Abstract
Background
When medical therapy is unable to achieve biochemical control of secondary hyperparathyroidism, parathyroidectomy (PTX) is indicated, fortunately in a minority of patients. Thus, data on PTX prevalence and biochemical control are limited and, in particular in Italy, date back to 1999.
Methods
We designed a prospective, observational and multicenter study to collect data from dialysis units distributed throughout the Italian regions. Clinical data were collected with a dedicated data sheet.
Results
From January to December 2010, 149 Centers serving a total of 12,515 patients provided data on 528 living PTX cases (PTX prevalence = 4.2%). Prevalence was higher in hemo- than in peritoneal dialysis (4.5 vs. 1.9%, X2 = 21.52; p < 0.001), with non-significant regional differences (range 0.8–7.4%). PTX patients were younger (57.6 ± 12.5 vs. 67.1 ± 14.5 years; p < 0.001), more frequently female (56 vs. 38%, X2 = 68.05, p < 0.001) and had been on dialysis for a longer time (14.63 ± 8.37 vs. 4.8 ± 6.0 years, p < 0.001) compared to the 11,987 who did not undergo neck surgery. Median time since surgery was 6.0 years (3.0–9.0; 50%, IQR). The most frequent type of surgery was subtotal PTX (sPTX = 55.0%), significantly higher than total PTX (tPTX = 38.7%) or total PTX plus auto-transplantation (aPTX = 6.3%) (X2 = 5.18; Bonferroni post-hoc test, sPTX vs. tPTX + aPTX = p < 0.05). As for parathyroid hormone (PTH), calcium and phosphate control, cases targeting the KDOQI ranges were 18, 50.1 and 54.4%, respectively. The most prevalent biochemical condition was low PTH (62.7%).
Conclusion
PTX prevalence in Italy is stable compared to previous observations, is higher in hemodialysis than in peritoneal dialysis and results in a suboptimal biochemical control.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Secondary hyperparathyroidism (SHP) almost invariably affects end-stage renal disease (ESRD) patients and results in bone disease (renal osteodystrophy, ROD), bone fractures and vascular calcifications [1,2,3,4,5]. Further, in recent years the clinical relevance of divalent ions [6] and bone biomarkers [7] as prominent risk factors for all-cause and cardiovascular mortality has clearly emerged [8]. Since both high and low parathyroid hormone (PTH), calcium (Ca) and/or phosphate (P) levels are associated with poor cardiovascular outcomes [3, 6, 9], target ranges are recommended. Regrettably, available drugs like vitamin D, calcium sensing receptor activators and phosphate binders do not allow to reach the recommended ranges in many patients [6, 9,10,11] and parathyroid surgery (PTX) still remains a therapeutic option [12]. However, even surgical therapy does not guarantee optimal biochemical control and either persistently high or excessively low PTH values and/or off-target Ca and P are not rarely described following surgery [13, 14]. Also, country-specific differences secondary to variable therapeutic strategies are possible [15] which further widen the clinical outcome of patients who receive PTX. Intriguingly, despite suboptimal biochemical control, almost all available studies suggest that after PTX hemodialysis patients have a lower rate of all-cause and cardiovascular mortality [16, 17]. Regrettably, no explanation exists to justify improved survival after PTX and none of the published studies is randomized and controlled and therefore exempt from selection biases [18]. Since “there will never be a randomized controlled trial” on PTX [18], observational studies are still necessary. In particular, given that country-specific differences are possible and that the largest study on PTX prevalence in Italy dates back to 1999 and is limited to a single northern region of the country [19], we considered it useful to implement a large multicenter study involving all Italian regions. We present in this paper the preliminary results of our observational, multicenter study which aimed at evaluating, in Italian patients undergoing dialysis, the prevalence of PTX in more recent years, and its impact on the achievement of biochemical control. Results of the phase-II case-control prospective and observational study, dealing with all-cause and cardiovascular mortality, will be reported in a subsequent paper.
Methods
Study design
We designed an observational, prospective and multicenter study involving dialysis units spread throughout the country. The study protocol was characterized by two distinct phases. The first one was simply observational and aimed at identifying the main clinical characteristics of the prevalent PTX patients. The second phase was prospective and case-control and aimed at evaluating possible differences in mortality between PTX and non-PTX patients. The protocol was submitted to and approved by the local Ethics Committee (Policlinico Umberto I, Rome, Italy, protocol: No. 888/09). The study complied with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Inclusion criteria were: age older than 18 years, history of PTX, dialysis treatment (either hemodialysis or peritoneal dialysis), and capacity to give informed consent.
