Abstract
The fate and the accumulation kinetics of mercurychloride (HgCl2) were investigated in the invasive copepod species Pseudodiaptomus marinus, which originates from the North-Western Pacific Ocean and was recently recorded from the Atlantic and Mediterranean coasts. The main objective of this study was to determine lethal concentrations (LC50 %) of HgCl2 in P. marinus and to study its bioaccumulation kinetics in the laboratory. Lethality experiments were performed for 24, 48, 72, and 96 h. Experiments in presence and absence of food source using one sub-lethal concentration of HgCl2 (14.15 μg/L) were carried out to study the uptake, the accumulation and the influence of exposure pathways of HgCl2 in P. marinus. LC50 for 96 h was calculated as 42.4 μg/L in response to HgCl2. The uptake and bioaccumulation kinetics of HgCl2 in P. marinus are not depending on the exposure pathways, where no significant differences were depicted between the uptake/accumulation of HgCl2 from the micro-algal diet and from the seawater medium. Those results could be helpful in the understanding of mercury uptake, bioaccumulation and bio-amplification processes especially concerning invasive copepod species.
Article Highlights
-
LC50 for 96h was calculated as 42.4 μg/L HgCl2.
-
I. galbana uptaked and accumulated Hg more than starved and fed P. marinus.
-
The uptake and the bioaccumulation kinetics of Hg was slow during the 1st and 4th days in starved and fed P. marinus.
-
The uptake and bioaccumulation kinetics of HgCl2 in P. marinus were not depending on the exposure pathway.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mercury (Hg) is a ubiquitous element and is considered as one of the most toxic environmental pollutants (Daye et al. 2013; Øverjordet et al. 2014; Lescord et al. 2015). Mercury contamination is a relevant topic in ecotoxicology, since its fate and consequences in marine organisms depend on different factors such as its physicochemical form or the exposure pathway. The mercury toxicity and accumulation tendency is determined by its speciation (Lopes et al. 2014; Øverjordet et al. 2014). For example, methylmercury (MeHg) has a striking neurotoxic effect while inorganic mercury primarily disturbs the cellular redox balance (Stohs and Bagchi 1995). In the aquatic environment, mercury undergoes transformations among different chemical forms such as elementary mercury (Hg0), inorganic mercury (Hg2+), monomethylmercury (MeHg+), dimethylmercury (Me2Hg), and other organomercury species (Daye et al. 2013). Among marine invertebrates, copepods play a critical role in pelagic food webs by their function as the main transfer link between primary producers and higher trophic levels (Kwok et al. 2015; Steele 1974). Since several pollutants are lipophilic this suggests the potential for a strong biomagnification of calanoid food sources (Kwok et al. 2015). Indeed, copepods have the capacity to accumulate trace metals in their tissues at higher levels than those detected in their ambient aquatic environments but are still able to survive (Barka 2007). Due to this and other ecological and physiological characteristics (e.g., wide geographic distribution, adaptability, tolerance), copepods can be useful for ecotoxicological testing (Kwok et al. 2015).
Despite this importance, only limited information on mercury bioaccumulation in marine planktonic copepods is available (Hsiao and Fang 2013). Mercury bioaccumulation in plankton is of great concern, as it would provide further information about the trophic transfer and biomagnification of this contaminant (Hsiao et al. 2010; Hsiao and Fang 2013). Pseudodiaptomus marinus (Sato 1913) is a marine calanoid copepod originating from the Indo-Pacific region and has successfully colonized new geographic ocean areas such as the Mediterranean Sea (Galil 2009) and the North Sea in the last five decades (Sabia et al. 2012) and was recently recorded in the Belgian part of the North Sea (Deschutter et al. 2018). In natural habitats, P. marinus is detritivorous and herbivorous (Uye and Kasahara 1983). From another part, P. marinus is easy to rear under laboratory conditions in comparison to other calanoid species (Uye and Onbe 1975). In fact, experimental feeding studies demonstrated that flagellated microalgae, such as Isochrysis galbana, a widely used species in aquaculture, could be used as a major food source during all P. marinus developmental stages (Sabia et al. 2014). P. marinus is further characterized by a high physiological adaptability to environmental conditions (Arias et al. 2016; Deschutter et al. 2018; Sabia et al. 2015). For those reasons, P. marinus, seems to be an appropriate model for the study of the eco-physiological adaptability facing environmental variations and contaminants effects. Indeed, since the observation of this species in new invaded environments, the majority of studies were focused on its distribution and eco-biological endpoints such as reproduction, feeding strategies and swimming behavior (e.g. Brylinski et al. 2012; Deschutter et al. 2018; Sabia et al. 2012, 2015). In contrast, only few studies were available concerning the tolerance and responses to environmental pollutants in P. marinus (e.g. Arias et al. 2016; Huang et al. 2006; Tlili et al. 2016). In marine organisms, the entrance pathways of trace metallic elements (ETMs) into copepods could be by: (1) direct absorption from water through permeable body surfaces and from the gut; (2) ingestion of pre-contaminated phytoplankton (Wang et al. 2002). Many studies highlighted the potential influence of those pathways of exposure on the uptake and accumulation kinetics in marine copepods (Lee and Fisher 2017). In this context, the main objectives of the present work were the characterization of mortality effects and uptake/bioaccumulation kinetics of mercury chloride in the invasive copepod P. marinus by comparing different exposure pathways of dissolved inorganic mercury. Thus, the present study can contribute to a better characterization of trophic transfer of mercury into the alien species, P. marinus.
