Abstract
Electron beam (EB) degradation of perfluorooctanoic acid (PFOA) and perfluorooctane sulfonate (PFOS) in aqueous solutions was studied. It suggested that PFOA and PFOS degradation followed the pseudo-firstorder kinetics, and degradation rates increased with increasing initial pH. Radical scavengers’ experiments indicated that hydrated electron and hydrogen radical were important in the electron beam degradation of PFOA and PFOS, especially hydrated electron. The decomposition efficiencies were 95.7% for PFOA and 85.9% for PFOS, by EB irradiation in an anoxic alkaline solution (pH = 13). The potential degradation pathways of PFOA and PFOS by electron beam irradiation through defluorination and the removal of CH2 unit were proposed.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Perfluorooctane sulfonate (PFOS) and perfluorooctanoate (PFOA) are among the perfluorinated compounds (PFCs) that received a great deal of concern in recent years. Because of their extraordinary stability, hydrophobicity, oleophobicity and excellent surfactant performance, many PFCs have been widely applied to industrial and domestic production for over sixty years. PFOA and PFOS have been found almost ubiquitously in various environmental media including waters [1,2,3,4], air [5,6,7], sediments [8, 9], wildlife [10, 11] and human [12, 13]. Moreover, toxic and bioaccumulative effects of contaminants also have been confirmed by extensive studies [14,15,16,17]. These indicate that the toxic pollutants can accumulate in human body and may pose a risk to human health. As a result, PFOS was listed as a persistent organic pollutant under the Stockholm Convention in 2009, calling for restricted production and use worldwide.
Recently, the degradation of PFOA and PFOS has received increasing attention. Various techniques including adsorption [18, 19], photolytic methods [20,21,22,23], photoelectrocatalysis [24], ultrasonic irradiation [25, 26], thermolysis [27], gamma irradiation [28], etc. [29,30,31,32,33], were used to remove PFOA and PFOS. However, PFCs are chemically inert due to the unique properties of fluorine and relatively strong carbon–fluorine bond (C–F, 116 kcal/mol) making them resistant to conventional advanced oxidation processes. Therefore, new techniques are desirable to efficiently decompose PFOA and PFOS.
Electron beam (EB) irradiation has been recognized as effective method to treat organic pollutants [34,35,36,37,38]. Water is degraded into hydroxyl radicals (·OH), hydrogen radical (\( {\text{H}}^{ \cdot } \)) and hydrated electron (e −aq ) with different G-values (μmol/J) under EB irradiation as Eq. (1). The radicals react with the pollutants and cause their degradation [39]. While hydroxyl radical can oxidize the organic compounds in aqueous solutions, hydrogen radical and hydrated electron have reductive ability to reduce the targeted organic compounds [40, 41]. Hydrated electron is a strong reducing agent, reacting rapidly with halogenated organic compounds [41,42,43,44].
In the present study, the degradation kinetics of EB-irradiated PFOA and PFOS were determined. The effects of initial concentrations and initial pH on the degradation of contaminants were discussed. The roles of oxidative or reductive species in PFOA and PFOS degradation were investigated by selectively adding specific promoters or inhibitors, such as O2, N2 and tert-butanol (t-BuOH). Possible mechanisms of the radiolysis degradation of PFOA and PFOS were proposed. Our findings may bring an efficient and environmentally friendly technique for PFCs degradation.
2 Materials and methods
2.1 Materials
Perfluorooctanoic acid (PFOA, C8F15HO2, 95.5%) and perfluorooctane sulfonate (PFOS, C8F17SO3K, 98.0%) were supplied by Dr. Ehrenstorfer (Augsburg, Germany). Ammonium acetate (LC grade, 99%), fluoride standard (99.99%) and methanol (HPLC grade, 99.99%) were provided by Shanghai Anpel scientific instrument corporation. Sodium carbonate (Na2CO3), sodium bicarbonate (NaHCO3), hydrogen chloride (HCl), sodium hydroxide (NaOH) and tert-butanol (t-BuOH) were purchased from Sinopharm Chemical Reagent Co., Ltd. All aqueous solutions were prepared by a Millipore Milli-Q system (resistance [18.2 MΩ]).
