Abstract
Introduction
Awake prone positioning has the potential to improve oxygenation and decrease respiratory rate, potentially reducing the need for intubation in patients with acute hypoxemic respiratory failure. We investigated awake prone positioning-induced changes in oxygenation and respiratory rate, and the prognostic capacity for intubation in patients with COVID-19 pneumonia.
Methods
International multicenter prospective observation study in critically ill adult patients with COVID-19 receiving supplemental oxygen. We collected data on oxygenation and respiratory rate at baseline, and at 1 h after being placed in prone positioning. The combined primary outcome was oxygenation and respiratory rate at 1 h. The secondary endpoint was treatment failure, defined as need for intubation within 24 h of start of awake prone positioning.
Results
Between March 27th and November 2020, 101 patients were enrolled of which 99 were fully analyzable. Awake prone positioning lasted mean of 3 [2–4] h. In 77 patients (77.7%), awake prone positioning improved oxygenation, and in 37 patients (54.4%) it decreased respiratory rate. Twenty-nine patients (29.3%) were intubated within 24 h. An increase in SpO2/FiO2 of < 10 (OR 5.1, 95% CI 1.4–18.5, P = 0.01), a failure to increase PaO2/FiO2 to > 116 mmHg (OR 3.6, 95% CI 1.2–10.8, P = 0.02), and a decrease in respiratory rate of < 2 breaths/min (OR 3.6, 95% CI 1.3–9.5, P = 0.01) were independent variables associated with need for intubation. The AUC–ROC curve for intubation using a multivariable model was 0.73 (95% CI 0.62–0.84).
Conclusions
Awake prone positioning improves oxygenation in the majority of patients, and decreases respiratory rate in more than half of patients with acute hypoxemic respiratory failure caused by COVID-19. One in three patients need intubation within 24 h. Awake prone position-induced changes in oxygenation and respiratory rate have prognostic capacity for intubation within 24 h.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Why carry out the study? |
Studies have not yet provided clinicians with tools to predict which patients with acute respiratory failure due to COVID-19 are most likely to improve with awake prone positioning. |
Awake prone positioning has the potential to avoid the need for intubation in patients with acute respiratory failure, therefore preventing its complications (e.g., reducing the risk of lung injury caused by invasive ventilation, associated infections and factors that may lead to post-intensive care unit syndrome). |
We hypothesized that awake prone positioning improves oxygenation and decreases the respiratory rate, and that changes in these parameters have associations with need for intubation. |
What was learned from this study? |
(1) Awake prone positioning improved oxygenation within 1 h in the majority of patients receiving noninvasive respiratory oxygen therapy; (2) Awake prone positioning decreased respiratory rate in more than half of patients; and (3) changes in these parameters have prognostic value for need for intubation within 24 h. |
Awake prone positioning improves physiologic measurements in patients with acute hypoxemic respiratory failure related to COVID-19. One-third of the patients needed intubation within 24 h. The effects of awake prone positioning had prognostic capacity for intubation within 24 h. |
Introduction
Patients with coronavirus disease 2019 (COVID-19) may develop severe and progressive acute hypoxemic respiratory failure, requiring intubation for invasive ventilation [1, 2]. Prone positioning has the potential to improve oxygenation, and also to improve outcome in these patients [3]. Awake prone positioning, i.e., prone positioning in nonintubated patients, may also improve oxygenation, and even reduce respiratory rate. Awake prone positioning therefore has the potential to reduce the need for intubation, preventing its complications [4], reducing the risk of lung injury caused by invasive ventilation [5], and associated infections [6].
In the prone position, the anterior chest wall expansion is restricted, resulting in a more homogeneous chest wall compliance. Gravitational forces on lung parenchyma allow greater recruitment of the posterior zones, enabling homogenization of transpulmonary pressure improving the ventilation to perfusion ratio [7]. This results in a more significant proportion of alveoli participating in gas exchange because the dorsal areas, which anatomically have an increased number of alveolar units, are no longer compressed by the weight of the abdominal cavity and the mediastinum. An equal distribution of stress forces onto the lungs by the diaphragm also occurs in prone position, which may help reduce lung injury in spontaneously breathing patient, possibly minimizing patient self-inflicted lung injury (P–SILI) [8, 9].