Material and Methods
Copepod and Microalgal Culture
Cultures of P. marinus were maintained in the Marine Station of Wimereux since 2011 (Tlili et al. 2016). The original strain was collected from Lake Faro, Sicily (Italy) (Sabia et al. 2014) and a stock culture used for experiments was performed in an incubator at 18 °C and a photoperiod cycle of 12L:12D. The protocol of maintaining P. marinus populations during several generations is slightly modified from those used for the estuarine copepod Eurytemora affinis (Souissi et al. 2010, 2015). To guarantee the production of high number of copepods for the need of experiments, a stock culture was maintained in 20 L Nalgene flasks and 40 L transparent Plexiglas flat bottom tank with moderate aeration.
Copepods were fed with the microalgae Isochrysis galbana cultivated in Conway medium with the same method described in Sadovskaya et al. (2014). The original strain of the microalgae Isochrysis galbana (I. galbana) was obtained from the Roscoff algal collection (Roscoff, France) and mass cultures were performed in the Marine Station of Wimereux (Wimreux, France) following to the protocol described previously in Sadovskaya et al. (2014) and Tlili et al. (2016). The original strain was maintained in the laboratory in 250 mL Erlenmeyer flasks and was shaken automatically at slow speed using an automatic shaker (KS 250 basic, IKA Labortechnik). The culture was kept in an incubator (SANYO model MLR-351) at 20 ± 2 °C and a photoperiod of 12L:12D cycle under a fluorescent light with an intensity of 2500 lx. Batch cultures in 2–6 L flasks were used to grow the microalgae for this study. The culture flasks were filled with autoclaved seawater of salinity 32 ± 2 PSU and enriched with Conway medium. The composition of Conway medium was composed by: 100 mg NaNO3, 20 mg NaH2PO4, 45 mg Na2EDTA, 33.6 mg H3BO3, 0.36 mg MnCl2, 1.3 mg FeCl3, 0.021 mg ZnCl2, 0.02 mg CoCl2·6H2O, 0.02 mg CuSO4·5H2O, 0.09 mg (NH4)6Mo7O24·4H2O, 0.2 mg thiamine HCl (vitamin B1) and 0.01 mg cyanocobalamin (vitamin B12), for each liter of autoclaved seawater. Cultures were aerated with sterile air and incubated in the same condition as the strain inoculum.
The seawater used for copepods, algae culture and experiments was pumped from the English Channel near Wimereux Marine Station, and was filtered several times up to 1 µm. The salinity of the water was 32 PSU ± 2 and temperature was 20 ± 2 °C. The detailed chemical composition (major and trace elements) is presented in Table 1.
Chemicals and Reagents
HgCl2 used for the experiments was obtained from Merck (Darmstadt, Germany; purity 99.9%). Experimental solutions were obtained by adding appropriate volumes of dissolved mercury to filtered seawater.
Lethal Concentration Experiments
Mortality %, and lethal concentration experiments were performed at standard conditions, in the absence of feeding and at controlled physic-chemical parameters (ambient temperature = 20 ± 2 °C; pH = 8.4 ± 0.2; dissolved oxygen = 8±1 mg/L, salinity: 32 ± 2 PSU) as described and recommended in previous studies (Lassus et al. 1984; Tlili et al. 2016).
Different concentrations of mercury (15, 35 and 75 μg/L) were prepared in 100-mL beakers for a total of 4 treatments including the control group (0 μg/L Hg). All treatments were performed in triplicate. For each concentration tested, a group of adult P. marinus specimens (n = 100) were placed in their respective pre-labeled beakers. Dead copepods were identified under a stereomicroscope every 24 h; they were identified as those that were not moving for few seconds and by further touching them gently with a very fine and tiny glass tip to stimulate movement. If there was still no movement, the copepods were considered dead, recorded, and immediately discarded. For each mercury concentration tested, resulting individual mortality was recorded for 24, 48, 72 and 96 h to calculate LC50 using Probit analysis.
