Abstract
Lignin is a major by-product of the wood and paper industries. Valorization of this complex organic polymer, through the creation of novel high-value side products, is a key aspiration of these industries. Due to its unique chemical composition, lignin can be efficiently used to produce many fine chemicals. In this review, we discuss various techniques and strategies used to synthesize lignin-derived functional materials. The transformation of lignin during pyrolysis and hydrothermal carbonization is also reported. Furthermore, current applications and surface properties of lignin-based carbonaceous materials in the fields of catalysis, bio-adsorption removal of pollutants, and supercapacitors are summarized.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Imprudent fossil fuel consumption has polluted the environment and warmed the globe, necessitating the need for bio-renewable energy sources that are clean and cost-effective. Energy sources such as hydroelectric, solar, wind, and biomass reduce the use of fossil fuels and resulting environmental issues. However, many renewable energy sources generate electricity and require high-energy storage capacities. Bio-materials, such as lignin, can play a role in sustainable energy storage. Development of novel organic energy storage systems is complex and requires that the synthesis of these value-added materials should be cost-effective, environmentally benign and readily scalable into mass production. Carbon-based biomaterials have been used for CO2 capture [69, 167] in catalysis [88, 94, 142], and pollutant capture [4, 37]. In addition, they have found application in gas storage [1, 133, 166], as electrode materials, supercapacitors and in fuel cells [124, 178].
Carbon-based materials can be broadly classified as amorphous or crystalline. Amorphous carbon compounds include carbon black, activated carbon and likewise, crystalline carbon materials include carbon nanotubes, nanofibers and graphene. Both amorphous and crystalline carbon-based materials are synthesized by carbonization of polymers (synthetic and natural) [174], chemical vapor deposition [87] and arc discharge [156] leading to the development of materials with various porosities and functionalities. However, those strategies are typically time-consuming and require hazardous organic solvents at high temperatures, and complicated electrochemical analysis for characterization [162]. In addition, most of these methods need fossil fuel-based precursors such as methane or ethane and also require metal catalysts. Due to these reasons, these methods are currently not considered to be cost effective or environmentally friendly for synthesizing carbon-based materials [148].
Lignocellulosic biomass, a naturally abundant bio-renewable resource, can be used to synthesize functional carbon materials [62]. The three major components of lignocellulosic biomass include cellulose, lignin, and hemicellulose [176]. Among these, lignin, an abundant source of aromatic compounds, accounts for 15–35% of the lignocellulosic biomaterial [10]. Despite its abundance, lignin is usually treated as a waste or a low-value material from the paper and pulp industry. The global annual production of chemical pulp is about 150 million tons per year, and the estimated amount of lignin from the pulp making process is about 70 million tons per year [18]. Utilization of lignin for generating heat or energy constitutes about 95% of the total lignin market. Some of the disadvantages of utilizing lignin for generating heat or energy include formation/release of organic pollutants such as polycyclic aromatic hydrocarbons [28, 120], oxygenated polycyclic aromatic hydrocarbons and dioxins, which are generated during incomplete combusion of lignin [147]. Incomplete combustion of lignin can also cause the formation of particulate matter which in turn leads to both environmental and health issues [110].
Lignin is also used as a food and concrete additive, dispersants, resin, and binding material, contributing to about 65% of the remaining 5% lignin market [18]. Finally, industrial lignin is likewise utilized for some conventional applications in the water treatment industry, and as cleaners, emulsifiers, and additives in the rubber industry and expanders in battery generation [60].
More recently, there has been a rapid development towards the synthesis of lignin-based carbon fiber materials using fiber-spinning technology. Lignin-based carbon fibers have the potential to be used for diverse applications, including high-temperature insulating materials [147, 176], and the capture of carbon dioxide [110] and other gases [18]. Moreover, these materials can be applied for synthesizing supercapacitors and macromolecules [13]. Nevertheless, obtaining such high-value-added structural lignin-based carbon fibers is very challenging due to limitations in current spinning technology, leading to carbon fibers with inferior mechanical properties (low tensile strength and elastic modulus) [99].
Incorporation of lignin as an additive into polymeric materials is another valorization strategy that can be used for modifying the polymers towards specific applications [40]. For example, lignin can be incorporated into thermoset materials such as phenol–formaldehyde, urea–formaldehyde, and epoxy resins [70, 180]. Likewise, lignin can be blended with thermoplastic polymers such as polyesters, polyamides, polyacrylonitrile, and polyethylenes, based on compatibility with chemical matrices, for facilitating the extrusion process [27, 85, 109, 112]. Therefore, the strong chemical compatibility of lignin and synthetic polymers can result in blends, which are more hydrophobic than lignin or polymers, in turn making these blends suitable for external applications such as making roofs [85].
There is significant interest in finding renewable replacements for unsaturated petrochemicals (ethylene, propylene, and butadiene) and aromatic compounds (benzene, toluene, and xylene derivatives), from lignin. For example, the US Department of Energy has estimated the consumption of biomass in the biorefinery industry to be about 1.3 billion tons, out of which 22.5 million tons to have potential for conversion towards chemicals and fuels production [116]. If we assumed that 20% of the biorefinery lignin can be utilized for production of aromatic and linear hydrocarbons, we could reduce petrochemicals use in the US by approximately 10% [118].
Significant efforts are currently underway towards the development of an environmentally friendly path to utilize and recycle lignin in a sustainable fashion. However, the sustainable recycling of lignin has not yet been achieved and is challenging due to the depolymerization process and difficulties in product separation [169]. Furthermore, complex structures present in lignin vary with species and growth conditions, this means that a lignin depolymerization strategy will not translate across various plant species.
The most significant bond in a lignin matrix is the β-O-4 linkage, which is very difficult to cleave via conventional mild methods such as hydrolysis or micro-biological techniques [28, 176]. Therefore, lignin depolymerizations are almost always carried out under harsher conditions. One such route is by the use of metal catalysts such as Pt, Pd, or Ni, which can catalytically cleave the β-O-4 linkages under high pressure and temperature [72, 139, 151]. Similarly, a homogenous acid–base catalytic process can also be used to cleave β-O-4 linkages at high temperatures [113, 123]. These strategies require rigorous conditions, complex instrumentation, and expensive catalysts, therefore are not ideal for scalable industrial production.
An alternative to catalyst-based depolymerization is the thermo-chemical conversion of lignin into value-added fine chemicals or compounds. This is a sustainable, cost effective and environmentally benign process [39, 155]. For instance, pyrolysis, hydrothermal carbonization, and gasification are thermo-chemical conversion processes that can be used for generating fine chemicals and materials from lignin [2, 6, 9, 12, 16, 21, 25, 49, 58, 66, 73, 86, 96, 150, 154, 159, 179]. Thermochemical conversion technologies have benefits compared to direct burning, for example, the generation or emission of hazardous gases is much lower in thermo-chemical processes than with direct burning. Also, the production of value-added chemicals through thermochemical conversion are possible while none are obtained by incineration [20]. Generating valuable materials and chemicals from lignin depolymerization through thermo-chemical processes has therefore become a hot research area in the past few years as several phenolic materials can be generated in high yields. This is due to the presence of aromatic functional groups in high concentrations in the lignin macromolecule [80, 123, 139, 158].
The effective valorization of lignin can help reduce the emission of greenhouse gases. For instance, lignin processing can generate bio-renewable fuels and value-added chemicals which have low carbon content and consequently a minimal effect on human food. In addition, lignin conversion to value-added fine chemicals and materials will contribute to the growth of the lignocellulosic biorefinery industry for conversion of biomass (carbohydrates) to biofuels [10].
1.1 Functional materials via thermochemical conversion of lignin
For the rational and effective design of the thermochemical process and catalysts for lignin valorization, an in-depth understanding of the chemical structure, and properties along with possible reactions and mechanisms of lignin are necessary. Value-added fine chemicals, fuels and different types of materials can be achieved by various separations, isolation, and chemical conversions techniques. These bio-derived chemicals can be obtained by a catalytic reaction pathway as part of a biorefinery process [121]. Therefore, fundamental information regarding lignin chemistry, structure, and possible depolymerization mechanisms will be discussed in detail.
1.2 Lignocellulosic biomass
Lignocellulosic biomass is a renewable and abundant resource of three biopolymers: cellulose, hemicellulose, and lignin. Lignin is a complex aromatic bio-polymer made of hydroxycinnamyl alcohol building blocks, and is typically not soluble in water [122]. Various chemical bonds are present in lignin in a non-regular sequence. However, in cellulose, the sequence of monomeric units is well defined by regular β-1,4-glycosidic bonds, making any elucidation of chemical reactions harder in the case of lignin [122]. Lignins typically have higher carbon concentration and lower oxygen content than polysaccharides, including cellulose. Because of its high carbon content, lignins are an attractive feedstock for biofuels and production of chemicals, including benzene, toluene, and xylenes. The key challenge, however, is to efficiently separate the oxygenated aromatic compounds [121, 122].
1.3 Lignin chemistry and its structure
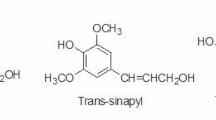
Lignin has a complex phenolic chemical structure containing phenyl propane type motifs. As can be seen in Fig. 1, lignin acts as a filler between cellulose and hemicelluloses and serves to hold the lignocellulosic matrix together [176]. The lignin biopolymer is composed of the three main monomers (p-coumaryl alcohol, sinapyl alcohol, and coniferyl alcohol) (Fig. 2). The biomass constitutes 40% of these three monomers [176].

(Reprinted with permission from Zakzeski et al. [176]. Copyright (2010) American Chemical Society)
Schematic representation of lignin structure and its location in the lignocellulosic biomass
Structural elucidation of lignin has revealed that 50% of lignin’s constituents contain aromatic rings. Five major types of bonds (linkages) in the lignin matrix include β-O-4 ether, β-5 phenylcoumaran, β-β′ pinoresinol, diphenyl ether 4-O5′, and β-1′ diphenyl methane [176]. Linear or cyclic C–O–C bonds are considered to be the most important bonding pattern in lignin. Likewise, the most abundant linkage in both softwood and hardwood-lignin is the β-O-4 linkage. The lignin composition of alcohols (p-coumaryl, coniferyl, and sinapyl alcohols) varies depending on the type of lignin species (softwood vs. hardwood). For instance, coniferyl alcohols account for ca. 90% of soft wood lignin, whereas in the case of hard wood lignin, coniferyl, and sinapyl alcohols exists in equal composition [14]. Hard wood lignin also contains more linear linkages than soft wood lignin due to the existence of additional methoxy groups on aromatic rings, in turn preventing the formation of 5-5 and dibenzodioxocin bonds.
