110 whales, representing approximately 25–30 mature females, < 0.4% of their original total estimated abundance of approximately 35,000 whales (Carroll et al. 2015; Jackson et al. 2016). Considered extirpated around mainland New Zealand (Gaskin 1964), the small remnant population found at the sub-Antarctic Auckland Islands was further impacted by illegal Soviet whaling from 1963 to 1966 (Tormosov et al. 1998). Scientific interest in SRWs in New Zealand waters began in 1995 (Patenaude et al. 1998) following a New Zealand Air Force surveillance flight which reported substantial numbers of right whales at the Auckland Islands during the winter of 1992 (Jackson et al. 2009). Since then, studies have focussed on Port Ross, a sheltered harbour where several hundred right whales gather to calve during the austral winter (Childerhouse et al. 2010; Carroll et al. 2011, 2013, 2014, 2015, 2016; Davidson et al. 2017; Rayment et al. 2012, 2015, 2017; Webster et al. 2016, 2019). Detailed analysis of the impact of whaling captures and subsequent recovery estimates the current population of New Zealand is at approximately 12% of its original size; the most recent abundance estimate is for 2009 at around 2200 individuals and is recovering at around 5–7% per year (Carroll et al. 2013; Jackson et al. 2016).
SRWs spend most of the year in offshore foraging grounds before migrating to inshore calving grounds during winter months (Jackson et al. 2016). Their diet is primarily composed of copepods (Calanus sp.) and small krill (Euphausia sp.; Tormosov et al. 1998), typically the dominant components of oceanic zooplankton, and taxa that are strongly impacted by ocean warming (Hayes et al. 2005; Nicol et al. 2008). With a gestation period of around 12 months (Best 1994), SRWs rely almost exclusively on endogenous energy stores while travelling to, residing in, and then travelling from their calving grounds. Breeding females therefore finance the late stages of gestation and early lactation almost exclusively from stored fat reserves, a period of around 7–11 weeks (Burnell and Bryden 1997; Best 2000). Thus, it follows that the body condition of breeding females arriving at their calving grounds is largely dictated by their success in the previous foraging season, and in turn is likely to be a major driver of calf growth and survival (Miller et al. 2011, 2012; Christiansen et al. 2018).
Photogrammetry, the photographic measurement of size and shape, is commonly used in the morphometric analyses of animals (e.g., Zelditch et al. 2004; Frederich et al. 2008; Burnett et al. 2018) and has been applied to living whales for over four decades (e.g., Whitehead and Payne 1981; Gordon 1990; Ratnaswamy and Winn 1993; Dawson et al. 1995; Jaquet 2006; Growcott et al. 2012; Christiansen et al. 2019). The most practical of these approaches is the recent development of single-camera photogrammetry from Unmanned Aerial Vehicles (UAVs) in which image scale is provided via accurate measurement of altitude (e.g., Durban et al. 2015, Dawson et al. 2017). UAVs are becoming increasingly sophisticated and less expensive, offering new opportunities as platforms for data collection (e.g., Linchant et al. 2015; Fiori et al. 2017; Horton et al. 2019). In combination with quality optics and precise altitude measurement, we can now quantify body size and shape, and thus assess the body condition of large whales from UAVs (e.g., Dawson et al. 2017; Christiansen et al. 2019).
Individual body condition has been assessed previously in studies of other right whale populations (e.g., North Pacific right whales—Klumov 1962; Omura et al. 1969; North Atlantic and southern right whales—Miller et al. 2012; southern right whales—Christiansen et al. 2018). Most recently, Christiansen et al. (2020) analysed photogrammetric data gathered from North Atlantic right whales (NARWs) and four populations of SRWs, revealing the poor nutritional condition typical of individual NARWs, sharpening the focus on their critically endangered conservation status (e.g., Kraus et al. 2016; Corkeron et al. 2018). Given the paucity of information on foraging areas for almost all populations, and the small number of links in the food web between primary productivity and right whales (Tomosov et al. 1998; Leaper et al. 2006), assessing the size and body condition of individuals within discrete populations provides insight not only into the health of the population, but on the nutritional conditions they experience. This in turn can aid researchers in the identification of feeding environments which, for the SRW, remain elusive.
With this in mind, the aims of this study were (1) to describe the size structure, in terms of total lengths, of southern right whales in Port Ross, the primary calving ground in New Zealand, and (2) to identify key areas of the body that are important for endogenous energy storage in this recovering population. The size and body condition structure of this population has not previously been described in detail. Thus, by doing so we aim to lay the foundation from which future studies can follow.
Methods
An expedition to Port Ross, Auckland Islands (Fig. 1), was timed to coincide with the annual peak in numbers of SRWs (Rayment et al. 2017). During a three-week period (26/07/2016–17/08/2016), images of individual right whales were gathered via a “prosumer” UAV (DJI Inspire 1 Pro) modified for single image photogrammetry. An onboard custom-built datalogger recorded lidar measurements of altitude and the tilt of the drone (when correcting for wind) collected via an Inertial Measurement Unit (IMU) continuously during flight (Dawson et al. 2017). The UAV was flown from the deck of a 20 m research vessel, Polaris II, out towards nearby whales. Images were taken at altitudes of 20–30 m, at which height noise from the DJI Inspire 1 Pro is below that of ambient noise within the water (Christiansen et al. 2016b). Flights lasted 10–12 min; we adopted a conservative approach to battery use, landing the UAV at 40% battery remaining. Several images were taken of each whale encountered, with only those that met strict quality criteria retained for analysis. Images were first graded on sharpness (1 being “pin-sharp”; 4 being out of focus), tail orientation was recorded (straight, at the surface or drooped), and whether the sides of the whale were clear and undistorted by surface ripples was noted. Images were retained in measurement analysis if they were sharp (i.e., grades 1 or 2), with the sides of the whale clearly visible, and flukes either straight or at the surface, showing minimal tailstock flexion (which would otherwise cause a downward bias in length measurements). To ensure consistency among retained images in quality and measurements, the same researcher conducted quality assessment and measurement procedures.