Experimental Setup
Uptake and Accumulation Kinetics in P. marinus
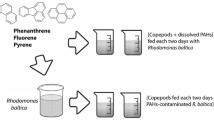
For the monitoring of mercury uptake and accumulation kinetics, two types of experiments were done (starvation and feeding experiments). For starved experiments, copepods were exposed to one selected sub-lethal concentration of HgCl2 (14.2 μg/L, corresponding to the 1/3 of 96 h LC50 previously determined) for 1, 2, 3, 5, 7, 10 and 15 days without feeding. To attend this purpose, a group of P. marinus (n = 1000) was transferred into a 2 L glass beaker containing the selected above concentration dissolved in filtered seawater (salinity: 32 ± 2 PSU). Experimental tanks were maintained at constant temperature (20 ± 2 °C) with very moderate and continuous artificial aeration. For the feeding experiment, a group of P. marinus (n = 1000) was transferred into a 2 L glass beaker containing filtered seawater and a pre-contaminated algae solution (during 24 h) used as the unique feeding source. The initial cell density of the micro-algae I. galabana was 105 cells/mL.
For both experiments (starvation and feeding), at each sampling period (n = 3) groups of copepods (each group was composed by 30–50 specimens) were sampled using a glass pipette, carefully filtered, weighted and directly dried at ambient temperature, under hood, during 72 h for mercury determination. For water analysis, samples (n = 3) of 5 mL each were directly taken from the experimental tanks, carefully filtered and stoked in analytic tubes with 50 µL of HNO3 until analysis. For all experiments, a comparison with a non-exposed group, considered as a control, was performed.
Uptake and Accumulation Kinetics of the Diet
For the accumulation assessment of the diet used, an aliquot of I. galbana, from the exponential growth phase (concentration: 105 cells/mL) was placed into 200 mL glass beaker containing the above concentration of HgCl2 under continuous shaking and aeration. At each sampling period, 3 suspensions of I. galbana were taken using a glass pipette, carefully filtered, through cellulose nitrate filters (0.22 mm porosity) weighted and directly dried at ambient temperature, under hood, during 72 h for mercury determination. For water analysis, samples (n = 3) of 5 mL each were directly taken from the experimental tanks, carefully filtered and the resulting water was stoked in analytic tubes with 50 µL of HNO3 until analysis.
Hg-Analysis
Total HgCl2 concentrations in P. marinus, I. galbana and water samples were determined according to the protocol of Kadlecova et al. (2011). Total mercury was analyzed without any pre-treatment using a one-purpose atomic absorption spectrometer Advanced Mercury Analyser (Model AMA 254; Altec Ltd., Plzen, Czech Republic). The limit of detection (LOD) and the limit of quantification (LOQ) of the analyser were 0.1 and 0.3 μg kg−1, respectively. Quality assurance (QA) vs quality control of mercury measurements were validated using certified reference materials (certified from the international Atomic Energy Agency). A good agreement was observed between the obtained and the certified values for total mercury (data non-shown).
Uptake Kinetics and Accumulation Factor Determination
To assess the uptake kinetics of mercury in P. marinus and I. galbana, the model proposed by Luoma and Rainbow (2005) was used. The uptake kinetic model (μg g−1 day−1) followed the following equation:
where “Cin” is the Hg concentration in tissues (μg g−1 dry weight), “ku” the uptake rate constant (g dry weight/L/day), “Cw” the Hg concentration in water (μg/L) and “T” the exposure time (days). The accumulation factor was assessed as the ratio between mercury concentration in tissues and the water concentration (Landrum et al. 1992).
Statistical Analysis
All results were expressed as mean ± standard deviation (SD) from at least triplicate. To compare two independent groups, a Mann–Whitney test was used and for the comparisons of two dependant group, a Wilcoxon matched-pairs signed-ranks test was used. For all cases, the differences were considered significant at p < 0.05. Statistical comparisons were performed using XLSTAT (ver. 2015.2.02. ). The kinetics of bioaccumulation of mercury was directly fitted with an exponential model (Y = a × exp(− b × x) + c) using Curve Fitting Toolbox of Matlab Software (Mathworks Inc., vers 7.2).