Lignin has a non-crystalline (amorphous) structure due to its highly complex branched configuration [120]. Cellulose and hemicellulose, are hydrophilic and soluble in water, due to the presence of aromatic functional groups through the β-O-4 linkage. Lignin is hydrophobic and insoluble in water at acidic or neutral pH. However, lignin can be dissolved in a variety of organic solvents, including ethanol, acetone, and chloroform, as well as in alkali solutions [169]. Since lignin carbon atoms possess a lower chemical valence than those in polysaccharides, the average redox number of a carbon atom in lignin is negative (− 0.4), while the average carbon atom redox in the polysaccharides is zero. This indicates that lignin has a higher energy content than polysaccharides [100].
Lignin in secondary cell wall can be characterized through high resolution solution NMR and solid state NMR techniques [51, 117]. The solution NMR characterization significantly changes the physical states and various interactions of lignin with other cell wall motifs which in turn leads to unreliable information towards understanding of cell wall structure [75, 117]. Therefore, a new technique called solid state NMR has been intorduced in order to get more precise and certain information about interactions of lignin and polysaccharides and how they are packed in intact secondary cell walls [75, 117]. The solid state NMR spectroscopy combined with dynamic nuclear polarization (DNP) technique provide useful information about motifs interactions in the secondary cell walls. For instance, as can be seen in Fig. 3, the prominent interactions of lignin units with xylan are observed but the direct interactions of cellulose with lignin are less dominant. These findings can facilitate our understanding towards molecular structure of secondary cell walls in plants which in turn modify the biomass depolymerization process and its conversion to fine chemicals [75, 117].

(Reprinted with permission from Kang et al. [75], Copyright (2019) Nature)
The structural model of secondary cell wall. Cellulose microfibrils, two-fold and three-fold xylan, and lignin are described in red, purple, blue and yellow, respectively. The secondary cell wall contains two hydrophobic cores of lignin and cellulose which are bridged by xylans. The interaction of polar motifs of xylan with lignin are boxed in blue. Polysaccharides are abbreviated as polysac
1.4 Lignin depolymerization mechanisms
Due to the complex nature of the β-O-4 linkages in lignin, there exists no effective way to biochemically cleave ether bonds via enzymes and microorganisms for producing value-added materials and chemicals [128]. One alternate approach is to use thermochemical processes such as hydrothermal carbonization, pyrolysis, and gasification, for producing value-added compounds from lignin. Irrespective of lignin type (alkali, sulfite, and Kraft) or origin (soft wood and hard wood), lignin can be thermochemically processed to generate value-added aromatic compounds and phenols [65]. However, the physical changes and chemical processes that occur during thermochemical processes are complicated. Several processes have been reported with variations in reaction conditions such as the type of reactor, heating rate, and reaction temperature. For optimized conversion of lignin to the desired product, it is necessary to know thermochemical conversion mechanisms. Therefore, in the following section, physical and chemical changes in lignin that occur during thermochemical processes such as pyrolysis and hydrothermal carbonization are discussed [65, 181].
1.5 Pyrolysis
Pyrolysis is a thermal decomposition process that typically takes place in a fluidized bed reactor under oxygen-free conditions [181]. Based on the heating rates, pyrolysis can be classified as slow and fast. Significant products generated during lignin pyrolysis include volatile organic compounds such as methanol, ethanol, acetone, acetaldehyde, and phenol derivatives like guaiacol, syringol, catechol, and other mono-lignols. Gaseous compounds such as H2, CH4, C2H4, C2H6, CO, and CO2 are also produced during lignin pyrolysis. In addition to producing bio-renewable energy products such as bio-oil and synthesis gas, large scale biochar production is feasible from the lignin pyrolysis process. Biochar is a thermally stable solid material, which can have a large surface area known for possessing abundant hydroxyl and carbonyl groups that can undergo further functionalization processes [172].
Depolymerization or pyrolysis of hemicellulose and cellulose is a simpler process than pyrolysis because cleavage of lignocellulosic biomass occurs within a narrow range of temperatures. Lignin depolymerization through pyrolysis occurs over a wider range of temperatures, beginning with the cleavage of weaker bonds such as hydrogen and C–OH bonds at low temperatures. Subsequently, the process proceeds through the cleavage of stronger linkages such as β-O-4 bonds at higher temperatures [172]. Initial cleavage of β-O-4 linkage can generate free radicals, in turn leading to the production of aldehydes, styrenes, toluols and guaiacyl hydroxyls. In the later stages of the depolymerization process, p-hydroxy phenols, catechols, and cresols are typically generated [17, 33]. All lignin decomposition stages can be monitored by nuclear magnetic resonance (NMR) spectroscopic methods, in particular, using 1H and 13C NMR [82]. Auxiliary products can be obtained during the lignin decomposition process. For example, the generated free radicals can capture protons leading to the formation of small molecules such as vanillin and 2-methoxy-4-methylphenol. These radicals can be used for facilitating the chain propagation of small molecules into oligomers. The propagating species then undergo a termination step when two radicals collide generating stable oligomeric materials. Via a similar route, polymeric biochar regenerates via re-polymerization of random radicals at higher temperatures (above 350 °C) as schematically shown in Fig. 3 [33].
Pyrolysis of lignin produces biochar and a range of other fine chemicals. Phenolic materials and H2 gas are also considered to be sustainable products of lignin pyrolysis [24]. When a Ni-based catalyst was employed for the production of bio-oil from steam reforming of lignin at 800 °C, a maximum yield of ca. 17-gram hydrogen for 100 g bio-oil was obtained. Likewise, the concentration of phenolic compounds in the organic phase of bio-oil was about 0.5 g per gram of bio-oil, indicating generation of high concentrations of bio-oil as well as phenolic compounds [92]. Some of the prominent reactions that occur in the catalytic generation of hydrogen gas from bio-oil involve the following reactions [46]:
A summary of the chemical processes involved in the generation of liquid fuels, syngas, biochar, and chemicals from lignin is illustrated in Fig. 4.

(Reprinted with permission from Chu et al. [33], Copyright (2013), Royal Chemical Society)
Mechanism of lignin decomposition during the pyrolysis process
1.6 Hydrothermal carbonization
Hydrothermal carbonization takes place under high temperatures and in liquified conditions to transform both the physical and chemical characteristics of biomass [119]. Under high pressure, lignin has an increase in solubility, and enhanced chemical and physical interaction with water. Hydrothermal carbonization typically involves the hydrolysis and cleavage of ether linkages (C–O–C) and C–C bonds, dealkylation reactions such as demethylation and condensation also occur, with these processes and reactions competing with each other [15, 74].
In the hydrothermal carbonization of lignin, the β-O-4 linkages and αC-βC bonds are cleaved from 330 to 400 °C and pressure of 50–115 MPa, however, C–C bonds in the aromatic rings are not affected [41]. At lower temperatures and shorter times, the hydrothermal carbonization of lignin produces both phenolic monomers and dimers via primary hydrolysis and cleavage of ether and aliphatic bonds [136]. After prolonged times and at high temperatures, demethoxylation and alkylation reactions of phenolic materials occur leading to the production of several alkyl phenols (Fig. 5) [74].
Only recently, structural changes in the char residue were observed after hydrothermal carbonization. The results suggest the promotion of lignin decomposition at high temperatures. The char formed at these high temperatures were observed to possess rough surfaces with highly ordered crystalline structures, albeit with few vesicles and low surface area. Furthermore, most functional groups present on the char surface were eliminated at temperatures above 350 °C, hydroxyl groups were an exception and can subsequently be utilized for further functionalization [63].
A key advantage of hydrothermal carbonization over pyrolysis is improved lignin solubility, without the associated pretreatment cost [136]. Unlike the pyrolysis process which has been industrially used for decades, hydrothermal carbonization is not yet commercially utilized due to the necessity for high capital investment [36]. Feeding feedstock into the reactor under harsh reaction conditions (high pressure), along with the heat and mass transfer phenomena, are some of the issues associated with the process [136]. Issues associated with the heat and mass transfer phenomena could lead to the Wall effect [119]. Despite these drawbacks, when the abovementioned issues are resolved, the hydrothermal carbonization technology offers excellent potential for producing value-added compounds and functional carbon compounds from lignin.
1.7 Biochar functionalization
Thermochemical transformation of lignin produces a low functional group char which restricts its widespread application. Biochar surface functionalization can create active sites for catalysis and bio-adsorption of pollutants. For example, biochar with an ordered porous structure, functional groups, and large surface area can be beneficial towards high mass transfer capacity for energy storage applications or for actively loading capacity for catalysis applications.
1.8 Surface functionalization
Surface modification is carried out to increase functional groups, such as amine, hydroxyl, carbonyl, carboxylic and sulfonic groups, on the bio-char surface. For example, the presence of carboxylic and hydroxyl functional groups on the surface considerably increases the adsorption of heavy metals via hydrogen bonding or complexation by other means [59, 89, 170]. Likewise, the presence of amine group on the biochar surface provides active alkaline (basic) sites on the char surface thereby greatly enhancing carbon dioxide capture and removal of acidic contaminants [1, 32, 175]. Similarly, the introduction of an acidic functional group like SO3H via high concentrations of sulfuric acid or its derivative, chlorosulfuric acid [5, 38, 77, 90, 164], affords acidic properties to the bio-char surface, which can then be employed as a solid catalyst to accelerate chemical reactions [146]. Other surface modification reactions include surface oxidation using hydrogen peroxide, ozone, potassium permanganate and nitric acid; amination; and sulfonation [3, 52, 144, 165]. Oxygen containing functional groups such as carboxyl, phenolic, hydroxyl, lactones, and peroxides can be added to the surface of biochar through exposure to ozone using an ozone generator and oxygen using a plasma reactor.
Lignin-based acid catalysts have also been prepared via slow pyrolysis [107]. In this approach, the pyrolysates from slow pyrolysis are sulfonated to obtain the acid-catalysts. Lignin-based basic catalysts can be prepared using a conventional strategy in order to introduce amine functional groups to the biochar surface with ammonia at high temperatures. This process is accompanied by a pre-oxidation process to enhance the reactivity by increasing the oxygen content on the char surface [67, 131, 138]. Alternatively, chemical functionalization of the char surface can also be performed using amine-containing compounds such as 3-chloropropylamine, tris(2-aminoethyl)amine, and polyethyleneimine that are more cost effective, economically viable, and environmentally benign than ammonia (NH3) treatment [97]. Amino containing bio-chars can be used as powerful tools (materials) for selective adsorption of acidic contaminants or pollutants.
As mentioned earlier, surface modification of lignin-derived biochar provides diverse opportunities in many different fields such as use as catalysts and in removing pollutants from the environment. Nevertheless, complexity in obtaining a large heterogeneous surface area remains a major hurdle for its effective utilization in these applications.