Recent studies of awake prone position in COVID-19 patients receiving non-invasive respiratory oxygenation therapies (NIROT), i.e., conventional oxygen therapy, high flow nasal oxygen (HFNO) noninvasive ventilation delivered either with continuous positive airway pressure (CPAP) or bilevel positive pressure ventilation (NIV), have shown encouraging results with respect to prevention of intubation [3, 10, 11]. Delayed intubation, however, can also worsen outcomes [12, 13]. It could be helpful to predict when awake prone positioning during NIROT cannot prevent intubation. The primary objective of this study was therefore to determine the effect of prone positioning on oxygenation and the respiratory rate in critically ill non-intubated COVID-19 patients receiving any type of NIROT. The secondary objective was to study the prognostic capacity of these changes for need for intubation. We hypothesized that awake prone positioning improves oxygenation and decreases the respiratory rate, and that changes in these parameters have associations with need for intubation.
Methods
Design
This is an investigator-initiated, observational, prospective, international, and multicenter study in six hospitals in Europe, one hospital in Brazil, and one hospital in the United States. Study sites were recruited through direct contact by the principal investigator. The institutional review boards of the participating centers approved the study protocol, and signed informed consent was obtained in every patient. Study coordinators monitored the study according to the International Conference on Harmonization Good Clinical Practice-guidelines. Integrity and timely completion of data collection was ensured by the principal investigator. A statistical analysis plan for the analysis was finalized before cleaning and closing of the study database.
The study was approved by all institutions and the names of all the ethics committees are provided. This study was performed in accordance with the Helsinki Declaration of 1964 and its later amendments.
Participants
Consecutive non-intubated patients were eligible for participation if (i.) aged ≥ 18 years, (ii.) admitted to the emergency department, ward, or ICU (i.e., intermediate and intensive care unit) of a participating hospital, and (iii.) having acute hypoxemic respiratory failure caused by COVID-19. Acute hypoxemic respiratory failure was defined as presence of any of the following criteria: (a) peripheral oxygen saturation (SpO2) < 92% with oxygen > 4 l/min; (b) indirect signs of increased work of breathing with use of accessory respiratory muscles, and a respiratory rate > 25/min; (c) PaO2/FiO2 ratio < 200 mmHg. Patients were excluded if there was a contraindication for prone positioning [14], if no signed informed consent was obtained, or in case of acute hypercapnic respiratory failure.
The decision to commence with invasive ventilation was to be based on clinical judgment rather than isolated gasometrical criteria. Herein, the following criteria had to be considered: any respiratory or cardiac arrest, or hemodynamic instability; any respiratory pauses; any altered level of consciousness including uncontrolled agitation not responding to medical treatment, or a worsening Glasgow Coma Scale score; evidence of exhaustion including subjective increase in use of accessory muscles, or a thoracoabdominal paradox; and development of inability to clear secretions from the airway in patients with abundant sputum production, or evidence of aspiration.
Collected Data
After enrollment, baseline data including demographic and anthropometric data, a baseline arterial blood gas measurement, vitals, and ventilation parameters including respiratory rate, and fractional concentration of oxygen in inspired air (FiO2) were collected.
In addition, we collected detailed information regarding oxygen support, including the type of oxygen interface, i.e., non-rebreather mask or Venturi mask or nasal prong (conventional oxygen therapy: COT), high-flow nasal oxygen (HFNO), continuous positive pressure airway (CPAP), and noninvasive bilevel ventilation. If a patient received noninvasive ventilation, we also collected the applied level of positive end-expiratory pressure (PEEP).
The percentage of the fractional concentration of inspired oxygen (FiO2) was calculated by a 4% increase in FiO2 for every liters/min use of oxygen from the oxygen device used in case of non-rebreather mask or nasal prong device. If a Venturi system was in used, the masks used had different color-changeable plastic pieces in which each color signifies the amount of oxygen delivery as a percentage. In the case of HFNO, a humidification chamber was set at 37 °C or 31 °C or 34 °C, according to patient’s comfort. The flow rate and FiO2 were independently titrated, and was based on SpO2 levels. HFNO, CPAP, and NIV were delivered through a high flow system or adapted to the ventilator. In the of CPAP or NIV, different interfaces were used (i.e., complete face mask, full face mask, helmet). All data were collected before start of prone positioning, and again after 1 h.
