Abstract
To understand how the intercropping system alleviates stressful conditions, this study was aimed at elucidating the effects of legume–cereal intercropping in enhancing phosphorus (P) availability in P-deficient soil under the semi-arid climate of southern Tunisia. During the two growing seasons––2018–2019 and 2019–2020––two experimental fields were employed to explore the effect of soil P availability on the growth of durum wheat and chickpea under different cropping systems. A randomized complete block design was used in this experiment, with three blocks each divided into three plots. Each plot was used for one of the following four cropping systems with three replications: (i) monocrop chickpea (Ck-M); (ii) monocrop durum wheat (DW-M); (iii) durum wheat–chickpea intercrop (DWCk-IC); and (iv) vacant soil without plants as a control. Compared to the vacant soil, we found a significant increase in the Olsen P concentration in the soil rhizosphere by about 16%, 48%, and 36% for the DW-M, Ck-M, and DWCk-IC, respectively. Also, the increase in soil P availability was associated with a pH decrease of −0.73 and −0.37 units for Ck-M and DWCk-IC, respectively. In addition, the soil microbial biomass P increased significantly (P < 0.05), by about 27%, 22%, and 18% for the Ck-M, DWCk-IC, and DW-M, respectively, compared to the vacant soil. The increased soil P availability improved the P contents in the durum wheat and chickpea roots and shoots, although it mostly enhanced the intercropped dry weight. These findings demonstrate that durum wheat–chickpea intercropping could be a practical cropping system for improving the soil P availability through enhanced activity in the microbial community and soil acidification.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Phosphorus (P), one of the essential nutrients required by crops, is involved in an array of processes, such as photosynthesis and respiration, and is an integral component of several crop structures, such as phospholipids (Adhikari and Pandey 2019). The availability of P often limits crop growth and is a significant constraint on improving agricultural production in order to meet the increasing global food demand (Duchene et al. 2017). Soils can contain pools of P that could be several thousand times greater than the amount necessary for crop growth. Indeed, only a tiny soluble fraction might be available for crop uptake (Gong et al. 2020). The P in soil can either be adsorbed to the soil particle surfaces (Hamdi et al. 2015, 2022) or precipitated with calcium (Ca2+) or aluminum (Al3+) and iron (Fe3+) (Mpanga et al. 2018). Applied mineral P fertilizer can enhance the P availability in soils, but it is costly and can have negative impacts on the environment because it can cause eutrophication (Jia et al. 2020). Therefore, there is a need to develop alternative management practices that better exploit soil P resources by increasing P bioavailability in agro-ecosystems (Neji et al. 2019; Zhang et al. 2021a). The benefits of the intercropping system in optimizing soil P use have been reported in numerous studies (e.g., Lian et al. 2019). This system can involve a mix of two different crop species growing simultaneously in the same field area for a period of their respective life cycles (Lian et al. 2019). The system has enough potential to enable agriculture sustainability through crop diversification, improve crop yields, enhance soil biological activity, and optimize nutrient cycling. Intercropping systems have been widely practiced, globally, including wheat–maize (Mupangwa et al. 2021), maize–fava bean (Xu et al. 2018), wheat–pea and maize–soybean (Fu et al. 2019), and legume–durum wheat (Cong et al. 2015). Legume–durum wheat intercropping has shown P acquisition advantages under adverse conditions (Cheng et al. 2014; Gong et al. 2020). Several studies have also indicated that legumes can develop biochemical mechanisms that solubilize organic soil P (Layek et al. 2018; Li and Wu 2018; Dai et al. 2019; Zhang et al. 2021b), thereby increasing its availability to the intercropped species, including durum wheat (Gong et al. 2020), through root-induced processes, such as acidification of the rhizosphere following the exudation of certain organic acids, and/or indirectly by microbial activity (Farid et al. 2019). Among legume species, the ability of chickpea to enhance soil P availability to cereal roots under soil P deficiency has been studied (Atienza and Rubiale 2017; Gong et al. 2020; Jia et al. 2020). With its ability to form taproots and exude vast amounts of organic acids (Lian et al. 2019; Liao et al. 2020; Tang et al. 2021), chickpea is able mobilize the soil P. Also, chickpea has been identified as an essential plant species for cultivation in soils where higher amounts of P are stored in poorly available forms for different crops. However, other research works have reported no significant effects (Anand et al. 2016; Ning et al. 2017). These contrasting results support the need to further investigate the mechanisms responsible for P availability under a durum wheat–chickpea intercrop (DWCk-IC). Despite the sizeable literature on intercropping, little information is available on the effect of a DWCk-IC in an arid region, such as Tunisia (North Africa). Such regions cover nearly 40% of the total land area in Tunisia and host more than 30% of the total population (Aziz et al. 2016). However, they present limits to increasing agricultural production due to the loss of ecosystem productivity caused by the degradation in water quality and the low nutrient content in the arable soil, which reduces the number of crop varieties suitable for agriculture. Moreover, our thorough literature review revealed that precise information is deficient concerning the effects of chickpea on the soil P status and on durum wheat production in an intercrop system under the conditions of alkaline soils in arid regions of Tunisia. We hypothesized that durum wheat growth would be enhanced under an intercropping system with chickpea as a result of the rhizosphere effects of the chickpea roots. The main objectives of the current study were to (i) evaluate the impacts of intercropping on dry weight, grain yield, and soil pH variation compared to monocrops; (ii) investigate soil P availability and uptake by crops under intercropping systems; and (iii) analyze the potential of the soil microbial biomass phosphorus (SMBP) to contribute to P availability.