UAV-derived aerial images were used to identify individual whales and establish a new aerial photo-ID catalogue. For ID, images were retained only if they clearly showed the whales’ callosities and/or other distinguishing features used in identification (e.g., dorsal blazes or “grey-morph” colouration; Payne et al. 1983; Schaeff et al. 1999; Carroll et al. 2014). As with established techniques for lateral photo-ID of right whales (Cooke et al. 2003; Bannister 2010; Brandão et al. 2012), a suite of 17 distinguishing features was used to identify individuals, including presence or absence of lip callosities, the number of rostral islands and the nature of the bonnet (Fig. 2; Pirzl et al. 2009). Individuals were catalogued in a custom-written database, Bigfish (Pirzl et al. 2009), with lactating females categorised as those with a closely and consistently accompanying calf, subadults as those individuals with total length (TL) measurements smaller than the smallest lactating female identified, and apparent non-breeding adults as those with TL measurements greater than the smallest lactating female, but without a closely and consistently accompanying calf (Christiansen et al. 2020).
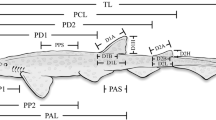
Example aerial image of a female southern right whale with measurement planes as per Dawson et al. (2017). The turquoise line is total length (TL), the yellow lines are width measurements at 10% increments along TL, the purple line is eye to eye, the red line is rostrum to eye, the dashed black line is rostrum to blowhole and the green line is measurement from fluke tip to fluke tip
Photogrammetric measurements of individuals included TL, widths at 10% intervals along the body’s length, as well as the distances from eye to eye, rostrum to eye, rostrum to blowhole and between fluke tips (Fig. 2). Measurement of the aerial images was achieved using a custom-written Graphical User Interface (“Whalength”), written in MatLab. This software corrected for measured camera and lens distortions, using altitude and tilt data recorded at the time of the photograph. Repeated measurements of a floating known-size target indicated a total system accuracy of approximately 99% (see Dawson et al. 2017 for system details, hardware building guide, testing and software).
We attempted to get several measurable images from each encounter with an individual whale, choosing only the best image on any given day to avoid pseudo-replication. Thus, means of measurements for individuals are calculated from the best images captured on different days; the precision of our measurements was assessed by calculating coefficients of variation (CVs). As the number of survey days was less than the minimum number of days required between sightings to detect changes in body condition in right whales (20 days; Christiansen et al. 2018), all photographs of each individual that met the quality criteria were included in averaging.
To understand the relative contribution of each measurement to overall variation within the dataset, measurement data were analysed via Principal Component Analysis (PCA). PCA is a relatively simple multivariate method which reduces the dimensionality of a dataset of correlated variables via orthogonal transformation into a new set of uncorrelated principal components (PCs), while retaining as much of the variation as possible within the original dataset. The resulting PCs are then ranked based on the amount of variation they explain in the original dataset (Jolliffe 2002; Tabachnick and Fidell 2014). Preliminary assessment of absolute measurements indicated high correlation between TL and all but one width/measurement variable. To ensure that the PCA was not dominated by variation in absolute size, but allowed quantification of variation in shape, the data analysed were normalised for body length using ratios of each measurement to total length (Miller et al. 2012). Principal component analysis on these normalised data was conducted in JMP Pro (v 14.0; SAS institute 2018), retaining all factors accounting for > 10% of the variance in the dataset. This was followed by Varimax rotation, chosen to simplify interpretation of the resulting factors (Tabachnick and Fidell 2014), using the factor analysis option within JMP’s PCA module. We used the recommendations of Comrey and Lee (1992) to help interpretation of the factor loadings (correlations between the original measured variables and the extracted factors), and hence what the factors represent.
Results
Images of sufficient quality for use in photogrammetric analysis were obtained from 136 flights. A total of 108 uniquely identifiable whales were entered into a new aerial photo-ID catalogue. Of those, 48 (~ 44%) were lactating females (observed with an accompanying calf), 10 (~ 9%) were apparent non-breeding adults and 50 (~ 46%) were subadults. Given the lack of permanence of callosities in calves, they were not entered into the catalogue. Therefore, the number of calves equates to the number of lactating females (i.e., 48). The discovery curve for the cumulative number of uniquely identifiable individuals encountered per day does not appear to reach an asymptote, suggesting that more individuals were available for identification in the study area during the period of study than were identified. The curve for lactating females is suggestive of approaching an asymptote at approximately day 9, with only slight increases up until the final sampling day (Fig. 3).
Photographs of 69 uniquely identifiable whales met the quality protocols for measurement of total length (Figs. 4 and 5). Lactating females averaged 13.74 m long (min = 11.84 m, max = 15.22 m; SD = 0.73 m, n = 32). Non-breeding adults averaged 13.17 m (min = 11.96 m, max = 14.92 m; SD = 1.07 m, n = 9). Subadults had a mean TL of 10.52 m (min = 8.82 m, max = 11.72 m; SD = 0.74 m, n = 28). Calves had a mean TL of 6.13 m (min = 5.15 m, max = 7.53 m; SD = 0.58 m, n = 29). Across all demographic groups maximum body width was at the position of 30% along TL.
Box and whisker plots for lengths of lactating female (LF; n = 32), apparent non-breeding adult (NBA; n = 9), subadult (SA; n = 28) and calf (C; n = 29) southern right whales at Port Ross, Auckland Islands, during the calving season of 2016. For each demographic, the median, upper and lower quartiles, greatest and lowest values are displayed
Frequency distributions of length (m) measurements of southern right whales in Port Ross, Auckland Islands during a three-week long expedition in 2016. Length bins are 0.5 m. N for lactating females (green, n = 32), non-breeding adults (orange, n = 9), subadults (purple, n = 28) and calves (blue, n = 29)
While measured total lengths spanned from 5.14 to 15.22 m, the main demographic groups (calves, subadults, adults) are identified by clear peaks in the distribution (Fig. 5). The low number of non-breeding (unaccompanied) adults, and a preponderance of mothers and calves reflects the importance of this habitat for parturition and lactation.
Principal component analyses were restricted to 63 of the 69 whales measured; these were individuals for which the full suite of measurements was obtained. After applying the quality criteria, 53 individuals had measurements from a single encounter; ten individuals were measured on two separate days (1–11 days apart). Additionally, we were able to obtain some measurements of 29 calves, though these were not included in PCA analysis for two reasons: (a) size (in both length and width) is highly variable at this early developmental stage, and (b) the active behaviour of calves meant that white water often prevented acquisition of the full suite of body measurements from UAV images.
