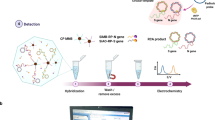
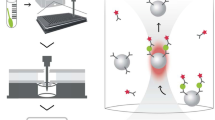
Abstract
Coronavirus disease 2019 (COVID-19), caused by SARS-CoV-2, has rapidly spread and caused a severe global pandemic. Because no specific drugs are available for COVID-19 and few vaccines are available for SARS-CoV-2, accurate and rapid diagnosis of COVID-19 has been the most crucial measure to control this pandemic. Here, we developed a portable bifunctional electrical detector based on graphene fieldeffect transistors for SARS-CoV-2 through either nucleic acid hybridization or antigen-antibody protein interaction, with ultra-low limits of detection of ~0.1 and ~1 fg mL−1 in phosphate buffer saline, respectively. We validated our method by assessment of RNA extracts from the oropharyngeal swabs of ten COVID-19 patients and eight healthy subjects, and the IgM/IgG antibodies from serum specimens of six COVID-19 patients and three healthy subjects. Here we show that the diagnostic results are in excellent agreement with the findings of polymerase chain reaction-based optical methods; they also exhibit rapid detection speed (~10 min for nucleic acid detection and ~5 min for immunoassay). Therefore, our assay provides an efficient, accurate tool for high-throughput point-of-care testing.

摘要
新型冠状病毒肺炎(COVID-19)正在多个国家快速传播, 已 经导致了严重的全球大流行. 由于目前没有针对此类病人的特效 药和针对此病毒的疫苗, 准确、快速地进行新冠病人检测成为了 控制大流行最有效的措施. 本文中我们开发了一种基于石墨烯场 效应晶体管的便携式双功能电检测仪, 其通过核酸互补杂交或者 抗原-抗体特异性结合作用, 能分别进行病毒核酸序列检测和抗体 检测, 检测限分别低至0.1和1 fg mL−1. 我们通过临床样品检测进一 步评估了此方法: 从10个新冠病人和8个正常人咽拭子中提取RNA 直接用于核酸检测; 从6个新冠病人和3个正常人血清中提取抗体 用于抗体检测. 临床样品检测结果和基于聚合酶链反应的光学方 法结果完全吻合, 同时此方法拥有超快的检测速度(核酸检测需 10 min, 抗体检测需5 min). 因此, 我们的实验提供了一种有效、准 确、高通量的新冠现场即时检测工具.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Zhu N, Zhang D, Wang W, et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med, 2020, 382: 727–733
Wang C, Horby PW, Hayden FG, et al. A novel coronavirus outbreak of global health concern. Lancet, 2020, 395: 470–473
de Wit E, van Doremalen N, Falzarano D, et al. SARS and MERS: Recent insights into emerging coronaviruses. Nat Rev Microbiol, 2016, 14: 523–534
Lu R, Zhao X, Li J, et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: Implications for virus origins and receptor binding. Lancet, 2020, 395: 565–574
WHO Coronavirus Disease 2019 (COVID-19): Situation Report-52. https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200312-sitrep-52-covid-19.pdf?sfvrsn=e2bfc9c0_4
WHO Coronavirus Disease (COVID-19) Dashboard. https://covid19.who.int
Talebian S, Wallace GG, Schroeder A, et al. Nanotechnology-based disinfectants and sensors for SARS-CoV-2. Nat Nanotechnol, 2020, 15: 618–621
Broughton JP, Deng X, Yu G, et al. CRISPR-Cas12-based detection of SARS-CoV-2. Nat Biotechnol, 2020, 38: 870–874
Chan JFW, Yip CCY, To KKW, et al. Improved molecular diagnosis of COVID-19 by the novel, highly sensitive and specific COVID-19-RdRp/Hel real-time reverse transcription-PCR assay validated in vitro and with clinical specimens. J Clin Microbiol, 2020, 58: e00310–20
Real-time RT-PCR panel for detection 2019-nCoV (US Centers for Disease Control and Prevention, 2020). https://www.who.int/docs/default-source/coronaviruse/uscdcrt-pcr-panel-for-detection-instructions.pdf
WHO Laboratory testing for coronavirus disease 2019 (Covid-19) in suspected human cases: Interim guidance, 2020. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/technicalguidance/laboratory-guidance
Xie X, Zhong Z, Zhao W, et al. Chest CT for typical coronavirus disease 2019 (COVID-19) pneumonia: Relationship to negative RT-PCR testing. Radiology, 2020, 296: E41–E45
Zhang Q, Zhao Q. Inactivating porcine coronavirus before nuclei acid isolation with the temperature higher than 56°C damages its genome integrity seriously. BioRxiv, 2020, doi: https://doi.org/10.1101/2020.02.20.958785
Lu CH, Yang HH, Zhu CL, et al. A graphene platform for sensing biomolecules. Angew Chem Int Ed, 2009, 48: 4785–4787
He G, Li J, Ci H, et al. Direct measurement of single-molecule DNA hybridization dynamics with single-base resolution. Angew Chem Int Ed, 2016, 55: 9036–9040
Ke G, Duan C, Huang F, et al. Electrical and spin switches in single-molecule junctions. InfoMat, 2020, 2: 92–112
Su D, Gu C, Guo X. Functional molecular electronic devices through environmental control. Sci China Mater, 2019, 62: 1–7
Syu YC, Hsu WE, Lin CT. Review-Field-effect transistor biosensing: Devices and clinical applications. ECS J Solid State Sci Technol, 2018, 7: Q3196–Q3207
Liang Y, Xiao M, Wu D, et al. Wafer-scale uniform carbon nanotube transistors for ultrasensitive and label-free detection of disease biomarkers. ACS Nano, 2020, 14: 8866–8874