2.2 The samples and their irradiation
Four initial concentrations of PFOA and PFOS solutions in 100 mL polypropylene vessels were stored at 4 °C before use. Samples pouched in high-density polyethylene (HDPE) bags were irradiated to 100–500 kGy at ambient temperatures by 1.8 MeV electron beams of up to 10 mA. The samples were placed about 30 cm away under the beam-scan horn.
2.3 Analytical methods
Concentrations of PFOA, PFOS and their degradation products were determined by liquid chromatography–tandem mass spectrometry (LC–MS/MS) using an Agilent 1260 LC chromatograph coupled to an Agilent 6460 mass spectrometer with electron spray ionization (ESI) interface and a heated nebulizer. HPLC separation was carried out by ZORBAX SB-C18 reversed-phase column (4.6 × 50 mm, 5 μm, Agilent) and EC-C18 (3.0 × 100 mm, 2.7 μm, Agilent) at 40 °C using a gradient composition of solvent A (methanol) and solvent B (water containing 2 mmol/L of ammonium acetate). The injection volume was 5 μL. The gradient (%A) was as follows: 0–0.75 min, a linear increase from 5 to 60%; 0.75–5 min, increase to 92% A; 5–5.1 min, further increase to 100% A; 5.1–8.5 min, being held at 100% A; 8.5–9.0 min, the mobile phase returning to the initial conditions; and 9–10 min, being held at 5% A. The flow rate was 0.4 mL/min. Quantification was performed using multiple reaction monitoring (MRM) of the transitions m/z 499 → 80.1 (PFOS) and m/z 413 → 368.9 (PFOA). Mass spectrometry full scanning analysis (m/z 100–550) was used to identify the intermediate products of PFOS and PFOA after irradiation. The instrument conditions were as follows: capillary voltage, 4.0 kV; drying gas flow, 3 L/min; drying gas temperature, 350 °C; and source gas flow, 10 L/min; nebulizer pressure, 38 psi and nozzle voltage of 1 kV.
An ion-chromatograph system (Dionex ICS-1100, USA) was used to determine the concentration of F−. It consisted of an automatic sample injector (sample injection volume 25 μL), a degasser, a pump, a guard column (Dionex AG22, 4 × 50 mm, USA), a separation column (Dionex AS22, 4 × 250 mm, USA), a column oven (30 °C) and a conductivity detector with a suppressor device. The mobile phase was composed of 4.5 mmol/L Na2CO3 and 1.4 mmol/L NaHCO3, and the flow rate was set at 1.0 mL/min. The lowest detection limit of F− was 0.02 mg/L.
3 Results and discussion
3.1 Kinetic studies of PFOA and PFOS under electron beam radiolysis
PFOA and PFOS aqueous solutions in initial concentrations of 10–40 mg/L were irradiated to 100–500 kGy. As shown in Fig. 1a, the PFOA and PFOS contents decreased with increasing doses. At initial concentrations of 10, 20, 30 and 40 mg/L, the PFOA degradation rates at 500 kGy were 63.6, 51.4, 48.2 and 41.7%, respectively; and the PFOS degradation rates were 57.1, 42.4, 35.5 and 32.4%, respectively. These results indicate that PFOA and PFOS degradation rates decreased with increasing initial concentrations. The PFOA and PFOS concentration changing rate was plotted in natural logarithm versus the irradiation dose, and each of the data sets could be fitted linearly with R 2 > 0.90, suggesting that the EB degradation of PFOA and PFOS followed pseudo-first-order kinetics at different initial concentrations. Also, as shown in Fig. 1b, certain amounts of fluoride ions were detected at different doses, implying that the EB degradation of PFOA and PFOS was mainly through the defluorination.
3.2 Effect of initial pH on PFOA and PFOS degradation
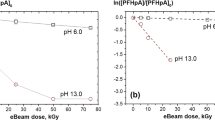
Initial pH is an important factor in the degradation processes, as hydroxyl radicals, hydrated electron and hydrogen radical may react with OH− and H+ in aqueous solution. PFOA and PFOS aqueous solutions (20 mg/L) in initial pH 3–13 were irradiated to 100–500 kGy. The degradation (Fig. 2a) and the fluoride release (Fig. 2b) increased with the pH value. At pH13 and 500 kGy, the degradation rates of PFOA and PFOS were 88.1 and 63.4%, respectively; and the concentration of fluoride ions increased rapidly to 239.5 and 352.6 μmol/L with the corresponding defluorination percentage of 37.5 and 51.8%, respectively. Decomposition of PFOS is more difficult than that of PFOA, as oxidation of sulfonate to sulfate is more difficult. However, the generated fluoride ions concentrations of PFOS were slightly higher than those of PFOA. This can be attributed to hydrogen radical, which can be inverted into hydrated electron in alkaline condition, and the loss of hydrated electron in acidic condition, as shown in Eqs. (2) and (3). Thus, the solutions in alkaline conditions may be helpful to the degradation of PFOA and PFOS.
