Abstract
The seasonal population dynamics of Fusarium solani f. sp. phaseoli (Fsp) in naturally infested soil and the severity of Fusarium root rot were studied. Areas were sequentially planted (2 to 4 sowings) with the same legume crop (common bean, pigeonpea, sunn hemp, and jack bean) or cereal crop (corn, pearl millet and signalgrass) during the year, which were then compared to background bare soil. Counts of colony forming units (CFU) of Fsp populations during 12 sampling times spaced around one month were high at the first sowing and decreased in the bare soil and cereal crops. CFU reached zero after 9 months in the maize and pearl millet plots and after 11 months in the bare soil and signalgrass plots. The count of CFU in the legume plots peaked during the summer and decreased during the winter, in a pattern that followed monthly air temperatures. Disease severity was higher in common bean than in the other crops and tended to increase over time. Planting of legumes should be avoided in infested soil and cereal crops may be an option to reduce and even eradicate inoculum as part of an integrated disease management of Fusarium root rot.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Fusarium solani f. sp. phaseoli W.C. Snyder & H.N. Hansen (Fsp) is a soil-borne fungus that causes Fusarium root rot in common beans (Phaseolus vulgaris L.). This fungus is widespread in all common bean production areas of Brazil (Toledo-Souza et al. 2009), where severe epidemics of Fusarium root rot are commonly observed (Paula Júnior et al. 2006). The disease causes damping-off of seedlings and root rot on adult plants, both leading to yield losses. The fungus is able to survive in the form of chlamydospores for many years in the soil which eventually colonize roots of non-host crops without causing disease (Abawi and Pastor-Corrales 1990).
Root rots are generally difficult to control (Paula Júnior et al. 2006) and biological control using Trichoderma spp. (Teixeira et al. 2012) and genetic resistance (Nicoli et al. 2012) may help to reduce crop losses. Although the former has been stated as a long-lasting and effective strategy for root rot control (Teixeira et al. 2012), some isolates of Trichoderma spp. are not effective against soil-borne pathogens in regions of Brazil with average temperature below 20 °C, since this condition favors the pathogen more than the antagonist (Paula Júnior et al. 2012). Materials with complete resistance to Fusarium root rot remain a great challenge to common bean breeders. Thus, for pathogens such as Fsp that have a very narrow host range (Abawi and Pastor-Corrales 1990), the use of non-host crops in rotational systems may lead to improved control (Paula Júnior et al. 2006; Toledo-Souza et al. 2008). Generally, cereal crops planted prior to common bean are suppressive to Fusarium spp. populations in soils whereas leguminous crops can maintain or increase population size (Toledo-Souza et al. 2008). However, the benefits of using cereal crops as well as the harm of using legumes have been not well quantified in terms of their effects on the population dynamic of Fsp in soil. Thus, this study aimed to quantify year-round survival and population dynamics of Fsp in soil and the severity of Fusarium root rot in field plots sequentially planted with several cereal and legume crops over 1 year.
Different crops (treatments) were planted simultaneously in two neighboring fields in an experimental research area of Empresa de Pesquisa Agropecuária de Minas Gerais (EPAMIG), located in Oratórios (20°24′11″ S, 42°49′08″ W, elevation 478 m), state of Minas Gerais, Brazil. Experiments were conducted for 1 year starting on November 20, 2006. The soil in the areas selected for the experiments was naturally infested with Fsp due to the production of common bean over the previous 6 years. Four legume crops were examined: common bean (Phaseolus vulgaris cv. Pérola, the most widely planted in Brazil), jack bean (Canavalia ensiformis), pigeonpea (Cajanus cajan) and sunn hemp (Crotalaria juncea). Furthemore, three cereal crops were also examined: maize (Zea mays, cv. AG-1051), pearl millet (Pennisetum glaucum) and signalgrass (Brachiaria decumbens). An additional bare soil (no crop) treatment was included. Each plot was weeded by hand on a weekly basis. At the end of a crop cycle, shoots of the crops were either pulled or cut, depending on the crop, and the next crop cycle was planted on the same day. Each trial used a randomized complete-block design with six replicates (plots). Plots consisted of four 2 m-long rows, spaced 0.5 m apart, with 10 plants per meter. In signalgrass plots, seeds were hand-sown at plots (6 kg seed/ha). At each replanting, a commercial fertilizer (4 N:14P2O5:8K2O) at 600 kg/ha was applied in the furrows along with the seeds. On November 21, 2006, glyphosate (4 kg/ha) was applied once for weed control in the entire experimental area. Insect control was performed as needed. The experimental area was irrigated with overhead sprinklers positioned 1.5 m above ground level. Irrigation was provided as needed to promote good seedling emergence, and thereafter at a rate of approximately 40 mm of water per week, which was an amount similar to that of the region.