1.9 Modulating the porosity and surface area
One of the major challenges with the introduction of different functional groups onto the biochar surface is their effect on biochar porosity and surface area. In general, bio-char generated from thermochemical conversion of lignin results in fewer micropores with a lower specific surface area. To improve the numbers of micro pores, a pre-activation process is employed for facilitating their use as high performance materials for energy storage, catalysis, and pollutant removal applications. Highly ordered porous structures with large surface areas are typically obtained by employing specific chemicals such as zinc chloride and phosphoric acid as a catalyst during pyrolysis [50, 145] or by performing post-modification reactions through chemical and physical means [114, 173]. Phosphoric acid (H3PO4) and zinc chloride (ZnCl2) are some of the common catalysts that are employed in order to form a regular porous structure in the char. In addition, these catalysts also inhibit the formation of tar during pyrolysis and decrease the required pyrolysis temperature, which in turn leads to the formation of more highly ordered porous structures.
When the mechanistic catalytic activity of phosphoric acid during pyrolysis of Kraft lignin was investigated using thermogravimetry (TGA) and differential thermogravimetry (DTG) techniques, the results indicated that the pyrolysis process proceeds via a set of pseudo-first order reactions involving the formation of water and P2O5. Further analyses revealed that pyrolysis leads to the evaporation of these moieties and subsequent formation of highly ordered biochar with high surface areas and micropores [104]. Like H3PO4, ZnCl2 can also act as a catalyst for biochar activation. ZnCl2 accomplishes this by expanding the lignin fiber at low temperature (below 200 °C) as boiling and melting points of ZnCl2 are low, while also facilitating the depolymerization process. Finally, the production of a well-defined microporous structure of bio-char is realized by removing uniformly distributed ZnCl2 from the char matrix. Different concentrations of ZnCl2 at various temperatures have been impregnated into the kraft lignin. The effect of various parameters, such as concentrations and temperatures, on the ultimate porosity and surface area of biochar generated from pyrolysis of lignin have been evaluated [53]. The general observation is that the increase in temperature from 400 to 500 °C leads to the generation of larger and broader distribution of micropores with volumes up to 1.0 cm3/g. Increasing lignin ZnCl2 content results in an increase in pore volume and surface area. The largest micropore volume (1.8 cm3 g−1) and surface area (1800 m2 g−1) that can be generated is from an impregnation ratio (ZnCl2 to lignin) of 3–2 at 500 °C.
Another commonly used strategy to achieve high porosity is by post-activation or modification of the bio-char. This can be achieved using physical or by chemical means. In physical method, the lignin derived char undergoes a controllable gasification process resulting in the formation of steam and carbon dioxide or a combination of both. The removal of carbon containing reactive functional groups often increases biochar surface area and porosity. The physical process typically encompasses the following reactions [168].
The synthesis of activated carbon with steam at 450 °C is a prime example of physical activation of bio-char [47]. Activated carbon materials-derived from lignin have been synthesized with various pore structures using different reaction conditions, with pore size and volume considerably influenced by pyrolysis and activation temperatures, and activation times [83, 126, 132]. Kraft and hydrolytic lignins have been used as a precursor for obtaining physically activated carbons. Results indicate that inorganic impurities such as sodium carbonate and sodium sulfate in the Kraft lignin greatly influence the pyrolysis mechanism (behavior) in N2 atmosphere and subsequent activation in carbon dioxide or water steam. Due to Kraft lignin’s high reactivity during activation at 750 °C, activated carbons possessed very low micropore volumes. On the other hand, the reduced reactivity of hydrolytic lignin during activation by carbon dioxide, resulted in high surface areas (1400–1600 m2 g−1), micropore volumes (0.5–0.6 cm3 g−1) and micropore widths (1.2–1.4 nm) of char compounds as evaluated by BET analyses [26]. Furthermore, reduction in activation times were observed when sodium chloride and potassium chloride (6.2% and 50 wt%) were impregnated onto the surface of demineralized kraft lignin and subsequently activated at 750 °C in carbon dioxide. This effect was more pronounced with potassium chloride as the impregnating reagent with lower activation temperatures, and under optimal conditions led to the preparation of ultra-microporous char materials with mean pore width of 0.53–77 nm. Potassium has a higher radius to charge ratio than sodium, and it can more effectively activate char in a short period of time [143].
Potassium hydroxide is one of the most frequently used reagents for the chemical activation process. The following reactions occur during the chemical activation process:
In this process, decomposition of the lignin-derived char’s carbonyl, hydroxyl, and carboxyl functional groups, at high activated temperatures, produces volatile compounds (H2O and CO2) and non-volatile potassium carbonate (K2CO3). The in situ formation of CO2 and H2O during chemical activation process plays a role in the growth of pores. The release of carbon monoxide and hydrogen gases along with the gasification of metallic potassium (boiling point; 774 °C) generates a highly ordered porous bio-char [8]. In addition, the in situ formation and efficient intercalation of metallic potassium into the carbon matrix result in expanded carbon lattices [55]. An increase in the activated temperature further leads to the reaction between the newly formed K2CO3 and the carbon matrix forming gaseous species such as CO and K(g), which in turn generates large pores in the bio-char [157]. The activated biochar with highly ordered porous structure and large surface area was obtained.
Electrospinning and templating are other techniques that can be utilized to obtain highly ordered microporous and mesoporous activated carbon fibers from alkaline lignin or lignin- polyethylene oxide hybrid solution. Subsequent, pyrolysis and activation with NaOH or KOH at low impregnation ratio under N2 flow at 850 °C transformed non-porous lignin-hybrid precursor fibers to highly ordered porous activated carbon fibers with a specific surface area of 1400 m2 g−1 and pore volume of 0.91 m2 g−1, respectively [61]. It is clearly evident from this study as well as other studies [11, 44, 126] that KOH concentration and activation temperatures are two key factors during the activation process for modulating the porosity and surface area of biochar [130]. Low concentration KOH leads to the formation of microporous materials with a narrow pore size distribution, while high concentration KOH results in high porosity and increased surface area and pore volumes [11, 42, 43, 126, 163].
With templating, porous structures are created in the lignin-derived char when a colloidal silicate solution, for example, is first infiltrated into a lignin precursor followed by pyrolysis [45] (Fig. 6). In the next step, the template is removed, resulting in the introduction of a mesoporous structure into the char framework. Finally, physical activation, for example, with CO2 results in a mesoporous material with micropores in the framework [45]. Such activated lignin-derived chars typically have a high specific surface area (~ 2000 m2 g−1), with their porosity readily modulated by changing the activation conditions. KOH concentration and activation temperatures have an effect on the microporous structure, while silica concentrations and colloidal silica particle sizes play vital roles in obtaining highly ordered materials with high porosity (Fig. 7).

(Reprinted with permission from Kang et al. [74], Copyright (2011), American Chemical Society)
The production of lignin-derived biochar from the hydrothermal carbonization process

(Reprinted with permission from Fierro et al. [45], Copyright (2013), Elsevier Ltd.)
A schematic representation of the colloidal silica templated synthesis of microporous–mesoporous carbons from kraft lignin
A surfactant, Pluronic F127, has been reported to be a suitable template for fabricating mesoporous activated carbon material from a pre-crossed link lignin gel similar to colloidal silica particles [127]. In this method, the impregnation of Pluronic F127 into the lignin framework is followed by pyrolysis. The resulting mesoporous carbon material are subsequently activated through either physical or chemical means. For example, CO2 and KOH activation results in activated mesoporous carbon materials with highly ordered porous structures with surface area and pore volume as high as 1148 m2/g and and 1.0 cm3/g, respectively. Templating reagents, zeolite Y, Zeolite ß, ZSM-5, and mordenite have also been impregnated into Alcell lignin solutions as carbon precursors for obtaining hierarchically porous carbons [152]. The results indicate that the cationic characteristic and abundant hydroxyl groups in the zeolite framework have profound effects on the surface chemistry of porous carbons. These porous carbons also possess high specific surface area with unique properties such as high adsorption of pollutant, high storage capacity and high active loading of materials, due to high concentrations of pyrrolic, pyridinic, OH, and carboxyl groups formed during the pyrolysis process.
1.10 Characterization of lignin derived functional materials
1.10.1 Surface chemistry characteristics
Various experimental techniques such as X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM) and surface area analysis (BET) are used for materials characterization. In Table 1, the different characterization techniques and their applications are explained.
Lignin-based carbon materials possess different acidic or basic hydrophobic and hydrophilic functional surface groups contributing to material reactivity. The incorporation of heteroatoms such as N, O, P, and S into the aromatic framework of char change the surface of the lignin derived char [23]. The surface acidity can be determined by Boehm titration [22]. Strong acidic functional groups such as COOH can react with NaHCO3, however, there is no reaction between intermediate acidic functional groups such as phenolic OH and lactone and NaHCO3. The intermediate acidic functional groups are able to react with Na2CO3 and NaOH while the weak acidic functional groups such as C=O can only react with NaOH. Therefore, all functional groups can be quantified.
The synthesis of ZnCl2 activated lignin derived char has been reported [98]. The impregnation ratio of ZnCl2 to industrial waste lignin and temperature can significantly affect on the surface acidity of the activated bio-chars. When lignin/ZnCl2 ratio (w/w) was 1/1.5, the highest surface area was obtained. If the pyrolysis temperature increases, the content of oxygen containing functional groups on the surface will decrease [137].
Studies show that there is a considerable surface difference with oxidative pyrolyzed lignin derived activated carbons. When pyrolyzed at about 500 °C with 5% oxygen in helium, there is a 15% decrease in char yield and the surface area of the resulting biochar was very low (5 m2/g). 13C cross polarized magic angle spinning nuclear magnetic resonance spectroscopy (13CP-MAS NMR) clearly shows that the concentration of functional groups such as aromatic OH and aromatic O-CH3 gradually decreases with increasing pyrolysis temperature.1 Increasing pyrolysis temperature above 450 °C leads to elimination of the aromatic-O (150 ppm) and carboxyl (180 ppm) functional groups which in turn results in an increase in aromatic carbon structures on the surface (128 ppm) [31]. The aromatic carbon structure is produced as the main component in bio-char at pyrolysis temperatures higher than 450 °C.
FTIR, temperature-programmed desorption (TPD), and x-ray photoemission spectroscopy (XPS) are used to investigate surface characteristics of biochar obtained from lignin [34, 35, 48]. At different temperatures, TPD by mass spectroscopy can identify the gaseous components such as H2O, CO, and CO2 evolving from the heated solid surface. It has been found that the important functional groups such as phenolic OH, ether bonds (C–O–C), carboxylic group, lactone, and carboxylic anhydrides are present in the surface of biochar [79]. The FTIR spectra of pyrolyzed lignin revealed that hydroxyl containing functional groups gradually decreased upon increasing pyrolysis temperature. Furthermore, an increase in activation time leads to extensive oxidation of alcohols to carboxylic and aldehyde groups. This is illustrated by a gradual increase in the intensity of the absorption peaks at about 1700 cm−1 (C=O stretching vibrations) and a gradual decrease of the absorption peaks around 3330 cm−1 where O–H stretching vibrations are observed [115].
