Abstract
Polyethylene terephthalate (PET) is widely used as a raw material for water bottles. However, since PET is an ester, water can react with it to release toxic molecules such as formaldehyde. This work provides a quantitative analysis, by high-performance liquid chromatography (HPLC–UV–Vis) after derivatization with dinitrophenylhydrazine (DNPH) of formaldehyde (FA) in flat natural or carbo-gaseous mineral waters bottled coming from different regions of Morocco, bought at Oujda in a local supermarket and analyzed in the Université of Oujda. At 18 °C, formaldehyde was not detected in 9 of the 12 bottles purchased at the supermarket. For bottles of flat natural mineral water incubated at 30 °C, FA concentrations were below the detection limit, reaching 0.38 µg/L in carbo-gaseous mineral water. At 40 °C, the concentration range was from 12.9 µg/L for flat natural to 31.5 µg/L for carbo-gaseous minerals water. After a 24-month storage at room temperature and away from light, flat natural mineral waters showed traces of FA, and in carbo-gaseous mineral water, the concentration of FA could reach 13.9 µg/L. The results show that temperature, storage duration, and the presence of CO2 influence the migration of formaldehyde from PET, but the rate of migration does not pose a threat to consumer health. Statistical parameters validating the analytical method used are presented with accuracy and precision. The repeatability of several stability statistics of the analytical technique was evaluated.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Production and variety of bottled waters in Morocco have experienced an increase in recent years in relationship with a growing consumption. Moroccan law distinguishes three types of bottled water: table water (ET), sources water (ES), and natural mineral water (EM) including flat natural mineral water and carbo-gaseous mineral water (EMG) (Gharibi et al. 2018; Schymanski et al. 2018).
The bottled water quality depends not only on the water quality captured but also on the conditions of its packaging and storage in a plastic container (Danopoulos et al. 2020). The water–plastic exchange process can influence the water chemistry (Koelmans et al. 2019). In this way, the exchange between organic material and water is always possible during the physical process, storage or transport under inadequate conditions (sun exposure, temperature rise, saturated atmosphere in gas) (Chen et al. 2023). Contamination of water by “secondary” micro-organic molecules and ions can also affect its quality and, as a result, have adverse effects on consumer health (Surana et al. 2022). Recent studies have shown that drinking water can be contaminated with a large number of plastic microparticles (10 000 particles per liter) between 6.5 and 100 µm of size (Marsden et al. 2019; Koelmans et al. 2019; Mason et al. 2018). In bottled water, packaging and/or the bottling process mainly cause this contamination (Bach et al. 2012).
Poly (ethylene terephthalate) (PET) is widely used to bottle water. The preference of this polyester to distribute water in bottles is due to its relative high chemical inertia, and its physical properties such as transparency, lightness, gas sealing, and its ease of recycling (Jin et al. 2020).
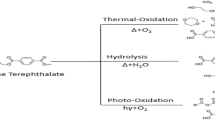
When interacting with water, the PET releases formaldehyde (FA), a toxic molecule (Coniglio et al. 2020) which is part of the family of volatile organic compounds (VOCs) (Zhang et al. 2020). Solubility of FA in water is 400 µg/L at 20 °C (Grützner and Hasse 2004) and increases with temperature (Kramer et al. 2012). Formaldehyde is a product of normal metabolism and is essential for the biosynthesis of certain amino acids in human metabolism cycle (Pietzke et al. 2020a, b). When FA is dissolved in water, it hydrates itself to give methylene glycol (or methanediol) (CH2(OH)2) which polymerizes to give a series of polymethylene glycols (Kramer et al. 2012; Abdu et al. 2014). The hydration reaction of formaldehyde to methylene glycol is reversible, both compounds are in equilibrium and are constantly and rapidly transformed from one to the other (Hazra et al. 2013). The conversion rate depends on temperature, pH, concentration, and the presence of other molecules such NH3, CH3OH et CO2 (Trincado et al. 2018). The photodegradation rate of formaldehyde was reduced in the presence of other VOCs such as benzene, toluene, ethylbenzene, o-xylene (Flueckiger et al. 2009), acetic acid, ethanol, and isooctane (Li et al. 2016). Therefore, even if they constitute two chemically distinct molecules, the Scientific Committee for Consumer Safety has considered that methylene glycol, in a solution, is equivalent to formaldehyde (Golden and Valentini 2014).
This organic compound can migrate from plastic to water during the packaging process. The amount of formaldehyde in water depends on the chemical quality of the raw material making the PET, the molecular weight of the polymer, and the manufacturing technology used for its production. The blowing temperature has a great impact on the FA transfer from the plastic to water (Mutsuga et al. 2005). Migration also depends on external factors such as temperature and storage duration, interactions between containers, and the concentration of carbon dioxide in the water (Nawrocki et al. 2002; Mutsuga et al. 2006; Abboudi et al. 2016).
The health risks resulting from the consumption of formaldehyde come from its cytotoxicity, its genotoxicity, and its endocrine disruptor characterization (Pfuhler and Wolf 2002). FA is considered carcinogenic to humans (Santana et al. 2019), and it is identified as one of the substances causing sick house syndrome (Agarwal et al. 2011). The formaldehyde is rapidly absorbed by the gastrointestinal and upper respiratory tracts (Dehghani et al. 2018). Gastrointestinal tract tumors in rats increased when given 1500 mg/L FA (Nielsen and Wolkoff 2010). The 50 mg/(kg*day) was identified for histopathological changes to the stomach from a well-conducted 2-year oral study in the rat (Til et al. 1989). Exogenous formaldehyde is taken up into the human body via ingestion, inhalation, and dermal absorption. Formaldehyde (FA) can cause allergic contact dermatitis, upper respiratory tract irritation, and coughing (Aung et al. 2021). Severe exposure may result in serious lower respiratory effects, such as bronchitis and pneumonia (Kumar et al. 2023). Acute ingestion exposure can produce hypotension and cardiovascular collapse. Nausea, vomiting, diarrhea, and abdominal pain may occur following ingestion of formaldehyde (Pandey et al. 2000). Ulceration and damage along the aerodigestive tract, including oral and gastrointestinal mucosa, have been reported in cases where formaldehyde had been ingested in excess quantities (Kundu et al. 2020). Humans can be affected by sensory of irritation at levels of 0.5 ppm formaldehyde and above contained in the air. Based on the available data, there is no conclusive evidence of systemic toxicity following oral administration of formaldehyde. Concerning the water bottled, the assessment of plastic inertia in contact with water is governed by the EU, the regulation states that the specific migration limit of FA in foodstuffs must be less than 15 mg/kg (Baiguini et al. 2011).