Legume crops were evaluated for Fusarium root root rot severity 11 times at approximately 1 month spacing using a 1–9 disease scale (Van Schoonhoven and Pastor-Corrales 1987). On each assessment, soil was sampled and the population of Fsp was estimated as the number of colony-forming units (CFU) per gram of soil. Ten soil samples (30 g) were randomly collected in a plot from the upper 10 cm of soil. Samples were thoroughly mixed before 1.0 g of soil was suspended in water to a concentration of 0.1 mg/L. Afterward, each sample was placed on Petri dishes with PCNB-peptone-agar (Abawi and Pastor-Corrales 1990). Dishes were incubated in darkness for 48 h at 22 °C. An initial assessment of Fsp population density on November 10 2006 indicated a homogenous distribution of the inoculum in the experimental area, with an average of 4.2 × 104 CFU/g of soil. Such inoculum density is considered the minimum level to be able to cause disease in this pathosystem (Costa and Costa 2004).
A visual evaluation of the data indicated that the mean CFU counts over the year in some treatments (legume crops) had a multimodal pattern, and a preliminary fit of a linear regression model indicated a poor fit, although one that could be considered statistically significant (data not shown). Hence, we fitted a generalized additive model (GAM), which includes both a linear term as well as a non-parametric smoothing term that attributes local fluctuations to unexplained random noise in order to highlight the seasonal trend (Wood 2006). A thin plate regression spline was used as the smoothing of the model fitted to Poisson distributed (count) data with a square-root link function to relate the random component and the additive component. The dimension of the basis used to represent the smooth term (k) was chosen as 6 after increasing k value at each model fit until a non-statistically important change was observed. The first model runs included crop and trial and their interaction as linear terms and one smooth term by each level of the factor. To test whether the data exhibited significant seasonal structure depending on treatments, significance of the parametric coefficients was tested via F test and the approximate significance of the smooth term was tested via chi-square test from the analysis of deviance. All analyses were conducted using R (R Development Core Team). The GAMs were fitted using the mgcv package. The fitted values of seasonal trends and respective 95 % confidence interval (using k = 6) for each treatment were depicted using the ggplot2 package.
Severity data was also used to calculate AUDPC based on the trapezoidal integration method using the agricolae package of R. Treatment effects were tested and compared by analyses of variance (ANOVA) and Fisher’s protected LSD tests. Since the factor “experimental site” was not significant in the preliminary ANOVA (data not shown), data from the two experimental sites were combined.
Mean daily air temperature were obtained form a standard weather station located at the experimental site. Temperatures were summarized as monthly averages, or the average temperature for an approximately 30-day period prior to planting date and prior to each assessment date, totaling 12 averages over the year. The overall trend for the 12 monthly averages was also depicted using values estimated by a GAM (k = 6).