Results
Lethal Concentration
Mortality observed % in P. marinus exposed to different mercury concentrations are presented in Table 2. Then, mortality percentages were probit transformed and lethal concentration (LC50 %) values extrapolated from regression lines. LC50% values of mercuric chloride in adult P. marinus, were 172.4; 90.72; 60.65 and 42.42 μg/L, respectively, for 24, 48, 72 and 96 h.
Bioaccumulation of Mercury in P. marinus Through Waterborne and Dietary Pathways
Mortality observed in control P. marinus group at the 0% mercury concentration was presented in Table 3. For starved experiments, the mortality % was low from day 1 to day 4 to attend a maximum of 25% at the end of experiment (15th day). Concerning the feeding experiments, the mortality % in the control group was low during the first week to reach a maximum of 11.5% at the 15th day.
HgCl2 concentration was measured in P. marinus tissues and the seawater medium (Fig. 1). After 1 day of exposure, the concentration of Hg in P. marinus tissues was 2.4 μg/g dry weight, and then increased gradually to 3.9, 4.6, 5.3, and 7.2 μg/g dry weight after 3, 4,7 and 10 days, respectively. No significant changes were detected until the end of the experiment after 15 days. Statistical analysis showed significant differences between the control and the exposed copepods group, except the two first sampling dates of mercury uptake. The concentration of Hg in water was 11.3 μg/L at the 1st day and gradually decreased to 5.3 and 5.8 μg/L at 10 and 15 days of the experiment. Statistical analysis showed no significant differences between days 1 and 2 of the experiment.
Mercury accumulation kinetics in P. marinus measured in tissues and water. White column present Hg concentration P. marinus and grey column the Hg concentration in water Results are expressed as mean ± standard deviation. Different letters above histograms indicate a significant difference among exposure time at P < 0.05
The mercury bioaccumulation in P. marinus tissues and water after feeding by precontaminated microalgae is shown in Fig. 2. After 1 day of exposure, the concentration of Hg was 1 μg/g dry weight, and then an increase was recorded after 2 and 7 days of exposure (2.3 and 4.3 μg/g dry weight, respectively). On the other hand, the Hg concentration in water was 11.8 μg/L and did not show any significant difference between exposure times until 7 days of experiment, followed by a concentration decrease at 10 days (7.1 μg/L). No significant differences were detected between the 1st and 5th days.
Mercury accumulation kinetics in P. marinus tissues fed with pre-contaminated algae and water. Results are expressed as mean ± standard deviation. White column represent Hg concentration P. marinus fed with contaminated microalgae and grey column represents the Hg concentration in water. Different letters indicate a significant difference among exposure time at P < 0.05
Mercury Accumulation in I. galbana
Accumulation kinetics of mercury in I. galbana cells and water are presented in Fig. 3. At 1 day the concentration of Hg in I. galbana was 2.2 μg/g dry weight and gradually increased to reach 6.8, 8.1, and 10 μg/g dry weight after 2, 3, and 4 days, respectively. No significant changes were observed then between the 4th day to the end of the experiment (15 days of exposure). For water, the concentration of Hg was 12.9 μg/L at 1 day and gradually decreased to reach 0.75 μg/L at 10 days.
Mercury accumulation kinetics in I. galbana measured in cells and water. Results are expressed as mean ± standard deviation. White column present Hg concentration in I. galabana and grey column the Hg concentration in water. Different letters above histograms indicate a significant difference among exposure time at P < 0.05
Uptake and Accumulation Kinetics
The uptake rate constant (Ku) from seawater solution was presented in Fig. 4. The microalgae I. galbana showed the highest uptake rate (0.67 g dry weight/L/day) compared to starved copepod P. marinus (0.086 g dry weight/L/day) and copepods fed by the contaminated diet (0.038 g dry weight/L/day). No significant differences were observed between the uptake of mercury in copepods from water or from diet. The accumulation factor (AF) from aqueous solution to tissues is given in Fig. 5. I. galbana has the highest bioaccumulation factor of mercury (0.53 L g−1 day−1) followed by the fed P. marinus (0.39 L g−1 day−1) and the starved P. marinus (0.26 L g−1 day−1). No significant differences were observed concerning the bioaccumulation factor of mercury in copepods depending of exposure pathways (water/food).
Discussion
The determination of lethal concentrations of Trace Elements in aquatic organisms had revealed their quantitative responses and sensitivity levels, providing guidelines for the standardization and regulation of those environmental pollutants influx into different aquatic ecosystem components (Kadiene et al. 2017; Zidour et al. 2019).
