Abstract
Serotypes and antimicrobial susceptibility of Salmonella’s isolates from broiler production chain were determined. A total of 239 isolated strains from chicken, carcasses, breeding environments, and slaughter was analyzed by disk diffusion test, in the period of 2009 to 2010. For antibiotics with a high number of resistant strains, the minimal inhibitory concentration (MIC) was performed. We identified 24 serotypes, being the most frequent, Minnesota (31.4%) and Infantis (22.6%). The highest percentages of resistance were obtained for sulfonamide (42.7%), followed by tetracycline with 37.6% and amoxicillin with 27.6%. From the total, 32 resistance profiles were identified, being 60.7% of the strains were resistant to at least one antibiotic. Of these, 31.7% of the isolates showed multidrug resistance profiles belonging to serovar Minnesota, Saintpaul, and S. enterica. The highest resistance was found in isolates from slaughterhouse (66.9%) and aviary (58.7%). A large number of strains showed MIC above the maximum tested concentration for the antibiotics amoxicillin and sulfamethoxazole. The high number of Salmonella’s resistant strains indicates the need for prudent use of these drugs in poultry production in order to reduce the occurrence and spread of antibiotic resistance profiles, and the risk that multiresistant strains isolated from broilers may pose a risk to human health.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Salmonellosis is a public health problem because it is a common cause of human gastroenteritis and the major cause of foodborne hospitalizations worldwide [1, 2]. It is estimated that Salmonella causes 93.8 million human infections and 155,000 deaths annually worldwide [3].
In Brazil, from 1999 to 2008, 6602 outbreaks of foodborne illness were reported, and Salmonella spp. were responsible for 43% of outbreaks where the etiologic agent was identified [4]. Although the genus is composed of multiple serotypes, some predominates in the epidemiology of human disease, such as, Typhimurium, Enteritidis, Newport, Heidelberg, and Infantis [5]. Salmonella is typically found in poultry products, and chicken meat has been an important vehicle for foodborne illnesses [6].
The indiscriminate use of antibiotics, inserted in the production process of food of animal origin, can act as a selection pressure for some Salmonella serovars and their antimicrobial resistance. Thus, in addition to constant monitoring and the identification of the serovars of this pathogen along the production chain, it is important to evaluate the resistance to antibiotics, in order to verify the occurrence and dissemination of resistant strains in the environment and in the animal, especially those with a multiresistant profile [7].
The progressive increase of Salmonella strains isolated from humans that are resistant to multiple antimicrobials has been associated with the widespread use of these agents in the production of animals for human consumption [8]. Thus, resistant Salmonella can be transmitted from animals to humans via food chain, associated mainly to the consumption of chicken meat and eggs [9, 10].
It is known that poultry farm widely uses antibiotics as a prophylactic measure and/or growth promoter. Therefore, it is suspected that multiresistant bacteria found in human is at least in part, of animal origin, and their genes may have acquired resistance during production, before being transmitted to humans through food consumption [11]. Although Salmonella infections are often asymptomatic in chickens, the colonization of these pathogens in poultry poses a significant risk to human health by eating contaminated meat, especially when it comes to contamination with microorganisms that exhibit resistance profiles to antibiotics [12].
The production and distribution of food in a globalized form rapidly spreads pathogens. This situation combined to the challenge of multidrug resistance creates new barriers for control and prevention of Salmonella infection in human and veterinary medicines with serious implications for public health. With this, we aimed to evaluate the spread of the serotypes of Salmonella spp. at various stages of an industrial poultry production chain, from the farm to the slaughter stage, and also to check the antibiotic resistance profiles of the obtained isolates.
Material and methods
The study comprised 239 strains of Salmonella previously isolated in two slaughter broiler plants, with full production cycle and integration system in the states of São Paulo and Mato Grosso do Sul, Brazil, during the period of 2009 to 2010. The slaughter plants were inspected by the federal inspection service, from the Brazilian Ministry of Agriculture, and the chicken meat produced was destined to retail sale in Brazil and other countries (mainly Europe and Asia). The samples were collected during all the production cycle, since broiler farms and breeder hens up to the industrialized final product are ready to trade, including environmental samples.