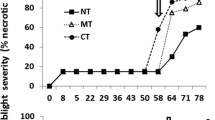
Inoculum density of Fsp was significantly affected by treatment (p < 0.001). The deviance explained by this reduced model (equivalent to R2) was 82.9 %. Overall, there was a consistent decrease in Fsp inoculum over the year in bare soil and in the non-host cropping systems. The CFU reached zero after 9 months in the maize and pearl millet rotations, and was zero after 11 months in the bare soil and signalgrass treatments (Fig. 1). However, a seasonal pattern was found in all legume crops sown in the infested soils. In general, and consistently across the four legume crops, the inoculum increased to a peak during mid-summer (February); decreased afterwards reaching the lowest levels in mid-winter (July); and started a new peak until the end of the experiment. Both the linear and smoothing terms in the GAM were influenced by crop (p < 0.05), with coefficients for common bean differing significantly from jack bean, which differed from pigeonpea and sunn hemp (data not shown). The CFU patterns over the year followed the smoothed trend of monthly average temperatures between assessment times (Fig. 1, dashed line).
Dynamics of the number of colony forming units (CFU) of Fusarium solani f. sp. phaseoli in soil of two experimental trials (circles) where three cereal crops (left graphs) and four legume crops (right graphs) were planted. Solid line is the fit of a generalized additive model (GAM) for the mean CFU of the two trials combined. The vertical arrows on the x axes indicate the date of the replanting for each species. Dashed line is the smoothed trend line (GAM) fitted to the approximately monthly average temperature between assessment times during the year. Note that the dashed line is the same for each crop
The pattern of root rot severity in the four legume crops over the year was similar to Fsp inoculum density, with a peak during summer, a decrease during winter, and another peak in spring. Mean AUDPC (of the two experiments combined) varied significantly among the crops: common bean > jack bean > pigeon pea = sunn hemp (Fig. 2).
Rotation with non-host cerealcrops has been recommended for Fsp-infested soils (Abawi and Pastor-Corrales 1990; Paula Júnior et al. 2006). Toledo-Souza et al. (2008) reported that maize, pearl millet, sorghum, guinea grass, and signalgrass may be suppressive to soil populations of Fusarium spp. when used as previous crops incorporated into the soil (conventional tillage) or maintained on the soil surface (no-tillage). Our results confirm that growing non-host cereal crops is an effective measure to reduce Fsp inoculum in the soil. In addition to crop rotation with a non-host cereal crop, different soil management practices have been recommended for managing Fusarium root rot in common bean (Abawi and Pastor-Corrales 1990; Berni et al. 2002; Paula Júnior et al. 2006). Toledo-Souza et al. (2008) found that soil-borne Fusarium spp. inoculum was higher in no-till than in conventionally tilled fields, except when beans were planted on residues of Brachiaria brizantha. Hall and Phillips (1992) showed evidence that the population dynamics of Fsp in soil depends on the effects of crop sequence on parasitic activities of the pathogen. In a rotation trial conducted by these authors over 14 years, population densities of Fsp in soil remained low in fallow, maize and soybean treatments. When common bean was grown every 3rd year, and especially every year, Fsp population densities increased significantly. Our results confirmed that not using common bean or other legume crops is a beneficial strategy to suppress and eradicate Fsp population in the soil after 9 months.
Viable chlamydospores of Fsp can remain in soil for long periods of time (Burke and Hall 1991; Mondal and Hyakumachi 1998). However, in the absence of hosts, their germination may be reduced by nutrient deficiency in the soil and by formation of inhibitory substances produced from decomposing residues (Lewis and Papavizas 1977). Conversely, the continuous planting of susceptible crops may keep or even increase the Fsp population density in the soil. We found that this increase in the pathogen population varied across crops, with pigeonpea and sunn hemp leading to lower increases of the population compared to common bean and jack bean, which is in agreement with previous studies (Berni et al. 2002; Toledo-Souza et al. 2008).
Our data suggest that fluctuations in population densities and disease severity over the year were associated with seasonal temperatures. According to Abawi and Pastor-Corrales (1990), disease severity in the field is higher at 22 °C and lower at 32 °C. Probably, at temperatures below 18 °C, as those registered between June and August, the pathogen’s metabolism was reduced.
Pigeonpea, sunn hemp and jack bean are used either as green manure or in rotation systems involving common bean. Results from this study and from previous studies (Berni et al. 2002; Toledo-Souza et al. 2008) indicated that they should not be used in rotation systems is soils infested by Fsp. In contrast, crop rotation with non-host cereal crops would be an effective measure under an integrated disease management strategy. We conclude that the use of cereals in crop rotation with common bean may effectively reduce the risk of root rot epidemics by reducing Fsp primary inoculum.
