Abstract
Introduction
Correct use of inhalation devices is critical for optimal drug delivery to the lower lung. This Delphi process was conducted to compile lists of potential handling errors when using a Spiromax® or Turbuhaler® inhalation device, as determined by an independent panel.
Methods
A Delphi process was used to obtain consensus on potential handling errors for each device from ten independent respiratory device experts. In Round 1, advisors listed potential errors with the devices. In Round 2, a severity rating was assigned to each error based on erroneous device handling negatively affecting functionality and treatment effectiveness (error [score 0–3]; potentially critical [4–7]; critical [8–10]). In Round 3, advisors revised their ratings based on the group scores and voted on whether to accept the median severity score as the consensus in Round 4.
Results
A total of 29 potential errors for Spiromax and 31 for Turbuhaler were identified in Round 1. After Round 4, consensus was reached for 69% of the Spiromax errors and 94% of the Turbuhaler errors. After completion of the Delphi process, some anomalies were identified in the list of handling errors, which were then investigated with the panel via teleconferences. After teleconferences to discuss discrepancies in the results, there were 22 errors for Spiromax (four critical, 12 potentially critical, and six errors) and 27 for Turbuhaler (nine critical, 14 potentially critical, and four errors). Not inhaling through the mouthpiece, exhaling into the device, and incorrect mouth positioning were identified as critical errors for both devices.
Conclusion
Through the Delphi process, advisors independently identified and reached consensus on handling errors for Spiromax and Turbuhaler. Fewer Spiromax errors were classified as critical or potentially critical than with Turbuhaler, indicating that there may be less potential for handling errors with Spiromax.
Funding
Teva Branded Pharmaceutical Products R & D, Inc.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Respiratory diseases such as asthma and chronic obstructive pulmonary disease (COPD) are routinely managed using inhaled bronchodilators and corticosteroids [1, 2]. However, the medication must reach the receptors in the lower airways to exert its effect. To ensure this happens, correct use of the inhaler device is critical for optimal drug delivery [3].
Patient-related factors such as poor adherence and inhaler handling errors have a negative impact on asthma control [4]. Studies have shown that poor inhaler technique is common and inhaler use is often highly suboptimal [5]. Estimates of improper inhaler use range from 20% to 82% of patients [4, 6–9]. There is increasing evidence in support of the claim that correct inhaler technique is fundamental for effective therapy and asthma management [5, 10, 11]. Errors in the use of inhalers have been shown to have serious effects in terms of the management of the disease [6, 8, 9]. Poor inhaler technique in patients with asthma is associated with increased hospitalization, more emergency department visits, increased use of oral corticosteroids and antimicrobials, and poorer asthma control [6, 8, 9]. Some errors are device-independent and include not breathing out before actuation of the device [12].
Although correct inhaler technique involves some common steps for all devices, the optimal inhalation pattern differs between devices [3, 13]. Inhalers that are perceived as difficult to use are usually associated with poor inhalation technique and low treatment adherence, leading to worsening in asthma control [14]. As such, the technical characteristics of an inhaler device can determine how well it is handled, and consequently, how often it is correctly used. An inhaler that is easy to use, intuitive, and preferred by the patient could ultimately improve adherence [14–16].
In patients with inadequately controlled asthma, the combination therapy involving an inhaled corticosteroid (ICS) and a long-acting β2 agonist (LABA) is a recommended therapy [17]. The fixed-dose combination of the ICS/LABA, budesonide/formoterol (BF), has proven to be an effective therapy by improving pulmonary function and asthma control [1, 2] in patients with asthma and COPD. DuoResp® (BF) Spiromax® (Teva Pharmaceuticals) is a BF dry powder inhaler designed to provide dose equivalence with enhanced user-friendliness compared to Symbicort® (BF) Turbuhaler® (AstraZeneca) [18]. Both inhalers are approved in Europe for the treatment of patients with asthma and COPD (for whom an ICS/LABA combination is indicated).