Sampling was conducted in aviaries with the aid of swabs and Prope drag when the chickens were approximately 30 days.
Samples were collected at the required points of the slaughterhouse according to Pathogen Reduction Program—PRP (Programa de Redução de Patógenos) [13]—added to other points that presents a higher isolation frequency of Salmonella during the routine of the studied industries.
The antigenic typifying was performed by the Fundação Instituto Oswaldo Cruz in the state of Rio de Janeiro (FIOCRUZ). This antigen characterization was done based on serological classification of Kauffmann-White and Le Minor, with representation in accordance with the criteria of Grimont and Weill [14].
The antibiotic susceptibility test was performed at the Laboratório de Biotecnologia Animal Aplicada, Universidade Federal de Uberlândia (LABIO/UFU). The antibiotic susceptibility of the isolates was evaluated by the disk diffusion technique using the protocol recommended by the Clinical and Laboratory Standards Institute (CLSI) [15].
The criterion of antibiotic choice was based on the use of these drugs in human and veterinary medicines and the resistance occurrence in both areas. Antibiotics and concentrations in micrograms tested were amoxicillin (10 μg) (β-lactam/penicillin), norfloxacin (10 μg) (fluoroquinolone), neomycin (30 μg) (aminoglycoside), gentamicin (10 μg) (aminoglycoside), trimethoprim (5 μg) (pyrimidine), ceftazidime (30 μg) (β-lactam/cephalosporin), chloramphenicol (30 μg) (fenicol), imipenem (10 μg) (β-lactam/carbapenem), tetracycline (30 μg) (tetracycline), and sulfonamide (300 μg) (sulfonamide) (LaborClin®). The zones of inhibition were measured and classified as intermediate, sensitive, or resistant according to the CLSI recommendations [15]. Salmonella isolates that were resistant to more than two antibiotic’s classes were defined as multiresistant [16].
Salmonella isolates that presented resistance or intermediate resistance to sulfonamide, tetracycline, and amoxicillin were submitted to the minimum inhibitory concentration (MIC) test by the use of Ɛ test (BioMérieux Brazil®) antibiotic gradient strips, following the manufacturer’s recommendations to perform the technique and interpret the results. In this study, the MIC technique for antibiotics sulfamethoxazole (SX, 8–1024 μg/mL) (equivalent to sulfonamide), tetracycline (TC, 1–256 μg/mL), and amoxicillin (AC, from 0.75 to 256 μg/mL) was performed. The interpretation criteria of MIC were tetracycline (≤ 4 μg/mL: susceptible; ≥ 16 μg/mL: resistant), sulfamethoxazole (≤ 256 μg/mL: susceptible; ≥ 512 μg/mL: resistant), and amoxicillin (≤ 8 μg/mL: susceptible; ≥ 32 μg/mL: resistant). Escherichia coli ATCC 25922 was used as a control strain in both techniques.
Results
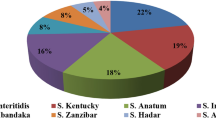
Twenty-four serovars were identified among the 239 isolates of Salmonella originated from industrial plants, with S. Minnesota (31.4%) and S. Infantis (22.6%) being the most frequent (Table 1), presenting a higher prevalence in the slaughter environment, compared to the other serovars. The frequency and distribution of the serovars at the slaughterhouse did not differ according to the source of isolation of the strains, being carcass, meat cuts, or the environment of slaughter.
Considering all serotypes, the highest percentages of resistance were to sulfonamide antimicrobials with 102 strains (42.7%), followed by tetracycline with 90 (37.6%) and amoxicillin with 66 (27.6%) (Table 2) belonging, respectively, to the classes of sulfonamides, tetracyclines, and beta lactam benzylpenicillanic, which was banned in Brazil as growth promoters [17]. All isolates were susceptible to imipenem.