In carbo-gaseous mineral waters, the solubility of formaldehyde is greater; CO2 reacts with CH2O (FA) to form clusters of hydrated methanediol, which are less volatile, so the FA content increases (Huang et al. 2014). When CO2 reacts with CH2O, it consumes CH2O molecules, leading to the formation of larger molecules such as methanediol. These larger molecules are less volatile compared to CH2O, which means they are less likely to evaporate or escape into the atmosphere. As a result, the concentration of CH2O decreases while the concentration of the larger molecules, including methanediol, increases, leading to an increase in FA content (Huang et al. 2014). The pressure exerted by CO2 in the water on the sides of the bottles can also accelerate the aldehydes migration (Dąrowska et al. 2003). Storing carbonated mineral water for more than 8 to 9 months decreases the amount of aldehydes (Nawrocki et al. 2002).
In term of methodology used, the analysis of formaldehyde (FA) in water will be carried out by derivation with 2,4-dinitrophenylhydrazine (2,4-DNPH), then pre-concentration by liquid–liquid extraction (ELL), and analysis by HPLC–UV–Vis according to the methods (US-EPA 1992, 1996). This approach was chosen because it is used as a reference method, it is simple to implement, and it does not require sophisticated equipment.
The main objective of this work is to determine the migration of formaldehyde in quantity in two types of bottled water in Morocco [flat naturel mineral water (EM2) and carbo-gaseous mineral water (EMG)]. Some parameters influencing the migration of formaldehyde from PET to water will be studied. The focus will be on parameters that directly affect consumers, namely storage conditions (temperature and duration) and the presence of CO2 in natural waters.
Materials and methods
Instrumentation and reagents
The dinitrophenyl hydrazone (FA-DNPH) was quantified using an Agilent-type HPLC system with a diode array detector (waters 2996), equipped with a 20 µL injection loop. The Alliance eW2695 was used, it is a model of HPLC separation module, which likely includes components for sample injection, solvent delivery, column compartment, and other modules necessary for chromatographic separation. The separation is carried out on a C18 type column 250 mm long, 4.6 mm internal diameter filled with particles 5 μm in diameter. An isocratic mobile phase consisting of a 60:40 (v:v) ACN/H2O mixture was pumped into the column at a flow rate of 1 mL/min. The column was maintained at 25 °C. Acquisition by HPLC–UV was carried out at 360 nm for FA-DNPH (Qian et al. 2017). The wavelength was chosen based on the UV–visible spectra plotted from hydrazone. The hydrazone was eluted after 5 min.
The total organic carbon (TOC) analysis (TOC/ TN Analyzer multi-N/C 2100/2100) is available at the physical measurements room of the Oujda Faculty of Science. This test is not specific to carbonyl group (formaldehyde) but it gives an indication of the enrichment of water in the organic fraction of C. The tests commonly used to identify carbonyl compounds are the Tollens test, the Fehling test, and the test by Benedict (Asatkar and Basak 2023).
Samples
In this study, two types of water were chosen: flat natural mineral water (EM2) and carbo-gaseous mineral water (EMG). The effect of the duration of bottle storage on the migration of formaldehyde into the water has been followed and analyzed. Moreover, the impact of storage temperature has been determined. Table 1 shows the samples studied, their date of bottling marked on the corks, and the date of carrying out the analyses.
The experimentation concerned 18 EM2 and 18 EMG bottles (each EM2 and each EMG bottles are issue from the same brand) to evaluate, on the one hand, the evolution of the concentration of the organic molecules with the temperature (18 °C—room temperature, 30°, 40°, and 50 °C) and with the storage time (10 days, 1 year, and 2 years) and, on the other hand, to check the reproducibility of the process, with 3 EM2 and 3 EMG bottles for each experiment. Note that the same group (3 EM2 and 3 EMG), stored for 10 days at 18 °C, was used as a common control for the temperature and storage time tests. The high temperatures, 40 and 50 °C, were chosen, given the summer temperatures in many regions of Morocco which exceed 40 °C. Likewise, water bottles are displayed by small traders outside shops in conditions that can exceed 50 °C. The different bottles were stored in a place sheltered from the sun and rising temperatures. The water bottles were conserved, as far as possible, in natural conditions in terms of temperature and external humidity, and away from any exposure of odors. For long-term storage, all samples were stored at room temperature with artificial lighting and controlled temperature for 24 months, mimicking typical conditions in retail outlets, supermarkets, and homes.
Effect of storage time on formaldehyde migration
To study the effect of storage, 18 bottles of water (bottled in Morocco) divided into 3 groups were analyzed. The first group contained bottles of 0.5L of water stored for 2 years: three flat natural mineral water (EM2) and three carbo-gaseous mineral water (EMG)). The second group included bottles of 0.5L of water stored a year: three flat natural mineral water and three carbo-gaseous mineral water. The third and last group was composed of six non-stored bottles (V = 0.5L) as control samples (in fact 10 days of storage at room temperature): three flat natural mineral water and three carbo-gaseous mineral water.