XPS technique can be applied to analyze the chemical composition of the biochar surface. The biochar surface contains carbon (near 90%) with a graphite structure (C–C bonds with a binding energy of 284.4 eV). C–O bonds in hydroxyl groups, ether bonds and C=O make up the remaining bonds in the char surface [11]. It is well known that reaction conditions such as slow pyrolysis, fast pyrolysis, and hydrothermal carbonization and the type of lignin strongly contributed to the compositions and type of functional groups on the resulting surface of biochar. The major components in the fast pyrolysis process include oxygen containing functional groups such as OH, C=O and COOH in char but those functional groups are considered as minor components from slow pyrolysis. In the slow pyrolysis process, the aromatic C–H bonds are found to be dominant.
1.10.2 Biochar structure
Lignin biochar structure characterization can be elucidated using the various techniques. For example, nitrogen adsorption–desorption isotherms are employed to determine surface area and pore volume [91, 177]. X-ray diffraction (XRD), Raman spectroscopy, and energy dispersive X-ray (EDX) spectroscopy are used to characterize the char microstructure. Surface morphology can be observed through scanning electron microscopy (SEM) and transmission electron microscopy (TEM) [29, 137, 160]. XRD results indicate that biochar obtained from lignin is primarily amorphous. However, low crystalline content from a conjugated aromatic structure, has been occasionally observed in the structure of the carbon matrix [11]. A more ordered crystalline char structure and increase in char crystal size can be generated with increasing pyrolysis temperature [95]. Nitrogen adsorption desorption isotherms revealed that CO2 activation (physical activation) results in increased microporous volume and distribution. Also, the produced activated carbon possesses a regular and well developed mesoporous and microporous structure [125].
Lignin derived biochar which is activated by ZnCl2 (chemical activation) demonstrates a large micropore volume of 1 cm3 g−1. The content of its porous structure increases with activation temperature up to 500 °C. However, the porous structure of lignin derived char considerably decreases at 600 °C. The increasing impregnation ratio of ZnCl2 to lignin from 0.4 to 2.3 caused an increase in porosity and pore size of lignin derived char [53]. SEM images exhibit that the formation of melt, precipitation of inorganic salts, liquid phase, vesicles and surface etching can occur during the lignin pyrolysis process [69].
1.11 Applications of lignin-derived activated carbons
The thermochemical conversion of lignin to functional carbon or hybrid materials is considered to be a cost effective and environmentally benign process. These materials have various applications in the fields of catalysis, environmental pollutants removal and energy storage.
1.12 Use of lignin-derived char as a catalyst
Many chemical industries use metal compounds in chemical reactions because they can selectively catalyze organic reactions with high activity. However, these processes are not always cost effective due to high energy requirement [141]. Lignin derived carbon biomaterials are considered to be environmentally friendly compounds with good thermal conductivity. This combination means that they can be used as an alternative materials in some existing industrial chemical reactions.
The introduction of carboxyl, amine, hydroxyl and sulfonyl functional groups on the surface of lignin based biomaterials can induce prominent catalytic properties. For example, activated carbon based materials which contain acidic SO3H groups are employed as a metal free catalyst that can be used in different acid-catalyzed chemical processes such as dehydration, hydrolysis, and esterification [106, 134, 140]. The introduction of high density hydrophilic SO3H onto the biochar surface can create a hydrophilic environment in the carbon matrix that absorb hydrophilic molecules such as water and methanol [149]. Thus, the access of hydrophilic reactants to the SO3H functional groups in the surface of biochar is facilitated.
The incorporation of sulfonyl groups on the surface of the solid acid catalysts results in a considerable increase in the catalytic performance of char [57, 111]. For example, biodiesel (methyl ester) production from the reaction of acidified soybean soapstocks with methanol using lignin-derived carbonaceous acid catalyst has been reported [56]. The sulfonyl groups in the esterification reaction act as Brønsted acid sites for a donation of protons which catalyzes chemical reactions.
Lignin derived solid acid catalysts have been employed for catalytic esterification reaction of acetic acid with ethanol. This material can also catalyze the hydration of 2,3-dimethyl-2-butene [93]. Strong acid catalysts such as acidic cationic exchange resins (Amebrlyst 15 and sulfonated carbon) possess lower catalytic activity than lignin derived solid acid catalysts with lower sulfonyl functional group content. Thus, lignin based solid acid catalysts have an unusually high catalytic activity. This is likely due to its macroporous structure which allows for high mass transfer during a chemical reaction. Hydrolysis is another important reaction catalyzed by solid acid catalysts. Lignin derived solid acid catalyst, without pyrolysis, demonstrates a high catalytic activity for the hydrolysis of sugarcane bagasse, cellulose, and starch.
A solid base catalyst can be applied to catalyze an esterification reaction. The mixture of potassium carbonate with kraft lignin followed by activation at 800 °C under nitrogen flow has led to the synthesis of a solid base catalyst. The resulting lignin derived carbonaceous base catalyst demonstrated high catalytic activity for transesterification of methanol with rapeseed oil in order to produce biodiesel with a yield of 99.6%. The catalyst was reusable and biodiesel yield remained over 82% after four cycles [91]. The preparation of lignin derived base catalyst is an environmentally benign and cost-effective process which produces a reusable material with high catalytic activity. This type of catalyst can be considered as a heterogeneous carbonaceous base catalyst for large scale production of biodiesel.
Lignin based carbonaceous materials can also be used as supports in order to immobilize metals or oxide nanoparticles for catalyzing various chemical reactions. This is due to the high surface area and porosity of lignin derived char. The synthesis of iron nanoparticles incorporated onto activated carbonaceous material has been reported [177]. The FeCl3 has been used to chemically activate lignin at 800 °C and the catalyst was applied for wet peroxide oxidation of phenol. In particular, the introduction of FeCl3 creates a regular porous structure along with the formation of stable iron nanoparticles into the carbon matrix. The iron nanoparticles play an important role in the reduction of the iron leaching wet peroxide oxidation process. The lignin-based carbonaceous materials impregnated with FeCl3 exhibited good stability and catalytic activity. Moreover, the in situ preparation of iron nanoparticles encapsulated into the lignin-derived char has been reported [171]. The catalyst was employed to convert syngas to liquid fuels. The catalyst activity and selectivity towards the CO conversion and production of liquid hydrocarbons was approximately 95% and 68%, respectively after a 1500 h run. Furthermore, during biomass gasification, the full destruction of NOx precursors such as HCN and NH3 using a biochar supported iron particles has been reported [102, 103].
Lignin-derived solid catalysts have been shown to be effective in different chemical reactions. However, the surface area of the lignin-based material is complex and its heterogenous surface characteristics can significantly suppress catalytic activity and selectivity. Moreover, the existence of inorganic elements such as K, Na, Ca, and Mg can negatively affect the catalyst selectivity because they may promote side reactions [19]. However, in some reactions such as methane cracking reactions at high temperature, the inorganic species dispersed on the lignin-derived char greatly increase the catalytic activity of the catalyst [81].
1.13 Lignin-based materials for energy storage applications
Efforts are underway to efficiently employ and store renewable energies from wind, solar and biomass in order to overcome issues associated with a global reduction of fossil fuels and harmful emission of greenhouse gases [135]. Reversible supercapacitors have long lifetimes and high energy and power densities [101] and are able to quickly store intermittent renewable energy [7, 178]. Supercapacitors have been applied in various electrical fields such as mobile electrical systems [101, 135]. Lignin-based carbonaceous materials are considered to be favorable candidates for use in capacitors because the incorporation of surface functional groups can significantly increase performance and amount of energy stored in the supercapacitor. Moreover, many possible surface functional groups are electrochemically inert and can considerably improve the wettability of the carbon electrodes [129]. The abundance of oxygenated functional groups on the surface of char leads to prevention of further oxidation of the carbon matrix which in turn increases the cycle stabilities of electrode materials [105].
The synthesis of a high performance supercapacitor from binder-free and mechanically flexible material has been reported [84]. Alkali lignin in aqueous KOH has been used for the preparation of this material composed of electrospun carbon nanofibers.2 The electrospinning technique was employed in order to mix the aqueous solutions of alkali lignin with polyvinyl alcohol to generate composite nanofiber materials. Subsequently, they were stabilized and carbonized in air and in an inert atmosphere, respectively. The resulting carbon nanofibers containing 70 wt% lignin, have an average diameter of 100 nm and a surface area of 583 m2 g−1. The lignin-derived carbon nanofibers showed outstanding supercapacitor performance having high specific capacitance and superior cycle performance [2].
Lignin-based submicron activated carbon fibers have been used for fabrication of supercapacitors with high energy density [64]. The as-synthesized materials demonstrated favorable electrochemical performance due to their high surface area, high porous structure, hydrophilic surface and superior conductivity. These materials also possess outstanding gravimetric capacitance and cycle stability. Thus, these materials are good candidates for electrical energy storage. The preparation of lignin-based activated carbons containing nanoporous structures with a narrow and tunable pore size distribution has recently been reported. The materials were activated by KOH and applied to organic-based capacitors during their production [108]. The increase in the graphitization degree leads to a reduction in the non-Faradaic charge storage and can increase the specific capacitance values up to 87 F g−1 in an organic electrolyte [68].
In addition to electric double layer supercapacitors, another type of supercapacitors known as pseudocapacitance-based supercapacitors have been prepared by impregnation of electro-active compounds such as metal oxides on the char surface. For example, the self-assembly of nickel oxide nanoparticles incorporated into the mesoporous structure of lignin-based activated carbons for supercapacitors applications has been reported [30]. The as-synthesized materials demonstrate superior specific capacitance, favorable capacitance retention, and outstanding cycle stability.
Lithium ion batteries can be charged quickly and possess a durable life cycle with an outstanding energy density. Hence, they are employed in mobile phones and scalable energy storages. The carbon-based material can be used as the anode or cathode depending on the redox potential of the lithium intercalation compound applied in the batteries [124]. The electrospinning technique was employed to prepare a lignin-based nitrogen-doped free-standing fused carbon fibrous mat from a mixture of lignin/polyethylene oxide (90:10). Subsequently, the produced material was carbonized and thermally annealed with urea [161]. The materials were employed as the anode in the lithium-ion batteries and they demonstrated high electrochemical performance with a favorable specific capacity.
The lignin-derived nanofibers have been synthesized from a mixture of polyacrylonitrile and refined lignin through a simple electrospinning technique [71]. The produced materials were carbonized and stabilized in order to apply them in Na+-ion batteries as free-standing and binder-free anodes. The nanofibers showed a prominent cycle performance.