A high prevalence of sulfonamide-resistant isolates was found in the poultry broiler aviary (42.5%) and slaughterhouse (48.8%), the same occurring for tetracycline with 36.2% and 45.7%, respectively. As for amoxicillin, a high prevalence of resistant isolates from slaughterhouse (30.7%) and feed factory (33.3%) was found (Table 2).
Just one Salmonella isolate (0.4%) presented resistance to chloramphenicol (anfenicol). A total of 32 resistance profiles among the Salmonella isolates was identified, being 94 (39.3%) isolates susceptible to all tested antibiotics, while 145 (60.7%) presented resistance to at least one antibiotic. Considering only the 145 isolates that presented resistance, 46 (31.7%) were resistant to three or more classes of antibiotics and they were identified as belonging to serovars S. Minnesota, S. Saintpaul, and S. enterica nontypeable. The profiles had predominant resistance to tetracycline and sulfonamide (33 isolates), followed by intermediate resistance only for amoxicillin (25 isolates) (Table 3).
Comparing the resistance profiles and serotypes, the serovar Minnesota had the highest percentage of resistant strains (92.0%) (≥ 1 class of antibiotics), and 57.3% of isolates were multiresistant, highlighting a strain which showed resistance to six antibiotics (Tables 3 and 4).
Considering the origin of the samples, we observed that the slaughterhouse’s isolates had high resistance rate to at least one antibiotic (66.9%), followed by broiler aviary (58.7%), and feed factory (40.0%). Most of the multiresistant isolates was originated from the slaughterhouse (22.8%), followed by the broiler aviary (20.0%), and the feed factory (3.3%) (Table 5).
In order to check the MIC, the antibiotics selected were sulfamethoxazole (equivalent to sulfonamide), tetracycline, and amoxicillin, for which the highest percentages of isolates classified as resistant or intermediate resistance by disk diffusion test.
In this study, several isolates previously classified as resistant or with intermediate resistance by the diffusion test with disks were reclassified as sensitive by the MIC technique. For tetracycline, from the 90 Salmonella isolates previously classified as resistant in the disk diffusion test, eight were reclassified as sensitive. Among the 76 isolates that were resistant in both tests, most had a MIC of 96 μg/mL.
Considering the results of MICs for sulfamethoxazole, 24 of the 102 isolates were susceptible, while the remaining 78 strains showed MIC greater than the maximum concentration of the antibiotic tested (> 1024 μg/mL). As for amoxicillin, of the 66 isolates evaluated, 40 were classified as sensitive, with a large number of isolates with an MIC > 256 μg/mL.
Discussion
S. Enteritidis and S. Typhimurium were, for many years, the most frequently isolated serovars in poultry’s origin products [18]; however, in this study, the Minnesota and Infantis serovars were the most isolated along the production chain (Table 1). Probably, the intense control in poultry production focused in Enteritidis and Typhimurium [19] turned out to select other serotypes by a mechanism of competitive exclusion, which could explain the high frequency found for S. Infantis and S. Minnesota.
The high number of S. Infantis isolates resembles a study conducted in the EU, which proved that this serovar was more common in the environment of broiler chickens and in carcasses [20, 21]. Moreover, S. Infantis is among the serovars most involved in cases of human salmonellosis in Brazil [22] being considered important in human health as well in Japan [23]. This serovar is recognized for its pathogenic potential, which triggers gastroenteric disease, which can lead to septicemic infection in humans, especially in cases of severe infections in children [24]. A study conducted in Japan by Noda et al. [24] revealed that the genotypes of S. Infantis isolated from humans were similar to isolates from chicken meat, suggesting that cases of human infection with this serovar originated from the consumption and handling of chicken meat.