Data were collected from all the dialysis units that answered our call and agreed to participate. From January to December 2010 we received data from 149 dialysis units. The data sheet was filled by the referent physician, who identified patients with anonymous codes. The questionnaire addressed a variety of areas including medical history, timing of PTX, types of surgery (total, sub-total or total with auto-transplantation), laboratory data (calcium, phosphate, PTH, albumin, alkaline phosphatase, hemoglobin, ferritin, fibrinogen, C-reactive protein and erythrocyte sedimentation rate), and prescribed medications (vitamin D and calcium-based therapies, calcimimetic, phosphate binders and erythropoiesis stimulating agents). From each unit we also received information on the total number of treated patients, their sex, age, dialysis duration, comorbidities and cause of renal insufficiency. These data were used to select a comparable control-group of non-PTX patients for the second phase prospective study.
Endpoints
The primary endpoint of phase I of this study was to determine the prevalence of PTX in dialysis patients in Italy, its regional distribution and the extent to which PTX patients reach biochemical targets for mineral metabolism.
Statistical analysis
The prevalence of PTX in Italy was calculated by dividing the number of PTX cases by the total number of patients receiving dialysis treatment in the dialysis units participating in the study. We also calculated the percentage of patients achieving the Kidney Disease Outcomes Quality Initiative (KDOQI) [20] target ranges for Ca, P, and PTH. Data are expressed as mean ± standard deviation (SD) for Gaussian variables or median and interquartile range (IQR) when normality was not tenable. We used the Kolmogorov–Smirnov test to evaluate normality of continuous measurements. Parametric tests and chi-squared test were used for qualitative variables and t-test for quantitative variables, to compare measurements among groups. All tests were two tailed and (adjusted) p values < 0.05 were considered as statistically significant. When general r-by-c contingency tables yielded statistical significance, we proceeded to the evaluation of two-by-two sub-tables of interest. In that case, significance levels were Bonferroni-adjusted by multiplication by the number of two-by-two tables evaluated. The family-wise significance level was fixed at 5%, so that a Bonferroni-adjusted p value below 0.05 was considered as statistically significant after taking into account multiplicity. Analyses were performed using the open source software package R version 3.4.0 [21].
Results
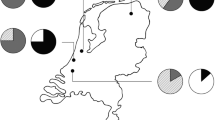
As illustrated in Fig. 1, the 149 units that positively answered our call were spread throughout the country. We received data pertinent to 528 living PTX cases, out of a total 12,515 prevalent patients. Table 1 reports the mean age, sex (%) and mean dialysis duration (years) of the PTX group compared to the 11,987 patients who did not undergo neck surgery. PTX patients were younger (57.6 ± 12.5 vs. 67.1 ± 14.5 years, p = 2.2e−16), more frequently female (56 vs. 38%, X2 68.05, p = 2.2e−16) and on dialysis for a longer time (14.63 ± 8.37 vs. 4.8 ± 6.0 years, p = 2.2e−16). Comorbidities, as illustrated in Table 1, were more common in non-PTX patients, except for diabetes which was significantly less frequent in PTX (7 vs. 17.3%, p = 8.08e−08). Compared to non-PTX, causes of ESRD in PTX patients were more commonly (Table 1) biopsy-proven or clinically-diagnosed glomerulonephritis (43 vs. 36%; p = 0.001) or tubular-interstitial nephropathies (12 vs. 8%; p = 4.7e−05), and less frequently nephroangiosclerosis (10 vs. 15%; p = 0.001). Prevalence of PTX was 4.2% for the whole country with small regional differences (regional range 0.8–7.4%, Fig. 2) that were not significantly different if we considered separately the three typical Italian macro regions: North (4.2%), Central (4.17%), and South (5.25%) Italy. As for dialysis technique, PTX prevalence was higher in hemodialysis than in peritoneal patients (4.5 vs. 1.9%, X2 = 21.52; p = 9.18e−07).
Median time since surgery was 6.0 years (3.0–9.0; 50%, IQR) and, as illustrated in Fig. 3, the distribution of PTX cases according to the year of surgery showed a drop starting from 2005. Indeed, from 2005 to 2009 the recorded cases were 34.4% of the total, which is lower than the 42.5% of the preceding 5 years (from 2000 to 2004). This drop is unexpected since we can hypothesize a stable mortality rate of the PTX patients. The most frequent type of surgery was subtotal PTX (sPTX = 55.0%), significantly higher than total PTX (tPTX = 38.7%) and total PTX plus auto-transplantation (aPTX = 6.3%) (X2 = 5.18; Bonferroni post hoc test, sPTX vs. tPTX + aPTXp = 0.022). In 1% of patients, the type of surgery was unknown.