The abovementioned findings show that lignocellulosic biomass can be used as in the fabrication of cheap, excellent performaning carbon-based electrode compounds for batteries. The porous structure, high density functional groups, high availability of electrolyte ions, and short distances for diffusion processes makes these lignin based materials favorable candidates for use in high performance in electrochemical energy storage.
1.14 Lignin-based materials as pollutant bio-absorbents
There have been many attempts to overcome issues associated with environmental pollution. The surface of lignin-derived materials can have high surface area and possess abundant functional groups and therefore can be used as a favorable bio-absorbents for hazardous heavy metals and organic pollutants. Anthropogenic heavy metals and organic materials (pharaceuticals and agricultural chemicasl) can find their way into the environment and can be toxic to plants, animals and humans. Porous materials can play an important role in the removal of environmental pollutants via adsorption processes. Lignin-based functional materials contain some reactive functional groups (primarily –OH and –COOH) which can be used directly or tuned via surface functionalization methods in order tailor the material for improved environmental pollutant adsorption. For example, a porous, high surface area, lignin-derived carbonaceous material was used to remove Ni(II) from wastewater. An increase in pH led to an increase in Ni(II) adsorption indicating an electrostatic adsorption mechanism [48].
Conventional carbon-based microporous materials demonstrate lower surface adsorption activity towards heavy metals such as Cu2+ than the lignin-derived carbonaceous materials obtained from microwave pyrolysis of lignin [98]. Chemically activated carbonaceous materials possess higher porosity and surface area than the lignin-based porous materials. Thus, it can be concluded that in addition to surface area and porosity, the content of functional groups containing oxygen can play a significant role in the adsorption process. Activated materials were synthesized from pyrolysis of phosphoric acid impregnated lignin resulting in a material with high surface area and ordered mesoporous and microporous structure [54]. This material was used as an adsorbents for the removal of Cr(IV) from wastewater with an adsorption capacity of Cr(IV) was 92.6 mg g−1.
Recently, the Douglas fir-derived biochar was magnetized through Fe3O4 precipitation onto the bio-char surface from an aqueous solution of Fe3+/Fe2+ upon NaOH treatment. The resulting material was used for the removal of pollutants such as 4-nitroaniline, salicylic acid, benzoic acid, and phthalic acid from water [76]. A series of activated carbons were synthesized from acid-precipitated eucalyptus kraft lignin that were carbonized and gasified under a N2 atmosphere at 350–800 °C and CO2 at 750–850 °C. The resulting materials were employed as adsorbents for removal of p-nitrophenol (PNP) which is an organic pollutant in wastewater. The adsorption capacity ranges from 1 to 4.4 mmol/g. The amount of PNP adsorbed varies considerably with initial concentration of adsorbate, pH, temperature, and contact time [35]. It has been found that lignin-derived carbon materials can play a great role as adsorbents for efficient removal of sodium dodecylbenzene sulfonate from wastewater [34]. Moreover, the activated carbonaceous materials prepared from kraft lignin via phosphoric acid chemical activation were employed as favorable adsorbents towards the removal of organic pollutants such as phenol and 2,4,5-trichloro-phenol. The as-synthesized materials present a high surface area with a highly ordered microporous and mesoporous structure. The maximum adsorption capacities for phenol and 2,4,5-trichloro-phenol were 227.3 and 476.2 mg g−1 [54].
Lignin-based carbonaceous materials have performed well when used for the removal of environmental pollutants such as heavy metals and organic chemicals. However, some polycyclic aromatic hydrocarbons and some heavy metals can be released from the char surface during the adsorption processes [78, 153]. Thus, future investigations should focused on the development of more efficient and sustainable techniques for char functionalization in order to decrease the release of pollutants during use as environmental adsorbents.
2 Conclusions
Lignin, a significant component of lignocellulosic biomass, can be an important resource for the production of various fine chemicals, fuels, and functional carbon materials. Lignin valorization is an important process in any modern biorefinery industry. The complex structure and composition of lignin requires many strategies for the production of fine chemicals. Major routes for lignin valorization include pyrolysis and hydrothermal carbonization processes. In this review, we have described the current approaches and techniques for conversion of lignin to a range of carbon-based functional materials. The lignin chemical structure and physical properties are significantly changed during these thermochemical processes. The complex structure of lignin and surface chemistry of lignin-derived carbonaceous materials are discussed. Lignin-derived thermochemial materials from pyrolysis or hydrothermal carbonization often have limited surface area with low functional groups content and low porosity. These materials can be tailored for specific applications following functionalizion and through methods aimed at increasing porosity and surface area. The introduction of abundant functional groups such as hydroxyl, amine, carboxylic, and sulfonyl groups into the surface of lignin-derived carbon materials through oxidation, amination, and sulfonation processes can create high value char materials with a high density of functional groups. The different methods of activation including physical and chemical activation create char materials with high surface area and controllable porosity. Thus, the functionalized lignin-derived carbon compounds have applications in different fields such as catalysis, energy storage, and removal of pollutants.
References
Adelodun AA, Lim YH, Jo YM (2014) Stabilization of potassium-doped activated carbon by amination for improved CO2 selective capture. J Anal Appl Pyrolysis 108:151–159
Akhtar J, Amin NAS (2011) A review on process conditions for optimum bio-oil yield in hydrothermal liquefaction of biomass. Renew Sustain Energy Rev 15:1615–1624
Anfruns A, García-Suárez EJ, Montes-Morán MA, Gonzalez-Olmos R, Martin MJ (2014) New insights into the influence of activated carbon surface oxygen groups on H2O2 decomposition and oxidation of pre-adsorbed volatile organic compounds. Carbon 77:89–98
Apul OG, Karanfil T (2015) Adsorption of synthetic organic contaminants by carbon nanotubes: a critical review. Water Res 68:34–55
Arancon RA, Barros HR Jr, Balu AM, Vargas C, Luque R (2011) Valorisation of corncob residues to functionalised porous carbonaceous materials for the simultaneous esterification/transesterification of waste oils. Green Chem 13:3162–3167
Arena U (2012) Process and technological aspects of municipal solid waste gasification. A review. Waste Manag 32:625–639
Arico AS, Bruce P, Scrosati B, Tarascon J-M, van Schalkwijk W (2005) Nanostructured materials for advanced energy conversion and storage devices. Nat Mater 4:366–377
Armandi M, Bonelli B, Geobaldo F, Garrone E (2010) Nanoporous carbon materials obtained by sucrose carbonization in the presence of KOH. Microporous Mesoporous Mater 132:414–420
Ateş F, Miskolczi N, Borsodi N (2013) Comparision of real waste (MSW and MPW) pyrolysis in batch reactor over different catalysts. Part I: product yields, gas and pyrolysis oil properties. Biores Technol 133:443–454
Azadi P, Inderwildi OR, Farnood R, King DA (2013) Liquid fuels, hydrogen and chemicals from lignin: a critical review. Renew Sustain Energy Rev 21:506–523
Babeł K, Jurewicz K (2008) KOH activated lignin based nanostructured carbon exhibiting high hydrogen electrosorption. Carbon 46:1948–1956
Baggio P, Baratieri M, Gasparella A, Longo GA (2008) Energy and environmental analysis of an innovative system based on municipal solid waste (MSW) pyrolysis and combined cycle. Appl Therm Eng 28:136–144
Baker DA, Rials TG (2013) Recent advances in low-cost carbon fiber manufacture from lignin. J Appl Polym Sci 130:713–728
Balakshin M, Capanema E, Berlin A (2014) Isolation and analysis of lignin-carbohydrate complexes preparations with traditional and advanced methods. Stud Nat Prod Chem 42:83–115
Barbier J, Charon N, Dupassieux N, Loppinet-Serani A, Mahé L, Ponthus J, Courtiade M, Ducrozet A, Quoineaud A-A, Cansell F (2012) Hydrothermal conversion of lignin compounds. A detailed study of fragmentation and condensation reaction pathways. Biomass Bioenergy 46:479–491
Bellomare F, Rokni M (2013) Integration of a municipal solid waste gasification plant with solid oxide fuel cell and gas turbine. Renew Energy 55:490–500
Ben H, Ragauskas AJ (2011) NMR characterization of pyrolysis oils from kraft lignin. Energy Fuels 25:2322–2332
Berlin A, Balakshin M (2014) Chapter 18—Industrial lignins: analysis, properties, and applications. In: Gupta VK, Tuohy MG, Kubicek CP, Saddler J, Xu F (eds) Bioenergy research: advances and applications. Elsevier, Amsterdam, pp 315–336
Bhandari PN, Kumar A, Bellmer DD, Huhnke RL (2014) Synthesis and evaluation of biochar-derived catalysts for removal of toluene (model tar) from biomass-generated producer gas. Renew Energy 66:346–353
Bhaskar T, Matsui T, Kaneko J, Uddin MA, Muto A, Sakata Y (2002) Novel calcium based sorbent (Ca-C) for the dehalogenation (Br, Cl) process during halogenated mixed plastic (PP/PE/PS/PVC and HIPS-Br) pyrolysis. Green Chem 4:372–375