The high frequency of isolates resistant to sulfonamide and tetracycline (Table 2) can be explained by the fact that these antibiotics are usually administered in the Brazilian poultry industry as therapeutic agents for bacterial infections [25]. In another study, in Thailand and Cambodia, the majority of the Thai isolates were resistant to ampicillin (72.4%), whereas most Cambodian isolates were resistant to sulfamethoxazole (71.0%) [26]. The only resistant isolate to chloramphenicol was due, probably, to the fact that in Brazil, Normative Instruction no. 9 [27] predicts the ban of manufacturing, handling, fractionation, marketing, import, and use of the chloramphenicol’s active ingredient for veterinary use. The higher number of resistant strains isolated from slaughterhouse and feed factory may be indicative of uncontrolled or unattended use of these antibiotics in poultry production, leading to the emergence of resistant strains, which hinders its control (Table 5).
At a conference held by FAO/WHO/OIE in Rome, Italy, in late of 2007, WHO and OIE presented two lists for antimicrobials of critical use for human and animal health, respectively [28, 29]. This meeting sought to identify the antimicrobials belonging to the two lists and, as much as possible, analyze the risk of them for human health. According to this study, three classes of antimicrobials were considered priorities for the development of measures and strategies for the management of bacterial resistance since appearance on both lists, which were cephalosporins of third and fourth generation, quinolones (including fluoroquinolones), and macrolides. In this study, it was observed that for ceftazidime (third generation of cephalosporin), there were five (2.1%) resistant isolates, while for norfloxacin (fluoroquinolone), only one strain (0.4%) was resistant, suggesting a good application of these drugs in veterinary therapy (Table 2). Data provided by PREBAF [16] showed higher rates of resistance found in this study, with 59.2% of isolates resistant to the amphenicols’s group, 44% for quinolones, and 22.8% to cephalosporins of third generation.
There are cases of human salmonellosis in which the infection may be severe leading to the compulsory use of antibiotics agents [20]. Amoxicillin and sulfonamide drugs are used in the treatment of human Salmonella infection [30]. Thus, the high percentages of isolates resistant to these drugs serve as a warning to the risk of resistance that may be transmitted to humans via contaminated food, making the treatment more difficult in cases of severe salmonellosis. Furthermore, there is evidence that the use of antibiotics for growth promotion agents, prophylaxis and treatment of production’s animals, increases the prevalence of resistance among human pathogens, such as some Salmonella serovars [31]. Thus, reducing the use of antimicrobials in veterinary area could contribute to reducing this problem in human medicine.
The serovar Minnesota had the highest percentage of multiresistant strains (Tables 3 and 4), probably due to improper or illegal use of antimicrobials in poultry industry. This situation could act by selecting certain serotypes of Salmonella that end up spreading in the environment [32]. Another hypothesis is that the spread may have occurred in genes of antimicrobial resistance among isolates of this serovar.
The opposition to the use of antibiotics at low concentrations in animal feed production originates from the concern for the development of antibiotic resistance by bacteria in animals and their possible transfer to human population through the food chain. Therefore, the occurrence of multiresistant isolates of Salmonella to antibiotics in animal products warns a risk condition to public health due to the increased of hospitalizations, mortality, and expenses incurred when compared to susceptible strains [12, 16]. Thus, the monitoring of Salmonella serotypes and resistance patterns circulating in the country should be assessed continuously in chicken, in order to protect the health of the population.
The feed and its raw materials have high contamination rates for Salmonella, representing an important sources of infection of poultry. In this context, the World Health Organization warns that there is an estimate that half of the total of antibiotics produced in the world is used in animal feed [32]. Thus, while the percentage of multiresistant isolates obtained from feed was low in this study, special attention should be given to the periodic monitoring of this type of sample, because of the possible spread of resistant Salmonella for the living poultry, carcass, and then to the final consumer product.
According to Thakur et al. [12], the prevalence of antimicrobial resistant Salmonella in broilers and in the authoring environment has important implications from the point of view of food safety, because the decrease of pathogen load in the field can reduce carcass contamination at slaughter. Thus, possibly, the high number of resistant isolates obtained in the broiler poultry favored the spread of this pathogen and residence within the slaughter room, justifying the high percentage of antibiotic resistance found in this study in both environments.