Intervention
Patients were managed in different hospitals settings: emergency room, hospital ward, ICU. The nurses helped the patient into a prone position, who was then encouraged to maintain in that position for as long as it was tolerable. For comfort, nurses offered pillows that could be placed around the chest, pelvis, and/or knees. Personal cell phone or tablets with internet connection were provided with the aim to further improve tolerance of the prone position. At the end of the first hour in a prone position, patients were free to resume the supine position or maintain the prone position at their discretion. Sessions of prone positioning were allowed thereafter according to clinicians’ indication and patients’ preference. In centers involved in this study, the caregivers assisted and stimulated patients to stay in prone position for as long as possible, with breaks if needed. At least two sessions lasting 30 min or longer had to be performed each day.
During these additional prone positioning sessions, we did not collect data. The decision to use each of the oxygenation devices was made according to the physician in charge. As well the decision to use analgosedation for poor tolerance to prone position was made according to the physician in charge.
Endpoints
The primary endpoint was the effects of awake prone positioning on oxygenation, and on the respiratory rate 1 h after start of awake prone positioning. The secondary endpoint was early intubation defined as the need for invasive ventilation within the first 24 h of start of awake prone positioning.
Power Calculation
We performed no a priori sample size calculation; the number of patients admitted in the participating centers served as the sample size.
Statistical Analysis
We report numbers and percentages for categorical variables, and the medians with interquartile ranges for continuous variables. The assumption of normality was checked by means of the Kolmogorov–Smirnov test. Categorical variables were compared using the chi-square test or the Fisher's exact test, whereas continuous variables were compared using the nonparametric Mann–Whitney test. The Wilcoxon signed-rank test was used to compare a continuous variable between paired groups (pre-post-analysis).
Univariate and multivariate logistic regression analyses [15] were performed to identify variables associated with intubation. The following variables were first analyzed univariately based on previous findings [3, 16] and clinical characteristics: sex, age, BMI, respiratory rate, PaO2/FiO2, SpO2/FiO2, PaO2, and ROX Index. Predictors for treatment failure were predefined, and included respiratory rate, oxygenation defined as the SpO2/FiO2 ratio and PaO2. Final variable selection was performed using the backward stepwise selection method. Odds ratios (ORs) and their 95% confidence intervals (CI) were calculated. Single collinearity was evaluated using the Pearson’s coefficient correlation (r). PaO2/FiO2 and was excluded because of collinearity with SpO2/FiO2 and ROX Index, while ROX Index was omitted because of collinearity with respiratory rate, PaO2/FiO2 and SpO2/FiO2. Multicollinearity was examined by means of the variance inflation factor (VIF). The model’s performance was assessed by studying calibration and discrimination. We evaluated calibration with the Hosmer–Lemeshow goodness-of-fit test, comparing observed and predicted risks across subgroups of the population by using the model. If the P value is large (> 0.05), then the model is well calibrated and fits the data well. The discriminative ability refers to the model's ability to distinguish patients intubated from those not intubated and is expressed as the area under the receiver operating characteristic (ROC) curve (AUC) ranging from 0.5 (no discriminative ability) to 1.0 (perfect discriminative ability).
The following variables were second analyzed univariately based on previous findings and clinical characteristics: sex, age, BMI, change from supine to prone position after 1 h in respiratory rate, change from supine to prone position after 1 h in PaO2/FiO2, change from supine to prone position after 1 h in SpO2/FiO2, change from supine to prone position after 1 h in PaO2, and change from supine to prone position after 1 h in ROX Index, and similar criteria were applied for multivariable analysis (P < 0.25).
ROC curves were constructed to determine the best cutoff for PaO2, PaO2/FiO2, SpO2/FiO2, respiratory rate, and ROX index to predict intubation or mortality. Youden’s index [17] was defined for all points along the ROC curve, and the maximum value of the index was used as a criterion for selecting the optimum cutoff.