The bottling dates marked on the bottles ranged from 9 to 14 days before the date of purchase (Table 1).
Preparation of calibration standards
A 1 mg/L stock solution of formaldehyde (FA) has been prepared in 1000 mL of ultra-pure water. Then seven calibration solutions were prepared from the initial solution added to 100 mL of ultra-pure water (Water for chromatography (LC–MS Grade) LiChrosolv®. Merck), with the following concentrations 0, 10, 20, 50, 100, 250, and 500 µg/L to draw the calibration curve. The stock solution was kept cold (4 ºC). A chemical derivatization was performed modify the structure of formaldehyde, in particular to make them visible by the detection system of the analysis instrument. Formaldehyde does not have a chromophore group in the ultraviolet. By reaction of hydrazine with formaldehyde in an acid medium, hydrazones are formed, which show a maximum absorption around 360 nm (Qian et al. 2017). The DNPH solution at 200 mg/L for the derivation was prepared in a 60:40 (v:v) acetonitrile/water mixture. The citrate buffer (1 mol/L, pH 3.4) was prepared by mixing 80 mL of citric acid solution (1 mol/L) and 20 mL of sodium citrate solution (1 mol/L). It was used to buffer the derivatization reaction mixture at pH 3.4 according to the method (US-EPA 1992, 1996). To be able to calculate the yield of the reaction of derivatization of formaldehyde into hydrazone, a calibration line was carried out by the standards of the hydrazones in acetonitrile/water 60:40 (v:v). All reagents used were obtained from Sigma–Aldrich Co. with a purity greater than 99.8%.
The procedure was carried out as follows: 100 mL of standard solution or commercial water was collected from glass beakers, and 4 mL of citrate buffer (1 mol/L) was added to adjust the pH of the sample to 3.4. Then 6 mL of 0.2 mg/L DNPH in ACN/H2O (60:40) was added and the sample was maintained for 2 h at 0 °C to produce hydrazones (Shi et al. 2022). Thus, the reaction medium has a total volume of 110 mL (Fig. 1).
Hydrazone solution concentration
The analytical methodology followed was utilized, after derivatization, to perform a liquid–liquid extraction with dichloromethane and to analyze them by liquid-phase chromatography coupled with a UV detector. The choice of the ELL was linked to its use in all laboratories. This technique will then be validated according to the AFNOR Standard (NF 2009) before finally being applied to a batch of mineral water samples. Once the formaldehyde is transformed into the hydrazone derivative, the classic pre-concentration technique was applied and the concentration of the sample was evaluated: liquid–liquid extraction. The extraction device (separation funnel, funnel, collection vial, and beaker) was rinsed with 20 mL of dichloromethane for 15 min. An ultra-pure water blank (water reputed to be free of traces of organic compounds) was carried out, following the same procedure as that applied for the samples to check the absence of contamination during the sample extraction procedure. Formaldehyde extraction using LLE was performed based on the method (US-EPA 1996). The solution containing the hydrazone was transferred to a 250 mL separating funnel and extracted with three 20 mL portions of dichloromethane. Each vial was mechanically shaken for 15 min. The three dichloromethane fractions were combined, 5 g of sodium sulfate was added to remove all traces of moisture, and the solution was filtered. The filtrate was completely evaporated under a stream of nitrogen, the residue was dissolved in 10 mL acetonitrile:water of 60:40 (v:v) and then filtered through a 0.45 µm nylon filter. The extracts obtained can then be stored in 15 mL glass tubes with screw caps and in the freezer (− 4 °C) while waiting for their analysis. The inner surface of the plugs (in contact with the extract) was covered with aluminum foil to avoid contamination.
Effect of temperature on formaldehyde migration
To study the influence of temperature on the transfer of organic pollutants to water, two groups of samples were selected. The first contains three flat natural mineral waters in transparent bottles and the water bottles are of the same brand and production lot. The second group is composed of three carbo-gaseous mineral waters packaged in transparent bottles. Water bottles are also of the same brand and production lot. And the same water quality and number of bottles (6) were reproduced at different temperature for a total of 24 water bottle.
Then the tests were carried out with respect to four conditions temperature, (1) the control samples, with the three flat natural mineral water and three carbo-gaseous mineral waters which were preserved at room temperature for 10 days. The experiment was planted during the spring season at a room temperature of around 18 °C (these bottles were the same used for the storage test), (2) the samples incubated at 30 °C in the oven for 10 days, (3) the samples incubated at 40 °C in the oven for 10 days, and 4) finally the samples incubated at 50 °C in the oven for 10 days.
The adopted analytical methodology comprises three steps (Baños and Silva 2009), derivatization of formaldehyde (Fig. 2). The first step is the process of derivatization which is a technique that allows the analysis of compounds that cannot be directly analyzed. Indeed, formaldehyde is trapped by 2,4-dinitrophenylhydrazine (or 2,4-DNPH) by chemisorption; chemical derivatization involves the reaction of hydrazine (2,4-DNPH) with aldehydes in an acid medium to give hydrazones. The second step is the liquid–liquid extraction process with dichloromethane from the hydrazone formed and the last step is the analysis by liquid chromatography coupled to a DAD detector of the product extracted in the organic solvent.
Aldehyde analysis method validation
The analysis of aldehydes by HPLC–UV–Vis after derivatization with DNPH and concentration by SPE has been validated according to the AFNOR Standard (NF 2009). For this, a calibration range in the EUP has been prepared. It is composed of six levels: 10, 20, 50, 100, 250, and 500 µg/L. This range was carried out over 5 consecutive days. Each point in the range was derived and extracted following the procedure described above. Statistical tests were performed to verify linearity: Fisher’s test for repeatability stability, and Cochran’s test. Dixon’s test was done to look for outliers.