To determine which of these two inhalers patients might find easiest to use correctly, it is necessary to establish objective criteria by which these inhaler devices are compared with regards to their ease-of-use and potential handling errors. In this study, practicing clinicians with a specialty in device handling in asthma or COPD were asked to identify potential handling errors for the Spiromax and the Turbuhaler device. Subsequently, a Delphi process was used to reach consensus on the errors most likely to be critical for effective drug delivery. A Delphi process was chosen because it is an established tool for reaching a consensus on a complex problem in a highly objective way [19–21]. In the Delphi process, a peer group of experts work together anonymously to reach consensus on a complex problem as objectively as possible.
The aims of this Delphi process study were to independently establish what is considered an error (for optimal drug delivery) when handling empty versions of DuoResp Spiromax or Symbicort Turbuhaler dry powder inhalers and to assign a level of severity to each of these errors.
Methods
Study Procedures
This study was conducted by InterPhase Consult, an independent strategic consulting firm, on behalf of Teva Pharmaceuticals Inc. InterPhase Consult ensured that the methodology and conduct of the study were free from bias and Teva Pharmaceuticals had no influence on the choice of advisors, design, and administration of the Round 1–4 questionnaires, or the data analysis.
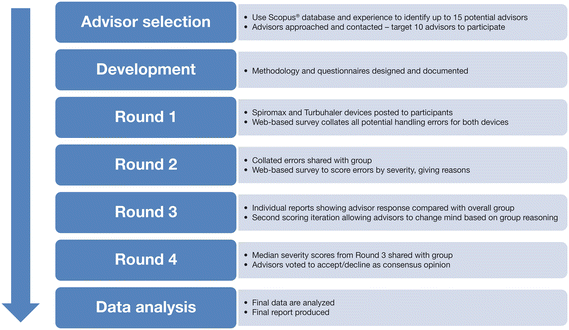
This Delphi process was initiated with selection of the advisors and development of the questionnaires, followed by four rounds of the Delphi process and then data analysis (Fig. 1). Per Office for Human Research Protections (OHRP) approved assurance created under 45 CFR 46.103, the protocol was exempt from informed written consent because (1) the research presented no more than minimal risk of harm to subjects, (2) the exemption did not adversely affect the rights and welfare of the subjects, (3) the research could not practicably be carried out without the exemption and (4) whenever appropriate, the subjects were provided with additional pertinent information after participation. (http://www.hhs.gov/ohrp/humansubjects/guidance/45cfr46.html).
Advisor Selection
Fifteen European advisors were identified. Advisors were invited to participate until 10 had accepted, per the recommendations of Delbecq et al. (1975) for panel selection when performing a Delphi methodology study [22]. Please refer to the Appendix in the supplementary material for additional details on the advisors selection.
Delphi Process (Rounds 1–4)
Using the Delphi process, advisors were asked via questionnaires to agree on a list of potential inhaler usage errors and then rate them according to the perceived impact each error would have on effective drug delivery. Advisors remained anonymous to each other until the teleconferences. Questionnaires were developed before the study by InterPhase analysts, but the wording could be adapted during the course of the study, if necessary, to ensure instructions to the advisors were clear and the Delphi process was able to achieve consensus. Advisors responded to the questionnaires online using the website SurveyMonkey® (Palo Alto, CA, USA). The Delphi process aimed to achieve consensus (see Data Analysis below) on ≥90% of all errors.
Round 1
Participating advisors were sent empty Spiromax and Turbuhaler devices along with the respective patient instruction leaflets. Once they were familiar with the devices and their instructions, the advisors were asked to list all of the potential handling errors they could think of for each device. Handling errors were defined as any way in which the inhaler could be used incorrectly during the inhalation process. The advisors were randomly divided into two groups; one group reviewed the Spiromax device first and the other reviewed the Turbuhaler device first. The resultant lists from the 10 advisors were consolidated by InterPhase Consult into one list of errors for each device.