We investigated missing data patterns for covariates and assumed missing at random (MAR) conditioned in the covariates [18]. We used multiple imputation to generate five datasets to evaluate the prediction performance for the intubation. The model for multiple imputation included all covariates of the risk models. For simplicity, for the performance evaluation we filled in missing values with the first set of imputed values from the multiple imputation.
The level of significance was set at 0.05 (two-tailed). All analyses were performed using IBM SPSS version 26.0 (IBM Corp., Armonk, NY, USA).
Results
Patients
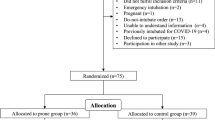
Between March 27 and November 2020, 101 patients fulfilled the inclusion criteria. In two patients, data collection was incomplete, leaving us with 99 fully analyzable patients. Patient characteristics are presented in Table 1. Most patients were admitted to an ICU at the moment of inclusion, and HFNO and CPAP were the most frequently used oxygen NIROT devices at start of awake prone positioning.
Effects of Prone Positioning
Awake prone positioning lasted mean of 3 h [2,3,4] . In 77 patients (77.7%), awake prone positioning resulted in an improved oxygenation, and in 37 patients (54.4%) it resulted in a lower respiratory rate at 1 h after start of the intervention (Fig. 1).
Cumulative frequency distribution of the study variables PaO2, PaO2/FiO2, SpO2/FiO2, and respiratory rate. The plots show the study parameters at baseline and 1 h after awake prone position of intubated and nonintubated patients. The lines in red are intubated patients at 1 h of awake proning; the dotted lines in red are intubated patients before awake proning; the lines in green are nonintubated patients at 1 h of awake proning; the dotted lines in green are nonintubated patients before awake proning
Need for Intubation
Twenty-nine patients (29.3%) were intubated for invasive ventilation within 24 h after start of awake prone positioning. Patients that were intubated received oxygen via HFNO more often and had worse oxygenation problems than patients that were not intubated (Table 1). Patients that were intubated were also more tachypneic (Figs. 1 and 2). Otherwise, there were no differences between the two groups, neither with respect to patient characteristics nor to other ventilatory parameters and hemodynamic parameters.
Cumulative frequency distribution of the study variables SpO2, FiO2, ROX index, and PaCO2. The plots show the study parameters at baseline and 1 h after awake prone position of intubated and nonintubated patients. The lines in red are intubated patients at 1 h of awake proning; the dotted lines in red are intubated patients before awake proning; lines in green are nonintubated patients at 1 h of awake proning; dotted lined in green are nonintubated patients before awake proning
Predictors of Intubation
Baseline characteristics that had an independent association with intubation within 24 h were a baseline respiratory rate > 26 breaths/min, SpO2/FiO2 < 147 and PaO2 < 100 mmHg (Table 1 online supplementary material). The AUC–ROC for the multivariable model using these baseline characteristics was 0.89 (95% CI 0.81–0.97) (Fig. 3A). The probability of intubation for patients without any of these characteristics was 1%, and 90% for patients having all three characteristics (Table 2).
A ROC curve for the multivariable model, based on respiratory rate, SpO2/FiO2 and PaO2 at baseline. Sensitivity 76%, specificity 87%, positive predictive value 71%, negative predictive value 89%; B ROC curve for the multivariable model, based on changes in respiratory rate, PaO2/FiO2, and PaO2 from supine to 1 h after prone position. Sensitivity 65%, specificity 61%, positive predictive value 41%, negative predictive value 81%
Responses to awake prone positioning that had an independent association with intubation were failure to lower the respiratory rate by at least two breaths/min, failure to increase PaO2/FiO2 > 116 mmHg, and failure to increase SpO2/FiO2 ≥ 10 mmHg (Table 1 online supplementary material). The AUC–ROC curve for the multivariable model using the physiological responses to prone positioning was 0.73 (Fig. 3B). The probability of intubation for patients without any of these responses was 2%, and 62% for patients having all three responses (Table 2).
Discussion
The findings of this study in critically ill patients with acute hypoxemic respiratory failure due to COVID-19 can be summarized as follows: (1) awake prone positioning improved oxygenation within one hour in the majority of patients receiving NIROT; (2) awake prone positioning decreased respiratory rate in more than half of patients; and (3) changes in these parameters have prognostic value for need for intubation within 24 h.