Mean CVs for the ten individuals measured on more than one day (eight lactating females and two subadults; Fig. 6) ranged from 0.8% (TL) to 9.1% (width at 90% TL). Similar to results from Miller et al. (2012), as the length of the measured feature decreased, CVs generally increased (Fig. 6).
Mean coefficients of variation (CV) of total length (TL) measurements and width measurements at 10% increments along TL, as well as other measurements from whales that were measured more than once during the three-week expedition to Port Ross, Auckland Islands, in 2016. N = 10. CVs are expressed as percentages; error bars are 95% confidence intervals
Three principal components each accounted for > 10% of the total variation, accounting for 73% in total (Table 1). Following varimax rotation, these principal components represent thoracic and abdominal body width (PC1), head and fluke width (PC2) and head length (PC3). We therefore interpret PC1 as measuring whale “condition”, while PCs 2 and 3 together represent aspects of structural size. The highest loadings in PC1 (> 0.90) were body width at 30–80% of total length. No strong negative relationships were observed.
Subadults and apparent non-breeding adults displayed far more variability across both PC1 and PC2; whereas lactating females showed far less variability in both of these PCs (Fig. 7). Subadults showed proportionately shorter heads than breeding and non-breeding adults, indicated by smaller scores in PC3, suggesting a prioritisation of body growth in this life stage.
Principal component scores, following varimax rotation, from photogrammetric measurements of individual southern right whales at Port Ross in 2016. Panels are a PC1 vs PC2, b PC2 vs PC3 and c PC1 vs PC3. Points are individual whales; green points are lactating females (LF; n = 30), orange are non-breeding adults (NBA; n = 9) and purple are subadults (SA; n = 24)
Discussion
This study provides a quantitative description of the morphometrics and body condition of southern right whales at Port Ross, Auckland Islands; the primary calving ground in New Zealand waters. The data were gained using modified “prosumer” UAVs to collect high-quality photogrammetric data from whales present during the peak of a calving season.
Total length measurements of SRWs in Port Ross were similar to those of SRWs from South Africa, and from North Atlantic right whales (NARW, Eubalaena glacialis) off eastern Canada reported by Miller et al. (2012; 11.46–14.63 m for adults of both species; 4.99–8.11 m among SRW calves), as well as SRWs in Peninsula Valdés, Argentina, reported by Christiansen et al. (2019; 12.83–15.05 m for lactating females, 9.26–11.88 m for subadults and 4.31–8.22 m for calves) and lactating female SRWs from the Head of Bight, Australia, reported by Christiansen et al. (2018; 13.00–14.90 m). Whales measured in the present study, as well as those reported by Miller et al. (2012) and Christiansen et al. (2019), were generally shorter in length than North Pacific right whales (NPRW, Eubalaena japonica) reported by Omura et al. (1969; females = 11.65–16.10 m, males = 12.40–17.10 m) and Klumov (1962; females = 16.30–17.40 m, males = 17.00–17.06 m), highlighting the overall larger size of this closely related species (Kenney 2002).
While other studies have developed approaches to estimate body area indices (Burnett et al. 2018) or whole-body volumes from UAV photogrammetry (Christiansen et al. 2018, 2019), we believe that PCA retains important advantages. Its purpose is to extract a smaller number of factors which combine several correlated variables. Hence it is ideal for the task of finding which measurements combine to measure nutritional condition, and which represent structural aspects of body proportions. Our PCA results were strikingly similar to those of Miller et al. (2012), who used a twin-engine aircraft to gain photogrammetric measurements of lengths and widths of 22 female NARWs at various stages of their reproductive cycle. Factor loadings in the present study show that PC1 largely represents width of the body at 30–80% along TL, with width at 50% of TL displaying the highest loading. Miller et al. (2012) found a very similar result in their PCA, finding a slightly narrower range of body widths (40–70% of TL) that explained the greatest variability. In their study, width at 60% of TL was the single measurement (other than TL) of greatest variability. Additionally, width measurements of SRWs measured by Christiansen et al. (2019), NARWs measured by Miller et al. (2012), and the present study, found width measurements at 30% along TL to be the greatest for all demographics. Combined, these results highlight the importance of mid-length areas as key sites for energy stores in right whales.
Sites where low storage of lipid is expected (e.g., the structural elements: dimensions of the head, etc.) are not highly influential in PC1 in our study, though are loaded highly in PC2 and PC3. We note that measurements of eye to eye are essentially width measurements at 25–30% along TL and are loaded relatively highly in PC1. Given the dominance of the mid-region in PC1, and that of head measurements in PC2 and PC3, we interpret PC1 as characterising individual body condition, while PC2 and PC3 characterise structural size, especially head size.
The large variability we observed for subadults in PC2, compared to lactating and non-breeding adults, likely reflects the increased resource allocation towards structural areas in this demographic. Subadults seem to allocate energy to structural growth first, then gain condition as they approach adulthood (Charnov 1993; Kozlowski and Weiner 1997). That apparent non-breeding adults and subadults show greater variability in PC1 (i.e. body condition) compared to lactating females may also reflect the lower energetic demands on these demographics that are not energetically tied to calves.
We recognise that, while a quantitative approach was adopted in the present study to define demographics, there remains a degree of uncertainty. Due to the timing of our expedition, we do not know at what stage females arrive at Port Ross, though most have clearly gone past full term and are therefore easy to identify. For apparent non-breeding adults and subadults, the apparent difference in the proportionate length of the head to total lengths, identified by PC3, provides confidence that these demographic designations are accurate. This approach has been used to define age classes in other whale species (e.g., Whitehead and Payne 1981), though not for right whales. This provides a potential avenue for future research into the use of this metric to define right whale age class and deserves further investigation, though is beyond the scope of this study.