Round 2
Advisors received the consolidated list of errors (ordered randomly) and were asked to score each error with respect to its severity using an integer scale of 0–10, (0, least critical; 10, most critical). The severity levels were then separated into three categories: ‘error', ‘potentially critical', and ‘critical' as shown in Fig. 2. Based on the advisors’ expert opinion with regards to the impact on treatment outcomes and/or inhaler device functionality, ‘error' indicated an error that would have minor or no impact, ‘potentially critical' indicated an error would have a mild-to-moderately negative impact, and ‘critical' indicated an error would have a negative impact. An error could be removed from the list if seven or more advisors provided approval. Advisors were also asked to give their reasoning behind the score they assigned for each error.
Round 3
Advisors were given an individualized report showing their responses relative to the consolidated group severity scores and were given the opportunity to re-evaluate their scores and revise them if they wished. If a consensus was not achieved for ≥90% of errors for each device, a fourth round was to be conducted.
Round 4
Advisors were provided with a list of all errors grouped within their corresponding categories as determined by the median score from Round 3. They were then asked to agree or disagree with the error category for each error based on the median error severity ranking from Round 3. The outcome from Round 3 was accepted as consensus group opinion if ≥90% of the advisors agreed in Round 4.
Post-hoc Study Amendment
In a post hoc study amendment, advisors were asked to participate in a series of teleconferences to discuss and refine the output from the Delphi process. A summary of the consensus from the calls was subsequently approved by the participating advisors.
Data Analysis
Scores obtained for each error in Rounds 2 and 3 were analyzed using descriptive statistics and median scores were calculated using Microsoft® Excel 2010® software (Microsoft, Redmond, USA). Errors were ranked according to median scores within each of the three severity categories. Interquartile ranges (IQRs) were calculated to give a measure of dispersion and, therefore, consensus. Consensus for each error was deemed to have been reached when the IQR was ≤2, or if ≥80% of advisors put the error into the same severity category.
Results
Conduct of the Study
Ten advisors participated in this study (Table S1). Full consensus was not reached for ≥90% of the errors after Round 3; a fourth round was conducted. Some changes were made to the wording and design of the questionnaire following Round 1 to emphasize the intended meaning and definition of a handling error.
Identification of Errors Using the Delphi Process (Rounds 1–4)
Round 1
A total of 29 potential errors were identified for Spiromax in Round 1 and 31 were identified for Turbuhaler (data on file, Teva Pharmaceuticals and InterPhase Consult).
Rounds 2 and 3
None of the errors was deemed to be invalid by seven or more advisors in Round 2 and all were retained.
Median severity scores were assigned to each of the errors and did not change significantly between Rounds 2 and 3 (Table 1). A critical severity score was given on fewer occasions for errors in using Spiromax compared with Turbuhaler.
The errors considered by the advisors to be most critical (median severity score of ≥9) when using Turbuhaler were ‘not inhaling through the mouthpiece (e.g., nose)’, ‘difficulty/failure to remove the cap prior to usage’, ‘forgetting to load the device prior to each dosage’, and ‘exhaling into the device’. The error ‘forgetting to load the device prior to each dosage’ was assigned to the critical error category by all ten advisors; nine advisors put ‘exhaling into the device’, ‘not inhaling through the mouthpiece (e.g., nose)’, ‘red grip is only rotated fully in one direction prior to using the device’, and ‘holding the mouthpiece instead of the barrel when twisting the red grip’ in the critical error category.
For Spiromax, the errors considered to be most critical were ‘not inhaling through the mouthpiece (e.g., nose)’ and ‘failure to read the dose indicator correctly, potentially leading to using an empty device’. None of the errors were classified as being critical by all 10 advisors; eight advisors classed ‘not inhaling through the mouthpiece (e.g., nose)’ as a critical error.