Marine copepods have a species-dependent sensitivity in response to acute Hg exposure with different LC50 values ranging between 10 and 600 μg/L (Øverjordet et al. 2014). Comparative data of Hg 96 h LC50 values in copepods are presented in Table 4. In the present study, P. marinus, the 96 h LC50 was about42.4 μg/L, whereas the 96 h LC10 was 3.1 μg/L. Accordingly, P. marinus is in the same range of sensitivity as other copepods except Eurytemora affinis (US EPA 1981). Different lethal concentration studies had highlighted the critical ecological effect of lethal and sublethal concentrations of toxic metals (Øverjordet et al. 2014; Zamora et al. 2015). Thus, it is necessary to perform those experiments at standard conditions, such as no feeding and the same life cycle status, to avoid factors to become confounding (Kadiene et al. 2017; Tlili et al. 2016; Zidour et al. 2019).
Research in metal ecotoxicology emphasizes the importance of the exposure pathways on accumulation and toxicity processes. Thus, dietary exposure is a significant factor for metal accumulation in zooplankton, and feeding strategies are very important for the bioaccumulation processes and metal transfers in marine food chains (Battuello et al. 2017). In the present study, the accumulation of total mercury in P. marinus exposed to contaminated diets did not show significant differences compared to the direct exposure. Some comparative studies confirmed that the diet pathway did not have a pronounced influence on the assimilation of methyl mercury in marine copepods (Williams et al. 2010; Lee and Fisher 2016). In contrast for others trace elements, e.g. cadmium and nickel, uptake and accumulation studies on marine copepods showed that the dietary exposure increased the assimilation for those metals by copepods (Kadiene et al. 2017; Tlili et al. 2016). In natural conditions, metals could be taken up directly from the aqueous medium or from the food. In the case of cadmium for example, more than 50% of accumulated metal comes from the dissolved phase (Wang and Fisher 1998). On the other hand, it is well demonstrated that uptake kinetics in copepods depended on the nature of metal (essential, non-essential) (Barka 2007). In comparison with nickel exposure at the same experimental conditions, mercury-exposed P. marinus showed a slow uptake rate (0.09 μg/L/day) of dissolved Hg in water, especially between the 1st and 3rd days compared to P. marinus exposed to nickel (0.14 μg/L/day) (Tlili et al. 2016). In marine copepods, the exposure to sub-lethal concentrations of dissolved Hg can cause multi-scale effects and damages with some direct ecological implications such as reduced fecundity (Valko et al. 2005). In fact, different Hg forms (organic and inorganic) are known to deplete biological systems of free sulfhydryl groups (SH) which are generally involved in protecting from cellular damage in response to oxidative stress (Valko et al. 2005). In addition, the inorganic form (Hg2+) can disrupt multiple steps in the metabolism of the endogenous antioxidant glutathione (GSH) involved in detoxification processes (Stohs and Bagchi 1995; Valko et al. 2005). Hg2+ can also cause rapid lipid peroxidation mainly due to a combination of antioxidant depletion and enhanced production of reactive oxygen species (Monteiro et al. 2010; Valko et al. 2005). In natural aquatic habitats, trace metal levels of the different trophic positions depend on several factors including physicochemical properties of the habitat (water and sediment geochemistry, pH, and metal speciation), feeding habits, and metal handling and storage strategies of respective species in the food web (Wang et al. 2002). In a recent study focusing on the determination of Hg levels in marine copepods in Taiwan (Hsiao and Fang 2013), the Hg bioaccumulation factor value of sampled copepods did not positively correlate with its ambient water concentration and vice versa. This confirms the notion that mercury is a non-essential element and highly toxic substance for marine organisms (Hsiao and Fang 2013). For example methylmercury has a rapid accumulation in protein-rich tissues and is excreted slowly. This explains why Hg can be bioaccumulated in organisms and biomagnified in food webs (Kidd et al. 2012). Moreover, the inorganic form of mercury (Hg2+) is transported into living organism via active transport systems through membranes rather than by passive diffusion (Morel et al. 1998). Several other factors are complicating this situation and the modes and kinetics of uptake rate such as ambient temperature (Tsui and Wang 2006). Microalgae represent an important food source for many marine copepods and consequently, metal bioaccumulation by phytoplankton and its subsequent transfer to marine copepods need to receive attention (Wang et al. 2007; Tlili et al. 2016). The marine microalgae and dinoflagellate Isochrysis galbana represent an important food source for copepods aquaculture, easily assimilated by marine animal larvae and useful for ecotoxicological experiments (Sadovskaya et al. 2014; Tlili et al. 2016). In the present study, I. galbana, exposed to a sub-lethal concentration of HgCl2, showed a fast uptake of dissolved Hg especially at the beginning of the experiment (days 1–2).