3.3 Effect of radical scavengers on PFOA and PFOS degradation
In order to investigate which active species played the leading role in degradation of PFOA and PFOS, the aqueous solutions (20 mg/L) were saturated by N2, O2 and 0.1 mol/L tert-butanol with N2. In N2-saturated solution, hydroxyl radicals, hydrated electron and hydrogen radical were all existed and should be considered in the reactions process. It is well known that the reducing radicals hydrated electron and hydrogen radical can react with O2 through Eqs. (4) and (5) and turn into activated radicals \( {\text{O}}_{2}^{ - \cdot } \) and \( {\text{HO}}_{2}^{ \cdot } \). Hydroxyl radicals were the main reactive species in O2 saturated solution. However, both hydrated electron and hydrogen radical existed in N2-saturated solution containing 0.1 mol/L tert-butanol, because hydroxyl radicals were scavenged by tert-butanol, as shown in Eq. (6).
Figure 3 shows the degradation and fluoride release of PFOA and PFOS under the different conditions. At 500 kGy, the degradation rates were higher in N2-saturated solutions, being 81.6% for PFOA and 71.3% for PFOS, and in N2-saturated solution containing 0.1 mol/L tert-butanol, being 87.4% for PFOA and 76.9% for PFOS, while the degradation rates were low in O2-situated solutions, being just 11.0% for PFOA and 8.7% for PFOS. For PFOA, the higher concentrations of fluoride ions were observed in N2-situated solution (200.3 μmol/L) and N2-saturated solution containing 0.1 mol/L tert-butanol (232.9 μmol/L) at 500 kGy, while it reached 27.5 μmol/L in O2-saturated solution. Similarly, the formation of fluoride ions of PFOS showed the same trends as that of PFOA. Lower degradation rates and fluoride ions concentrations of PFOA and PFOS were observed in O2-saturated solution indicated that hydroxyl radicals were not favorable for degrading PFOA and PFOS under electron beam irradiation. Consequently, higher degradation rates and concentrations of fluoride ions occurred in N2-saturated solution and N2-saturated solution containing 0.1 mol/L tert-butanol. Due to the strong reductive property and the relatively higher concentration of hydrated electron [41,42,43], we considered that hydrated electron, and hydrogen radical especially, played key roles in electron beam degradation of PFOA and PFOS.
Furthermore, the quantum yield of hydrated electron increases greatly in alkaline solutions as the transformation of hydrogen atoms into hydrated electrons, i.e., Eq. (2). As the results show, the PFOA and PFOS degradation rates and concentrations of fluoride ions are higher in alkaline condition. Thus, the effect of pH on PFOA and PFOS decomposition further implies that hydrated electrons play the key role in the decomposition of PFOA and PFOS.
In order to investigate the optimal EB degradation condition of PFOA and PFOS, the aqueous solutions (20 mg/L) were saturated by N2 and adjusted to pH 13. The results are shown in Fig. 3. At 500 kGy, the degradation rates of PFOA and PFOS in nitrogen atmosphere were 95.7 and 85.9%, and their fluoride ion concentrations increased rapidly to 324.6 and 416.9 μmol/L, respectively, indicating that PFOA and PFOS in an anoxic alkaline solution are efficiently decomposed by EB irradiation.
3.4 Mechanism of PFOA and PFOS degradation by irradiation
PFOA, PFOS and their degradation products in the aqueous solutions were identified using LC–MS/MS method. The results are shown in Fig. 4. C7F14HCOOH, C6F12HCOOH, C5F10HCOOH, C4F9COOH, C3F7COOH, C2F5COOH, C7F14HSO3 −, C6F12HSO3 −, C5F11SO3 −, C3F7SO3 −, C2F5SO3 − and other intermediate products appeared after irradiation.