After Round 3, consensus was reached for 38% of the errors for Spiromax and 68% of the errors for Turbuhaler.
Round 4
In Round 4, the median severity scores from Round 3 were accepted as the consensus for an additional nine errors for Spiromax and eight errors for Turbuhaler, resulting in 69% consensus for Spiromax and 94% for Turbuhaler.
Post-hoc Review and Analysis
A post-hoc review (by InterPhase Consult and Teva Pharmaceuticals) of the errors identified for Spiromax and Turbuhaler found some anomalies, such as: handling errors that had been scored differently for the two devices, but which may reasonably have been expected to be scored consistently; handling errors that had been scored consistently, but which may reasonably have been expected to have been scored differently; and handling errors that may be invalid based on the definition of a handling error. Following this review, all advisors were invited to attend a teleconference to discuss the results. Six advisors agreed to participate and two teleconferences were held to discuss the output of the Delphi process and the anomalies, and to give the advisors an opportunity to revise their scores. After the teleconferences, a summary of the consensus from the calls was sent to the participating advisors for their approval.
A total of 16 errors were identified as being anomalous and were discussed. After the calls, four of these retained their original classification and 12 were reclassified. For Spiromax, six errors were removed, as they were not thought to be valid handling errors for the device, and two potentially critical errors were reclassified as non-critical errors (not holding the breath for long enough following inhalation; device not held upright during loading/inhalation). For Turbuhaler, five errors were removed, as they were not thought to be valid handling errors for the device, and two errors that had originally only been identified for Spiromax were added (not holding the breath for long enough following inhalation; unable to/forgetting to breathe out fully prior to inhalation).
Final Outcome
Following the teleconferences, the number of potential handling errors was finalized as 22 for Spiromax and 27 for Turbuhaler (Table 1; Fig. 3). Based on the group median scores from Round 3 and the subsequent error categorization during the teleconferences, the 22 potential handling errors for Spiromax comprised four critical errors, 12 potentially critical errors, and six non-critical errors (Table 1; Fig. 3). In comparison, the 27 potential handling errors for Turbuhaler comprised nine critical errors, 14 potentially critical errors, and four non-critical errors (Table 1; Fig. 3).
Discussion
Despite the availability of a wide variety of medications, a significant portion of patients with asthma and COPD remain uncontrolled [14–16]. The correct use of an inhaler device is crucial for optimal drug delivery. As such, the inhaler device itself plays an important and active role in asthma management. It is therefore important to have devices which are intuitive to use and to train patients.
Using the Delphi process, advisors participating in this study independently identified potential handling errors for Spiromax and Turbuhaler and ranked them according to their severity. Spiromax was associated with fewer errors overall (22 vs. 27) and fewer errors that were classified as critical (four vs. nine) or potentially critical (12 vs. 14) than Turbuhaler. This suggests that there may be less potential for critical handling errors when using the Spiromax device compared with Turbuhaler.
Findings from this study are in agreement with outcomes reported in two independent device mastery studies among healthy Finnish volunteers and healthcare practitioners (HCPs) in Australia. In healthy adult Finnish volunteers [23] and Australian HCPs [24], Spiromax was associated with higher levels of device mastery and fewer errors by intuitive use/no instructions and after reading the patient information leaflet compared with Turbuhaler.
Training patients in inhaler use is a core component of good clinical practice [25]. Consequently, providing HCPs with an inhaler that is less prone to device handling errors and easy to use and teach patients could potentially reduce the number of device handling errors and ultimately improve asthma control. Results from this study highlight specific errors that could potentially be committed using Spiromax or Turbuhaler. Two errors for Turbuhaler received the maximum median severity score of 10; these were ‘not inhaling through the mouthpiece (e.g., nose)' and ‘difficulty/failure to remove the cap prior to usage'. A number of errors regarding priming of the device were classified as critical for Turbuhaler (which were not identified for Spiromax). These included, ‘forgetting to load the device prior to each dosage', ‘failing to fully rotate the red grip', ‘failure to read the dose indicator correctly, potentially leading to using an empty device', and ‘red grip is only rotated fully in one direction prior to using the device'. The highest median severity score for a Spiromax error was 9.5 for ‘not inhaling through the mouthpiece (e.g., nose)'. Thus, HCPs should focus on these potential errors when training patients to use Spiromax or Turbuhaler.