2 Material and Methods
2.1 Site Description
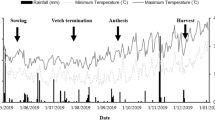
Field studies were conducted during the 2018–2019 and 2019–2020 growing seasons in the experimental station of the Institute of Arid Regions Medenine, Tunisia (33°2 9′ 56.71′′ N, 10° 38′ 41.50′′). The weather conditions relating to the Medenine province in Tunisia (average monthly rainfall and temperature values for the plant growth phase of the 2018–2019 and 2019–2020 growing seasons) are presented in Fig. 1. These data showed that highest precipitation was in February 2019 (20.66 mm), July 2019 being the driest month, with 2.1 mm. During the growing season, the coldest period was from December to February, with minimum temperatures of 15°C recorded. Samples were collected from the top 20 cm of the soil layer in order to determine various physicochemical properties before the beginning of the experiment. These were air-dried in the laboratory and sieved (through a 2-mm mesh). The soil was alluvial poorly evolved with limitations of N and P (Mtimet 2001), containing 70.2% sand, 20.1% loam, and 8.7% clay (Table 1). The topsoil was alkaline, with a pH of 7.75 and electrical conductivity (EC) of 1.7 mS cm–1. Its calcium carbonate (CaCO3) and organic matter (OM) contents were relatively low, with respective percentages of 2.4% and 1.21%. The calcium (Ca2+), magnesium (Mg2+), potassium (K+), and sodium (Na+) exchangeable cations were estimated as 624, 201, 225, and 344 mg kg–1, respectively. The topsoil was deficient in P, with its Olsen P and total P being estimated to be about 6.6 and 8.95 mg P kg−1, respectively. Its nitrogen (N) content was relatively low at 4. 04 mg N kg−1.
2.2 Cropping System and Field Plot Design
During the two cropping seasons, one chickpea cultivar (Cicer arietinum L. cv. Amdoun 1) and one durum wheat cultivar (Triticum turgidum durum L. cv. Simeto) were grown for the study; these are common crops in the Tunisia legume–cereal agro-ecosystem and which has been reported to possess excellent tolerance to low soil P. The experiment involved a randomized complete design of three blocks, each divided into three plots. Each plot was used for one of the following four cropping systems and each was replicated three times: (i) a chickpea monocrop (Ck-M); (ii) a durum wheat monocrop (DW-M); (iii) a DWCk-IC; and (iv) a vacant plot without plants as a control. The size of each plot was 4.5 m2 (3 × 1.5 m). The seeding density was that recommended for the region’s farming practices––50±5 seeds m–2 for the Ck-M, 250±10 seeds m–2 for the DW-M, with 150±10 and 30±3 seeds m–2 for the DWCk-IC, respectively. In both the monocrop and intercrop systems, the distance between the rows and plants of the chickpea crop was 25 and 20 cm, respectively. The two species were sown in the same row to maximize root proximity and chickpea–wheat rhizosphere interactions. The seeding of both species took place on January 15, 2018 and January 10, 2019. The plots were manually weeded and maintained weed-free. The experimental plots did not receive any fertilizers or herbicides. For crop management, intercropping is carried out under rainfed conditions but in order to avoid the effects of the water stress caused by the low rainfall and ensure a normal crop development, 100 mm of irrigation was applied to all treatments in the germination stages. The plants were harvested at maturity––the durum wheat on June 25, 2018 and June 20, 2019, and the chickpea on July 20, 2018 and July 15, 2019.
2.3 Characterization of Plant and Soil Samples
During the plant cycle of the two cropping seasons, plant samples were taken during two sampling periods corresponding to complete flowering (about 70 days after sowing) and at crop maturity (the harvest stage of the cropping cycle). Three sub-replicates (0.25 m2 quadrats) were chosen from each plot replicate, excluding the border rows, from which all the plants were harvested. The durum wheat shoots were cut at the shoot–root junction. However, the chickpea roots were severed at the cotyledonary node. The roots and shoots were oven-dried for 48 h at 60°C and then weighed. The total plant root and shoot dry weights of each species were determined.
In order to determine the land-equivalent ratio (LER), Eq. (1) was applied:
where Yaa and Ybb = yields from the monocrops of species a and b, respectively; and Yab and Yba = yields from the intercrop of species a and b, respectively. For the N determination, the shoot and root samples were digested in hot concentrated sulfuric acid, following the Kjeldahl method. After digestion with nitric acid and perchloric acid, the total P contents (shoots and roots) were determined following the malachite green method. The rhizosphere soil adhering to the durum wheat and chickpea roots was sampled by brushing off the <1–4-mm aggregates. These samples were thoroughly mixed and pooled to make a composite sample for each plot. A vacant plot sample was taken as a control. All composite soil samples were air-dried, sieved through a 2-mm mesh), and the following properties were determined using standard methods. The soil pH was determined from a soil suspension in deionized water (soil:water ratio = 1:5) using a pH meter. A conductivity meter was used to measure the electrical conductivity in a 1:1 soil:water slurry. The total P and available P were determined by digestion using perchloric and nitric acids, and by the Olsen method. The total N concentrations were determined using the Kjeldahl procedure. The soil Ca, Mg, K, and Na contents were assessed using an atomic absorption spectrophotometer (PerkinElmer, Inc., Shelton, CT, USA). The OM content was determined using the Walkley and Black (1934) method. The soil CaCO3 content was estimated using the Horton and Newson method. Finally, the SMBP was determined from the difference between the amount of inorganic P (Pi) extracted by 0.5 (Spm) sodium bicarbonate (pH 8.5) from fresh soil fumigated with chloroform and the amount extracted from unfumigated soil.