In Port Ross, females with calves also show a strong preference for calm waters in the lee of the shore (Rayment et al. 2015). While the UAV can fly safely in winds of up to 20 knots, quality photogrammetry requires the water surface to be calm, as surface roughness obscures or distorts the sides of the animal in the image (Christiansen et al. 2018). Also, because we flew the UAVs from our anchored research vessel, our effort was concentrated in the sheltered waters near our anchorages. Hence our sampling is probably not representative of all whales present and likely to be biased towards females and their calves. Certainly, our sample cannot represent the size structure of the greater population. Adult non-breeders (especially males) are underrepresented, as are subadults. The subadults present were highly social, suggesting that this could be the principal reason for their presence, as hypothesized by Torres et al. (2017) for the Campbell Islands. Indeed, although sexual behaviour among subadults is frequently observed, the low numbers of adults, the strong bias towards females already with calves, and reproductively immature subadults, suggests that functional mating may occur elsewhere.
With one exception, calves were between 5 and 7 m in total length, smaller than recorded in other studies. We attribute this to the period of time, relative to birth, we spent at the remote Auckland Islands. The studies cited above are also not so restricted in duration, hence calves have time to grow larger while researchers are present. The duration of lactation in SRWs is not well understood, and the length of care can vary dramatically. For example, skim-feeding by calves that are accompanying their mothers at around 6 months old has been observed in some populations (Best 2007), while mother-calf associations for some NARWs have also been documented at over 14 months (Hamilton and Cooper 2010). The lactation phase of mammalian reproduction is the most energetically demanding (Gittleman and Thompson 1988), with costs coming directly at the expense of the mother. Weaning age is largely dictated by her ability to support the growth of her calf with endogenous stores until a threshold calf length relative to maternal size is reached (Lee et al. 1991; Huang et al. 2009). The brief and intense nursing periods of mysticetes, in which up to 30% of endogenous stores can be converted into milk (Tyack 1986; Oftedal 2000; Christiansen et al. 2018), is therefore reliant on her foraging success during previous foraging seasons. Given the relatively short duration of our expedition, resulting in the lack of concurrent aerial photographs of individuals over the entire breeding season, quantification of the relative cost of lactation to mothers in this population was beyond the scope of this study, though deserves further investigation.
There is no evidence that right whales feed while on calving grounds. Individuals must therefore ensure they meet a minimum body condition before migrating from foraging grounds. The reported residency times in calving areas for various SRW populations are (mean ± SD, maximum): South Africa = 59 ± 3.9, 105 days (Best 2000); Argentina = 77 ± 36, 170 days (Rowntree et al. 2001); Australia = 70.9 ± 30, 108 days (Burnell and Bryden 1997). For NARWs, reported residency time is 50 ± 20 days (range 14–96; Right Whale Consortium 2011). Additionally, feeding resumes only when whales arrive back at their respective foraging grounds. For NARWs, Firestone et al. (2008) have estimated an average travel time of 21–24 days, while Mate et al. (2011) found SRWs travel for 9–22 days from calving to foraging grounds. Moreover, SRW calf growth rate has been estimated at 2.8 ± 0.7 cm d−1 (Best and Rüther 1992). Christiansen et al. (2019) modelled an increase in body mass of 6–7 tonnes over the first three months of life. Breeding females therefore not only rely on endogenous energy stores to meet their own energetic demands but also that of a late-stage foetus or a nursing calf for extended periods of time.
The identification of the mid and caudal regions as key areas of resource storage has been identified in other species of baleen whales, including fin (Balaenoptera physalus), sei (B. borealis), minke and humpback (Lockyer et al. 1985; Lockyer 1987; Christiansen et al. 2016a), as well as SRWs off the Head of Bight, Australia (Christiansen et al. 2018). Though patterns of lipid storage, utilisation and nutritive condition are poorly understood in right whales, external appearance can be used as a proxy for nutritive condition (Pettis et al. 2004; Miller et al. 2012). Body condition can vary among individuals of different reproductive status (Lockyer et al. 1985; Lockyer 1987; Perryman and Lynn 2002; Christiansen et al. 2018) and is a fundamental aspect of their life history (Brodie 1975; Lockyer 1981; Christiansen et al. 2018; Goldbogen and Madsen 2018).
Since the diet of right whales consists largely of copepods and small krill (Tormosov et al. 1998), typically the dominant components of oceanic zooplankton and species that have been shown to be strongly impacted by ocean warming (Hayes et al. 2005; Nicol et al. 2008), SRWs feed closer to the base of the Southern Ocean food web than any other megavertebrate. Baleen whales also play a key role in ecosystem functioning, aiding in the transfer of nutrients from highly productive high-latitude foraging grounds to less productive lower-latitude calving areas (Roman et al. 2014). Thus, by tracking changing body condition of individuals through time we can gain a direct indicator of population health, as well as an indirect indicator of the health of the ecosystems they inhabit.