3.4.1 Mechanism of PFOA degradation by irradiation
Based on these results, a mechanism for PFOA degradation by EB irradiation is proposed in Fig. 5a. Under nitrogen atmosphere, the hydrated electrons attack PFOA, induce the fluoride elimination and react with hydrogen radical to yield the C6F13CHFCOOH radical via Eqs. (7–9) [45, 46]. The electrophilic radicals are attacked by hydrated electrons to cause an intramolecular defluorination and react further with hydrogen radical to yield C6F13CH2COOH, via Eqs. (10–12) [47, 48]. The generated C6F13CH2COOH can transform to C6F13COOH, via Eqs. (13) and (14) [46]. Other short-chain perfluorocarboxylic acid such as PFHxA, PFPA and PFBA is formed successively by similar reaction.
3.4.2 Mechanism of PFOS degradation by irradiation
On the basis of the intermediate products identified, the possible degradation pathway of PFOS is proposed in Fig. 5b. The degradation pathway of PFOS is similar to that of PFOA. Reaction of PFOS with hydrated electron yields the radical anion C8F17SO3·2– via Eq. (15). The radical anion is decomposed by fluoride elimination and reacts further with hydrogen radical to yield C7F15CHFSO3 − via Eqs. (16–19). The \( {\text{C}}_{7} {\text{F}}_{15} {\text{CHSO}}_{3}\cdot^{ - } \) may react with hydrogen radical to form C7F15CH2SO3 −, via Eq. (20) [48, 49]. The generated C7F15CH2SO3 − transforms to C7F15SO3 − via Eqs. (21) and (22) and finally complete degradation.
The proposed degradation mechanisms of PFOA and PFOS were different from previous studies [23, 28, 30, 50]. The present degradation mechanisms have two main differences from those in Ref. [50]. One is the main reactive species. While the hydroxyl radical took part in the mineralization process, hydrated electron and hydrogen radical played key roles in the EB degradation, especially hydrated electron. Another is the main reaction pathway. In electrolysis system, \( {\cdot} {\text{OH}} \)-initiated process was the main degradation pathway, and decarboxylation reaction occurred on anode first in electrolysis system. However, direct defluorination was the important pathway in electron beam degradation.
4 Conclusion
Electron beam radiolysis of PFOA and PFOS was performed. The degradation of PFOA and PFOS followed the pseudo-first-order kinetics. At 500 kGy, the degradation rates of PFOA and PFOS in N2 atmosphere were 95.7 and 85.9% at pH13, and the fluoride ion concentration rapidly increased to 324.6 and 416.9 μmol/L with corresponding defluorination rates of 46.8 and 71.4%, respectively. The results showed that PFOA and PFOS could be efficiently decomposed by EB irradiation in an anoxic alkaline solution. The work with radical scavengers demonstrated hydrated electron, and hydrogen radical especially, played key roles in EB degradation of PFOA and PFOS. Based on effects of the radicals and detected intermediate products, the EB degradation pathways of PFOA and PFOS were proposed. Further work may include adding catalyst to improve the degradation efficiencies at low dose, and toxicity of PFCs by-products.