Despite the importance of monitoring the bacterial resistance by diffusion test, whenever possible, it should be taken to ensure the use of MIC technique, which is regarded as a more reliable way to establish the resistance or to calculate the therapeutic dose.
The high number of isolates with MIC above the maximum of the antibiotic concentration (> 256 μg/mL for amoxicillin and > 1024 μg/mL for sulfamethoxazole) indicates that primitive properties of these microorganisms may have undergone changes with the emergence of resistance acquired in these strains. According to Palermo Neto [33], acquired resistance is one that arises in a primitively sensitive bacteria to an antibiotic and that at any given time shall no longer suffer the action of medications that showed to be effective against the original population of this bacteria.
The emergence of multiresistant Salmonella is a serious problem because it limits the therapeutic possibilities in cases of invasive infections in humans and animals and increases danger of transfer by recombination of this resistance to enteric pathogens. These findings reinforce the need for continuous monitoring of antibiotic resistance among bacterial pathogens present in food of animal origin and the importance of conducting susceptibility tests as a basis for clinical treatment decisions. The high number of resistant isolates may be due to widespread use of antibiotics in the world in different areas, which has led to enormous pressure in the selection of resistance among bacterial pathogens [34]. Furthermore, the monitoring of prevalence and resistance to Salmonella spread by food can target interventions for more effective treatments, demonstrate the importance of these zoonotic foodborne pathogens on public health, and also assist in the implementation of policies for its control [35].
These results emphasize the need for judicious use of antibiotics in animal production, based on previous tests to assess the strength and determination of appropriate doses to be applied by analyzing the minimum inhibitory concentration, thus avoiding the emergence of strains with acquired resistance to other antibiotics, especially those of common use in human medicine. For this, a continuous effort and collaboration between several professionals, especially the professionals of health and agricultural areas, which should also act in the awareness of the whole population, are necessary.
References
Scallan E, Hoekstra RM, Angulo FJ, Tauxe RV, Widdowson MA, Roy SL, Jones JL, Griffin PM (2011) Foodborne illness acquired in the United States-major pathogens. Emerg Infect Dis 17:7–15
Shao D, Shi Z, Wei J, Ma Z (2011) A brief review of foodborne zoonosis in China. Epidemiol Infect 139:1497–1504
Majowicz SE, Musto J, Scallan E, Angulo FJ, Kirk M, O’Brien SJ, Jones TF, Fazil A, Hoekstra RM, International Collaboration on Enteric Disease ‘Burden of Illness’ Studies (2010) The global burden of nontyphoidal Salmonella gastroenteritis. Clin Infect Dis 50:882–889
Brasil (2008) Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância Epidemiológica. Manual integrado de vigilância e controle da febre tifoide. Brasília: Editora do Ministério da Saúde, 92 p. (Série A. Normas e Manuais Técnicos). Available at: http://bvsms.saude.gov.br/bvs/publicacoes/manual_vigilancia_controle_febre_tifoidel.pdf. Accessed 14.02.2017
CDC (2009) Centers for Disease Control and Prevention. National Salmonella Surveillance Annual Summary. p 1–96, Available at http://www.cdc.gov/ncezid/dfwed/PDFs/SalmonellaAnnualSummaryTables2009.pdf. Accessed 24.03.2016
WHO. World Health Organization. Food safety and foodborne illness. 2002. Available at: http://www.who.int/mediacentre/factsheets/fs237/en/print.html. Accessed 15.08.2015