References
Aguilar A, Borrell A (1990) Patterns of lipid content and stratification in the blubber of fin whales (Balaenoptera physalus). J Mammal 71:544–554. https://doi.org/10.2307/1381793
Bannister JL (2010). Population trend in right whales off southern Australia. Rep Int Whal Comm Spec Issue SC/S11/RW1, 1–8. https://archive.iwc.int/pages/search.php?search=%21collection73&k=. Accessed 11 Aug 2021
Beck GG, Smith TG, Hammill MO (1993) Evaluation of body condition in the Northwest Atlantic harp seal (Phoca groenlandica). Can J Fish Aquat Sci 50:1372–1381. https://doi.org/10.1139/f93-157
Best PB (1994) Seasonality of reproduction and the length of gestation in southern right whales Eubalaena australis. J Zool Soc Lond 232:175–189. https://doi.org/10.1111/j.1469-7998.1994.tb01567.x
Best PB (2000) Coastal distribution, movements and site fidelity of right whales Eubalaena australis off South Africa, 1969–1998. S Afr J Mar Sci 22:43–55. https://doi.org/10.2989/025776100784125618
Best PB (2007) Whales and dolphins of the southern African subregion. Cambridge University Press, Cape Town, South Africa
Best PB, Rüther H (1992) Aerial photogrammetry of southern right whale, Eubalaena australis. J Zool Soc Lond 228:595–614. https://doi.org/10.1111/j.1469-7998.1992.tb04458.x
Bonnet X, Lourdais O, Shine R, Naulleau G (2002) Reproduction in a typical capital breeder: costs, currencies, and complications in the aspic viper. Ecology 83:2124–2135. https://doi.org/10.1890/0012-9658(2002)083[2124:RIATCB]2.0.CO;2
Bradford AL, Weller DW, Punt AE, Ivashchenko YV, Burdin AM, VanBlaricom GR, Brownell RL (2012) Leaner leviathans: body condition variation in a critically endangered whale population. J Mammal 93:251–266. https://doi.org/10.1644/11-MAMM-A-091.1
Brandão A, Butterworth DS, Müller A, Best PB (2012) Application of a photoidentification based assessment model to southern right whales in South African waters. Rep Int Whal Comm Spec Issue SC/64/BRG2, 1–15. https://archive.iwc.int/pages/search.php?search=%21collection73&k=. Accessed 11 Aug 2021
Brodie PF (1975) Cetacean energetics, an overview of intraspecific size variation. Ecology 56(1):152–161. https://doi.org/10.2307/1935307
Burnell SR, Bryden MM (1997) Coastal residence periods and reproductive timing in southern right whales. J Zool 241:613–621. https://doi.org/10.1111/j.1469-7998.1997.tb05736.x
Burnett JD, Lemos L, Barlow D, Wing MG, Chandler T, Torres LG (2018) Estimating morphometric attributes of baleen whales with photogrammetry from small UASs: A case study with blue and gray whales. Mar Mamm Sci 35(1):108–139. https://doi.org/10.1111/mms.12527
Carroll EL, Patenaude N, Childerhouse S, Kraus SD, Fewster RM, Baker CS (2011) Abundance of the New Zealand subantarctic southern right whale population estimated from photo-identification and genotype mark-recapture. Mar Biol 158(11):2565–2575. https://doi.org/10.1007/s00227-011-1757-9
Carroll EL, Childerhouse SJ, Fewster RM, Patenaude NJ, Steel D, Dunshea G, Boren L, Baker CS (2013) Accounting for female reproductive cycles in a superpopulation capture-recapture framework. Ecol Appl 23(7):1677–1690. https://doi.org/10.1890/12-1657.1
Carroll EL, Rayment WJ, Alexander AM, Baker CS, Patenaude NJ, Steel D, Constantine R, Cole R, Boren LJ, Childerhouse S (2014) Reestablishment of former wintering grounds by New Zealand southern right whales. Mar Mamm Sci 30(1):206–220. https://doi.org/10.1111/mms.12031
Carroll EL, Baker CS, Watson M, Alderman R, Bannister J, Gaggiotti OE, Gröcke DR, Patenaude N, Harcourt R (2015) Migratory traditions shape the genetic structure of southern right whales around Australia and New Zealand. Sci Rep UK 5:16182. https://doi.org/10.1038/srep16182
Carroll EL, Fewster RM, Childerhouse S, Patenaude N, Boren LJ, Baker CS (2016) First direct evidence for natal wintering ground fidelity and estimate of juvenile survival in the New Zealand southern right whale Eubalaena australis. PLoS One 11(1):e0146590. https://doi.org/10.1371/journal.pone.0146590
Charnov EL (1993) Life history invariants. Oxford University Press, Oxford
Childerhouse S, Double M, Gales N (2010) Satellite tracking of southern right whales (Eubalaena australis) at the Auckland Islands, New Zealand. Rep Int Whal Comm 5. SC/62/BRG19
Christiansen F, Vikingsson GA, Rasmussen MH, Lusseau D (2013) Minke whales maximise energy storage on their feeding grounds. J Exp Biol 216:427–436. https://doi.org/10.1242/jeb.074518
Christiansen F, Vikingsson GA, Rasmussen MH, Lusseau D (2014) Female body condition affects foetal growth in a capital breeding mysticete. Funct Ecol 28:579–588. https://doi.org/10.1111/1365-2435.12200
Christiansen F, Dujon AM, Sprogis KR, Arnould JPY, Bejder L (2016a) Noninvasive unmanned aerial vehicle provides estimates of the energetic cost of reproduction in humpback whales. Ecosphere 7:e01468. https://doi.org/10.1002/ecs2.1468
Christiansen F, Rojano-Doñate L, Madsen PT, Bejder L (2016b) Noise levels of multi-rotor unmanned aerial vehicles with implications for potential underwater impacts on marine mammals. Front Mar Sci 3:227. https://doi.org/10.3389/fmars.2016.00277
Christiansen F, Vivier F, Charlton C, Ward R, Amerson A, Burnell S, Bejder L (2018) Maternal body size and condition determine calf growth rates in southern right whales. Mar Ecol Prog Ser 592:267–281. https://doi.org/10.3354/meps12522
Christiansen F, Marioni S, Moore M, Di Martino M, Ricciardi M, Warick HA, Irschick DJ, Gutierrez R, Uhart MM (2019) Estimating body mass of free-living whales using aerial photogrammetry and 3D volumetrics. Methods Ecol Evol 12:2034–2044. https://doi.org/10.1111/2041-210X.13298