References
M.K. So, S. Taniyasu, N. Yamashita et al., Perfluorinated compounds in coastal waters of Hong Kong, South China, and Korea. Environ. Sci. Technol. 38(15), 4056–4063 (2004). doi:10.1021/es049441z
Y.L. Mak, S. Taniyasu, L.W.Y. Yeung et al., Perfluorinated compounds in tap water from China and several other countries. Environ. Sci. Technol. 43(13), 4824–4829 (2009). doi:10.1021/es900637a
L. Ahrens, S. Felizeter, R. Sturm et al., Polyfluorinated compounds in waste water treatment plant effluents and surface waters along the River Elbe, Germany. Mar. Pollut Bull. 58(9), 1326–1333 (2009). doi:10.1016/j.marpolbul.2009.04.028
L. Ahrens, J.L. Barber, Z. Xie et al., Longitudinal and latitudinal distribution of perfluoroalkyl compounds in the surface water of the Atlantic Ocean. Environ. Sci. Technol. 43(9), 3122–3127 (2009). doi:10.1021/es803507p
M. Shoeib, T. Harner, G.M. Webster et al., Indoor sources of poly- and perfluorinated compounds (PFCS) in Vancouver, Canada: implications for human exposure. Environ. Sci. Technol. 45(19), 7999–8005 (2011). doi:10.1021/es103562v
E. Goosey, S. Harrad, Perfluoroalkyl substances in UK indoor and outdoor air: spatial and seasonal variation, and implications for human exposure. Environ. Int. 45, 86–90 (2012). doi:10.1016/j.envint.2012.04.007
B. Liu, H. Zhang, D. Yao et al., Perfluorinated compounds (PFCs) in the atmosphere of Shenzhen, China: spatial distribution, sources and health risk assessment. Chemosphere 138, 511–518 (2015). doi:10.1016/j.chemosphere.2015.07.012
C.P. Higgins, J.A. Field, C.S. Criddle et al., Quantitative determination of perfluorochemicals in sediments and domestic sludge. Environ. Sci. Technol. 39(11), 3946–3956 (2005). doi:10.1021/es048245p
H. Nakata, K. Kannan, T. Nasu et al., Perfluorinated contaminants in sediments and aquatic organisms collected from shallow water and tidal flat areas of the Ariake Sea, Japan: environmental fate of perfluorooctane sulfonate in aquatic ecosystems. Environ. Sci. Technol. 40(16), 4916–4921 (2006). doi:10.1021/es0603195
C.G. Pan, J.L. Zhao, Y.S. Liu et al., Bioaccumulation and risk assessment of per- and polyfluoroalkyl substances in wild freshwater fish from rivers in the Pearl River Delta region, South China. Ecotoxicol. Environ. Saf. 107, 192–199 (2014). doi:10.1016/j.ecoenv.2014.05.031
M. Houde, A.O. De Silva, D.C.G. Muir et al., Monitoring of perfluorinated compounds in aquatic biota: an updated review PFCs in aquatic biota. Environ. Sci. Technol. 45(19), 7962–7973 (2011). doi:10.1021/es104326w
A. Karrman, J.L. Domingo, X. Llebaria et al., Biomonitoring perfluorinated compounds in Catalonia, Spain: concentrations and trends in human liver and milk samples. Environ. Sci. Pollut. R 17(3), 750–758 (2010). doi:10.1007/s11356-009-0178-5
L.W.Y. Yeung, M.K. So, G.B. Jiang et al., Perfluorooctanesulfonate and related fluorochemicals in human blood samples from China. Environ. Sci. Technol. 40(3), 715–720 (2006). doi:10.1021/es052067y
F. Suja, B.K. Pramanik, S.M. Zain, Contamination, bioaccumulation and toxic effects of perfluorinated chemicals (PFCs) in the water environment: a review paper. Water Sci. Technol. 60(6), 1533–1544 (2009). doi:10.2166/wst.2009.504
K.S. Guruge, L.W.Y. Yeung, N. Yamanaka et al., Gene expression profiles in rat liver treated with perfluorooctanoic acid (PFOA). Toxicol. Sci. 89(1), 93–107 (2006). doi:10.1093/toxsci/kfj011
N. Johansson, A. Fredriksson, P. Eriksson, Neonatal exposure to perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA) causes neurobehavioural defects in adult mice. Neurotoxicology 29(1), 160–169 (2008). doi:10.1016/j.neuro.2007.10.008