EFSA. European Food Safety Authority (2008) Scientific opinion of the panel on biological hazards on a request from the European Food Safety Authority on foodborne antimicrobial resistance as a biological hazard. EFSA J 765:1–87
WHO. World Health Organization (2004) 1st Joint FAO/OIE/WHO Expert workshop on non-human antimicrobial usage and antimicrobial resistance: scientific assessment, Geneva, pp 1–5 December 2003. Available at: http://www.who.int/foodsafety/publications/micro/nov2003/en/. Accessed 21.02.2015
Jay JM, Loessner MJ, Golden DA (2005) Modern food microbiology, 7th edn. Springer, New York
Le Hello S, Hendriksen RS, Doublet B et al (2011) International spread of an epidemic population of Salmonella enterica serotype Kentucky ST198 resistant to ciprofloxacin. J Infect Dis 204(5):675–684
Ungemach FR, Müller-Bahrdt D, Abraham G (2006) Guidelines for prudent use of antimicrobials and their implications on antibiotic usage in veterinary medicine. Int J Med Microbiol 296:33–38
Thakur S, Brake J, Keelara S, Zou M, Susick E (2013) Farm and environmental distribution of Campylobacter and Salmonella in broiler flocks. Res Vet Sci 94:33–42
Brasil (2003) Ministério da Agricultura, Pecuária e Abastecimento. Instrução Normativa n. 70, de 6 de outubro de 2003. Institui o Programa de Redução de Patógenos Monitoramento Microbiológico e Controle de Salmonella sp. em Carcaças de Frangos e Perus. Available at: http://extranet.agricultura.gov.br/sislegis-consulta/consultarLegislacao.do?operacao=visualizar&id=3136. Accessed 12.08.2015
Grimont PAD, Weill FX. Antigenic formulae of the Salmonella serovars. Paris: WHO Collaborating Centre for reference and research on Salmonella; 2007
CLSI (2013) Clinical and laboratory standards institute. Performance standards for antimicrobial susceptibility testing: twenty-third informational supplement. CLSI M100-S23, Wayne
Brasil (2008) Ministério da Saúde. Agência Nacional de Vigilância Sanitária - Anvisa. Relatório do Monitoramento da Prevalência e do Perfil de Suscetibilidade aos Antimicrobianos em Enterococos e Salmonelas Isolados de Carcaças de Frango Congeladas Comercializadas no Brasil. Programa Nacional de Monitoramento da Prevalência e da Resistência Bacteriana em Frango (PREBAF). Brasília,. 186 p
Brasil (2009) Ministério da Agricultura, Pecuária e Abastecimento. Instrução Normativa N° 26, de 09 de julho de 2009. Aprova o regulamento técnico para a fabricação, o controle de qualidade, a comercialização e o emprego de produtos antimicrobianos de uso veterinário. Diário Oficial da União, seção 1, p. 14
Berchieri A Jr, Freitas Neto OC (2009) Salmoneloses. In: Berchieri A Jr et al (eds) Doenças das aves, 2nd edn. FACTA, Campinas, pp 435–454
Brasil (2003) Ministério da Agricultura, Pecuária e Abastecimento. Instrução Normativa n. 78, de 3 de novembro de 2003. Aprova as Normas Técnicas para Controle e Certificação de Núcleos e Estabelecimentos Avícolas como livres de Salmonella Gallinarum e de Salmonella Pullorum e Livres ou Controlados para Salmonella Enteritidis e para Salmonella Typhimurium. Available at: http://extranet.agricultura.gov.br/sislegis-consulta/consultarLegislacao.do?operacao=visualizar&id=3864. Accessed 10.08.2015
EFSA. European Food Safety Authority (2007) Report of the Task Force on Zoonosis Data Collection on the Analysis of the baseline survey on the prevalence of Salmonella in broiler flocks of Gallus gallus, in the EU, 2005-2006, Part A: Salmonella prevalence estimates. EFSA J 98:1–85
EFSA. European Food Safety Authority (2010) Analysis of the baseline survey on the prevalence of Campylobacter in broiler batches and of Campylobacter and Salmonella on broiler carcasses in the EU, 2008, Part A: Campylobacter and Salmonella prevalence estimates. EFSA J 8(3):1–99