The chosen limit of quantification, the lowest concentration in the calibration curve which is 10 µg/L, was considered acceptable when: the mean concentration measured (n = 10) + (2 × standard deviation) < LOQ + 0 0.6 × LOQ, and when the mean concentration (n = 10) – (2 × standard deviation) > LOQ – 0.6 × LOQ. For the preparation, six standard solutions were prepared from EUP at concentration levels of 10 µg/L. These solutions were analyzed on 5 different days at the rate of 2 determinations per day.
Additional two checks were performed to monitor the validity and quality of the trial. The first consists doping of two samples. This doping makes it possible to highlight a possible matrix effect within the sample analyzed. Matrix effects are due to the presence of interfering substances in the sample, which are extracted together with the analytes. These substances alter the performance of the method by decreasing or enhancing the signal of the analytes. To detect this type of interference, doping is carried out in the matrix. These doping consist of the addition of a known quantity of analytes in the sample submitted for analysis. The second was an intra-series control for each series of samples. This control consists in preparing two control solutions of theoretical concentration of formaldehyde at 10 and 250 µg/L in the EUP. These solutions are intercalated between the series of samples, to assess the accuracy and drift of the analytical method. The calculated experimental concentration must not deviate from the theoretical value by 10%, to validate the series.
Results and discussion
Total organic carbon (TOC) test
To check for the presence of organic compounds in the water non-stored and stored for 2 years, such as carbonyls, we used the total organic carbon (TOC) test. The results obtained show that the EM2 flat natural mineral water contains a concentration of 1.22 mg/L, while the EMG carbo-gaseous mineral waters show a concentration of 7.78 mg/L. Carbonyl compounds such as formaldehyde found in bottled water are more likely to have originated from PET than from aquifers (Nawrocki et al. 2002). The presence of CO2 increases this concentration.
Effect of storage time on formaldehyde migration
The effect of storage time on the migration of FA in water is shown in Fig. 3 and Table 2. It is noted that formaldehyde is detected in the different waters analyzed only after 2 years of room condition storage.
The concentration of formaldehyde in samples not stored and analyzed immediately after purchase shows values below the detection limit. For flat natural mineral water and carbo-gaseous mineral water bottles stored over 12 months, formaldehyde was not detected in any of the 12 samples studied. Only after 24 months of storage, three samples showed traces of the organic molecule with a maximum concentration of 13.9 µg/L found in flat natural mineral water. After 24 months of storage, the amount of formaldehyde in the bottle of carbo-gaseous mineral water (EMG) is lower than those found in flat natural mineral water (EM2). This drop is probably due to the CO2 leakage by the cap causing also the departure of the predisposing FA molecules which volatilize.
Formaldehyde concentrations in Morocco PET-bottled water remain low compared to levels in other countries. In Japan, levels of FA detected in bottled water samples range from 10.1 to 27.9 µg/L (Li et al. 2016), in Taiwan, the concentration found can reach 200 µg/L (Tsai et al. 2003).
Effect of temperature on formaldehyde migration
Figure 4 and Table 3 represent results of the effect of temperature on the concentration of the formaldehyde. In the carbo-gaseous naturel water (EMG), the detection of formaldehyde occurs from 30 °C and disappears at 50 °C. However, in flat natural mineral water (EM2), formaldehyde is only detected starting 40 °C and increases with temperature. In water EMG, several other phases appear as the temperature increases.
At room temperature (T = 18 °C), formaldehyde was not detected in both types of water. However, when the temperature is close to 30 °C, the summer temperature in Oujda city, traces of formaldehyde appeared in the carbo-gaseous mineral waters (Table 3). Formaldehyde occurs in both type of water at higher temperatures (T = 40 °C). In EM2 flat natural mineral water, the value of formaldehyde is on average 12.9 µg/L. This value obtained after 10 days at 40 °C in flat mineral water is close to that found after 24 months of storage at room temperature in the same flat mineral water (Fig. 3). This is due to the solubility of formaldehyde which increases with temperature, accelerating the migration of this plastic molecule to water. Compared to other studies, the concentration found in flat natural mineral water (5.75 µg/L) is lower than the maximum value (39.4 µg/L) detected in PET-bottled naturel mineral water in Lebanon exposed to 40 °C for 10 days (Rayes et al. 2012). When the temperature reaches 50 °C, the amount of formaldehyde in flat natural mineral water decreases, and in carbo-gaseous mineral water, this value is below the detection threshold. When the temperature rises (T = 50 °C), close to PET's vitreous transition temperature (61–77 °C), the polymer becomes soft and the sealing of the caps is less close off. The saturated vapor pressure of water in the bottle increases sharply with the temperature. It rises from 2.3 kPa at 20 °C to 12.3 kPa at 50 °C (Mokdad et al. 2012). This pressure is exerted on the walls of the PET bottle and especially on the cap, allowing dissolved gases in water to escape. The departure of carbon dioxide drains and decreases the concentration of formaldehyde molecules dissolved in gaseous water.
The comparison of formaldehyde concentrations obtained in flat natural mineral and carbo-gaseous mineral waters from Moroccan PET-bottled is given in Table 4. It should be noted that the small discrepancy observed in the values found by the different studies in waters from different countries might be due to the chemistry of the waters studied and the PET composition of the bottles. However, all the values are of the same order of magnitude as those found in the waters bottled in Morocco and they are all lower than the guide values. The formaldehyde concentrations expressed in µg/L are in the range of 5.75–13.9 and 0.38–31.5 for flat natural mineral water and carbo-gaseous mineral waters, respectively.
Effect of CO2 on formaldehyde migration
The comparison between carbo-gaseous mineral water and flat mineral natural water clearly shows that the presence of CO2 combined with an increase of temperature accelerates the migration of formaldehyde in the water (Table 3). In fact, it is the combination of two parameters which induces this increase of FA, because in Table 2, it shows that, in room temperature condition, no matter the time period, the CO2 content and the FA affect more the flat mineral water.