2.4 Statistical Analyses
The statistical analysis was conducted using XLSTAT statistical software (Premium Version, 2017, Addinsoft, Long Island, NY, USA). The mean values of the results obtained from the two cultivation seasons are reported as main effects and interactions. The means of the soil and plant parameters were compared using Fisher’s least significant difference test at P < 0.05, with significantly different means indicated by different letters.
3 Results
3.1 Soil P Rhizosphere Characterization
Figure 2 shows the average concentration of total P and Olsen P measured in the rhizosphere (0–20 cm) soil of the different cropping systems during the two cropping seasons. The rhizosphere exhibited a significant increase in total P of 18% (DW-M), 36% (Ck-M), and 24% (DWCk-IC) compared to the vacant soil. The pattern was similar for the Olsen P, with a systematic and significant increase in P concentrations in the rhizosphere of 16% (DW-M), 48% (Ck-M), and 36% (DWCk-IC) compared to the vacant soil. This increase in the Olsen P concentration in the rhizosphere was most significant for the Ck-M and DWCk-IC. In most cases, the measured total P and Olsen P concentrations increased compared to the vacant soil.
Mean total P and Olsen P values in vacant soil and rhizosphere of durum wheat and chickpea grown as sole crops or intercrops during the two cropping seasons. DW-M: sole-cropped durum wheat, Ck-M: sole-cropped chickpea, DWCk-IC: durum wheat intercropped with chickpea, vacant (soil without plants). Values are the mean values of three replicates for the two cropping seasons. The error bars represent the standard error of the mean. Different letters indicate a significant difference at p < 0.05 between the rhizosphere and the vacant of different species under different cropping treatments
3.2 Soil P Status and pH Variation
The average P contents and the variation in pH during the two cultivation seasons are indicated in Fig. 3. The rhizosphere of the Ck-M was notably acidified compared to the vacant soil, with a pH decrease of about 0.7 units. Similarly, the soil pH of the DWCk-IC rhizosphere declined by about 0.4 units. There was no significant pH difference (P > 0.05) in the rhizosphere of the DW-M. It is worth mentioning that the Olsen P contents increased with decreasing pH in the rhizosphere of the DWCk-IC (Fig. 3). These findings confirm that acidification can increase the P availability in neutral to alkaline soils.
Effect of pH variation (mean values) on Olsen P (mean values) in vacant soil and rhizosphere soil of durum wheat and chickpea grown as sole crops or intercrops during the two cropping seasons. DW-M: sole-cropped durum wheat, Ck-M: sole-cropped chickpea, DWCk-IC: durum wheat intercropped with chickpea, vacant (soil without plants). Values are the mean values of three replicates for the two cropping seasons. The error bars represent the standard error of the mean
3.3 Soil Microbial Biomass P
The average values of the SMBP from the two growing seasons were significantly (p < 0.05) higher in the rhizosphere of all the crops systems relative to the vacant soil (Fig. 4), measured at 27%, 22%, and 18% for the Ck-M, DWCk-IC, and DW-M, respectively. The lines representing the relationship between the Olsen P and the SMBP on Fig. 5 reveal a positive correlation for the vacant soil (slope = 0.718, p < 0.05, R2 = 0.100), Ck-M (slope = 1.007, p < 0.05, R2 = 0.8), and DWCk-IC (slope = 0.675, p < 0.05, R2 = 0.740). By contrast, the same parameters were negatively correlated in the DW-M (slope = −0.694, p < 0.05, R2 = 0.31).
The response of the soil mass microbial phosphorus (SMBP) (mean values for the two cropping seasons) to the cropping systems. DW-M: sole-cropped durum wheat, Ck-M: sole-cropped chickpea, DWCk-IC: durum wheat intercropped with chickpea, vacant (soil without plants). Bars with the different letters indicate significant differences at p < 0.05 between the rhizosphere and the vacant of different species under different cropping treatments
Relationship between average Olsen P content and soil mass microbial phosphorus (SMBP) in the rhizosphere and the vacant (soil without plants) across all treatments during the two cropping seasons. DW-M: sole-cropped durum wheat, Ck-M: sole-cropped chickpea, DWCk-IC: durum wheat intercropped with chickpea. Lines represent the relationship between Olsen P and SMBP in vacant (slope = 0.718, p <0.05, R2 = 0.100), DW-M (slope = 0.694, p <0.05, R2 = 0.31), Ck-M (slope = 1.007, p <0.05, R2 = 0.8) and in the DWCk-IC (slope = 0.675, p < 0.05, R2 = 0.740)
3.4 Root and Shoot Dry Weight P Contents, Grain Yield, and Land Equivalent Ratio
Figure 6 shows the average values of the root (A) and shoot (B) dry weights of the durum wheat and chickpea for all treatments for the two growing seasons. The root and shoot dry weight of the durum wheat from the DWCk-IC was 15% higher than that of the DW-M. However, there was no significant difference between the root and shoot dry weights of the chickpea from the DWCk-IC and that of the Ck-M. The root and shoot dry weight significantly correlated (P < 0.05) with the weight of the root and shoot P contents at the peak vegetation growth stage for all treatments. The average grain yields of the durum wheat and chickpea under the different cropping systems from the two growing seasons is indicated in Fig. 7. The grain yield of the durum wheat from the DWCk-IC was significantly higher (+21%) than from the DW-M (Fig. 7A). For chickpea, the grain yield was lower (−15%) in the DWCk-IC than in the Ck-M (Fig. 7B). In addition, the LER values were significantly greater than 1 (Fig. 8), ranging from 1.05 to 1.3 in almost all cases for the different treatments. The LER for the DWCk-IC significantly exceeded that of their corresponding sole crops.