J. Jeon, K. Kannan, H.K. Lim et al., Bioaccumulation of perfluorochemicals in pacific oyster under different salinity gradients. Environ. Sci. Technol. 44(7), 2695–2701 (2010). doi:10.1021/es100151r
E.S. Darling, M. Reinhard, Nanofiltration for trace organic contaminant removal: structure, solution, and membrane fouling effects on the rejection of perfluorochemicals. Environ. Sci. Technol. 42(14), 5292–5297 (2008). doi:10.1021/es703207s
Q. Zhou, S. Deng, Q. Yu et al., Sorption of perfluorooctane sulfonate on organo-montmorillonites. Chemosphere 78(6), 688–694 (2010). doi:10.1016/j.chemosphere.2009.12.005
H. Hori, A. Yamamoto, E. Hayakawa et al., Efficient decomposition of environmentally persistent perfluorocarboxylic acids by use of persulfate as a photochemical oxidant. Environ. Sci. Technol. 39(7), 2383–2388 (2005). doi:10.1021/es0484754
M. Sansotera, F. Persico, V. Rizzi et al., The effect of oxygen in the photocatalytic oxidation pathways of perfluorooctanoic acid. J. Fluor. Chem. 179, 159–168 (2015). doi:10.1016/j.jfluchem.2015.06.019
S. Gatto, M. Sansotera, F. Persico et al., Surface fluorination on TiO2 catalyst induced by photodegradation of perfluorooctanoic acid. Catal. Today 241, 8–14 (2015). doi:10.1016/j.cattod.2014.04.031
J. Ling, P. Zhang, S. Tian et al., Ferric ion mediated photodecomposition of aqueous perfluorooctane sulfonate (PFOS) under UV irradiation and its mechanism. J. Hazard. Mater. 271(4), 9–15 (2014). doi:10.1016/j.jhazmat.2014.01.061
Q. Zhuo, S. Deng, B. Yang et al., Efficient electrochemical oxidation of perfluorooctanoate using a Ti/SnO2–Sb–Bi anode. Environ. Sci. Technol. 45(7), 2973–2979 (2011). doi:10.1021/es1024542
H. Moriwaki, Y. Takagi, M. Tanaka et al., Sonochemical decomposition of perfluorooctane sulfonate and perfluorooctanoic acid. Environ. Sci. Technol. 39(9), 3388–3392 (2005). doi:10.1021/es040342v
J. Cheng, C.D. Vecitis, H. Park et al., Sonochemical degradation of perfluorooctane sulfonate (PFOS) and perfluorooctanoate (PFOA) in landfill groundwater: environmental matrix effects. Environ. Sci. Technol. 42(21), 8057–8063 (2008). doi:10.1021/es8013858
H. Hori, Y. Nagaoka, M. Murayama et al., Efficient decomposition of perfluorocarboxylic acids and alternative fluorochemical surfactants in hot water. Environ. Sci. Technol. 42(19), 7438–7443 (2008). doi:10.1021/es800832p
Z. Zhang, J.J. Chen, X.J. Lyu et al., Complete mineralization of perfluorooctanoic acid (PFOA) by γ-irradiation in aqueous solution. Scientific Reports 4, 7418 (2014). doi:10.1038/srep07418
O.S. Arvaniti, Y. Hwang, H.R. Andersen et al., Reductive degradation of perfluorinated compounds in water using Mg-aminoclay coated nanoscale zero valent iron. Chem. Eng. J. 262, 133–139 (2015). doi:10.1016/j.cej.2014.09.079
S.W. Yang, J.H. Cheng, J. Sun et al., Defluorination of aqueous perfluorooctanesulfonate by activated persulfate oxidation. PLoS ONE 8(10), e74877 (2013). doi:10.1371/journal.pone.0074877
Y.C. Lee, S.L. Lo, P.T. Chiueh et al., Microwave-hydrothermal decomposition of perfluorooctanoic acid in water by iron-activated persulfate oxidation. Water Res. 44(3), 886–892 (2010). doi:10.1016/j.watres.2009.09.055
Y.C. Lee, S.L. Lo, P.T. Chiueh et al., Efficient decomposition of perfluorocarboxylic acids in aqueous solution using microwave-induced persulfate. Water Res. 43(11), 2811–2816 (2009). doi:10.1016/j.watres.2009.03.052
C.E. Schaefer, C. Andaya, A. Urtiaga et al., Electrochemical treatment of perfluorooctanoic acid (PFOA) and perfluorooctane sulfonic acid (PFOS) in groundwater impacted by aqueous film forming foams (AFFFs). J. Hazard. Mater. 295, 170–175 (2015). doi:10.1016/j.jhazmat.2015.04.024
G. Xu, J.Z. Yao, L. Tang et al., Electron beam induced degradation of atrazine in aqueous solution. Chem. Eng. J. 275, 374–380 (2015). doi:10.1016/j.cej.2015.04.063