Christiansen F, Dawson SM, Durban JW, Fearnbach H, Miller CA, Bejder L, Uhart M, Sironi M, Corkeron P, Rayment WJ, Leunissen E, Haria E, Ward R, Warick HA, Kerr I, Lynn MS, Pettis HM, Moore MJ (2020) Population comparison of right whale body condition reveals poor state of the North Atlantic right whale. Mar Ecol Prog Ser 640:1–16. https://doi.org/10.3354/meps13299
Clutton-Brock T, Sheldon BC (2010) Individuals and populations: the role of long-term, individual-based studies of animals in ecology and evolutionary biology. Trends Ecol Evol 25:562–573. https://doi.org/10.1016/j.tree.2010.08.002
Comrey AL, Lee HB (1992) A first course in factor analysis. Lawrence Eribaum Associates, Hillsdale, NJ
Cooke JG, Rowntree VJ, Payne RS (2003) Analysis of inter-annual variation in reproductive success of South Atlantic right whales (Eubalaena australis) from photo-identifications of calving females observed off Peninsula Valdés, Argentina, during 1971 - 2000. Rep Int Whal Comm Spec Issue SC/55/O23, 1–16. https://archive.iwc.int/pages/search.php?search=%21collection73&k=. Accessed 11 Aug 2021
Corkeron PJ, Hamilton P, Bannister J, Best P, Charlton C, Groch KR, Finfdlay K, Rowntree V, Vermeulen E, Pace RM III (2018) The recovery of North Atlantic right whales, Eubalaena glacialis, has been constrained by human-caused mortality. R Soc Open Sci 5(11):180892–180912. https://doi.org/10.1098/rsos.180892
Darwin CR (1859) On the origin of species by means of natural selection, or the preservation of favoured races in the struggle for life. John Murray, London
Davidson AR, Rayment W, Dawson SM, Webster T, Slooten E (2017) Estimated calving interval for the New Zealand southern right whale (Eubalaena australis). N Z J Mar Freshw Res 52(3):1–11. https://doi.org/10.1080/00288330.2017.1397034
Dawson SM, Bowman MH, Leunissen E, Sirguey P (2017) Inexpensive aerial photogrammetry of whales and large marine mammals. Front Mar Sci 4:e01468-e1477. https://doi.org/10.3389/fmars.2017.00366
Dawson SM, Chessum CJ, Hunt PJ, Slooten E (1995) An inexpensive, stereophotoraphic technique to measure sperm whales from small boats. Rep Int Whal Comm 45(43):1–436. https://archive.iwc.int/pages/search.php?search=%21collection73&k=. Accessed 11 Aug 2021
Durban JW, Fearnbach W, Barrett-Lennard LG, Perryman WL, Leroi DJ (2015) Photogrammetry of killer whales using a small hexacopter launched at sea. J Unmanned Veh Syst 3(3):131–135. https://doi.org/10.1139/juvs-2015-0020
Durban JW, Moore MJ, Chiang G, Hickmott LS, Bocconcelli A, Howes G, Bahamonde PA, Perryman WL, Leroi DJ (2016) Photogrammetry of blue whales with an unmanned hexacopter. Mar Mamm Sci 32:1510–1515. https://doi.org/10.1111/mms.12328
Festa-Bianchet M, Gaillard JM, Jorgenson JT (1998) Mass- and density-dependent reproductive success and reproductive costs in a capital breeder. Am Nat 152:367–379. https://doi.org/10.1086/286175
Fiori L, Doshi A, Martinez E, Orams MB, Bollard-Breen B (2017) The use of unmanned aerial systems in marine mammal research. Remote Sens 9(6):543–613. https://doi.org/10.3390/rs9060543
Firestone J, Lyons SB, Wang C, Corbett JJ (2008) Statistical modelling of North Atlantic right whale migration along the mid-Atlantic region of the eastern seaboard of the United States. Biol Conserv 141:221–232. https://doi.org/10.1016/j.biocon.2007.09.024
Frederich B, Adriaens D, Vandewalle P (2008) Ontogenetic shape changes in Pomacentridae (Teleostei Perciformes) and their relationships with feeding strategies: a geometric morphometric approach. Biol J Linn Soc 95:92–105. https://doi.org/10.1111/j.1095-8312.2008.01003.x
Gaillard JM, Festa-Bianchet M, Yoccoz NG, Loison A, Toïgo C (2000) Temporal variation in fitness components and population dynamics of large herbivores. Ann Rev Ecol Syst 31:367–393. https://doi.org/10.1146/annurev.ecolsys.31.1.367
Gaskin DE (1964) Return of the Southern Right Whale (Eubalaena australis) to New Zealand waters 1963. Tuatara 12:2–5
Gittleman JL, Thompson SD (1988) Energy allocation in mammalian reproduction. Am Zool 28:863–875. https://doi.org/10.1093/icb/28.3.863
Goldbogen JA, Madsen PT (2018) The evolution of foraging capacity and gigantism in cetaceans. J Exp Biol. https://doi.org/10.1242/jeb.166033
Gordon JCD (1990) A simple photographic technique for measuring the length of whales from boats at sea. Rep Int Whal Comm 40:581–587. https://archive.iwc.int/pages/search.php?search=%21collection73&k=. Accessed 11 Aug 2021
Growcott A, Sirguey P, Dawson S (2012) Development and assessment of a digital stereo photogrammetric system to measure cetaceans at sea. Photogramm Eng Remote Sens 78(3):237–246. https://doi.org/10.14358/PERS.78.3.237
Hamilton PK, Cooper LA (2010) Changes in North Atlantic right whale (Eubalaena glacialis) cow-calf association times and use of the calving ground: 1993–2005. Mar Mamm Sci 26:896–916. https://doi.org/10.1111/j.1748-7692.2010.00378.x
Hayes JP, Shonkwiler JS (2001) Morphometric indicators of body condition: worthwhile or wishful thinking? In: Speakman JR (ed) Body composition analysis of animals: a handbook of non-destructive methods. Cambridge University Press, Cambridge, pp 8–38
Hayes GC, Richardson AJ, Robinson C (2005) Climate change and marine plankton. Trends Ecol Evol 20(6):337–344. https://doi.org/10.1016/j.tree.2005.03.004
Horton TW, Hauser N, Cassel S, Klaus KF, Fettermann T, Key N (2019) Doctor drone: non-invasive measurement of humpback whale vital signs using unoccupied aerial system infrared thermography. Front Mar Sci 6:53. https://doi.org/10.3389/fmars.2019.00466
Huang SL, Chou LS, Ni IH (2009) Comparable length at weaning in cetaceans. Mar Mamm Sci 25:875–887. https://doi.org/10.1111/j.1748-7692.2009.00288.x
Ichii T, Shinohara N, Fujise Y, Nishiwaki S, Matsuoka K (1998) Interannual changes in body fat condition index of minke whales in the Antarctic. Mar Ecol Prog Ser 175:1–12. https://doi.org/10.3354/meps175001