Relationship between the average dry weight values of shoots (A) and roots (B) of cereals and chickpea and the average P contents of intercrops and in sole crop during the two cropping seasons. DW-M: sole-cropped durum wheat, Ck-M: sole-cropped chickpea, DW-IC: durum wheat intercropped with chickpea, Ck-IC chickpea intercropped with durum wheat. The vertical bars represent 95% confidence intervals of estimates
Mean values of grain yield of A DW-M: sole-cropped durum wheat and DW-IC: durum wheat intercropped with chickpea and of B Ck-M: sole-cropped chickpea, and Ck-IC chickpea intercropped with durum wheat during the two cropping seasons. Bars with the different letters compare cropping systems and years within a crop and are significantly different at p < 0.05
Estimation of average land-equivalent ratio (LER) values under different cropping systems over the two cropping seasons. DW-M: sole-cropped durum wheat, Ck-M: sole-cropped chickpea, DW-IC: durum wheat intercropped with chickpea, Ck-IC chickpea intercropped with durum wheat (the horizontal bars represent 95% confidence intervals of estimates)
4 Discussion
The analysis of the various parameters related to rhizosphere soil P processes indicated that the soil P under intercropping was significantly greater than that under the DW-M. Similar results have been reported for durum wheat intercropped with chickpea in field experiments on fertile soils (Ning et al. 2017). Chickpea roots are known to exude several acid components, which cause acidification of the chickpea rhizosphere (Schaap et al. 2021; Kutamahufa et al. 2022). Acidification of the rhizosphere caused by chickpea exudates likely benefitted the intercropped durum wheat by increasing P availability through the dissolution of P minerals (Mitran et al. 2018). Similarly, Lian et al. (2019) reported that chickpea species secrete higher phosphatase levels in P-deficient soil conditions via their root systems. Our results support the concept of cluster root formation playing a significant role in the acquisition of P from soils. In addition, it should be noted that the roots of chickpea are known to exude substantial amounts of organic acids or hydrogen (H+) in comparison to wheat (Atienza and Rubiale 2017). Therefore, for the DWCk-IC, the reduction in soil pH likely enhanced cation removal from the soil due to the higher yield in the intercropping system. In this context, Anand et al. (2016) reported that root-induced pH changes are mainly influenced by H+/hydroxide (OH−) exudation, depending on the cation–anion balance, which is mainly driven by N nutrients and changing chemical properties in the soil, and thus by the surface charge of the minerals influencing the partitioning of the P ions (Mkuhlani 2021). It is well known that a decrease in pH can alter the P availability through either the dissolution of P minerals, such as Ca phosphates, or the desorption of phosphate ions bound onto soil constituents (Abbasi and Manzoor 2018). Also, it may be that legumes, such as chickpea, take up more Ca2+ than monocotyledonous (grass) species, such as durum wheat (Atienza and Rubiale 2017). This may explain the differences between durum wheat and chickpea when considering root-induced changes in pH and P availability in their respective rhizospheres (Pierre et al. 2022). For the rhizosphere of the Ck-M, the relationship between the Olsen P concentrations and pH values indicate that root-induced pH changes resulted from a prominent rhizosphere process driving P availability. For the DWCk-IC, there was no significant correlation between the pH values and the Olsen P content (P < 0.05). This might suggest that the effect of pH on the Olsen P has been masked by the effect of pH change, resulting in a decrease in P availability over a significant distance from the plant roots. For the soil microbial biomass P, our results demonstrated that the Ck-M and DWCk-IC enhanced SMBP activity compared to the DW-M, which indicates that chickpea may utilize more organic soil P and improve nutrient cycling, and that soil fertility can positively affect the microbial biomass (Gong et al. 2020). Moreover, our results concur with those of previous studies that have indicated that chickpea plant species could influence the SMBP through the abundance, activity, and composition of the soil decomposer communities in their rhizospheres (Abd-Alla et al. 2019; Anand et al. 2016). The correlation between Olsen P and SMBP revealed; however, contrasting results that remind us that there are several processes controlling microbial communities. Previous studies have indicated that resource availability may have determined the composition of the microbial biomass in the vacant soil (Rakhimova et al. 2021; Schwerdtner and Spohn 2021; Sridhar and Salakinkop 2021). By contrast, more-complex processes, such as predation or phage dynamics, could also have been involved in the rhizosphere, especially in the intercrops (Nasto et al. 2017; Tang et al. 2021). In the DWCk-IC, the increase in SMBP appeared to be the result of microbial growth in the rhizosphere of the vacant soil. This suggests differences in resource allocation within microbial communities. The microorganisms around chickpea roots can enhance durum wheat root growth by mobilizing P storage even when the chickpea and durum wheat are being intercropped. In this context, Zhao and Wu (2014) posited that the organic soil P could comprise up to 65% of the total P in organic soil reservoirs, and that microorganisms can mineralize this significant fraction and enhance the available P in soil solution. Commonly, the results of this study showed that P was the limiting element in determining the productivity of durum wheat and chickpea in intercrop and sole systems. Similarly, a positive correlation has been established between biomass and chickpea plant P contents from field experiments in India (Saini et al. 2004). Other studies have also reported significant positive relationships between P contents and DWCk-IC yields in ferrosols in the sub-Sahara of Kenya (Ayaga et al. 2006). In addition, it can be seen that the P contents significantly varied under the intercropping systems compared with the monoculture systems (P < 0.05). The order Ck-M > DWCk-IC > Ck-M was found for the P in shoots and roots, suggesting that P is an essential resource for chickpea, and the P fixed by this plant