Distribution of the recorded PTX cases according to the year of surgery. In the most recent 5 years (from 2005 to 2009), the recorded cases are lower (34.4%) than those of the preceding 5 years (42.5% from 2000 to 2004). This drop is unexpected (we can hypothesize a stable mortality rate) and is by chance coincident with cinacalcet introduction in Italy
As for PTH, average values were within the KDOQI ranges (181.42 ± 292.09 pg/ml). However, cases at target were only 18.6%, in particular due to a significant percentage of cases with low PTH (62.7%) (low PTH vs. non-low PTH: X2 = 34; Bonferroni adjusted p = 5.3e−09). We also considered the prevalence of patients at KDOQI target according to the different types of surgery. As illustrated in Table 2, the prevalence of low PTH was highest with tPTX and the prevalence of cases within the range was highest with sPTX.
As for calcium and phosphate, average values were 8.76 ± 0.87 and 4.90 ± 1.36 mg/dl, respectively. Although the average values were within the KDOQI ranges, cases at target were only 50.1% for calcium and 54.4% for phosphate. Calcium and phosphate control was similar among different types of surgery, in the whole population and, separately, within hemodialysis and peritoneal dialysis patients.
Discussion
The first finding of our study is that, in more recent years, PTX prevalence in Italian dialysis patients averages 4.2%, similarly to the 4.5% reported by Malberti et al. in 2001 which was limited to a specific Northern region of Italy [17]. Their result included patients receiving dialysis treatment between 1983 and 1996 and who thus were mainly treated with calcium- and aluminum-based phosphate binders, natural vitamin D and no calcium sensing receptor (CaSR) activators. Compared to the PTX prevalence in the United States between 1990 and 1999 (average 3.1%), or in Sweden 1991–2009 (average 3.3%) [22, 23] the value in Italy seems a little bit higher. Also, our data suggest a limited variability of PTX rate among different Italian regions, a finding in agreement with an unchanged surgical strategy. Indeed, the 2009 Kidney Disease Improving Global Outcomes (KDIGO) guidelines [24] (which are those pertinent to our study, and have been confirmed by the most recent 2017 review [25]) generically recommend surgery for patients resistant to PTH-lowering therapies without specific biochemical targets. On practical grounds, shifts in the rate of PTX have been reported, in particular when new therapies (like i.v. vitamin D or cinacalcet) were introduced [22, 26], and in our observation a drop in PTX cases appeared after 2005, coincident with the introduction of cinacalcet in Italy. In agreement, an on-line survey including 39 Italian dialysis units, evidenced significant heterogeneity in the clinical approach to PTX and a lower indication for parathyroid surgery in very recent years [27].
The second finding is related to the clinical characteristics of our PTX population. Patients were mainly of female gender, younger, and had a longer duration of dialysis therapy compared to the whole prevalent sample. All these differences are in line with data in the literature. Female gender is reported to be more susceptible to parathyroid hyperactivity and is a risk factor for high PTH levels in both secondary [28] and primary hyperparathyroidism [29]. Specifically, previous studies recognized female gender as a risk factor for PTX [18, 19]. Other recognized risk factors for the development of severe and unresponsive SHP are younger age and longer stay on dialysis. However, we are aware that our finding in this regard could have resulted from a possible selection bias, since our study included prevalent patients. Elderly people with increased comorbidities and lower dialysis survival rate are less likely eligible for surgery and more frequently affected with low bone turnover and low PTH levels [30].
We also found a low prevalence of diabetes (7%) compared to the high proportion among dialysis patients averaging 17% in our population and ranging between 24 and 51% in the literature [31]. This finding is in line with previous studies, and could be hypothetically related to low PTH levels secondary to PTH secretion inhibition by advanced glycation end products [32], skeletal resistance to PTH [33] and high prevalence of adynamic bone disease (ABD) [34]. Further, the high cardiovascular risk of diabetic patients hampers PTX surgery.
Finally, the prevalence of PTX was low also in peritoneal dialysis, a finding at variance with other reports [19, 22]. However, a high incidence of low PTH and ABD in this class of patients (averaging 50%), has been recently described and was related to increased bone sclerostin expression [35].