Jackson JA, Carroll EJ, Smith TD, Zerbini AN, Patenaude NJ, Baker CS (2016) An integrated approach to historical population assessment of the great whales: case of the New Zealand southern right whales. R Soc Open Sci 3:150669. https://doi.org/10.1098/rsos.150669
Jackson J, Carroll EL, Smith TD, Patenaude N, Baker CS (2009) Taking stock: the historical demography of the New Zealand right whale. Unpub Report, 1–72
Jaquet N (2006) A simple photogrammetric technique to measure sperm whales at sea. Mar Mamm Sci 22(4):862–879. https://doi.org/10.1111/j.1748-7692.2006.00060.x
Jolliffe IT (2002) Principal component analysis. Springer-Verlag, New York Inc, USA
Kenney RD (2002) North Atlantic, North Pacific, and Southern Right Whales. In: Perrin WF, Würsig B, Thewissen JGM (eds) Encyclopedia of marine mammals. Academic Press, USA, pp 306–813
Klumov SK (1962) Gladkiye (Yaponskiye) kity Tikhovo Okeana (Russian). Trudy Inst Okeanol 58:202–297
Kozlowski J, Weiner J (1997) Interspecific allometries are by-products of body size optimization. Am Nat 149:352–380. https://doi.org/10.1086/285994
Kraus SD, Kenney RD, Mayo CA, McLellan WA, Moore M, Nowacek DP (2016) Recent scientific publications cast doubt on north atlantic right whale future. Front Mar Sci 3:1401–1403. https://doi.org/10.3389/fmars.2016.00137
Leaper R, Cooke J, Trathan P, Reid K, Rowntree V, Payne R (2006) Global climate drives southern right whale (Eubalaena australis) population dynamics. Biol Lett 2(2):289–292. https://doi.org/10.1098/rsbl.2005.0431
Lee PC, Majluf P, Gordon IJ (1991) Growth, weaning and maternal investment from a comparative perspective. J Zool 225:99–114. https://doi.org/10.1111/j.1469-7998.1991.tb03804.x
Lemos LS, Burnett JD, Chandler TE, Sumich JL, Torres LG (2020) Intra- and inter-annual variation in gray whale body condition on a foraging ground. Ecosphere 11(4):3094. https://doi.org/10.1002/ecs2.3094
Linchant J, Lisein J, Semeki J, Lejeune P, Vermeulen C (2015) Are unmanned aircraft systems (UASs) the future of wildlife monitoring? A review of accomplishments and challenges. Mamm Rev 45(4):239–252. https://doi.org/10.1111/mam.12046
Lockyer C (1981) Estimation of the energy costs of growth, maintenance and reproduction in the female Minke whale, (Balaenoptera acutorostrata), from the southern hemisphere. Rep Int Whal Comm 31:337–343
Lockyer C (1986) Body fat condition in northeast Atlantic fin whales, Balaenoptera physalus, and its relationship with reproduction and food resource. Can J Fish Aquat Sci 43:142–147. https://doi.org/10.1139/f86-015
Lockyer C (1987) The relationship between body fat, food resource and reproductive energy costs in North Atlantic fin whales (Balaenoptera physalus). Symp Zool Soc Lond 57:343–361. https://doi.org/10.1139/f86-015
Lockyer C (2007) All creatures great and smaller: a study in cetacean life history energetics. J Mar Biol Assoc UK 87(4):1035–1045. https://doi.org/10.1017/S0025315407054720
Lockyer C, McConnell LC, Waters TD (1985) Body condition in terms of anatomical and biochemical assessment of body fat in North Atlantic fin and sei whales. Can J Zool 63:2328–2338. https://doi.org/10.1139/z85-345
Mate BR, Best PB, Lagerquist BA, Winsor MH (2011) Coastal, offshore, and migratory movements of South African right whales revealed by satellite telemetry. Mar Mamm Sci 27:455–476. https://doi.org/10.1111/j.1748-7692.2010.00412.x
Miller CA, Reeb D, Best PB, Knowlton AR, Brown MW, Moore MJ (2011) Blubber thickness in right whales Eubalaena glacialis and Eubalaena australis related with reproduction, life history status and prey abundance. Mar Ecol Prog Ser 438:267–283. https://doi.org/10.3354/meps09174
Miller CA, Best PB, Perryman WL, Baumgartner MF, Moore MJ (2012) Body shape changes associated with reproductive status, nutritive condition and growth in right whales Eubalaena glacialis and E. australis. Mar Ecol Prog Ser 459:135–156. https://doi.org/10.3354/meps09675
Niæss A, Haug T, Nilssen E (1998) Seasonal variation in body condition and muscular lipid contents in Northeast Atlantic minke whale Balaenoptera acutorostrata. Sarsia 83(3):211–218. https://doi.org/10.1080/00364827.1998.10413682
Nicol S, Worby A, Leaper R (2008) Changes in the Antarctic sea ice ecosystem. Mar Freshw Res 59:361–382. https://doi.org/10.1071/MF07161
Oftedal OT (2000) Use of maternal reserves as a lactation strategy in large mammals. Proc Nutr Soc 59:99–106. https://doi.org/10.1017/S0029665100000124
Omura H, Oshuni S, Nemoto T, Nasu K, Kasuya T (1969) Black right whales in the North Pacific. Sci Rep Whal Res Inst 21:1–78
Patenaude NJ, Baker CS, Gales NJ (1998) Observations of southern right whales on New Zealand’s subantarctic wintering grounds. Mar Mamm Sci 14(2):350–355. https://doi.org/10.1111/j.1748-7692.1998.tb00726.x
Payne R, Brazier O, Dorsey EM, Perkins JS, Rowntree VJ, Titus A (1983) External features in southern right whales (Eubalaena australis) and their use in identifying individuals. In: Payne R (ed) Communication and behaviour of whales. AAAS Selected Symposium 76. Westview Press, Boulder Co, pp 371–445
Perryman WL, Lynn MS (2002) Evaluation of nutritive condition and reproductive status of migrating gray whales (Eschrichtius robustus) based on analysis of photogrammetric data. J Cetacean Res Manag 4:155–164
Pettis HM, Rolland RM, Hamilton PK, Brault S, Knowlton AR, Kraus SD (2004) Visual health assessment of North Atlantic right whales (Eubalaena glacialis) using photographs. Can J Zool 82:8–19. https://doi.org/10.1139/z03-207
Pirzl R, Patenaude NJ, Burnell S, Bannister J (2009) Movements of southern right whales (Eubalaena australis) between Australian and subantarctic New Zealand populations. Mar Mamm Sci 25(2):455–461. https://doi.org/10.1111/j.1748-7692.2008.00276.x