can be used by durum wheat in the intercropping system during both plants’ growth stages. This finding was be attributed to the complex biological diversity that exists under intercropping systems, where P is transferred to the soil via ions and root exudates, further facilitating the accumulation and decomposition of the soil P pool (Bhowmik et al. 2018; Tripathi et al. 2021). However, Balemi and Negisho (2012) reported that mixed chickpea intercropping increases P availability, providing higher P levels for P utilization by the adjacent crop. We found that the P contents in the shoots and roots of the durum wheat and chickpea under DWCk-IC increased by 28.8% and 23.4% and declined by 8.2 and 5.4%, respectively, compared to the DW-M. This shows that higher soil fertility might be expected to result in greater plant growth in chickpea, resulting in the exudation of a diverse range of organic acids by the chickpea into the soil, thereby facilitating P assimilation. Thus, use of the intercropping system could produce a large amount of P in the rhizosphere soil and increase nutrient accumulation. Furthermore, the dry weight at the completed flowering stage showed a significant increase in the durum wheat from the intercropping system compared to its monoculture. This increase could be attributed to the effect of the added N fixed by the chickpea crop in the intercrop system. However, the high chickpea dry weight observed in the Ck-M treatment compared to the intercropping system could be attributed to competition between the crops for growth resources, such as light, nutrients, and water, in the latter (Zhang et al. 2021a). This suggests that competition between durum wheat and chickpea in the intercropping system significantly affects chickpea growth compared to chickpea growth in sole cropping. Previous studies have reported that differences in the depth of the roots, lateral root spread, and root densities may result from competition for nutrients between the component crops in an intercropping system (Fernández-Juárez et al. 2020; Khanal et al. 2021). Also, a fertile soil rhizosphere contains a large supply of resources, which could thus reduce competition between plants compared to less fertile rhizospheres, where chickpea can enrich the soil nutrient base through the fixation of atmospheric N into the soil (Elhaissoufi et al. 2020; Chenene et al. 2021). According to Mitran et al. (2018), shading the chickpeas species component in intercropping systems can enhance N2 symbiotic fixation and photosynthesis, especially during the full flowering stage. This operation might offer opportunities for sustaining the enhancement of plant dry weight in intercropped species (Tiziani et al. 2020). This kind of system improves the health of the planting, reduces the spread of disease, and enhances plant growth (Arshad 2021; Amraei 2022). Furthermore, intercropping systems can suppress weeds (Ullah et al. 2017), thereby reducing the competition between cultivated plants and weeds for water and nutrients, and they favor the growth of cultivated plants. Several researchers have observed a decline in durum wheat growth due to competition with legumes at the end of the plant growth period (Ullah et al. 2017; Tindwa et al. 2019). However, the grain yield of durum wheat is significantly increased in intercropping than in sole cropping. This increased grain yield may be due to improved efficiency in the use of resources via both functional complementarity and facilitation between the intercropped chickpea and durum wheat. A previous study has also reported the benefit of intercropping cereals with chickpea due to the facilitation mechanisms the chickpea affords––increasing the P and N through rhizosphere acidification during N2 fixation––which may help sustain increased grain yield in intercropped durum wheat. For the chickpea, our data revealed that grain yield was lower for intercropping than sole cropping. In our field experiment, the chickpea and durum wheat were sown at the same time in the two seasons and matured at about the same time (the chickpea matured around 25 days after the durum wheat), thus maximizing the competition and resource (e.g., N and P) use by the durum wheat, facilitated by the intercropped chickpea. These results confirmed the findings of Latati et al. (2019), who reported a reduction in chickpea dry weight when intercropped with durum wheat. Finally, our results strongly correlated with the definition of the LER, where the combination of component species in the intercropping system was more productive than the same species grown as sole crops. Indeed, the LER values for the intercropping system was higher than for the sole cropping, highlighting the advantage of intercropping. In this context, Mahallati et al. (2015) and Homulle et al. (2021) reported that an LER of 1.0 or less indicates no difference between intercropped and monoculture yields, while any value greater than 1.0 indicates a yield advantage for intercropping. Moreover, Ajala et al. (2019) posited that a LER value of 1.2 indicates that the area planted with monocultures would need to be 20% greater than the area planted with intercrops to produce the same combined yields.
5 Conclusion
Reliable with the aims of this work, we revealed that durum wheat intercropped with chickpea, resulted in significantly improved P available for plant growth and biomass performance in low-P soils during the two growing seasons. Communally, reduction in pH and increased microbes activity appear to be the most important determinants of the P soil available status and improved biomass production, when durum wheat and chickpea are cultivated as intercrops in alluvial soil under semi-arid climatic conditions. This study will provide significant enhancement in our knowledge to well understand agriculture production in semi-arid climate, particularly in the context of climate change and expanding global populations in these geographic regions where food security is anticipated to be a challenge in the future. However, given the complexity of soil P availability, which largely depends on the soil properties and agricultural practices, additional research is required to understand the interactive relationships between soil P, agricultural practices, their history, and management.
Data Availability
Authors declare that data are available on request.