As for PTH control, a limited 18.6% patients were within the KDOQI recommended PTH target levels. In particular, the prevailing biochemical outcome was low PTH, with the highest prevalence in total PTX cases. Such a high prevalence of low PTH in the long term after PTX raises the question of the indication, if any, for neck surgery in ESRD patients. Indeed, the development of ABD becomes highly possible, and ABD is now regarded as the most prevalent type of bone disease in dialysis patients [34], even in non PTX cases. ABD is considered a clinical condition with increased mortality due to cardiovascular disease and bone fractures [3]. However, the potential risk of low PTH values and ABD after PTX seems to be counter-balanced by the improved survival rate almost invariably reported in the literature [16, 17]. Potentially positive effects of PTX include improvements in left ventricular hypertrophy [36], blood pressure, erythropoietin-resistant anemia [37], nutritional status and humoral and cellular immunity [38].
As for calcium and phosphate control, only 50% of patients were within the recommended target range, thus confirming that PTX does not guarantee biochemical control of mineral metabolism.
Conclusion
PTX prevalence in Italy is stable compared to previous observations, is higher in hemodialysis than in peritoneal dialysis, and results in a suboptimal biochemical control with prevailing low PTH values.
References
Torres PA, De Broe M (2012) Calcium-sensing receptor, calcimimetics, and cardiovascular calcifications in chronic kidney disease. Kidney Int 82:19–25
Bover J, Evenepoel P, Urena-Torres P, on behalf of the CKD-MBD Working group of ERA-EDTA et al (2015) Pro: Cardiovascular calcifications are clinically relevant. Nephrol Dial Transplant 30(3):345–351
London GM, Marty C, Marchais SJ, Guerin AP, Metivier F, de Vernejoul MC (2004) Arterial calcifications and bone histomorphometry in end-stage renal disease. J Am Soc Nephrol 15(7):1943–1951
Yamamoto S, Fukagawa M (2017) Uremic toxicity and bone in CKD. J Nephrol 30(5):623–627
Tartaglione L, Pasquali M, Rotondi S, Muci ML, Covic A, Mazzaferro S (2017) Positioning novel biologicals in CKD-mineral and bone disorders. J Nephrol 30(5):689–699
Tentori F, Blayney MJ, Albert JM et al (2008) Mortality risk for dialysis patients with different levels of serum calcium, phosphorus, and PTH: the Dialysis Outcomes and Practice Patterns Study (DOPPS). Am J Kidney Dis 52:519–530
Mazzaferro S, Tartaglione L, Rotondi S, Bover J, Goldsmith D, Pasquali M (2014) News on biomarkers in CKD-MBD. Semin Nephrol 34(6):598–611
Block GA, Klassen PS, Lazarus JM et al (2004) Mineral metabolism, mortality, and morbidity in maintenance hemodialysis. J Am Soc Nephrol 15:2208–2218
Fernández-Martín JL, Martínez-Camblor P, Dionisi MP, COSMOS group et al (2015) Improvement of mineral and bone metabolism markers is associated with better survival in haemodialysis patients: the COSMOS study. Nephrol Dial Transplant 30(9):1542–1551
Mazzaferro S, Brancaccio D, MessaP,et al on behalf of the FARO Study Group (2011) Management of secondary hyperparathyroidism in Italy: results of the Italian FARO survey. J Nephrol 24(02):225–235
Gallieni M, De Luca N, Santoro D et al (2016) Management of CKD-MBD in non-dialysispatients under regular nephrology care: a prospectivemulticenterstudy. J Nephrol 29(1):71–78
Minuto M, Vera L, Ansaldo GL et al (2014) Modernapproach to parathyroidectomy. G Ital Nefrol 31(6):1–18
Mazzaferro S, Pasquali M, Farcomeni A et al (2008) Parathyroidectomy as a therapeutic tool for targeting the recommended NKF-K/DOQI ranges for serum calcium, phosphate and parathyroid hormone in dialysis patients. Nephrol Dial Transplant 23(7):2319–2323
Kovacevic B, Ignjatovic M, Zivaljevic V et al (2012) Parathyroidectomy for the attainment of NKF-K/DOQI™ and KDIGO recommended values for bone and mineral metabolism in dialysis patients with uncontrollable secondary hyperparathyroidism.Langenbecks Arch Surg. 397(3):413–420
Wetmore JB, Liu J, Dluzniewski PJ, Ishani A, Block GA, Collins AJ (2016) Geographic variation of parathyroidectomy in patients receiving hemodialysis: a retrospective cohort analysis. BMC Surg 16(1):77
Kestenbaum B, Andress DL, Schwartz SM et al (2004) Survival following parathyroidectomy among United States dialysis patients. Kidney Int 66(5):2010–2016
Ivarsson KM, Akaberi S, Isaksson E et al (2015) The effect of parathyroidectomy on patient survival in secondary hyperparathyroidism. Nephrol Dial Transplant 30(12):2027–2033