Björklund A, Melaina M, Keoleian G (2001) Hydrogen as a transportation fuel produced from thermal gasification of municipal solid waste: an examination of two integrated technologies. Int J Hydrog Energy 26:1209–1221
Boehm HP (1994) Some aspects of the surface chemistry of carbon blacks and other carbons. Carbon 32:759–769
Brennan JK, Bandosz TJ, Thomson KT, Gubbins KE (2001) Water in porous carbons. Colloids Surf A 187:539–568
Bu Q, Lei H, Zacher AH, Wang L, Ren S, Liang J, Wei Y, Liu Y, Tang J, Zhang Q, Ruan R (2012) A review of catalytic hydrodeoxygenation of lignin-derived phenols from biomass pyrolysis. Biores Technol 124:470–477
Buah WK, Cunliffe AM, Williams PT (2007) Characterization of products from the pyrolysis of municipal solid waste. Process Saf Environ Prot 85:450–457
Carrott PJM, Carrott MMLR, Guerrero CI, Delgado LA (2008) Reactivity and porosity development during pyrolysis and physical activation in CO2 or steam of kraft and hydrolytic lignins. J Anal Appl Pyrolysis 82:264–271
Casenave S, Aït-Kadi A, Riedl B (1996) Mechanical behaviour of highly filled lignin/polyethylene composites made by catalytic grafting. Can J Chem Eng 74:308–315
Chakar FS, Ragauskas AJ (2004) Review of current and future softwood kraft lignin process chemistry. Ind Crops Prod 20:131–141
Chatterjee S, Clingenpeel A, McKenna A, Rios O, Johs A (2014) Synthesis and characterization of lignin-based carbon materials with tunable microstructure. RSC Adv 4:4743–4753
Chen F, Zhou W, Yao H, Fan P, Yang J, Fei Z, Zhong M (2013) Self-assembly of NiO nanoparticles in lignin-derived mesoporous carbons for supercapacitor applications. Green Chem 15:3057–3063
Chen C-P, Cheng C-H, Huang Y-H, Chen C-T, Lai C-M, Menyailo OV, Fan L-J, Yang Y-W (2014) Converting leguminous green manure into biochar: changes in chemical composition and C and N mineralization. Geoderma 232:581–588
Chen Y, Chen J, Chen S, Tian K, Jiang H (2015) Ultra-high capacity and selective immobilization of Pb through crystal growth of hydroxypyromorphite on amino-functionalized hydrochar. J Mater Chem A 3:9843–9850
Chu S, Subrahmanyam AV, Huber GW (2013) The pyrolysis chemistry of a β-O-4 type oligomeric lignin model compound. Green Chem 15(1):125–136
Cotoruelo LM, Marqués MD, Rodríguez-Mirasol J, Rodríguez JJ, Cordero T (2009) Lignin-based activated carbons for adsorption of sodium dodecylbenzene sulfonate: equilibrium and kinetic studies. J Colloid Interface Sci 332:39–45
Cotoruelo LM, Marqués MD, Díaz FJ, Rodríguez-Mirasol J, Rodríguez JJ, Cordero T (2012) Adsorbent ability of lignin-based activated carbons for the removal of p-nitrophenol from aqueous solutions. Chem Eng J 184:176–183
Czernik S, Bridgwater AV (2004) Overview of applications of biomass fast pyrolysis oil. Energy Fuels 18:590–598
Dąbrowski A, Podkościelny P, Hubicki Z, Barczak M (2005) Adsorption of phenolic compounds by activated carbon—a critical review. Chemosphere 58:1049–1070
Dehkhoda AM, West AH, Ellis N (2010) Biochar based solid acid catalyst for biodiesel production. Appl Catal A 382:197–204
Dodds DR, Gross RA (2007) Chemicals from biomass. Science 318:1250–1251
Duval A, Lawoko M (2014) A review on lignin-based polymeric, micro- and nano-structured materials. React Funct Polym 85:78–96
Ehara K, Saka S, Kawamoto H (2002) Characterization of the lignin-derived products from wood as treated in supercritical water. J Wood Sci 48:320–325
Fierro V, Torné-Fernández V, Celzard A (2007) Highly microporous carbons prepared by activation of kraft lignin with KOH. Stud Surf Sci Catal 160:607–614
Fierro V, Torné-Fernández V, Celzard A (2007) Methodical study of the chemical activation of Kraft lignin with KOH and NaOH. Microporous Mesoporous Mater 101:419–431
Fierro V, Torne V, Celzard A (2007) Methodical study of the chemical activation of Kraft lignin with KOH and NaOH. Microporous Mesoporous Mater 101(3):419–431
Fierro CM, Górka J, Zazo JA, Rodriguez JJ, Ludwinowicz J, Jaroniec M (2013) Colloidal templating synthesis and adsorption characteristics of microporous–mesoporous carbons from Kraft lignin. Carbon 62:233–239
Florin N, Harris A (2007) Hydrogen production from biomass. Environmentalist 27:207–215
Fu K, Yue Q, Gao B, Sun Y, Zhu L (2013) Preparation, characterization and application of lignin-based activated carbon from black liquor lignin by steam activation. Chem Eng J 228:1074–1082
Gao Y, Yue Q, Gao B, Sun Y, Wang W, Li Q, Wang Y (2013) Preparation of high surface area-activated carbon from lignin of papermaking black liquor by KOH activation for Ni(II) adsorption. Chem Eng J 217:345–353
García AN, Font R, Marcilla A (1995) Kinetic study of the flash pyrolysis of municipal solid waste in a fluidized bed reactor at high temperature. J Anal Appl Pyrolysis 31:101–121
Gaspard S, Passe-Coutrin N, Durimel A, Cesaire T, Jeanne-Rose V (2014) Chapter 2 Activated carbon from biomass for water treatment, biomass for sustainable applications: pollution remediation and energy. The Royal Society of Chemistry, London, pp 46–105
Giummarella N, Pu Y, Ragauskas AJ, Lawoko M (2019) A critical review on the analysis of lignin carbohydrate bonds. Green Chem 21:1573–1595
Gokce Y, Aktas Z (2014) Nitric acid modification of activated carbon produced from waste tea and adsorption of methylene blue and phenol. Appl Surf Sci 313:352–359
Gonzalez-Serrano E, Cordero T, Rodríguez-Mirasol J, Rodríguez JJ (1997) Development of porosity upon chemical activation of kraft lignin with ZnCl2. Ind Eng Chem Res 36:4832–4838
Gonzalez-Serrano E, Cordero T, Rodriguez-Mirasol J, Cotoruelo L, Rodriguez JJ (2004) Removal of water pollutants with activated carbons prepared from H3PO4 activation of lignin from kraft black liquors. Water Res 38:3043–3050
Gratuito MKB, Panyathanmaporn T, Chumnanklang RA, Sirinuntawittaya N, Dutta A (2008) Production of activated carbon from coconut shell: optimization using response surface methodology. Biores Technol 99:4887–4895
Guo F, Xiu Z-L, Liang Z-X (2012) Synthesis of biodiesel from acidified soybean soapstock using a lignin-derived carbonaceous catalyst. Appl Energy 98:47–52
Hara M, Yoshida T, Takagaki A, Takata T, Kondo JN, Hayashi S, Domen K (2004) A carbon material as a strong protonic acid. Angew Chem Int Ed 43:2955–2958
He M, Xiao B, Liu S, Guo X, Luo S, Xu Z, Feng Y, Hu Z (2009) Hydrogen-rich gas from catalytic steam gasification of municipal solid waste (MSW): influence of steam to MSW ratios and weight hourly space velocity on gas production and composition. Int J Hydrog Energy 34:2174–2183
He H, Qian T-T, Liu W-J, Jiang H, Yu H-Q (2014) Biological and chemical phosphorus solubilization from pyrolytical biochar in aqueous solution. Chemosphere 113:175–181
Holladay JE, Bozell JJ, White JF, Johnson D (2007) Top value-added chemicals from biomass volume II-results of screening for potential candidates from biorefinery lignin DOE Report PNNL, 16983
Hu S, Hsieh Y-L (2013) Ultrafine microporous and mesoporous activated carbon fibers from alkali lignin. J Mater Chem A 1:11279–11288
Hu B, Wang K, Wu L, Yu S-H, Antonietti M, Titirici M-M (2010) Engineering carbon materials from the hydrothermal carbonization process of biomass. Adv Mater 22:813–828
Hu J, Shen D, Wu S, Zhang H, Xiao R (2014) Effect of temperature on structure evolution in char from hydrothermal degradation of lignin. J Anal Appl Pyrolysis 106:118–124
Hu S, Zhang S, Pan N, Hsieh Y-L (2014) High energy density supercapacitors from lignin derived submicron activated carbon fibers in aqueous electrolytes. J Power Sources 270:106–112
Huber GW, Iborra S, Corma A (2006) Synthesis of transportation fuels from biomass: chemistry, catalysts, and engineering. Chem Rev 106:4044–4098
Hwang I-H, Aoyama H, Matsuto T, Nakagishi T, Matsuo T (2012) Recovery of solid fuel from municipal solid waste by hydrothermal treatment using subcritical water. Waste Manag 32:410–416
Jansen RJJ, van Bekkum H (1994) Amination and ammoxidation of activated carbons. Carbon 32:1507–1516
Jeon J-W, Zhang L, Lutkenhaus JL, Laskar DD, Lemmon JP, Choi D, Nandasiri MI, Hashmi A, Xu J, Motkuri RK, Fernandez CA, Liu J, Tucker MP, McGrail PB, Yang B, Nune SK (2015) Controlling porosity in lignin-derived nanoporous carbon for supercapacitor applications. Chemsuschem 8:428–432
Jiménez V, Ramírez-Lucas A, Díaz JA, Sánchez P, Romero A (2012) CO2 capture in different carbon materials. Environ Sci Technol 46:7407–7414
Jin Y, Cheng X, Zheng Z (2010) Preparation and characterization of phenol–formaldehyde adhesives modified with enzymatic hydrolysis lignin. Biores Technol 101:2046–2048
Jin J, Yu B-J, Shi Z-Q, Wang C-Y, Chong C-B (2014) Lignin-based electrospun carbon nanofibrous webs as free-standing and binder-free electrodes for sodium ion batteries. J Power Sources 272:800–807
Jongerius AL, Bruijnincx PCA, Weckhuysen BM (2013) Liquid-phase reforming and hydrodeoxygenation as a two-step route to aromatics from lignin. Green Chem 15:3049–3056
Kageyama H, Osada S, Nakata H, Kubota M, Matsuda H (2013) Effect of coexisting inorganic chlorides on lead volatilization from CaO–SiO2–Al2O3 molten slag under municipal solid waste gasification and melting conditions. Fuel 103:94–100
Kang S, Li X, Fan J, Chang J (2011) Classified separation of lignin hydrothermal liquefied products. Ind Eng Chem Res 50(19):11288–11296
Kang X, Kirui A, Dickwella Widanage MC, Mentink-Vigier F, Cosgrove DJ, Wang T (2019) Lignin-polysaccharide interactions in plant secondary cell walls revealed by solid-state NMR. Nat Commun 10(1):347–355
Karunanayake AG, Todd OA, Crowley ML, Ricchetti LB, Pittman CU, Anderson R, Mlsna TE (2017) Rapid removal of salicylic acid, 4-nitroaniline, benzoic acid and phthalic acid from wastewater using magnetized fast pyrolysis biochar from waste Douglas fir. Chem Eng J 319:75–88