Ratnaswamy MJ, Winn HE (1993) Photogrammetric estimates of allometry and calf production in fin whales, Balaenoptera physalus. J Mammal 74:323–330. https://doi.org/10.2307/1382387
Rayment W, Davidson A, Dawson S, Slooten E, Webster T (2012) Distribution of southern right whales on the Auckland Islands calving grounds. N Z J Mar Freshw Res 46(3):431–436. https://doi.org/10.1080/00288330.2012.697072
Rayment W, Dawson S, Webster T (2015) Breeding status affects fine-scale habitat selection of southern right whales on their wintering grounds. J Biogeogr 42:463–474. https://doi.org/10.1111/jbi.12443
Rayment W, Webster T, Brough T, Jowett T, Dawson S (2017) Seen or heard? A comparison of visual and acoustic autonomous monitoring methods for investigating temporal variation in occurrence of southern right whales. Mar Biol. https://doi.org/10.1007/s00227-017-3264-0
Right Whale Consortium (2011) North Atlantic right whale consortium identification database v. 02/25/2011. New England Aquarium, Boston, MA
Roman J, Estes JA, Morissette L, Smith C, Costa D, McCarthy J, Nation JB, Nicol S, Pershing A, Smetacek V (2014) Whales as marine ecosystem engineers. Front Ecol Environ 12:377–385. https://doi.org/10.1890/130220
Rowntree VJ, Payne RS, Schell DM (2001) Changing patterns of habitat use by southern right whales (Eubalaena australis) on their nursery ground at Peninsula Valdes, Argentina, and in their long-range movements. J Cetacean Res Manag 2 Spec Issue 133–143. https://journal.iwc.int/index.php/jcrm/article/view/298. Accessed 24 Mar 2020
Schaeff CM, Best PB, Rowntree VJ, Payne R, Jarvis C, Portway VA (1999) Dorsal skin color patterns among southern right whales (Eubalaena australis): genetic basis and evolutionary significance. J Hered. https://doi.org/10.1093/jhered/90.4.464
Stevenson RD, Woods A Jr (2006) Condition indices for conservation: new uses for evolving tools. Integr Comp Biol 46(6):1169–1190. https://doi.org/10.1093/icb/icl052
Tabachnick BG, Fidell LS (2014) Using multivariate statistics, 6th edn. Pearson Education, p 1055
Tormosov D, Mikhaliev Y, Best PB, Zemsky V, Sekiguchi K, Brownell R (1998) Soviet catches of southern right whales Eubalaena australis, 1951–1971. Biological data and conservation implications. Biol Conserv 86:185–197. https://doi.org/10.1016/S0006-3207(98)00008-1
Torres L, Rayment W, Olavarría C, Thompson DR, Graham B, Baker CS, Patenaude N, Bury SJ, Boren L, Parker G, Carroll EL (2017) Demography and ecology of southern right whales Eubalaena australis wintering at sub-Antarctic Campbell Island. New Zealand Polar Biol 40(1):95–106. https://doi.org/10.1007/s00300-016-1926-x
Tyack P (1986) Population biology, social behavior and communication in whales and dolphins. Trends Ecol Evol 1:144–150. https://doi.org/10.1016/0169-5347(86)90042-X
Vikingsson GA (1990) Energetic studies on Fin and Sei whales caught off Iceland. Rep Int Whal Comm 40:365–373
Vikingsson GA (1995) Body condition of fin whales during summer off Iceland. In: Blix AS, Walløe L, Ulltang Ø (eds) Whales, seals, fish and man. Elsevier Science, Amsterdam, The Netherlands, pp 361–369
Webster TA, Dawson SM, Rayment W, Parks SE, Van Parijs S (2016) Quantitative analysis of the acoustic repertoire of southern right whales in New Zealand. J Acoust Soc Am 140(1):322–333. https://doi.org/10.1121/1.4955066
Webster TA, Van Parijs SM, Rayment WJ, Dawson S (2019) Temporal variation in the vocal behaviour of southern right whales in the Auckland Islands. New Zealand Roy Soc Open Sci 6(3):181487–181515. https://doi.org/10.1098/rsos.181487
Whitehead H, Payne R (1981) New Techniques for Measuring Whales from the Air. US Mar Mamm Comm Rep MMC-76/22. Washington, DC, pp 36
Zelditch ML, Swiderski DL, Sheets HD, Fink WL (2004) Geometric morphometrics for biologists: a primer. Elsevier Academic Press, San Diego
Acknowledgements
We thank the skipper and crew of the RV Polaris II. Expedition members Liz Slooten, Bob Wallace, Steve Bradley and Julian Tyne helped with data collection. Hamish Bowman, Eva Leunissen and Pascal Sirguey helped develop our UAV photogrammetry system. Tim Jowett provided statistical advice.
Funding
This research was principally funded by the New Zealand Antarctic Research Institute (NZARI), with additional support from the University of Otago and the New Zealand Whale and Dolphin Trust (NZWDT). It was carried out under permit 50094-MAR from the Department of Conservation New Zealand.
Author information
Authors and Affiliations
Contributions
DJ: conceptualization; data curation; formal analysis; investigation; methodology; project administration; visualization; writing-original draft; writing-review and editing. WR: conceptualization; data curation; methodology; project administration; resources; supervision; validation; writing-review and editing. SD: conceptualization; data curation; formal analysis; funding acquisition; methodology; project administration; resources; supervision; validation; writing-review and editing.
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Handling editors: Leszek Karczmarski and Stephen C.Y. Chan.
This article is a contribution to the special issue on “Individual Identification and Photographic Techniques in Mammalian Ecological and Behavioural Research – Part 2: Field Studies and Applications” — Editors: Leszek Karczmarski, Stephen C.Y. Chan, Scott Y.S. Chui and Elissa Z. Cameron.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Johnston, D.R., Rayment, W. & Dawson, S.M. Morphometrics and body condition of southern right whales on the calving grounds at Port Ross, Auckland Islands. Mamm Biol 102, 1525–1536 (2022). https://doi.org/10.1007/s42991-021-00175-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42991-021-00175-6