Namdari P, Daraee H, Eatemadi A. Recent advances in silicon nanowire biosensors: Synthesis methods, properties, and applications. Nanoscale Res Lett, 2016, 11: 406
Szunerits S, Boukherroub R. Graphene-based biosensors. Interface Focus, 2018, 8: 20160132
Jia C, Li H, Jiang J, et al. Interface-engineered bistable [2]rotaxanegraphene hybrids with logic capabilities. Adv Mater, 2013, 25: 6752–6759
Campos R, Borme J, Guerreiro JR, et al. Attomolar label-free detection of DNA hybridization with electrolyte-gated graphene field-effect transistors. ACS Sens, 2019, 4: 286–293
Seo G, Lee G, Kim MJ, et al. Rapid detection of COVID-19 causative virus (SARS-CoV-2) in human nasopharyngeal swab specimens using field-effect transistor-based biosensor. ACS Nano, 2020, 14: 5135–5142
Van Elslande J, Houben E, Depypere M, et al. Diagnostic performance of seven rapid IgG/IgM antibody tests and the Euroimmun IgA/IgG ELISA in COVID-19 patients. Clin Microbiol Infection, 2020, 26: 1082–1087
Tuaillon E, Bolloré K, Pisoni A, et al. Detection of SARS-CoV-2 antibodies using commercial assays and seroconversion patterns in hospitalized patients. J Infection, 2020, 81: e39–e45
Li Z, Yi Y, Luo X, et al. Development and clinical application of a rapid IgM-IgG combined antibody test for SARS-CoV-2 infection diagnosis. J Med Virol, 2020, 92: 1518–1524
Hou H, Wang T, Zhang B, et al. Detection of IgM and IgG antibodies in patients with coronavirus disease 2019. Clin Transl Immunol, 2020, 9: e1136
Malard LM, Pimenta MA, Dresselhaus G, et al. Raman spectroscopy in graphene. Phys Rep, 2009, 473: 51–87
Ye J, Craciun MF, Koshino M, et al. Accessing the transport properties of graphene and its multilayers at high carrier density. Proc Natl Acad Sci USA, 2011, 108: 13002–13006
Xin N, Li X, Jia C, et al. Tuning charge transport in aromatic-ring single-molecule junctions via ionic-liquid gating. Angew Chem Int Ed, 2018, 57: 14026–14031
Chen JH, Ishigami M, Jang C, et al. Printed graphene circuits. Adv Mater, 2007, 19: 3623–3627
Xu S, Zhan J, Man B, et al. Real-time reliable determination of binding kinetics of DNA hybridization using a multi-channel graphene biosensor. Nat Commun, 2017, 8: 14902
Cai B, Wang S, Huang L, et al. Ultrasensitive label-free detection of PNA-DNA hybridization by reduced graphene oxide field-effect transistor biosensor. ACS Nano, 2014, 8: 2632–2638
Acknowledgements
This work was supported by the National Key R&D Program of China (2017YFA0204901), the National Natural Science Foundation of China (21727806, 21772003 and 21933001), the Tencent Foundation through the XPLORER PRIZE, Guangdong Major Project of Basic and Applied Basic Research (2019B030302007), and Beijing National Laboratory for Molecular Sciences (BNLMS201901).
Author information
Authors and Affiliations
Contributions
Guo X, Mo F, Wang P and Huang F conceived and designed the experiments; Ke G, Su D and Li Y fabricated the devices and performed the device measurements; Zhao Y, Wang H, Xiao F and Yuan Y designed and built the measurement machines; LiuWand Yang Z provided the antigen protein; Li M and Wang P provided the clinical samples; Guo X, Mo F, Wang P, Ke G and Su D analyzed the data and wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding authors
Additional information
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary information
Experimental details and supporting data are available in the online version of this paper.
Guojun Ke received his BS degree in 2012 and PhD degree in 2017 from the School of Chemistry, Sun Yat-Sen University, respectively. From 2013 to 2016, he was a visiting student at the University of Basel. He is currently working as a postdoctoral fellow in South China University of Technology. His current research focuses on device physics of single-molecule junctions.
Dingkai Su received his BS degree in 2017 from the College of Nano Science and Technology, Soochow University. He is currently a PhD candidate at the College of Chemistry and Molecular Engineering, Peking University, under the guidance of Prof. Xuefeng Guo. His research interest focuses on single-molecule devices and dynamics.
Xuefeng Guo received his PhD degree in 2004 from the Institute of Chemistry, Chinese Academy of Sciences. From 2004 to 2007, he was a postdoctoral research scientist at the Columbia University Nanocenter. He joined the faculty as a professor under the “Peking 100-Talent” Program at Peking University in 2008. His research focuses on functional nanometer/molecular devices.
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Ke, G., Su, D., Li, Y. et al. An accurate, high-speed, portable bifunctional electrical detector for COVID-19. Sci. China Mater. 64, 739–747 (2021). https://doi.org/10.1007/s40843-020-1577-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-020-1577-y