Although training in inhaler technique is important and assessing technique is recommended at every visit [25], in everyday life this might not prove practical or possible. Additionally, training alone might not be sufficient to ensure proper technique mastery. Some patients fail to achieve correct inhaler technique despite repetitive training [3]. The patient’s opinion on a particular device, psychosocial factors such as preference and willingness to use a particular device, and the device choice are also important aspects to consider [26]. The ability to use the inhaler correctly can influence adherence [3]. An intuitive device that is easy to use and preferred by the patient might also improve asthma control by minimizing the risk of handling error committed by the patient and improving adherence. However, it is important to note that a device that is shown to potentially be associated with fewer errors in an experimental setting does not necessarily translate to less potential errors and better inhalation technique in real life. Also, a device with less potential errors is not automatically the preferred device by the patient. Real-life scenarios can be different and several patient-related factors such as comorbidities, dexterity, familiarity with inhaler devices, and other lifestyle factors may play a role [8, 27]. Matching the correct patient with the correct inhaler is now thought to be essential when prescribing an inhaler that a patient can and will use correctly at every use [28, 29].
The Delphi process methodology was deemed most suitable for this study because it is an established tool for reaching consensus on a complex problem as objectively as possible. It has been proven to be effective for reaching a consensus when factual data do not exist or cannot easily be obtained, or when the problem is so subjective that an individual may give an opinion that conforms to those of other participants rather than maintaining their own critical opinion [19, 30]. Among the strengths of this study are the key characteristics of the Delphi process: the involvement of an expert panel, the anonymity of the advisors and their responses to other participants, the use of a series of questionnaires administered in rounds, the summarization of information from each round via a moderator and sharing of anonymized group opinion, the progressive convergence of opinions with administration of each questionnaire, and quantification of the extent of agreement using simple statistics.
This study has a number of limitations, one of which was the requirement for the teleconferences after Round 4 of the Delphi process to finalize the lists of potential handling errors. It was felt necessary to conduct the teleconferences for a number of reasons: there was < 90% consensus for the errors for Spiromax; some of the listed errors were not thought to be valid handling errors while others were thought to unfairly disadvantage one device over another; and some errors were listed for one device that should be applicable to both. Examples of invalid handling errors that were removed include: ‘washing the device using liquids/getting the device wet', ‘chewing the mouthpiece of the device', ‘information leaflet inaccurate/difficult to understand', and ‘attempting to fit a spacer device'. The need for the teleconferences may be interpreted as interference in the Delphi process; however, neither device was unfairly prejudiced by the teleconferences—both devices had some errors removed by the advisors. The aim of the teleconferences was to ensure the final error lists were accurate and representative of handling characteristics of the devices in the real world.
Other limitations include that the respiratory device experts chosen as advisors may not have been able to identify all of the handling errors that may occur in daily usage of the devices owing to their inherent expertise. As such, the findings from this study rely on the judgment of a select group who are experts in the field and may not be representative of the wider healthcare community. Additionally, the Delphi process employs a consensus approach. Extreme positions are usually eliminated and the outcome may be a diluted version of the overall expressed opinions.
Conclusions
Through the Delphi process, experts in respiratory devices independently identified and reached consensus on potential handling errors for Spiromax and Turbuhaler and their severity. ‘Not inhaling through the mouthpiece (e.g., nose)' was one of the most highly rated critical errors for both devices. Critical errors in priming the device were identified for Turbuhaler, but not for Spiromax, as might be expected given the difference in operation of the two devices. There were fewer errors identified overall for Spiromax compared with Turbuhaler, and fewer errors with Spiromax were classified as critical or potentially critical than with Turbuhaler. Altogether, this indicates that there may be less potential for critical errors with Spiromax than Turbuhaler.