Indeed, the presence of formaldehyde was detected in carbo-gaseous mineral water at 30 °C, while it was absent in flat natural mineral water. At 40 °C, the concentration increased to 31.5 µg/L in carbo-gaseous mineral water, almost triple what was detected in flat natural mineral water. This value (31.5 µg/L) is higher than the referenced threshold of odor and taste in waters (between 20 and 40 µg/L) (Rayes et al. 2012). This result, therefore, confirms other studies highlighting the accelerator effect of the migration of formaldehyde in water containing carbon dioxide (Dąrowska et al. 2003; Redžepović et al. 2012).
The solubility of CO2 in water decreases with temperature, it is equal to 1.67 g/L and 0.76 g/L respectively, at 20 °C and 50 °C (Diamond and Akinfey, 2003). Thus, when the carbo-gaseous mineral water reaches 50 °C, the concentration of CO2 decreases by half.
The pressure exerted by the CO2 released on the sides of the bottle was calculated by assuming that CO2 is a perfect gas. The mass degassed at 50 °C is 0.93 mg, if the estimated unfilled volume in the bottle is 5 mL, the partial pressure of CO2 calculated is 68 Pa. This pressure adds to that exerted by the saturated water vapor (Redžepović et al. 2012).
Taking advantage of the relaxation of the plug, the CO2 escapes with a part of formaldehyde molecules. The formation reaction of methanediol clusters hydrated by CO2 is reversible and strongly influenced by temperature (Dąrowska et al. 2003). At a temperature (T = 50 °C), the concentration in carbo-gaseous mineral water is below the detection threshold, which agrees with other studies (Dąrowska et al. 2003).
With the increase of incubation temperature, other important peaks appear (Fig. 4). These peaks are likely due to other aldehydes (acetaldehyde) which also have migrated from plastic to water (Dąrowska et al. 2003). These peaks are more intense for carbo-gaseous mineral waters.
Compliance with drinking water standards
Based on a comparison of permissible limits set by various international organizations (Table 5), formaldehyde (FA) levels in this study were below the allowable limit for drinking water. In general, the formaldehyde levels of the present study are in agreement with the results of previous water studies in different geographic locations. Values of formaldehyde in all natural mineral water in this study are lower than those reported for water from US supply pipes (Liteplo et al. 2002), ozonated drinking water in bottles water of the US (Cogliano et al. 2005), water samples purchased from supermarkets and shops in bottles waters of Iran (Dehghani et al. 2018), commercial mineral water in Japanese PET bottle water and Northern American PET bottle water (Mutsuga et al. 2006), carbo-gaseous mineral water in bottles water of Turkey (Özlem 2008) and for natural spring water in bottled-Damascus in Syria (Abboudi and Odeh 2015). But it is higher than those reported for raw water in North Saskatchewan River of US (Bujaczek et al. 2021), and for ozonated water and carbo-gaseous mineral natural mineral water in Poland PET bottles (Dąrowska et al. 2003). On the other hand, formaldehyde levels in the flat natural mineral water of the present work are in agreement with the findings presented by Santana et al. (2019) for commercial mineral water in European PET bottles. Formaldehyde (FA) concentrations in carbo-gaseous mineral water from this study are in the range of bibliographic works (Özlem 2008).
Validation of the formaldehyde analysis method
The analysis of aldehydes by HPLC/DAD after derivatization with DNPH and concentration by SPE has been validated according to the AFNOR Standard (NF 2009). For this, a calibration range in the EUP has been prepared. It is composed of six levels: 10, 20, 50, 100, 250, and 500 µg/L. This range was carried out over 5 consecutive days. Each point of the range has been derived and extracted following the procedure described previously (Experimental). The limit of quantification (LQ) was validated at 3.5 µg/L.
The intra-series control is a control of accuracy and drift at the level of the extraction method and the analytical instrument. A control solution is prepared from an intermediate solution. The theoretical concentration of this solution is 4 µg/L. This solution is prepared in the EUP when controlling a series of still water samples and in bottled sparkling water. This control solution is treated in the same way as the test samples. To validate the analytical series, the difference between the experimental concentration and the theoretical concentration must be less than or equal to 10%.
Standard deviation values for the various tests carried out for formaldehyde calibration are shown in Table 6. All statistical tests were performed on the area in µV sec. Figure 5 shows some examples of calibration.
The linearity curve, with R2 = 0.997, shows a slope of 1.66, close to 1.36 found by Baños and Silva for formaldehyde A = 2.9 ± 0.3 + (1.36 ± 0.04)*C (Baños and Silva 2009) (Fig. 6A). The difference between the measured values and the fitted values is mostly less than 10% except for “20” the concentration of 20 µg/L which is 10.4% (Fig. 6B).
Table 7 summarizes the statistical data obtained from the various tests adopted to validate the analytical method.
Kruskal–Wallis ANOVA test (Fisher’s test) values deduced from the Fisher test are shown in Table 7. A very low value of Prob > Chi-square (3.2887 × 10–5) strongly suggests that the variables are dependent and that the observed differences between groups are not due to chance. It suggest a good fit of the model to the data. Chi-square equals 28.2 and DF equals 5.
Repeatability stability test shows that a high value for R-Square equal to 1 and a low coefficient of variation (close to 0) indicate a good fit. A low value of root mean square error (Root MSE = 0.008) indicates good accuracy of fit.
Cochran’s C test for extreme variances gives a p value equal to 1 which means that the data are probably normally distributed and that statistical analyses based on this assumption can be considered reliable and valid.
For the Dixon test, the significance is 0, at the 0.05 level, there are no significant outliers.