Kastner JR, Miller J, Geller DP, Locklin J, Keith LH, Johnson T (2012) Catalytic esterification of fatty acids using solid acid catalysts generated from biochar and activated carbon. Catal Today 190:122–132
Keiluweit M, Kleber M, Sparrow MA, Simoneit BRT, Prahl FG (2012) Solvent-extractable polycyclic aromatic hydrocarbons in biochar: influence of pyrolysis temperature and feedstock. Environ Sci Technol 46:9333–9341
Khezami L, Chetouani A, Taouk B, Capart R (2005) Production and characterisation of activated carbon from wood components in powder: cellulose, lignin, xylan. Powder Technol 157:48–56
Kijima M, Hirukawa T, Hanawa F, Hata T (2011) Thermal conversion of alkaline lignin and its structured derivatives to porous carbonized materials. Biores Technol 102:6279–6285
Klinghoffer NB, Castaldi MJ, Nzihou A (2015) Influence of char composition and inorganics on catalytic activity of char from biomass gasification. Fuel 157:37–47
Kosa M, Ben H, Theliander H, Ragauskas AJ (2011) Pyrolysis oils from CO2 precipitated Kraft lignin. Green Chem 13:3196–3202
Lai C, Kolla P, Zhao Y, Fong H, Smirnova AL (2014) Lignin-derived electrospun carbon nanofiber mats with supercritically deposited Ag nanoparticles for oxygen reduction reaction in alkaline fuel cells. Electrochim Acta 130:431–438
Lai C, Zhou Z, Zhang L, Wang X, Zhou Q, Zhao Y, Wang Y, Wu X-F, Zhu Z, Fong H (2014) Free-standing and mechanically flexible mats consisting of electrospun carbon nanofibers made from a natural product of alkali lignin as binder-free electrodes for high-performance supercapacitors. J Power Sources 247:134–141
Li Y, Sarkanen S (2002) Alkylated kraft lignin-based thermoplastic blends with aliphatic polyesters. Macromolecules 35:9707–9715
Li AM, Li XD, Li SQ, Ren Y, Chi Y, Yan JH, Cen KF (1999) Pyrolysis of solid waste in a rotary kiln: influence of final pyrolysis temperature on the pyrolysis products. J Anal Appl Pyrolysis 50:149–162
Li X, Cai W, An J, Kim S, Nah J, Yang D, Piner R, Velamakanni A, Jung I, Tutuc E, Banerjee SK, Colombo L, Ruoff RS (2009) Large-area synthesis of high-quality and uniform graphene films on copper foils. Science 324:1312–1314
Li B, Wang J, Yuan Y, Ariga H, Takakusagi S, Asakura K (2011) Carbon nanotube-supported RuFe bimetallic nanoparticles as efficient and robust catalysts for aqueous-phase selective hydrogenolysis of glycerol to glycols. ACS Catal 1:1521–1528
Li M, Liu Q, Guo L, Zhang Y, Lou Z, Wang Y, Qian G (2013) Cu(II) removal from aqueous solution by Spartina alterniflora derived biochar. Biores Technol 141:83–88
Li S, Gu Z, Bjornson BE, Muthukumarappan A (2013) Biochar based solid acid catalyst hydrolyze biomass. J Environ Chem Eng 1:1174–1181
Li X-F, Zuo Y, Zhang Y, Fu Y, Guo Q-X (2013) In situ preparation of K2CO3 supported Kraft lignin activated carbon as solid base catalyst for biodiesel production. Fuel 113:435–442
Li D, Briens C, Berruti F (2015) Improved lignin pyrolysis for phenolics production in a bubbling bed reactor—effect of bed materials. Biores Technol 189:7–14
Liang F, Song Y, Huang C, Zhang J, Chen B (2013) Preparation and performance evaluation of a lignin-based solid acid from acid hydrolysis lignin. Catal Commun 40:93–97
Liu W-J, Tian K, Jiang H, Yu H-Q (2014) Harvest of Cu NP anchored magnetic carbon materials from Fe/Cu preloaded biomass: their pyrolysis, characterization, and catalytic activity on aqueous reduction of 4-nitrophenol. Green Chem 16:4198–4205
Lua AC, Yang T, Guo J (2004) Effects of pyrolysis conditions on the properties of activated carbons prepared from pistachio-nut shells. J Anal Appl Pyrolysis 72:279–287
Luo S, Xiao B, Hu Z, Liu S (2010) Effect of particle size on pyrolysis of single-component municipal solid waste in fixed bed reactor. Int J Hydrog Energy 35:93–97
Ma Y, Liu W-J, Zhang N, Li Y-S, Jiang H, Sheng G-P (2014) Polyethylenimine modified biochar adsorbent for hexavalent chromium removal from the aqueous solution. Biores Technol 169:403–408
Maldhure AV, Ekhe JD (2011) Preparation and characterizations of microwave assisted activated carbons from industrial waste lignin for Cu(II) sorption. Chem Eng J 168:1103–1111
Maradur SP, Kim CH, Kim SY, Kim B-H, Kim WC, Yang KS (2012) Preparation of carbon fibers from a lignin copolymer with polyacrylonitrile. Synth Met 162:453–459
Matthews LR, Niziolek AM, Onel O, Pinnaduwage N, Floudas CA (2016) Biomass to liquid transportation fuels via biological and thermochemical conversion: process synthesis and global optimization strategies. Ind Eng Chem Res 55:3203–3225
Miller JR, Simon P (2008) Electrochemical capacitors for energy management. Science 321:651–652
Min Z, Zhang S, Yimsiri P, Wang Y, Asadullah M, Li C-Z (2013) Catalytic reforming of tar during gasification. Part IV. Changes in the structure of char in the char-supported iron catalyst during reforming. Fuel 106:858–863
Min Z, Lin J-Y, Yimsiri P, Asadullah M, Li C-Z (2014) Catalytic reforming of tar during gasification. Part V. Decomposition of NOx precursors on the char-supported iron catalyst. Fuel 116:19–24
Montané D, Torné-Fernández V, Fierro V (2005) Activated carbons from lignin: kinetic modeling of the pyrolysis of Kraft lignin activated with phosphoric acid. Chem Eng J 106:1–12
Mysyk R, Raymundo-Piñero E, Anouti M, Lemordant D, Béguin F (2010) Pseudo-capacitance of nanoporous carbons in pyrrolidinium-based protic ionic liquids. Electrochem Commun 12:414–417
Nakajima K, Hara M (2012) Amorphous carbon with SO3H groups as a solid brønsted acid catalyst. ACS Catal 2:1296–1304
Namchot W, Panyacharay N, Jonglertjunya W, Sakdaronnarong C (2014) Hydrolysis of delignified sugarcane bagasse using hydrothermal technique catalyzed by carbonaceous acid catalysts. Fuel 116:608–616
Navarro-Suarez AM, Carretero-Gonzalez J, Roddatis V, Goikolea E, Segalini J, Redondo E, Rojo T, Mysyk R (2014) Nanoporous carbons from natural lignin: study of structural–textural properties and application to organic-based supercapacitors. RSC Adv 4:48336–48343
Nitz H, Semke H, Mülhaupt R (2001) Influence of lignin type on the mechanical properties of lignin based compounds. Macromol Mater Eng 286:737–743
Obrist D, Moosmüller H, Schürmann R, Chen LWA, Kreidenweis SM (2008) Particulate-phase and gaseous elemental mercury emissions during biomass combustion: controlling factors and correlation with particulate matter emissions. Environ Sci Technol 42:721–727
Okamura M, Takagaki A, Toda M, Kondo JN, Domen K, Tatsumi T, Hara M, Hayashi S (2006) Acid-catalyzed reactions on flexible polycyclic aromatic carbon in amorphous carbon. Chem Mater 18:3039–3045
Over LC, Hergert M, Meier MAR (2017) Metathesis curing of allylated lignin and different plant oils for the preparation of thermosetting polymer films with tunable mechanical properties. Macromol Chem Phys 218(6):1700177
Papatheofanous MG, Billa E, Koullas DP, Monties B, Koukios EG (1995) Two-stage acid-catalyzed fractionation of lignocellulosic biomass in aqueous ethanol systems at low temperatures. Biores Technol 54:305–310
Passé-Coutrin N, Jeanne-Rose V, Ouensanga A (2005) Textural analysis for better correlation of the char yield of pyrolysed lignocellulosic materials. Fuel 84:2131–2134
Perezdrienko IV, Molodozhenyuk TB, Shermatov BE, Yunusov MP (2001) Effect of carbonization temperature and activation on structural formation of active lignin carbons. Russ J Appl Chem 74:1650–1652
Perlack RD, Wright LL, Turhollow AF, Graham RL, Stokes BJ, Erbach DC (2005) Biomass as feedstock for a bioenergy and bioproducts industry: the technical feasibility of billion ton annual supply DTIC Document
Perras FA, Luo H, Zhang X, Mosier NS, Pruski M, Abu-Omar MM (2017) Atomic-level structure characterization of biomass pre- and post-lignin treatment by dynamic nuclear polarization-enhanced solid-state NMR. J Phys Chem A 121:623–630
Petersen TWAG (2004) Top value added chemicals from biomass volume: Results of screening for potential candidates from sugars and synthesis gas DTIC Document
Peterson AA, Vogel F, Lachance RP, Froling M, Antal JMJ, Tester JW (2008) Thermochemical biofuel production in hydrothermal media: a review of sub- and supercritical water technologies. Energy Environ Sci 1:32–65
Prakash A, Singh R, Balagurumurthy B, Bhaskar T, Arora AK, Puri SK (2015) Chapter 16—Thermochemical valorization of lignin, recent advances in thermo-chemical conversion of biomass. Elsevier, Boston, pp 455–478
Ragauskas AJ, Beckham GT, Biddy MJ, Chandra R, Chen F, Davis MF, Davison BH, Dixon RA, Gilna P, Keller M, Langan P, Naskar AK, Saddler JN, Tschaplinski TJ, Tuskan GA, Wyman CE (2014) Lignin valorization: improving lignin processing in the biorefinery. Science 344:1246843
Rinaldi R, Jastrzebski R, Clough MT, Ralph J, Kennema M, Bruijnincx PCA, Weckhuysen BM (2016) Paving the way for lignin valorisation: recent advances in bioengineering, biorefining and catalysis. Angew Chem Int Ed 55:8164–8215
Roberts VM, Stein V, Reiner T, Lemonidou A, Li X, Lercher JA (2011) Towards quantitative catalytic lignin depolymerization. Chem A Eur J 17:5939–5948
Roberts AD, Li X, Zhang H (2014) Porous carbon spheres and monoliths: morphology control, pore size tuning and their applications as Li-ion battery anode materials. Chem Soc Rev 43:4341–4356
Rodriguez-Mirasol J, Cordero T, Rodriguez JJ (1993) Activated carbons from carbon dioxide partial gasification of eucalyptus kraft lignin. Energy Fuels 7:133–138
Ruiz-Rosas R, Valero-Romero MJ, Salinas-Torres D, Rodríguez-Mirasol J, Cordero T, Morallón E, Cazorla-Amorós D (2014) Electrochemical performance of hierarchical porous carbon materials obtained from the infiltration of lignin into zeolite templates. Chemsuschem 7:1458–1467
Saha D, Li Y, Bi Z, Chen J, Keum JK, Hensley DK, Grappe HA, Meyer HM, Dai S, Paranthaman MP, Naskar AK (2014) Studies on supercapacitor electrode material from activated lignin-derived mesoporous carbon. Langmuir 30:900–910