References
Abawi GS, Pastor-Corrales MA (1990) Root rots of beans in Latin America and Africa: diagnosis, research methodologies, and management strategies. CIAT, Cali
Berni RF, Silveira PM, Costa JLS (2002) Influência do preparo de solo e da rotação de culturas na severidade de podridões radiculares no feijoeiro comum. Pesq Agrop Trop 32:69–74
Burke DW, Hall R (1991) Fusarium root rot. In: Hall R (ed) Compendium of Bean Diseases. APS Press, St. Paul, pp 9–10
Costa G, Costa J (2004) Influência da densidade de inóculo de Fusarium solani f. sp. phaseoli na severidade da podridão radicular seca do feijoeiro. Pesq Agrop Trop 34:89–92
Hall R, Phillips LG (1992) Effects of crop sequence and rainfall on population dynamics of Fusarium solani f. sp. phaseoli in soil. Can J Bot 70:2005–2008
Lewis JA, Papavizas GC (1977) Effect of plant residues on chlamydospore germination of Fusarium solani f. sp. phaseoli and on Fusarium root rot of bean. Phytopathology 67:925–929
Mondal SN, Hyakumachi M (1998) Carbon loss and germinability, viability, and virulence of chlamydospores of Fusarium solani f. sp. phaseoli after exposure to soil at different pH levels, temperatures, and matric potentials. Phytopathology 88:148–155
Nicoli A, Zambolim L, Paula Júnior TJ, Vieira RF, Teixeira H, Carneiro JES (2012) Resistance of advanced common bean lines to Fusarium root rot. Trop Plant Pathol 37:393–398
Paula Júnior TJ, Lobo Júnior M, Sartorato A, Vieira RF, Carneiro JES, Zambolim L (2006) Manejo integrado de doenças do feijoeiro em áreas irrigadas - Guia Técnico. EPAMIG-CTZM, Viçosa
Paula Júnior TJ, Teixeira H, Vieira RF, Morandi MAB, Lehner MS, Lima RC, Carneiro JES (2012) Limitations in controlling white mold on common beans with Trichoderma spp. at the fall-winter season. Summa Phytopathol 38:337–340
Teixeira H, Paula Júnior TJ, Vieira RF, Silva MB, Ferro CG, Lehner MS (2012) Trichoderma spp. decrease Fusarium root rot in common bean. Summa Phytopathol 38:334–336
Toledo-Souza ED, Silveira PM, Lobo Junior M, Café Filho AC (2008) Sistemas de cultivo, sucessões de culturas, densidade do solo e sobrevivência de patógenos de solo. Pesq Agrop Brasileira 43:971–978
Toledo-Souza ED, Lobo Junior M, Silveira PM, Café Filho AC (2009) Interações entre Fusarium solani f. sp. phaseoli e Rhizoctonia solani na severidade da podridão radicular do feijoeiro. Pesq Agrop Trop 39:13–17
Van Schoonhoven A, Pastor-Corrales MA (1987) Standard system for the evaluation of bean germplasm. CIAT, Cali
Wood SN (2006) Generalized additive models: an introduction with R. Chapman and Hall/CRC Press, Boca Raton
Acknowledgments
HT, MSL and RCL wish to thank FAPEMIG for their fellowships. TJPJ, EMDP and RFV wish to thank CNPq for their fellowships. This work was supported by FAPEMIG and CNPq grants.
Author information
Authors and Affiliations
Corresponding author
Additional information
Section Editor: Paul Esker
Rights and permissions
About this article
Cite this article
Teixeira, H., Júnior, T.J.P., Vieira, R.F. et al. Seasonal dynamics of soil-borne inoculum and severity of Fusarium root rot of common beans affected by sequential planting of legume or cereal crops. Trop. plant pathol. 40, 335–338 (2015). https://doi.org/10.1007/s40858-015-0047-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40858-015-0047-3