Conclusion
This study investigated formaldehyde migration from PET bottles into flat natural mineral water or naturally carbo-gaseous mineral water, considering storage time, temperature, and water type. It was found that formaldehyde migrated into flat natural mineral water only after extended storage (24 months) at room temperature. Higher temperatures expedited formaldehyde migration, with a concentration equivalent to 2 years at room temperature reached in 10 days at 40 °C. This underscores the importance of proper storage and transport to prevent formaldehyde contamination, especially in hot climates like Morocco.
Temperature near PET’s glass transition (50 °C) softens the plastic, weakening cap seals and allowing formaldehyde escape through steam pressure. Similarly, carbo-gaseous mineral water showed temperature-dependent formaldehyde migration, with CO2 presence exacerbating it. At 40 °C, formaldehyde concentration was nearly three times higher than in flat natural mineral water due to CO2-induced methanediol cluster formation during degassing.
Overall, Moroccan PET-bottled flat natural and carbo-gaseous mineral waters appear safe for consumption regarding formaldehyde levels, below international standards. Statistical analyses validated the analytical method’s reliability, indicating good linearity, statistical significance, stability, and normal distribution of results, with no outliers. Thus, the method proves suitable for formaldehyde concentration determination in water.
In view of the results found, we could expand the study to look at the difference in possible values, taking into account the place of purchase in rural or urban areas, the bottling date, storage conditions.
References
Abboudi M, Odeh A (2015) Impact of sunlight/dark storage on natural spring water bottled in polyethylene terephthalate. J Water Supply Res Technol AQUA 64(2):149–156. https://doi.org/10.2166/aqua.2014.076
Abboudi M, Odeh A, Aljoumaa K (2016) Carbonyl compound leaching from polyethylene terephthalate into bottled water under sunlight exposure. Toxicol Environ Chem 98(2):167–178. https://doi.org/10.1080/02772248.2015.1116001
Abdu H, Kinfu Y, Agalu A (2014) Toxic effects of formaldehyde on the nervous system. Int J Anat Physiol 3:50–59
Agarwal M, Manan D, Upadhayaya S (2011) Adsorption of formaldehyde on treated activated carbon and activated alumina. Curr World Environ 6(1):53. https://doi.org/10.12944/CWE.6.1.06
Asatkar AK, Basak RK (2023) Carbohydrate: introduction and fundamentals. In: Verma C, Verma DK (eds) Handbook of biomolecules. Elsevier, pp 25–55. https://doi.org/10.1016/B978-0-323-91684-4.00020-7
Aung WY, Sakamoto H, Sato A, Yi EEPN, Thein ZL, Nwe MS, Shein S, Linn H, Uchiyama U, Kunugita N, Win-Shwe TT, Mar O (2021) Indoor formaldehyde concentration, personal formaldehyde exposure and clinical symptoms during anatomy dissection sessions, University of Medicine 1, Yangon. Int J Environ Res Public Health 18(2):712. https://doi.org/10.3390/ijerph18020712
Bach C, Dauchy X, Chagnon MC, Etienne S (2012) Chemical compounds and toxicological assessments of drinking water stored in polyethylene terephthalate (PET) bottles: a source of controversy reviewed. Water Res 46(3):571–583. https://doi.org/10.1016/j.watres.2011.11.062
Baiguini A, Colletta S, Rebella V (2011) Materials and articles intended to come into contact with food: Evaluation of the rapid alert system for food and feed (RASFF) 2008–2010. Ig Sanita Pubblica 67(3):293–305
Baños CE, Silva M (2009) In situ continuous derivatization/pre-concentration of carbonyl compounds with 2, 4-dinitrophenylhydrazine in aqueous samples by solid-phase extraction: Application to liquid chromatography determination of aldehydes. Talanta 77(5):1597–1602. https://doi.org/10.1016/j.talanta.2008.09.053. (42(10))
Bujaczek T, Kolter S, Locky D, Ross MS (2021) Characterization of microplastics and anthropogenic fibers in surface waters of the North Saskatchewan River, Alberta, Canada. Facets 6(1):26–43. https://doi.org/10.1139/facets-2020-0057
Chen Y, Li H, Yin Y, Shan S, Huang T, Tang H (2023) Effect of microplastics on the adherence of coexisting background organic contaminants to natural organic matter in water. Sci Total Environ 905:167175. https://doi.org/10.1016/j.scitotenv.2023.167175
Cogliano VJ, Grosse Y, Baan RA, Straif K, Secretan MB, Ghissassi FE (2005) Meeting report: summary of IARC monographs on formaldehyde, 2-butoxyethanol, and 1-tert-butoxy-2-propanol. Environ Health Perspect 113(9):1205–1208
Coniglio MA, Fioriglio C, Laganà P, Coniglio MA, Fioriglio C, Laganà P (2020) Non-intentionally added substances. Springer International Publishing, pp 43–58
Danopoulos E, Twiddy M, Rotchell JM (2020) Microplastic contamination of drinking water: A systematic review. PLoS ONE 15(7):e0236838. https://doi.org/10.1371/journal.pone.0236838
Dąrowska A, Borcz A, Nawrocki J (2003) Aldehyde contamination of mineral water stored in PET bottles. Food Addit Contam 20(12):1170–1177. https://doi.org/10.1080/02652030310001620441
Dehghani MH, Farhang M, Zarei A (2018) Investigation of carbonyl compounds (acetaldehyde and formaldehyde) in bottled waters in Iranian markets. Int Food Res J 25(2):876–879