This study has several strengths. To our best knowledge, our study is the first to assess the predictive value of physiologic responses to awake prone positioning in patients with COVID-19 pneumonia. We included consecutive patients that were admitted in normal wards or ICUs in various types of hospitals in three countries, all contributing to the generalizability of the findings. Loss to follow-up was minimal, despite the restrictions caused by the pandemic. We used predefined and objectives endpoints, and followed the predetermined analysis plan that was in line with the latest recommendations for predictive research [19, 20]. Although physiological changes in response to APP have been described in previous studies, we decided to search for prognostic parameters for early intubation for comparison with a previously published study [16].
Benefit with regard to oxygenation of awake prone positioning in nonintubated ICU patients was initially suggested in patients with acute hypoxemic respiratory failure that was not related to COVID-19 [21], and in patients with severe hypoxemia after lung transplantation [22, 23]. Since the onset of the COVID-19 pandemic, a rapidly growing number of mainly small observational studies suggest that awake prone positioning can improve oxygenation, improvements in oxygenation usually happens within minutes [24,25,26,27,28] and can lasts for up to 1 h after turning back to a supine position [29]. Our findings confirm these findings, by showing that awake prone positioning rapidly improved oxygenation in roughly four out of five COVID-19 patients.
Individual studies [28, 30,31,32] and one meta-analysis [29] show that awake prone positioning can decrease the respiratory rate, which may decrease the risk of patient self-inflicted lung injury [9, 30]. We could confirm this decrease in respiratory rate, albeit that it was only found in half of the patients.
A recently published meta-trial of six randomized clinical studies in critically ill COVID-19 patients that needed supplementary oxygen via HFNO showed awake prone positioning to reduce the need for intubation within 28 days [3]. Mortality at day 28 was not affected by the intervention. However, a nonrandomized clinical trial in patients with acute hypoxic respiratory failure due to COVID-19 related, suggested awake prone positioning to have no benefit, as its use was neither associated with reduced progression to invasive ventilation, nor with an increase in the number of ventilator-free days [33]. None of these studies reported on early intubation, i.e., need for intubation within one day, and there was also no analysis that determined whether changes induced by prone positioning could predict the need for intubation.
Studies have not yet provided clinicians with tools to predict which patients with COVID-19 are most likely to improve with awake prone positioning, nor have they proven whether awake proning can delay or avoid the need for invasive ventilation. In our study, the variables under investigation have some prognostic capacity that may help clinicians identify patients that may not respond to awake proning, preventing intubation delay and early transfer of the patient to the ICU for close monitoring. Besides, in our study the baseline respiratory parameters also had capacity, which in some way could reflect a more protracted course of the disease that may lead to a greater probability of intubation.
Our study has limitations. First, our study had a relatively small sample size. Second, we could not perform arterial blood gas analyses in nearly half of the patients. Third, we did not collect data on subsequent cycles of awake prone positioning that patients might have undergone. Fourth, the median time from hospital admission to the first session of awake proning was nearly 3 days, suggesting a relative late start of the intervention in our cohort. It has been suggested that earlier use of awake proning could have better clinical outcomes [11]. Fifth, the sample size of our study is too small to come to subgroups of patients with different types of respiratory support. We acknowledge additional limitations in our study. We acknowledge that seeking for prognostic parameters for early intubation could be a surrogate outcome. During the initial wave of the pandemic, it was challenging to collect data on comorbidities, disease severity scores, and laboratory results, which are all potential confounding factors. Therefore, it is important to interpret our results with caution. These relevant drawbacks might attenuate the strength of the findings of this study; however, we should consider the overwhelming workload for clinicians at the beginning of SARSCoV2 spreading and the lack of chances of increasing nowadays the number of enrolled patients provided with a full set of physiological data due to the changed epidemiologic, clinical, and biological features of COVID-19 infection.
Conclusions
Awake prone positioning improves oxygenation in most and decreases respiratory rate in more than half of the patients with acute hypoxemic respiratory failure related to COVID-19 receiving NIROT. In this cohort, one out of three patients needed intubation for invasive ventilation within 24 h. The effects of awake prone positioning on oxygenation and respiratory rate had prognostic capacity for intubation within 24 h.