References
Calverley PM, Boonsawat W, Cseke Z, Zhong N, Peterson S, Olsson H. Maintenance therapy with budesonide and formoterol in chronic obstructive pulmonary disease. Eur Respir J. 2003;22(6):912–9.
Rabe KF, Atienza T, Magyar P, Larsson P, Jorup C, Lalloo UG. Effect of budesonide in combination with formoterol for reliever therapy in asthma exacerbations: a randomised controlled, double-blind study. Lancet. 2006;368(9537):744–53.
Haughney J, Price D, Barnes N, Virchow J, Roche N, Chrystyn H. Choosing inhaler devices for people with asthma: current knowledge and outstanding research needs. Respir Med. 2010;104(9):1237–45.
Molimard M, Le Gros V. Impact of patient-related factors on asthma control. J Asthma. 2008;45(2):109–13.
Papi A, Haughney J, Virchow JC, Roche N, Palkonen S, Price D. Inhaler devices for asthma: a call for action in a neglected field. Eur Respir J. 2011;37(5):982–5.
Al-Jahdali H, Ahmed A, Al-Harbi A, et al. Improper inhaler technique is associated with poor asthma control and frequent emergency department visits. Allergy Asthma Clin Immunol. 2013;9(1):8.
Arora P, Kumar L, Vohra V, et al. Evaluating the technique of using inhalation device in COPD and bronchial asthma patients. Respir Med. 2014;108(7):992–8.
Melani AS, Bonavia M, Cilenti V, et al. Inhaler mishandling remains common in real life and is associated with reduced disease control. Respir Med. 2011;105(6):930–8.
Molimard M, Raherison C, Lignot S, Depont F, Abouelfath A, Moore N. Assessment of handling of inhaler devices in real life: an observational study in 3811 patients in primary care. J Aerosol Med. 2003;16(3):249–54.
Thomas M. Why aren’t we doing better in asthma: time for personalised medicine? NPJ Prim Care Respir Med. 2015;25:15004.
Crompton GK, Barnes PJ, Broeders M, et al. The need to improve inhalation technique in Europe: a report from the Aerosol Drug Management Improvement Team. Respir Med. 2006;100(9):1479–94.
Molimard M. How to achieve good compliance and adherence with inhalation therapy. Curr Med Res Opin. 2005;21(Suppl. 4):S33–7.
Inhaler Error Steering Committee, Price D, Bosnic-Anticevich S, et al. Inhaler competence in asthma: common errors, barriers to use and recommended solutions. Respir Med. 2013;107(1):37–46.
Virchow JC, Akdis CA, Darba J, et al. A review of the value of innovation in inhalers for COPD and asthma. J Mark Access Health Policy. 2015;3:28760.
Smith IJ, Bell J, Bowman N, Everard M, Stein S, Weers JG. Inhaler devices: what remains to be done? J Aerosol Med Pulm Drug Deliv. 2010;23(Suppl 2):S25–37.
Ninane V, Brusselle GG, Louis R, et al. Usage of inhalation devices in asthma and chronic obstructive pulmonary disease: a Delphi consensus statement. Expert Opin Drug Deliv. 2014;11(3):313–23.
Lavorini F, Magnan A, Dubus JC, et al. Effect of incorrect use of dry powder inhalers on management of patients with asthma and COPD. Respir Med. 2008;102(4):593–604.
Weisfeld L, Shu Y, Shah TP. Bioequivalence of budesonide plus formoterol (BF) Spiromax(R) and BF Turbohaler(R) (with and without charcoal block) in healthy volunteers. Int J Clin Pharmacol Ther. 2015;53(7):593–602.