References
Abbasi KM, Manzoor M (2018) Bio-solubilization of phosphorus from rock phosphate and other P fertilizers in response to phosphate solubilizing bacteria and poultry manure in a silt loam calcareous soil. J Plant Nutr Soil Sci 181:345–356. https://doi.org/10.1002/jpln.201800012
Abd-Alla MH, Nafady NA, Bashandy SR, Hassan AA (2019) Mitigation of effect of salt stress on the nodulation, nitrogen fixation and growth of chickpea (Cicer arietinum L.) by triple microbial inoculation. Rhizosphere 10:100–148. https://doi.org/10.1016/j.rhisph.2019.100148
Adhikari P, Pandey A (2019) Phosphate solubilization potential of endophytic fungi isolated from Taxus wallichiana Zucc, Roots. Rhizosphere 9:2–9. https://doi.org/10.1016/j.rhisph.2018.11.002
Ajala RO, Awodun MA, Adeyemo AJ, Dada BF (2019) Assessment of wood ash application on yield advantage indices of maize and lima beans in an intercrop. J Exp Agric Int 34:1–11. https://doi.org/10.9734/jeai/2019/v34i130163
Amraei B (2022) Effects of planting date and plant density on yield and some physiological characteristics of single cross 550 hybrid maize as a second crop. Casp J Environ Sci 20:683–691. https://doi.org/10.2135/cropsci2002.1191
Anand K, Kumari B, Mallic MA (2016) Phosphate solubilizing microbes, an effective and alternative approach as biofertilizers. Int J Pharm Sci 8:37–40
Arshad M (2021) Fortnightly dynamics and relationship of growth, dry matter partition and productivity of maize based sole and intercropping systems at different elevations. Eur J Agron 130:126377. https://doi.org/10.1016/j.eja.2021.126377
Atienza S, Rubiale D (2017) Legumes in sustainable agriculture. Crop Past Sci 68:1–11. https://doi.org/10.1071/CPv68n11_FO
Ayaga G, Todd A, Brookes PC (2006) Enhanced biological cycling of phosphorus increases its availability to crops in low-input sub-Saharan farming systems. Soil Biol Biochem 38:81–90. https://doi.org/10.1016/j.soilbio.2005.04.019
Aziz A, Ahiabor B, Opoku A, Abaidoo R (2016) Contributions of rhizobium inoculants and phosphorus fertilizer to biological nitrogen fixation, growth and grain yield of three soybean varieties on a fluvicluvisol. Am J Exp Agric 10:1–11. https://doi.org/10.9734/AJEA/2016/20072
Balemi T, Negisho K (2012) Management of soil phosphorus and plant adaptation mechanisms to phosphorus stress for sustainable crop production, a review. J Soil Sci Plant Nutr 12:547–561. https://doi.org/10.4067/S0718-95162012005000015
Bhowmik P, Ellison E, Polley B, Bollina V, Kulkarni M, Ghanbarnia K, Song H, Gao C, Voytas DF, Kagale S (2018) Targeted muta-genesis in wheat microspores using CRISPR/Cas9. Sci Rep 8:6502. https://doi.org/10.1038/s41598-018-24690-8
Chenene Y, Blavet D, Belalmi M, Kaci G, Teffahi M, Ounane SM (2021) Variation of chickpea nodulation in a Mediterranean agroecosystem: relationship with soil characteristics and thresholds for significant contribution to plant growth. Agron Res 19:42–56. https://doi.org/10.15159/AR.21.015
Cheng LX, Tang CP, Vance PJ, White F, Zhang J, Shen J (2014) Interactions between light intensity and phosphorus nutrition affect the phosphate-mining capacity of white lupin (Lupinus albus L.). J Exp Bot 65:2995–3003 https://www.researchgate.net/publication/261605019
Cong W, Hoffland E, Li L, Six J, Sun J, Bao X, Zhang F, Van Der Werf W (2015) Intercropping enhances soil carbon and nitrogen. Glob Change Biol 21:1715–1726. https://doi.org/10.1016/j.rhisph.2023.100686
Dai J, Qiu W, Wang N, Wang T, Nakanishi H, Zuo Y (2019) From Leguminosae/Gramineae intercropping systems to see benefits of intercropping on iron nutrition. Front Plant Sci 10:605. https://doi.org/10.3389/fpls.2019.00605
Duchene O, Vian JF, Celette F (2017) Intercropping with legume for agro-ecological cropping systems, complementarity and facilitation processes and the importance of soil microorganisms. Agr Ecosyst Environ 240:148–161. https://doi.org/10.1016/j.agee.2017.02.019
Elhaissoufi W, Khourchi S, Ibnyasser A, Ghoulam C, Rchiad Z, Zeroual Y (2020) Phosphate solubilizing rhizobacteria could have a stronger influence on wheat root traits and aboveground physiology than Rhizosphere P solubilization. Front Plant Sci 11:1–15. https://doi.org/10.1016/j.jare.2021.08.014
Farid AS, Mohamed M, El-Dsouky M, El-Rewainy HM (2019) Isolation and characterization of phosphate solubilizing actinomycetes from rhizosphere soil. AJAS 49:125–137. https://doi.org/10.1007/s11356-018-2955-5
Fernández-Juárez V, Bennasar-Figueras A, Sureda-Gomila A, Ramis-Munar G, Agawin N (2020) Differential effects of varying concentrations of phosphorus, iron, and nitrogen in N2-fixing cyanobacteria. Front Microbiol 11:1–19. https://doi.org/10.3389/fmicb.2020.541558