Exposed to a sub-lethal concentration of the important trace metal nickel, I. galbana showed a similar uptake profile with only a slight difference between corresponding uptake rate constants (0.49 μg/L/day for Hg and 0.51 μg/L/day for Ni) (Tlili et al. 2016). I. galbana has the ability to pump water to extract minerals from the aquatic environment for growth and survival (Marchetti et al. 2012). In I. galbana, the bioaccumulation capacity of Cd, Pb, Zn, and Cu is likely associated with phytochelatin induction (Yap et al. 2004). Since phytoplankton is the entry point for mercury into the aquatic food web (Wu and Wan 2011), more attention should be given to bioaccumulation mechanisms of model species such as I. galbana to better understand and characterize bio-magnification of Hg at different levels of the aquatic trophic web, particularly those on direct relationships with human consumption. In the presence of a food source, P. marinus accumulated dissolved mercury from both microalgal diet and from water column. This result should be taken in account for a better understanding of the adaptability of invasive copepods such P. marinus.
Conclusion
Pseudodiaptomus marinus could be considered in the same range of sensitivity in response to dissolved mercury in comparison with other calonoid copepods. The accumulation kinetics of mercury in P. marinus was relatively slow, but the presence of food source do not seems to increase significantly HgCl2 uptake. I. galbana had a relatively important uptake and accumulation capacity of HgCl2 in aquatic environment. As recommended by recent reports (e.g. Battuello et al. 2017), it is necessary to take into account that different copepods had different feeding requirements and abilities to accumulate or excrete trace elements. Thus, finding of the present work could be helpful in the understanding of mercury bioaccumulation and bio-amplification processes especially concerning invasive species.
References
Arias AH, Souissi A, Roussin M, Ouddane B, Souissi S (2016) Bioaccumulation of PAHs in marine zooplankton: an experimental study in the copepod Pseudodiaptomus marinus. Environ Earth Sci 75:691
Barka S (2007) Insoluble detoxification of trace metals (copper, zinc, nickel, cadmium, silver and mercury) in a marine crustacean Tigriopus brevicornis (Müller). Ecotoxicology 16:491–502
Barka S, Pavillon JF, Amiard JC (2001) Influence of different essential and non-essential metals on MTLP levels in the copepod Tigriopus brevicornis. Comp Biochem Physiol 128C:479–493
Battuello M, Mussat Sartor R, Brizio P, Nurra N, Pessani D, Abete MC, Squadrone S (2017) The influence of feeding strategies on trace element bioaccumulation in copepods (Calanoida). Ecol Ind 74:311–320. https://doi.org/10.1016/j.ecolind.2016.11.041
Brylinski JM, Antajan E, Raud T, Vincent D (2012) First record of the Asian copepod Pseudodiaptomus marinus Sato, 1913 (Copepoda: Calanoida: Pseudodiaptomidae) in the southern bight of the North Sea along the coast of France. Aquat Invasions 7:577–584. https://doi.org/10.3391/ai.2012.7.4.014
Daye M, Ouddane B, Halwani J, Hamzeh M (2013) Solid phase extraction of inorganic mercury using 5-phenylazo-8-hydroxyquinoline and determination by cold vapor atomic fluorescence spectroscopy in natural water samples. Sci World J. https://doi.org/10.1155/2013/134565
Deschutter Y, Vergara G, Mortelmans J, Deneudt K, De Schamphelaere K, De Troch M (2018) Distribution of the invasive calanoid copepod Pseudodiaptomus marinus (Sato, 1913) in the Belgian part of the North Sea. BioInvasions Records 7:33–41. https://doi.org/10.3391/bir.2018.7.1.05
Galil BS (2009) Taking stock: inventory of alien species in the Mediterranean Sea. Biol Invasions 11:359–372
Hsiao SH, Fang TH (2013) Hg bioaccumulation in marine copepods around hydrothermal vents and the adjacent marine environment in northeastern Taiwan. Mar Poll Bull 74:175–182
Hsiao SH, Hwang JS, Fang TH (2010) The heterogeneity of the contents of trace metals in dominant copepod species in the seawater around northern Taiwan. Crustaceana 83:179–194