Sergeev AG, Hartwig JF (2011) Selective, nickel-catalyzed hydrogenolysis of aryl ethers. Science 332:439–443
Sevilla M, Mokaya R (2014) Energy storage applications of activated carbons: supercapacitors and hydrogen storage. Energy Environ Sci 7:1250–1280
Sevilla M, Fuertes AB, Mokaya R (2011) High density hydrogen storage in superactivated carbons from hydrothermally carbonized renewable organic materials. Energy Environ Sci 4:1400–1410
Shafeeyan MS, Wan Daud WMA, Houshmand A, Arami-Niya A (2012) The application of response surface methodology to optimize the amination of activated carbon for the preparation of carbon dioxide adsorbents. Fuel 94:465–472
Shen Q, Zhang T, Zhang W-X, Chen S, Mezgebe M (2011) Lignin-based activated carbon fibers and controllable pore size and properties. J Appl Polym Sci 121:989–994
Shevlin SA, Guo ZX (2009) Density functional theory simulations of complex hydride and carbon-based hydrogen storage materials. Chem Soc Rev 38:211–225
Shimizu K-I, Satsuma A (2011) Toward a rational control of solid acid catalysis for green synthesis and biomass conversion. Energy Environ Sci 4:3140–3153
Simon P, Gogotsi Y (2008) Materials for electrochemical capacitors. Nat Mater 7:845–854
Singh R, Prakash A, Balagurumurthy B, Bhaskar T (2015) Chapter 10—Hydrothermal liquefaction of biomass, recent advances in thermo-chemical conversion of biomass. Elsevier, Boston, pp 269–291
Spokas KA, Novak JM, Masiello CA, Johnson MG, Colosky EC, Ippolito JA, Trigo C (2014) Physical disintegration of biochar: an overlooked process. Environ Sci Technol Lett 1:326–332
Stöhr B, Boehm HP, Schlögl R (1991) Enhancement of the catalytic activity of activated carbons in oxidation reactions by thermal treatment with ammonia or hydrogen cyanide and observation of a superoxide species as a possible intermediate. Carbon 29:707–720
Sturgeon MR, O’Brien MH, Ciesielski PN, Katahira R, Kruger JS, Chmely SC, Hamlin J, Lawrence K, Hunsinger GB, Foust TD, Baldwin RM, Biddy MJ, Beckham GT (2014) Lignin depolymerisation by nickel supported layered-double hydroxide catalysts. Green Chem 16:824–835
Su F, Guo Y (2014) Advancements in solid acid catalysts for biodiesel production. Green Chem 16:2934–2957
Su DS, Zhang J, Frank B, Thomas A, Wang X, Paraknowitsch J, Schlögl R (2010) Metal-free heterogeneous catalysis for sustainable chemistry. Chemsuschem 3:169–180
Su DS, Perathoner S, Centi G (2013) Nanocarbons for the development of advanced catalysts. Chem Rev 113:5782–5816
Suhas S, Carrott PJM, Carrott MMLR (2009) Using alkali metals to control reactivity and porosity during physical activation of demineralised kraft lignin. Carbon 47:1012–1017
Sun C, Snape CE, Liu H (2013) Development of low-cost functional adsorbents for control of mercury (Hg) emissions from coal combustion. Energy Fuels 27:3875–3882
Taberna P-L, Gaspard S (2014) Chapter 9 Nanoporous carbons for high energy density supercapacitors, biomass for sustainable applications: pollution remediation and energy. The Royal Society of Chemistry, London, pp 366–399
Takagaki A, Tagusagawa C, Hayashi S, Hara M, Domen K (2010) Nanosheets as highly active solid acid catalysts for green chemical syntheses. Energy Environ Sci 3:82–93
Tian D, Hu Y, Wang Y, Boylan JW, Zheng M, Russell AG (2009) Assessment of biomass burning emissions and their impacts on urban and regional PM2.5: a Georgia case study. Environ Sci Technol 43:299–305
Titirici M-M, White RJ, Falco C, Sevilla M (2012) Black perspectives for a green future: hydrothermal carbons for environment protection and energy storage. Energy Environ Sci 5:6796–6822
Toda M, Takagaki A, Okamura M, Kondo JN, Hayashi S, Domen K, Hara M (2005) Green chemistry: biodiesel made with sugar catalyst. Nature 438:178
Toor SS, Rosendahl L, Rudolf A (2011) Hydrothermal liquefaction of biomass: a review of subcritical water technologies. Energy 36:2328–2342
Torr KM, van de Pas DJ, Cazeils E, Suckling ID (2011) Mild hydrogenolysis of in situ and isolated Pinus radiata lignins. Biores Technol 102:7608–7611
Valero-Romero MJ, Márquez-Franco EM, Bedia J, Rodríguez-Mirasol J, Cordero T (2014) Hierarchical porous carbons by liquid phase impregnation of zeolite templates with lignin solution. Microporous Mesoporous Mater 196:68–78
Van Wesenbeeck S, Prins W, Ronsse F, Antal MJ (2014) Sewage sludge carbonization for biochar applications. Fate of heavy metals. Energy Fuels 28:5318–5326
Velghe I, Carleer R, Yperman J, Schreurs S (2011) Study of the pyrolysis of municipal solid waste for the production of valuable products. J Anal Appl Pyrolysis 92:366–375
Vispute TP, Zhang H, Sanna A, Xiao R, Huber GW (2010) Renewable chemical commodity feedstocks from integrated catalytic processing of pyrolysis oils. Science 330:1222–1227
Volotskova O, Levchenko I, Shashurin A, Raitses Y, Ostrikov K, Keidar M (2010) Single-step synthesis and magnetic separation of graphene and carbon nanotubes in arc discharge plasmas. Nanoscale 2:2281–2285
Wang J, Kaskel S (2012) KOH activation of carbon-based materials for energy storage. J Mater Chem 22:23710–23725
Wang X, Rinaldi R (2012) Corrigendum: solvent effects on the hydrogenolysis of diphenyl ether with Raney nickel and their implications for the conversion of lignin. Chemsuschem 5:1335
Wang J, Cheng G, You Y, Xiao B, Liu S, He P, Guo D, Guo X, Zhang G (2012) Hydrogen-rich gas production by steam gasification of municipal solid waste (MSW) using NiO supported on modified dolomite. Int J Hydrog Energy 37:6503–6510
Wang D, Zhang W, Hao X, Zhou D (2013) Transport of biochar particles in saturated granular media: effects of pyrolysis temperature and particle size. Environ Sci Technol 47:821–828
Wang S-X, Yang L, Stubbs LP, Li X, He C (2013) Lignin-derived fused electrospun carbon fibrous mats as high performance anode materials for lithium ion batteries. ACS Appl Mater Interfaces 5:12275–12282
Wei L, Sevilla M, Fuertes AB, Mokaya R, Yushin G (2011) Hydrothermal carbonization of abundant renewable natural organic chemicals for high-performance supercapacitor electrodes. Adv Energy Mater 1:356–361
Wei H, Deng S, Hu B, Chen Z, Wang B, Huang J, Yu G (2012) Granular bamboo-derived activated carbon for high CO2 adsorption: the dominant role of narrow micropores. Chemsuschem 5:2354–2360
Wu Y, Fu Z, Yin D, Xu Q, Liu F, Lu C, Mao L (2010) Microwave-assisted hydrolysis of crystalline cellulose catalyzed by biomass char sulfonic acids. Green Chem 12:696–700
Wu L, Sitamraju S, Xiao J, Liu B, Li Z, Janik MJ, Song C (2014) Effect of liquid-phase O3 oxidation of activated carbon on the adsorption of thiophene. Chem Eng J 242:211–219
Xia Y, Yang Z, Zhu Y (2013) Porous carbon-based materials for hydrogen storage: advancement and challenges. J Mater Chem A 1:9365–9381
Xing W, Liu C, Zhou Z, Zhang L, Zhou J, Zhuo S, Yan Z, Gao H, Wang G, Qiao SZ (2012) Superior CO2 uptake of N-doped activated carbon through hydrogen-bonding interaction. Energy Environ Sci 5:7323–7327
Xiong X, Yu IKM, Cao L, Tsang DCW, Zhang S, Ok YS (2017) A review of biochar-based catalysts for chemical synthesis, biofuel production, and pollution control. Biores Technol 246:254–270
Xu C, Arancon RAD, Labidi J, Luque R (2014) Lignin depolymerisation strategies: towards valuable chemicals and fuels. Chem Soc Rev 43:7485–7500
Xu D, Zhao Y, Sun K, Gao B, Wang Z, Jin J, Zhang Z, Wang S, Yan Y, Liu X, Wu F (2014) Cadmium adsorption on plant- and manure-derived biochar and biochar-amended sandy soils: impact of bulk and surface properties. Chemosphere 111:320–326
Yan Q, Wan C, Liu J, Gao J, Yu F, Zhang J, Cai Z (2013) Iron nanoparticles in situ encapsulated in biochar-based carbon as an effective catalyst for the conversion of biomass-derived syngas to liquid hydrocarbons. Green Chem 15:1631–1640
Yang H, Yan R, Chen H, Lee DH, Zheng C (2007) Characteristics of hemicellulose, cellulose and lignin pyrolysis. Fuel 86:1781–1788
Yang K, Peng J, Srinivasakannan C, Zhang L, Xia H, Duan X (2010) Preparation of high surface area activated carbon from coconut shells using microwave heating. Biores Technol 101:6163–6169
Yang Z, Shen J, Jayaprakash N, Archer LA (2012) Synthesis of organic-inorganic hybrids by miniemulsion polymerization and their application for electrochemical energy storage. Energy Environ Sci 5:7025–7032
Yang G, Chen H, Qin H, Feng Y (2014) Amination of activated carbon for enhancing phenol adsorption: effect of nitrogen-containing functional groups. Appl Surf Sci 293:299–305
Zakzeski J, Bruijnincx PCA, Jongerius AL, Weckhuysen BM (2010) The catalytic valorization of lignin for the production of renewable chemicals. Chem Rev 110:3552–3599
Zazo JA, Bedia J, Fierro CM, Pliego G, Casas JA, Rodriguez JJ (2012) Highly stable Fe on activated carbon catalysts for CWPO upon FeCl3 activation of lignin from black liquors. Catal Today 187:115–121
Zhang LL, Zhao XS (2009) Carbon-based materials as supercapacitor electrodes. Chem Soc Rev 38:2520–2531
Zhang L, Champagne P, Xu C (2011) Bio-crude production from secondary pulp/paper-mill sludge and waste newspaper via co-liquefaction in hot-compressed water. Energy 36:2142–2150
Zhao B, Chen G, Liu Y, Hu K, Wu R (2001) Synthesis of lignin base epoxy resin and its characterization. J Mater Sci Lett 20:859–862
Zhou C-H, Xia X, Lin C-X, Tong D-S, Beltramini J (2011) Catalytic conversion of lignocellulosic biomass to fine chemicals and fuels. Chem Soc Rev 40:5588–5617
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The author(s) declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Erfani Jazi, M., Narayanan, G., Aghabozorgi, F. et al. Structure, chemistry and physicochemistry of lignin for material functionalization. SN Appl. Sci. 1, 1094 (2019). https://doi.org/10.1007/s42452-019-1126-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42452-019-1126-8