Fu Z, Zhou L, Chen P, Du Q, Pang T, Song C, Wang X, Liu W, Yang W, Yong T (2019) Effects of maize-soybean relay intercropping on crop nutrient uptake and soil bacterial community. J Integr Agric 18:2006–2018. https://doi.org/10.1016/S2095-3119(18)62114-8
Gong X, Dang K, Lv S, Zhao G, Tian L, Luo Y (2020) Interspecific root interactions and water-use efficiency of intercropped proso millet and mung bean. Eur J Agron 115:126034. https://doi.org/10.1016/j.eja.2020.126034
Hamdi W, Hamdi N, Jellali S, Seffen M (2022) Effect of background electrolytes on the adsorption of phosphorus (P) onto southern Tunisia natural clays. Phys Chem Earth 127:103160. https://doi.org/10.1016/j.pce.2022.103160
Hamdi W, Noura Z, Ernest K, Dlavet D, Seffen M (2015) Effect of the soils properties on the sorption capacity of phosphorus and ammonium by alkaline soils of the semi-arid areas. IOSR-J AC 5:34–42
Homulle Z, George TS, Karley AJ (2021) Root traits with team benefits: understanding belowground interactions in intercropping systems. Plant Soil:1–26. https://doi.org/10.1007/s11104-021-05165-8
Jia X, Zhong Y, Liu J, Zhu G, Shangguan Z, Yan W (2020) Effects of nitrogen enrichment on soil microbial characteristics, from biomass to enzyme activities. Geoderma 366:114256. https://doi.org/10.1016/j.geoderma.2020.114256
Khanal U, Stott KJ, Armstrong R, Nuttall JG, Henry F, Christy BP, Mitchell M, Riffkin PA, Wallace AJ, McCaskill M (2021) Intercropping-evaluating the advantages to broadacre systems. Agriculture 11:453. https://doi.org/10.3390/agriculture11050453
Kutamahufa M, Matare L, Soropa G, Mashavakure N, Svotwa E, Mashingaidze AB (2022) Forage legumes exhibit a differential potential to compete against maize and weeds and to restore soil fertility in a maize-forage legume intercrop. Acta Agric Scand B Soil Plant Sci 72:127–141. https://doi.org/10.1080/09064710.2021.1998593
Latati M, Dokukin P, Aouiche A, Rebouh NY, Takouachet R, Hafnaoui E (2019) Species interactions improve above-ground biomass and land use efficiency in intercropped wheat and chickpea under low soil inputs. Agro 9(11):765. https://doi.org/10.3390/agronomy9110765
Layek J, Das A, Mitran T, Nath C, Meena R, Yadav GS, Shivakumar G, Kumar S, Lal R (2018) Cereal + legume intercropping: an option for improving productivity and sustaining soil health. Legumes Soil Health Sustain Manag:347–386. https://doi.org/10.1007/978-981-13-0253-4_11
Li S, Wu F (2018) Diversity and co-occurrence patterns of soil bacterial and fungal communities in seven intercropping systems. Front Microbiol 9:1521. https://doi.org/10.3389/fmicb.2018.01521
Lian T, Mu Y, Jin J, Ma Q, Cheng Y, Cai Z (2019) Impact of intercropping on the coupling between soil microbial community structure, activity, and nutrient–use efficiencies. Peer J 7:6412. https://doi.org/10.7717/peerj.6412
Liao D, Zhang CC, Li H, Lambers H, Zhang FS (2020) Changes in soil phosphorus fractions following sole cropped and intercropped maize and faba bean grown on calcareous soil. Plant Soil 463:589–600. https://doi.org/10.1007/s11104-020-04460-0
Mahallati M, Koocheki A, Mondani F, Feizi H (2015) Amir moradi, S. Determination of optimal strip width in strip intercropping of maize (Zea mays L.) and bean (Phaseolus vulgaris L.). J Clean Prod 106:343–350. https://doi.org/10.1016/j.jclepro.2014.10.099
Mitran T, Meena RS, Lal R, Layek J, Kumar S, Datta R (2018) Role of soil phosphorus on legume production. Legum Soil Heal Sustain Manag:487–510. https://doi.org/10.1007/978-981-13-0253-4_15
Mkuhlani S (2021) Maize yields from rotation and intercropping systems with different legumes under conservation agriculture in contrasting agro-ecologies. Agric Ecosyst Environ 306:107170. https://doi.org/10.1016/j.agee.2020.107170
Mpanga I, Dapaah H, Geistlinger J, Ludewig U, Neumann G (2018) Soil type-dependent interactions of P-solubilizing microorganisms with organic and inorganic fertilizers mediate plant growth promotion in tomato. Agro 8:213. https://doi.org/10.3390/agronomy8100213
Mtimet A (2001) Soils of Tunisia. Options Méditerr 34:243–268
Mupangwa W, Nyagumbo I, Liben F, Chipindu L, Craufurd P, Mkuhlani S (2021) Maize yields from rotation and intercropping systems with different legumes under conservation agriculture in contrasting agro-ecologies. Agric Ecosyst Environ 306:107. https://doi.org/10.1016/j.agee.2020.107170
Nasto MK, Osborne BB, Lekberg Y, Asner GP, Balzotti CS, Porder S (2017) Nutrient acquisition, soil phosphorus partitioning and competition among trees in a lowland tropical rain forest. New Phytol 214:1506–1517. https://doi.org/10.1111/nph.14494
Neji M, Kouas S, Gandour M, Aydi S, Abdelly C (2019) Genetic variability of morpho-physiological response to phosphorus deficiency in Tunisian populations of Brachypodium hybridum. Plant Physiol Biochem 143:246–256. https://doi.org/10.1016/j.plaphy.2019.09.006