Data Availability
Availability of data and supporting materials section: All data generated or analyzed during this study are included in this published article and its supplementary material information files.
References
Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York city area. JAMA. 2020;323:2052–9.
CNARC. ICNARC report on COVID-19 in critical care, 10 April 2020. https://www.icnarc.org/. [Accessed 2020 Oct 14]
Ehrmann S, Li J, Ibarra-Estrada M, Perez Y, et al. Awake prone positioning for COVID-19 acute hypoxaemic respiratory failure: a randomised, controlled, multinational, open-label meta-trial. Lancet Respir Med. 2021;9:1387–95.
Russotto V, Myatra SN, Laffey JG, et al. Intubation practices and adverse peri-intubation events in critically Ill patients from 29 countries. JAMA. 2021;325:11164–72.
Slutsky AS, Ranieri VM. Ventilator-induced lung injury. N Engl J Med. 2013;369:2126–36.
Luyt CE, Bouadma L, Morris AC, et al. Pulmonary infections complicating ARDS. Intensive Care Med. 2020;46:2168–83.
Wang YX, Zhong M, Dong MH, et al. Prone positioning improves ventilation–perfusion matching assessed by electrical impedance tomography in patients with ARDS: a prospective physiological study. Crit Care. 2022;26:154.
Scholten EL, Beitler JR, Prisk GK, et al. Treatment of ARDS with prone positioning. Chest. 2017;151:215–24.
Telias I, Katira BH, Brochard L. Is the prone position helpful during spontaneous breathing in patients with COVID-19? JAMA. 2020;323:2265–7.
Li J, Luo J, Pavlov I, Perez Y, et al. Awake prone positioning for non-intubated patients with COVID-19-related acute hypoxaemic respiratory failure: a systematic review and meta-analysis. Lancet Respir Med. 2022;10(6): e53.
Musso G, Taliano C, Molinaro F, et al. Early prolonged prone position in noninvasively ventilated patients with SARS-CoV-2-related moderate-to-severe hypoxemic respiratory failure: clinical outcomes and mechanisms for treatment response in the PRO-NIV study. Crit Care. 2022;26:118.
Bellani G, Laffey JG, Pham T, et al. Noninvasive ventilation of patients with acute respiratory distress syndrome. Insights from the LUNG SAFE study. Am J Respir Crit Care Med. 2017;195:67–77.
Kang BJ, Koh Y, Lim CM, et al. Failure of high-flow nasal cannula therapy may delay intubation and increase mortality. Intensive Care Med. 2015;41:623–32.
Guérin C, Reignier J, Richard J-C, et al. Prone positioning in severe acute respiratory distress syndrome. N Engl J Med. 2013;368(23):2159–68.
Hosmer DW, Lemeshow S. Applied logistic regression. New York: NY John Wiley Sons; 1989.
Ibarra-Estrada M, Li J, Pavlov I, et al. Factors for success of awake prone positioning in patients with COVID-19-induced acute hypoxemic respiratory failure: analysis of a randomized controlled trial. Crit Care. 2022;26:84.
Jouden WJ. Index for rating diagnostic tests. Cancer. 1950;3:32–5.
Sterne JA, White IR, Carlin JB, et al. Multiple imputation for missing data in epidemiological and clinical research: potential and pitfalls. BMJ. 2009;338: b2393.
Moons KG, Kengne AP, Woodward M, et al. Risk prediction models: I. Development, internal validation, and assessing the incremental value of a new (bio)marker. Heart. 2012;98:683–90.
Moons KG, Kengne AP, Grobbee DE, et al. Risk prediction models: II. External validation, model updating, and impact assessment. Heart. 2012;98:691–8.
Valter C, Christensen AM, Tollund C, et al. Response to the prone position in spontaneously breathing patients with hypoxemic respiratory failure. Acta Anaesthesiol Scand. 2003;47:416–8.
Feltracco P, Serra E, Barbieri S, et al. Non-invasive ventilation in prone position for refractory hypoxemia after bilateral lung transplantation. Clin Transpl. 2009;23:748–50.
Feltracco P, Serra E, Barbieri S, et al. Noninvasive high-frequency percussive ventilation in the prone position after lung transplantation. Transpl Proc. 2012;44:2016–21.