G. Xu, T. Bu, M. Wu et al., Electron beam induced degradation of clopyralid in aqueous solutions. J. Radioanal. Nucl. Chem. 288(3), 759–764 (2011). doi:10.1007/s10967-011-0986-1
W. Park, M.H. Hwang, T.H. Kim et al., Enhancement in characteristics of sewage sludge and anaerobic treatability by electron beam pre-treatment. Radiat. Phys. Chem. 78(2), 124–129 (2009). doi:10.1016/j.radphyschem.2008.09.010
J. Criquet, N.K.V. Leitner, Reaction pathway of the degradation of the p-hydroxybenzoic acid by sulfate radical generated by ionizing radiations. Radiat. Phys. Chem. 106, 307–314 (2015). doi:10.1016/j.radphyschem.2014.07.016
C. Peng, Y. Ding, F. An et al., Degradation of ochratoxin A in aqueous solutions by electron beam irradiation. J. Radioanal. Nucl. Chem. 306(1), 39–46 (2015). doi:10.1007/s10967-015-4086-5
A. Kimura, M. Osawa, M. Taguchi, Decomposition of persistent pharmaceuticals in wastewater by ionizing radiation. Radiat. Phys. Chem. 81(9), 1508–1512 (2012). doi:10.1016/j.radphyschem.2011.11.032
E. Illes, E. Takacs, A. Dombi et al., Hydroxyl radical induced degradation of ibuprofen. Sci. Total Environ. 447, 286–292 (2013). doi:10.1016/j.scitotenv.2013.01.007
G.V. Buxton, C.L. Greenstock, W.P. Helman et al., Critical review of rate constants for reactions of hydrated electrons, hydrogen atoms and hydroxyl radicals (·OH/·O−) in aqueous solution. J. Phys. Chem. Ref. Data 17(2), 513–886 (1988). doi:10.1063/1.555805
O. Marsalek, F. Uhlig, J. Vandevondele et al., Structure, dynamics, and reactivity of hydrated electrons by ab initio molecular dynamics. Acc. Chem. Res. 45(1), 23–32 (2012). doi:10.1021/ar200062m
M.H. Elkins, H.L. Williams, A.T. Shreve et al., Relaxation mechanism of the hydrated electron. Science 342(6165), 1496–1499 (2013). doi:10.1126/science.1246291
D.C. Walker, in Radiation Chemistry, ed. by E.J. Hart (Vancouver: American Chemical Society, Canada, 1968), pp. 49–64
H. Park, Photolysis of aqueous perfluorooctanoate and perfluorooctane sulfonate. Rev. Roum. Chim. 55(10), 611–619 (2010)
S. Zhou, H. Tang, W. Nan et al., Reductive defluorination of perfluorooctanoic acid by hydrated electrons in a sulfite-mediated UV photochemical system. J. Hazard. Mater. 262(22), 332–338 (2013). doi:10.1016/j.jhazmat.2013.08.059
C. Combellas, F. Kanoufi, A. Thiebault, Reduction of polyfluorinated compounds. J. Phys. Chem. B 107(39), 10894–10905 (2003). doi:10.1021/jp034846b
H. Park, C.D. Vecitis, J. Cheng et al., Reductive defluorination of aqueous perfluorinated alkyl surfactants: effects of ionic headgroup and chain length. J. Phys. Chem. A 113(4), 690–696 (2009). doi:10.1021/jp807116q
L. Jin, P. Zhang, Photochemical decomposition of perfluorooctane sulfonate (PFOS) in an anoxic alkaline solution by 185 nm vacuum ultraviolet. Chem. Eng. J. 280, 241–247 (2015). doi:10.1016/j.cej.2015.06.022
J. Niu, H. Lin, C. Gong et al., Theoretical and experimental insights into the electrochemical mineralization mechanism of perfluorooctanoic acid. Environ. Sci. Technol. 47(24), 14341–14349 (2013). doi:10.1021/es402987t
Author information
Authors and Affiliations
Corresponding authors
Additional information
This work was supported by the National Natural Science Foundation of China (Nos. 11675098, 41430644, 41473090, 41373098) and Program for Changjiang Scholars and Innovative Research Team in University (No. IRT13078).
Rights and permissions
About this article
Cite this article
Ma, SH., Wu, MH., Tang, L. et al. EB degradation of perfluorooctanoic acid and perfluorooctane sulfonate in aqueous solution. NUCL SCI TECH 28, 137 (2017). https://doi.org/10.1007/s41365-017-0278-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41365-017-0278-8