Ning C, Qu J, He L, Yang R, Chen Q, Luo S (2017) Improvement of yield, pest control and Si nutrition of rice by rice–water spinach intercropping. Field Crops Res 208:34–43. https://doi.org/10.1016/j.fcr.2017.04.005
Pierre JF, Latournerie-Moreno L, Garruña-Hernández R, Jacobsen KL, Guevara-Hernández F, Laboski CA, Ruiz-Sánchez E (2022) Maize legume intercropping systems in southern Mexico: a review of benefits and challenges. Ciência Rural 52:11. https://doi.org/10.1590/0103-8478cr20210409
Rakhimova OV, Khramoy VK, Sikharulidze TD, Yudina IN (2021) Influence of nitrogen fertilizers on protein productivity of vetch-wheat grain under different water supply conditions. Casp J Environ Sci 19:951–954. https://doi.org/10.1016/j.agwat.2022.108000
Saini VK, Bhandari SC, Tarafdar JC (2004) Comparison of crop yield, soil microbial biomass C, N, and P, N-fixation, nodulation and mycorrhizal infection in inoculated and non-inoculated sorghum and chickpea crops. Field Crop Res 89:39–47. https://doi.org/10.1016/j.fcr.2004.01.013
Schaap KJ, Fuchslueger L, Hoosbeek MR, Hofhansl F, Martins NP, Valverde-Barrantes OJ (2021) Litter inputs and phosphatase activity affect the temporal variability of organic phosphorus in a tropical forest soil in the Central Amazon. Plant Soil 469:423–441. https://doi.org/10.1007/s11104-021-05146-x
Schwerdtner U, Spohn M (2021) Interspecific root interactions increase maize yields in intercropping with different companion crops. J Plant Nutr Soil Sci 184:596–606. https://doi.org/10.1002/jpln.202000527
Sridhar HS, Salakinkop SR (2021) Competitive functions, pest dynamics and bio economic analysis in traditional maize and legumes intercropping systems under rain fed situation of South India. Indian J Tradit Knowl 20:827–837. https://doi.org/10.9734/ijecc/2023/v13i82070
Tang X, Zhang C, Yu Y, Shen J, Van der Werf W, Zhang F (2021) Intercropping legumes and cereals increases phosphorus use efficiency; a meta-analysis. Plant Soil 460:89–104. https://doi.org/10.1007/s11104-020-04768-x
Tindwa HJ, Kachiguma A, Mrema JP (2019) Incubation of soil with agricultural lime and phosphorus enhances biological nitrogen fixation and yield of soybean (Glycine max L.) in an ultisol. J Cent Eur Agric 20:938–952. https://doi.org/10.5513/JCEA01/20.3.2118
Tiziani R, Mimmo T, Valentinuzzi F, Pii Y, Celletti S, Cesco S (2020) Root handling affects carboxylates exudation and phosphate uptake of white lupin roots. Front Plant Sci 11:584568. https://doi.org/10.3389/fpls.2020.584568
Tripathi SC, Venkatesh K, Meena RP, Chander S, Singh GP (2021) Sustainable intensification of maize and wheat cropping system through pulse intercropping. Sci Rep 11:1–10. https://doi.org/10.1038/s41598-021-98179-2
Ullah S, Khan MY, Asghar HN, Akhtar MJ, Zahir ZA (2017) Differential response of single and co-inoculation of Rhizobium leguminosarum and Mesorhizobium ciceri for inducing water deficit stress tolerance in wheat. Ann Microbiol 67:739–749. https://doi.org/10.1007/s13213-017-1302-2
Walkley A, Black IA (1934) An examination of Degtjareff method for determining soil organic matter and a proposed modification of the chromic acid titration method. Soil Sci 37:29–37. https://doi.org/10.1097/00010694-193401000-00003
XLSTAT X (2017) Data analysis and statistical solution for Microsoft Excel. Addinsoft, Paris
Xu Y, Lei B, Tang Y (2018) Effects of wheat-faba bean intercropping on soil microbial community structure in the rhizosphere. Agric Sci 09:1389–1400 https://www.scirp.org/journal/as
Zhang C, Yu Y, Shen J, Van der Werf W, Zhang F (2021a) Intercropping legumes and cereals increases phosphorus use efficiency; a meta-analysis. Plant Soil 460:89–104. https://doi.org/10.1007/s11104-020-04768-x
Zhang W, Zhang Y, An Y, Chen X (2021b) Phosphorus fractionation related to environmental risks resulting from intensive vegetable cropping and fertilization in a subtropical region. Environ Pollut 269:116098. https://doi.org/10.1016/j.envpol.2020.116098
Zhao K, Wu Y (2014) Rhizosphere calcareous soil P-extraction at the expense of organic carbon from root-exuded organic acids induced by phosphorus deficiency in several plant species. Soil Sci Plant Nutr 60:640–650. https://doi.org/10.1080/00380768.2014.934191
Funding
The authors extend their appreciation to the Deanship of Scientific Research at King Khalid University, Abha, Saudi Arabia, for funding this work through the Small Groups Project under grant number S.R.G.P./288/44.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hamdi, W., L’taief, B., Souid, A. et al. Durum Wheat–Chickpea Intercropping Improves Soil Phosphorus Status and Biomass Production Under Small Soil Inputs. J Soil Sci Plant Nutr 24, 1349–1361 (2024). https://doi.org/10.1007/s42729-024-01638-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42729-024-01638-8