Elharrar X, Trigui Y, Dols AM, et al. Use of prone positioning in non-intubated patients with COVID-19 and hypoxemic acute respiratory failure. JAMA. 2020;323:2336–8.
Despres C, Brunin Y, Berthier F, et al. Prone positioning combined with high-flow nasal or conventional oxygen therapy in severe COVID-19 patients. Crit Care. 2020;24:256.
Ng Z, Tay WC, Ho CHB. Awake prone positioning for non-intubated oxygen dependent COVID-19 pneumonia patients. Eur Respir J. 2020;56:2001198.
Caputo ND, Strayer RJ, Levitan R. Early self-proning in awake, non-intubated patients in the emergency department: a single ED’s experience during the COVID-19 pandemic. Acad Emerg Med. 2020;27:375–8.
Damarla M, Zaeh S, Niedermeyer S, et al. Prone positioning of nonintubated patients with COVID-19. Am J Respir Crit Care Med. 2020;202:604–6.
Ponnapa Reddy M, Subramaniam A, Afroz A, et al. Prone positioning of non–intubated patients with COVID-19—a systematic review and meta-analysis. Crit Care Med. 2021;49:e1001–14.
Sartini C, Tresoldi M, Scarpellini P, et al. Respiratory parameters in patients with COVID-19 after using noninvasive ventilation in the prone position outside the intensive care unit. JAMA. 2020;323:2338–40.
Zang X, Wang Q, Zhou H, et al. Efficacy of early prone position for COVID-19 patients with severe hypoxia: a single–center prospective cohort study. Intensive Care Med. 2020;46:1927–9.
Solverson K, Weatherald J, Parhar KKS. Tolerability and safety of awake prone positioning COVID-19 patients with severe hypoxemic respiratory failure. Can J Anaesth Anaesth. 2020;68(1):64–70.
Qian ET, Gatto CL, Amusina O, et al. Assessment of awake prone positioning in hospitalized adults with COVID-19: a nonrandomized controlled trial. JAMA Intern Med. 2022; (Published)
Yoshida T, Uchiyama A, Matsuura N, et al. Spontaneous breathing during lung–protective ventilation in an experimental acute lung injury model: high transpulmonary pressure associated with strong spontaneous breathing effort may worsen lung injury. Crit Care Med. 2012;40:1578–85.
Brochard L, Slutsky A, Pesenti A, et al. Mechanical ventilation to minimize progression of lung injury in acute respiratory failure. Am J Respir Crit Care Med. 2017;195:438–42.
Gattinoni L, Coppola S, Cressoni M, et al. COVID-19 does not lead to a “typical” acute respiratory distress syndrome. Am J Respir Crit Care Med. 2020;201:1299–300.
Funding
This article received partial economic support from the European Society of Intensive Care Medicine (ESICM).
Author information
Authors and Affiliations
Contributions
Luis Morales-Quinteros, Antonio Artigas, and Marcus J. Schultz carried out the concept and design. Luis Morales-Quinteros, carried out the statistical analysis. Antonio Artigas and Marcus J. Schultz carried out the supervision of the manuscript. Luis Morales-Quinteros and Antonio Artigas participated in the coordination of the study. All named authors contributed in the acquisition, analysis, interpretation of data and drafting of the manuscript. All authors contributed on the critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
Luis Morales-Quinteros, Raffaele Scala, Joao Manoel Silva, Antonio Leidi, Alexandre Leszek, Rodrigo Vazquez Guillamet, Sergi Pascual, Ary Serpa Neto, Antonio Artigas, and Marcus J. Schultz have nothing to declare.
Ethical Approval
The study was approved by all institutions and the names of all the ethics committees are provided. This study was performed in accordance with the Helsinki Declaration of 1964 and its later amendments.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Morales-Quinteros, L., Scala, R., Silva, J.M. et al. Associations of Awake Prone Positioning-Induced Changes in Physiology with Intubation: An International Prospective Observational Study in Patients with Acute Hypoxemic Respiratory Failure Related to COVID-19. Pulm Ther 9, 499–510 (2023). https://doi.org/10.1007/s41030-023-00242-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41030-023-00242-y