Huang Y, Zhu L, Liu G (2006) The effects of bis(tributyltin) oxide on the development, reproduction and sex ratio of the calanoid copepod Pseudodiaptomus marinus. Estuar Coast Shelf Sci 69:147–152
Kadiene EU, Bialais C, Ouddane B, Hwang JS, Souissi S (2017) Differences in lethal response between male and female calanoid copepods and life cycle traits to cadmium toxicity. Ecotoxicology 26:1227–1239
Kadlecova M, Ouddane B, Docekolova H (2011) Speciation of mercury in the strongly polluted sediments of the Deule River (France). J Environ Monit 14:961
Kidd KA, Muir DCG, Evans MS, Wang X, Whittle M, Swanson HK, Johnston T, Guildford S (2012) Biomagnification of mercury through lake trout (Salvelinus namaycush) food webs of lake with different physical, chemical, and biological characteristics. Sci Tot Environ 135:430–438
Kwok KWH, Souissi S, Dur G, Won E-J, Lee J-S (2015) Copepods as reference species in estuarine and marine waters. In: Amiard-Triquet C, Amiard JC, Mouneyrac C (eds) Aquatic ecotoxicology: advancing tools for dealing with emerging risks. Academic Press, New York, pp 281–308
Landrum PF, Lydy MJ, Lee H (1992) Environ. Toxicol Chem 11(12):1709–1725. https://doi.org/10.1002/etc.5620111205
Lassus P, Lebaut C, Le Dean L, Bardouil M, Truquet P, Bocquené G (1984) The use of harpacticoid copepods for testing the effects of chemicals on larval production. Inst Mar Sci Res 2:131–142
Lee CS, Fisher NS (2016) Methylmercury uptake by diverse marine phytoplankton. Limnol Oceanogr 61(5):1626–1639
Lee CS, Fisher NS (2017) Bioaccumulation of methylmercury in a marine diatom and the influence of dissolved organic matter. Mar Chem 197:70–79. https://doi.org/10.1016/j.marchem.2017.09.005
Lescord GL, Kidd KA, Kirk JL, O’Driscoll JN, Wang X, Muir DCG (2015) Factors affecting biotic mercury concentrations and biomagnification through lake food webs in the Canadian high Arctic. Sci Tot Environ 509(510):195–205
Lopes CB, Lopes I, Luciana SR, Duarte AC, Soares AMVM, Rocha J, Pereira E (2014) A multidisciplinary approach to evaluate the efficiency of a clean-up technology to remove mercury from water. Bull Environ Contam Toxicol 93:138–143
Luoma SN, Rainbow PS (2005) Why is metal bioaccumulation so variable? Biodynamics as a unifying concept. Environ Sci Technol 39:1921–1931
Marchetti J, Bougaran G, Le Dean L, Mégrier C, Lukomska E, Kaas R, Olivo E, Baron R, Robert R, Cadoret JP (2012) Optimizing conditions for the continuous culture of Isochrysis affinis galbana relevant to commercial hatcheries. Aquaculture 326(329):106–115
Monteiro DA, Rantin FT, Kalinin AL (2010) Inorganic mercury exposure: toxicological effects, oxidative stress biomarkers and bioaccumulation in the tropical freshwater fish matrinx, Brycon amazonicus (Spix and Agassiz, 1829). Ecotoxicology 19:105–123
Morel FMM, Kraepiel AML, Amyot M (1998) The chemical cycle and bioaccumulation of mercury. Annu Rev Ecol Syst 29:543–566
Øverjordet IB, Altin D, Berg T, Jenssen BM, Gabrielsen GW, Hansen BH (2014) Acute and sub-lethal response to mercury in Arctic and boreal calanoid copepods. Aquat Toxicol 155:160–165
Sabia L, Uttieri M, Pansera M, Souissi S, Schmitt FG, Zagami G, Zambianchi E (2012) First observations on the swimming behaviour of Pseudodiaptomus marinus from Lake Faro. Biol Mar Medit 19:240–241
Sabia L, Uttieri M, Schmitt FG, Zagami G, Zambianchi E, Souissi S (2014) Pseudodiaptomus marinus Sato, 1913, a new invasive copepod in Lake Faro (Sicily): observations on the swimming behaviour and the sex-dependent responses to food. Zool Stud 53:49
Sabia L, Zagami G, Mazzocchi MG, Zambianchi E, Uttieri M (2015) Spreading factors of a globally invading coastal copepod. Medit Mar Sci 16/2:460–47. https://doi.org/10.12681/mms.1154
Sadovskaya I, Souissi A, Souissi S, Grard T, Lencel P, Greene CM, Duin S, Dmitrenok PS, Chizhov AO, Shashkov AS, Usov AI (2014) Chemical structure and biological activity of a highly branched (1 → 3,1 → 6)-β-d-glucan from Isochrysis galbana. Carbohydr Polym 111:139–148