Diamond LW, Akinfiev NN (2003) Solubility of CO2 in water from− 1.5 to 100 C and from 0.1 to 100 MPa: evaluation of literature data and thermodynamic modelling. Fluid Phase Equilibr 208(1–2):265–290. https://doi.org/10.1016/S0378-3812(03)00041-4
Flueckiger J, Ko FK, Cheung KC (2009) Microfabricated formaldehyde gas sensors. Sensors 9(11):9196–9215. https://doi.org/10.3390/s91109196
Gharibi E, Ghalit M, Taupin JD, Patris N, Kouotou D (2018) Assessment of the quality of Moroccan bottled water by application of quality indices. J Water Supply Res Technol AQUA 67(6):576–585. https://doi.org/10.2166/aqua.2018.022
Golden R, Valentini M (2014) Formaldehyde and methylene glycol equivalence: critical assessment of chemical and toxicological aspects. Regul Toxicol Pharmacol 69(2):178–186. https://doi.org/10.1016/j.yrtph.2014.03.007
Grützner T, Hasse H (2004) Solubility of formaldehyde and trioxane in aqueous solutions. J Chem Eng Data 49(3):642–646. https://doi.org/10.1021/je030243h
Hazra MK, Francisco JS, Sinha A (2013) Gas phase hydrolysis of formaldehyde to form methanediol: impact of formic acid catalysis. J Phys Chem A 117(46):11704–11710. https://doi.org/10.1021/jp4008043
Huang PH, Hung SC, Huang MY (2014) Molecular dynamics investigations of liquid–vapor interaction and adsorption of formaldehyde, oxocarbons, and water in graphitic slit pores. Phys Chem Chem Phys 16(29):15289–15298. https://doi.org/10.1039/C4CP01922A
Jin Q, Shen Y, Cai Y, Chu L, Zeng Y (2020) Resource utilization of waste V2O5-based deNOx catalysts for hydrogen production from formaldehyde and water via steam reforming. J Hazard Mater 381:120934. https://doi.org/10.1016/j.jhazmat.2019.120934
Koelmans AA, Nor NHM, Hermsen E, Kooi M, Mintenig SM, De France J (2019) Microplastics in freshwaters and drinking water: Critical review and assessment of data quality. Water Res 155:410–422. https://doi.org/10.1016/j.watres.2019.02.054
Kramer ZC, Takahashi K, Vaida V, Skodje RT (2012) Will water act as a photocatalyst for cluster phase chemical reactions? Vibrational overtone-induced dehydration reaction of methanediol. J Chem Phys 136(16):164302. https://doi.org/10.1063/1.4704767
Kumar P, Singh AB, Arora T, Singh S, Singh R (2023) Critical review on emerging health effects associated with the indoor air quality and its sustainable management. Sci Total Environ 872:162163. https://doi.org/10.1016/j.scitotenv.2023.162163
Kundu A, Dey P, Bera R, Sarkar R, Kim B, Kacew S, Kacew S, Lee MB, Karmakar S, Kim HS (2020) Adverse health risk from prolonged consumption of formaldehyde-preserved carps in eastern region of Indian population. Environ Sci Pollut Res 27:16415–16425. https://doi.org/10.1007/s11356-020-07993-0
Li B, Wang ZW, Lin QB, Hu CY (2016) Study of the migration of stabilizer and plasticizer from polyethylene terephthalate into food simulants. J Chromatogr Sci 54(6):939–951. https://doi.org/10.1093/chromsci/bmw025
Liteplo RG, Beauchamp R, Chénier R, Meek ME (2002) Formaldehyde. Concise International Chemical Assessment Document 40. World Health Organization, Geneva
Marsden P, Koelmans AA, Bourdon-Lacombe J, Gouin, T, D'Anglada L, Cunliffe D, Jarvis P, Fawell J, De France J (2019) Microplastics in drinking water. Geneva: World Health Organization; 2019. Licence: CC BY-NC-SA 3.0 IGO.
Mason SA, Welch VG, Neratko J (2018) Synthetic polymer contamination in bottled. Water Front Chem. https://doi.org/10.3389/fchem.2018.00407
Mokdad S, Georgin E, Mokbel I, Jose J, Hermier Y, Himbert M (2012) On the way to determination of the vapor-pressure curve of pure water. Inter J Thermophys 33(8):1374–1389. https://doi.org/10.1007/s10765-012-1261-6
Mutsuga M, Tojima T, Kawamura Y, Tanamoto K (2005) Survey of formaldehyde, acetaldehyde and oligomers in polyethylene terephthalate food-packaging materials. Food Addit Contam 22(8):783–789. https://doi.org/10.1080/02652030500157593
Mutsuga M, Kawamura Y, Sugita-Konishi Y, Hara-Kudo Y, Takatori K, Tanamoto K (2006) Migration of formaldehyde and acetaldehyde into mineral water in polyethylene terephthalate (PET) bottles. Food Addit Contam 23(2):212–218. https://doi.org/10.1080/02652030500398361
Nawrocki J, Dąbrowska A, Borcz A (2002) Investigation of carbonyl compounds in bottled waters from Poland. Water Res 36(19):4893–4901. https://doi.org/10.1016/S0043-1354(02)00201-4
NF (2009) T 90–210, Qualité de l'eau-Protocole d'évaluation initiale des performances d'une méthode dans un laboratoire. Afnor 2009:1–43 (in french)
Nielsen GD, Wolkoff P (2010) Cancer effects of formaldehyde: a proposal for an indoor air guideline value. Arch Toxicol 84:423–446. https://doi.org/10.1007/s00204-010-0549-1
Özlem KE (2008) Acetaldehyde migration from polyethylene terephthalate bottles into carbonated beverages in Türkiye. Int J Food Sci Technol 43(2):333–338. https://doi.org/10.1111/j.1365-2621.2006.01443.x
Pandey CK, Agarwal A, Baronia A, Singh N (2000) Toxicity of ingested formalin and its management. Hum Exp Toxicol 19(6):360–366. https://doi.org/10.1191/096032700678815954