Loureiro ECB, Marques NDB, Ramos FLP, Reis EMF, Prazeres D (2010) Salmonella serovars of human origin identified in Pará State, Brazil from 1991 to 2008. Rev Pan-Amaz Saude 1(1):93–100
IDSC (2009) Infectious Disease Surveillance Center. Salmonellosis in Japan as of June 2009. Infectious Agents Surveillance Report. 30:203–204
Noda T, Murakami K, Ishiguro Y, Asai T (2010) Chicken meat is an infection source of Salmonella serovar Infantis for humans in Japan. Foodborne Pathog Dis 7:727–735
Oliveira SD, Flores FS, Santos LR, Brandelli A (2005) Antimicrobial resistance in Salmonella Enteritidis strains isolated from broiler carcasses, food, human and poultry-related samples. Int J Food Microbiol 97:297–305
Trongjit S, Angkititrakul S, Tuttle RE, Poungseree J, Padungtod P, Chuanchuen R (2017) Prevalence and antimicrobial resistance in Salmonella enterica isolated from broiler chickens, pigs and meat products in Thailand–Cambodia border provinces. Microbiol Immunol 61:23–33
Brasil (2003) Ministério da Agricultura, Pecuária e Abastecimento. Instrução Normativa N° 9, de 27 de Junho de 2003. Proibe a fabricação, a manipulação, o fracionamento, a comercialização, a importação e o uso dos princípios ativos cloranfenicol nitrofuranos e os produtos que contenham estes princípios ativos, para uso veterinário e suscetível de emprego na alimentação de todos os animais e insetos Diário Oficial da União, seção 1, p. 4
OIE (2007) World Organization for Animal Health. List of antimicrobials of veterinary importance. Report of the 75th general session (Resolution n° XXVIII). Paris, France, 25 p
WHO (2007) World Health Organization. Critically important antimicrobials for human medicine. Categorization for the development of risk management strategies to contain antimicrobial resistance due to non-human antimicrobial use. Report of the 2nd WHO Expert Meeting held in Copenhagen. Geneva, Switzerland, 34 p
Saraíva-Gomes J, Focaccia R, Lima VP (2006) Febres tifoide e paratifoide. In: Veronesi R (ed) Tratado de Infectologia, 3rd edn. Atheneu, São Paulo, pp 919–932
Singer R, Finch R, Wegener HC, Bywater R, Walters J, Lipsitch M (2003) Antibiotic resistance - the interplay between antibiotic use in animals and human beings. Lancet Infect Dis 3:47–51
WHO (2005) World Health Organization. Drug-resistant Salmonella. Fact Sheet No 139, revised, April. Avaiable at: http://www.who.int/mediacentre/factsheets/fs139/en/. Accessed 07.09.2016
Palermo Neto J (2011) Mecanismos de resistência aos antibióticos em saúde pública. In: Conferência Facta 2011 de Ciência e Tecnologia Avícolas, 2011, Santos. Anais... Santos: FACTA, p 39–50
Capita R, Alonso-Calleja C, Prieto M (2007) Prevalence of Salmonella enterica serovars and genovars from chicken carcasses in slaughterhouses in Spain. J Appl Microbiol 103:1366–1375
Mangen MJ, Batz M, Kassbohrer A et al (2010) Integrated approaches for the public health prioritization of foodborne and zoonotic pathogens. Risk Anal 30:782–797
Funding
This study was financially supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Ministério da Agricultura Pecuária e Abastecimento (MAPA), and the Secretaria de Defesa Agropecuária (SDA), approved with the grant number 578372/2008-8, according to the Notice CNPq/MAPA/SDA no. 064/2008.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Responsible Editor: Luis Augusto Nero
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mendonça, E.P., de Melo, R.T., Nalevaiko, P.C. et al. Spread of the serotypes and antimicrobial resistance in strains of Salmonella spp. isolated from broiler. Braz J Microbiol 50, 515–522 (2019). https://doi.org/10.1007/s42770-019-00054-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42770-019-00054-w