Sato F (1913) Pelagic copepods (no. 1). Sci Rep Hokkaido Fish Exp Stn 1:1–79
Sosnowski S, Gentile JH (1978) Toxicological comparison of natural and cultured population of Acartia tonsa to cadmium, copper and mercury. J Fish Res Board Can 35:1366–1369
Souissi A, Souissi S, Hansen BW (2015) Physiological improvement in the copepod Eurytemora affinis through thermal and multigenerational selection. Aquac Res 47:2227–2242
Steele J H (1974) The structure of marine ecosystems. Harvard University Press, Cambridge. https://doi.org/10.4319/lo.1974.19.6.1015
Stohs SJ, Bagchi D (1995) Oxidative mechanisms in the toxicity of metal ions. Free Rad Biol Med 18:321–336
Tlili S, Ovaert J, Souissi A, Ouddane B, Souissi S (2016) Acute toxicity, uptake and accumulation kinetics of nickel in an invasive copepod species: Pseudodiaptomus marinus. Chemosphere 144:1729–1737
Tsui TKT, Wang WX (2006) Acute toxicity of mercury to Daphnia magna under different conditions. Environ Sci Technol 40:4025–4030
U.S. Environmental Protection Agency (1981) Interlaboratory comparison acute toxicity tests using estuarine animals. EPA-600/4-81/003. Washington, DC
Uye S, Kasahara S (1983) Grazing of various developmental stages of Pseudodipatomus marinus (Copepoda: Calanoida) on natural occurring particles. Bull Plankton Soc Japan 30:147–158
Uye S, Onbe T (1975) The developmental stages of Pseudodiaptomus marinus SATO (Copepoda: Calanoida) reared in the laboratory. Bull Plankton Soc Japan 21:65–76
Valko M, Morris H, Cronin MTD (2005) Metals, toxicity and oxidative stress. Curr Med Chem 12:1161–1208
Wang WX, Fisher NS (1998) Accumulation of trace elements in a marine copepod. Limnol Oceanogr 43(2):273–283
Wang X, Hong H, Xu L, Chen W, Zhang Z (2002) Distribution and transportation of polycyclic aromatic hydrocarbons in suspended particulate matter and surface sediment from the Pearl River Estuary. J Environ Sci Health A 37:451–463
Wang MH, Wang DZ, Wang GZ, Huang XG, Hong HS (2007) Influence of N, P additions on the transfer of nickel from phytoplankton to copepods. Environ Pollut 148:679–687
Williams JJ, Dutton J, Chen CY, Fisher NS (2010) Metal (As, Cd, Hg, and CH3Hg) bioaccumulation from water and food by the benthic amphipod Leptocheirus plumulosus. Environ Toxicol Chem 29:1755–1761
Wu Y, Wan WX (2011) Accumulation, subcellular distribution and toxicity of inorganic mercury and methylmercury in marine phytoplankton. Environ Pollut 159:3097–3105
Yap CK, Ismail A, Omar H, Tan SG (2004) Toxicities and tolerances of Cd, Cu, Pb and Zn in a primary producer (Isochrysis galbana) and in a primary consumer (Perna viridis). Environ Int 29:1097–1104
Zamora LM, King CK, Payne SJ, Virtue P (2015) Sensitivity and response time of three common Antarctic marine copepods to metal exposure. Chemosphere 120:267–272
Zidour M, Boubechiche Z, Pan YJ, Bialais C, Cudennec B, Grard T, Drider D, Flahaut C, Ouddane B, Souissi S (2019). Population response of the estuarine copepod Eurytemora affinis to its bioaccumulation of trace metals. Chemosphere 220:505–513. https://doi.org/10.1016/j.chemosphere.2018.12.148
Acknowledgements
This work is a contribution to the Interdisciplinary Environmental Institute of Lille 1 University (IREPSE) as well as to Lille 1 Grant (BQR-Convergence-2014) aiming to reinforce multidisciplinary studies around copepods. This study was funded by an Erasmus Mundus (Fatima Al Fihri Lot 2) Post-Doctoral fellowship to S. Tlili. We would like to thank LASIR and LOG teams (University of Lille, France) for facilities and help in performing experiments and analysis. Authors thank reviewers for valuable suggestions and recommendations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tlili, S., Ovaert, J., Souissi, A. et al. Bioaccumulation of Mercury in the Copepod Pseudodiaptomus marinus: A Comparative Study Between Waterborne and Dietary Pathways. Int J Environ Res 13, 759–768 (2019). https://doi.org/10.1007/s41742-019-00202-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41742-019-00202-y