Pfuhler S, Wolf HU (2002) Effects of the formaldehyde releasing preservatives dimethylol urea and diazolidinyl urea in several short-term genotoxicity tests. Mutat Res Genet Toxicol Environ Mutagen 514(1–2):133–146. https://doi.org/10.1016/S1383-5718(01)00335-7
Pietzke M, Burgos-Barragan G, Wit N, Tait-Mulder J, Sumpton D, Mackay GM, Vazquez A (2020a) Amino acid dependent formaldehyde metabolism in mammals. Commun Chem 3(1):78
Pietzke M, Meiser J, Vazquez A (2020b) Formate metabolism in health and disease. Mol Metab 33:23–37. https://doi.org/10.1016/j.molmet.2019.05.012
Qian H, Pramanik S, Aprahamian I (2017) Photochromic hydrazone switches with extremely long thermal half-lives. J Am Chem Soc 139(27):9140–9143. https://doi.org/10.1021/jacs.7b04993
Rayes LA, Saliba CO, Ghanem A, Randon J (2012) BTES and aldehydes analysis in PET-bottled water in Lebanon. Food Addit Contam Part B 5(3):221–227. https://doi.org/10.1080/19393210.2012.698311
Redžepović AS, Ačanski MM, Vujić ĐN, Lazić VL (2012) Determination of carbonyl compounds (acetaldehyde and formaldehyde) in polyethylene terephthalate containers designated for water conservation. Chem Ind Chem Eng Q 18(2):155–161. https://doi.org/10.2298/CICEQ110606057R
Santana FO, Campos VP, Santos IF, Cruz LP, Brito AVS (2019) Seasonal quimiometric study of formaldehyde and acetaldehyde atmospheric levels and health risk assessment, in urban areas of Salvador-Bahia, Brazil. Microchem J 147:524–531. https://doi.org/10.1016/j.microc.2019.03.069
Schymanski D, Goldbeck C, Humpf HU, Fürst P (2018) Analysis of microplastics in water by micro-Raman spectroscopy: release of plastic particles from different packaging into mineral water. Water Res 129:154–162. https://doi.org/10.1016/j.watres.2017.11.011
Shi L, Li Y, Zhou X, Guo Y, Han Q, Xia W, Zhang W (2022) Isopropyl-naphthylamide-hydrazine as a novel fluorescent reagent for ultrasensitive determination of carbonyl species on UPLC. Microchem J 177:107308. https://doi.org/10.1016/j.microc.2022.107308
Surana D, Gupta J, Sharma S, Kumar S, Ghosh P (2022) A review on advances in removal of endocrine disrupting compounds from aquatic matrices: future perspectives on utilization of agri-waste based adsorbents. Sci Total Environ 826:154129. https://doi.org/10.1016/j.scitotenv.2022.154129
Swenberg JA, Kerns WD, Mitchell RI, Gralla EJ, Pavkov KL (1980) Induction of squamous cell carcinomas of the rat nasal cavity by inhalation exposure to formaldehyde vapor. Cancer Res 40(9):3398–3402
Til HP, Woutersen RA, Feron VJ, Hollanders VHM, Falke HE, Clary JJ (1989) Two-year drinking-water study of formaldehyde in rats. Food Chem Toxicol 27(2):77–87. https://doi.org/10.1016/0278-6915(89)90001-X
Trincado M, Grützmacher H, Prechtl MH (2018) CO2-based hydrogen storage–Hydrogen generation from formaldehyde/water. Phys Sci Rev 3(5):20170013. https://doi.org/10.1515/psr-2017-0013
Tsai CF, Shiau HW, Lee SC, Chou SS (2003) Determination of low-molecule-weight aldehydes in packed drinking water by high performance liquid chromatography. J Food Drug Anal 11(1):11. https://doi.org/10.38212/2224-6614.2734
US-EPA (1992) Method 554, Determination of carbonyl compounds in drinking water by dinitrophenylhydrazine dérivatisation and high-performance liquid chromatography, US Environmental Protection Agency. Revision 1.0, pp 1–24
US-EPA (1996) Method 8315A, Determination of carbonyl compounds by high performance liquid chromatography (HPLC). US Environmental Protection Agency Revision 1.0, pp 1–34
WHO (2017) Guidelines for drinking water quality: first addendum to the, 4th edn. World Health Organization, Geneva, p 631
Zhang ZF, Zhang X, Zhang XM, Liu LY, Li YF, Sun W (2020) Indoor occurrence and health risk of formaldehyde, toluene, xylene and total volatile organic compounds derived from an extensive monitoring campaign in Harbin, a megacity of China. Chemosphere 250:126324. https://doi.org/10.1016/j.chemosphere.2020.126324
Acknowledgements
The authors would like to thank the Physical Measurements Platform of the Chemistry Department of Faculty of Sciences, Mohammed First University Oujda (Morocco).
Funding
Funding information is not applicable/no funding was received.
Author information
Authors and Affiliations
Contributions
ElKhadir Gharibi ‘a’: conceptualization, validation, visualization, supervision, writing—review, funding acquisition, rewriting of the revised version, responses to reviewer comments. Mohammad Ghalit: experimental manipulation, conceptualization, investigation and formal analysis, writing—original draft, writing—review and editing. Mohamed Bouaissa: experimental manipulation, original draft, writing, investigation and formal analysis. Taupin J. Denis: validation, visualization, co-supervision, writing—review, rewriting of the revised version, responses to reviewer comments.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Responsible Editor: Mohamed Ksibi.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mohammad, G., Denis, T.J., Mohamed, B. et al. Formaldehyde contamination in PET-bottled mineral water: impact of CO2, temperature, and storage time with analytical method validation. Euro-Mediterr J Environ Integr 9, 1659–1671 (2024). https://doi.org/10.1007/s41207-024-00590-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41207-024-00590-4