Hsu C-C, Brian AS. The Delphi technique: making sense of consensus. Pract Assess Res Eval. 2007;12:1–8.
Rowe G, Wright G. The Delphi technique as a forecasting tool: issues and analysis. Int J Forecast. 1999;15:353–75.
Gordon TJ. The Delphi method. Washington, DC: American Council for the United Nations University; 1994.
Delbecq A, Van de Ven A, Gustafson D. Group techniques for program planning: a guide to nominal and Delphi processes. Glenview: Scott, Foresman & Company; 1975. p. 174.
Sandler N, Holländer J, Långström D, Santtila P, Saukkonen A, Torvinen S. Evaluation of inhaler technique mastery and handling errors with Spiromax®, Easyhaler®, and Turbuhaler® devices (FINHALER) [abstract]. In: Proceedings of the 17th Annual European congress of the International Society for Pharmacoeconomics and Outcomes Research (ISPOR), 8–12 Nov 2014, Amsterdam, The Netherlands.
Bosnic-Anticevich S, Lim D, Steel J, et al. Investigating handling errors over time for inhaler devices: the healthcare professional perspective [abstract 1785]. Allergy. 2015;70(Suppl 101):645.
(GINA) GIfA. Global strategy for asthma management and prevention 2014 [07/2015]. http://www.ginasthma.org/. Accessed Nov 2015.
Ovchinikova L, Smith L, Bosnic-Anticevich S. Inhaler technique maintenance: gaining an understanding from the patient’s perspective. J Asthma. 2011;48(6):616–24.
Price D, Brusselle G, Roche N, Freeman D, Chisholm A. Real-world research and its importance in respiratory medicine. Breathe (Sheff). 2015;11(1):26–38.
Lavorini F. The challenge of delivering therapeutic aerosols to asthma patients. ISRN Allergy. 2013;2013:102418.
Scichilone N, Benfante A, Bocchino M, et al. Which factors affect the choice of the inhaler in chronic obstructive respiratory diseases? Pulm Pharmacol Ther. 2015;31:63–7.
Linstone H, Turoff M. The Delphi method: techniques and approaches. New Jersey Institute of Technology; 2002. http://is.njit.edu/pubs/delphibook/. Accessed Nov 2015.
Acknowledgments
Sponsorship and article processing charges for this study were funded by Teva Branded Pharmaceutical Products R & D, Inc. (Frazer, PA). Medical writing support was provided by Frances Weir of GeoMed, an Ashfield company, part of UDG Healthcare plc. Support for this assistance was funded by Teva Branded Pharmaceutical Products R & D, Inc. Teva provided a full review of the article. GG and GS commissioned the study; CH and SB designed the study; SB and CH acquired the data; SB and CH analyzed the data; all authors interpreted the data and were involved in development of the manuscript and completed critical reviews. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval to the version to be published.
Disclosures
Guilherme Safioti is an employee of Teva Pharmaceuticals. Gokul Gopalan was an employee of Teva Pharmaceuticals at the time of this study. Charlie Hewitt and Sebastian Heinzmann are employees of Interphase Consult.
Compliance with ethics guidelines
Per Office for Human Research Protections (OHRP) approved assurance created under 45 CFR 46.103, the protocol was exempt from informed written consent because (1) the research presented no more than minimal risk of harm to subjects, (2) the exemption did not adversely affect the rights and welfare of the subjects, (3) the research could not practicably be carried out without the exemption, and (4) whenever appropriate, the subjects were provided with additional pertinent information after participation. (http://www.hhs.gov/ohrp/humansubjects/guidance/45cfr46.html).
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Hewitt, C., Heinzmann, S., Safioti, G. et al. Independent Identification of Handling Errors for Dry Powder Inhalers, Spiromax® and Turbuhaler®, Using a Delphi Process Involving Respiratory Device Experts. Pulm Ther 1, 77–90 (2015). https://doi.org/10.1007/s41030-015-0008-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41030-015-0008-5