Scialla JJ, WolfM (2015) When there will never be a randomized controlled trial. Kidney Int 88(2):220–222
Malberti F, Marcelli D, Conte F, Limido A, Spotti D, Locatelli F (2001) Parathyroidectomy in patients on renal replacement therapy: an epidemiologic study. J Am SocNephrol 12(6):1242–1248
National Kidney Foundation (2003) K/DOQI clinical practice guidelines for bone metabolism and disease in chronic kidney disease. Am J Kidney Dis 42(suppl 3):s1–s201
Farcomeni A (2008) A review of modern multiple hypothesis testing, with particular attention to the false discovery proportion. Stat Methods Med Res 17(4):347–388
Kestembaum B, Selinger SL, Gillen DL et al (2004) Parathyroidectomy rates among United States dialysis patients:1990–1999. Kidney Int 65:282–288
Akaberi S, Clyne N, Sterner G et al (2014) Temporal trends and risk factors for parathyroidectomy in the Swedish dialysis and transplant population—a nationwide, population-based study 1991–2009. BMC Nephrol 15:75
Kidney Disease: Improving Global Outcomes CKDMBDWG (2009) KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD). Kidney Int Suppl 76(S113):S1–S130
Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Update Work Group (2017) KDIGO 2017 clinical practice guideline update for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease–mineral and bone disorder (CKD-MBD). Kidney Int Suppl 7:1–59
Parfrey PS, Chertow GM, Block GA et al (2013) The clinical course of treated hyperparathyroidism among patients receiving hemodialysis and the effect of cinacalcet: the EVOLVE trial. J Clin Endocrinol Metab 98(12):4834–4844
Bellasi A, Morrone L, Mereu MC et al (2018) working group on CKD-MBD of the Italian Society of Nephrology. CKD-MBD management: what is the role of parathyroidectomy? Results from a nationwide survey in Italy. J Nephrol 31(4):585–591
upta A, Kallanbach LR, Zasuwa G et al (2000) Race is a major determinant of secondary hyperparathyroidism in uremic patients. J Am Soc Nephrol 11:330–334
Wermers RA, Khosla S, Atkinson EJ, Hodgson SF, O’Fallon WM, Meltron LJ (1997) The rise and fall of primary hyperparathyroidism: a population-based study in Rochester, Minnesota, 1965–1992. Ann Intern Med 126:433–440
Malluche HH, Monier-Faugere MC (1992) Risk of adynamic bone disease in dialyzed patients. Kidney Int Suppl38:s62–67
Bell S, Fletcher EH, Brady I, Looker HC, Levin D, Joss N et al (2015) End-stage renal disease and survival in people with diabetes: a national database linkage study. QJM 108(2):127–134
Yamamoto T, Ozono K, Miyauchi A et al (2001) Role of advanced glycation end products in adynamic bone disease in patients with diabetic nephropathy. Am J Kidney Int 38:s161–4
Iwasaki-Ishizuka Y, Yamato H, Nii-Kono T et al (2005) Downregulation of parathyroid hormone receptor gene expression and osteoblastic dysfunction associated with skeletal resistance to parathyroid hormone in a rat model of renal failure with low turnover bone. Nephrol Dial Transplant 20:1904–1911
Drüeke TB, Massy ZA (2016 Feb) Changing bone patterns with progression of chronic kidney disease. Kidney Int 89(2):289–302
de Oliveira RA, Barreto FC, Mendes M et al (2015) Peritoneal dialysis per se is a risk factor for sclerostin-associated adynamic bone disease. Kidney Int 87(5):1039–1045
Sato S, Ohta M, Kawaguchi Y et al (1995) Effects of parathyroidectomy on left ventricular mass in patients with hyperparathyroidism. Miner Electrolyte Metab 21:67–71
Rault R, Magnone M (1996) The effect of parathyroidectomy on hematocrit and erythropoietin dose in patients on hemodialysis. ASAIO J (Am Soc Artif Intern Organs 1992) 42(5):M901–M903
Yasunaga C, Nakamoto M, Matsuo K, Nishihara G, Yoshida T, Goya T (1999) Effects of aparathyroidectomy on the immune system and nutritional condition in chronic dialysis patients with secondary hyperparathyroidism. Am J Surg 178(4):332–336
Funding
This study was funded with an unrestricted grant by Amgen.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
SM received honoraria for Congress presentations by Vifor Fresenius; LT, CC, NDD, AP, MM, MF, MN, ML, MM, CG, SR and MP declare no CoI. MC gave lectures for Amgen, Abbvie, Shire, Vifor Fresenius, Baxter.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Statement of human rights
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Statement on welfare of animals
This article does not contain any studies with animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Mazzaferro, S., Tartaglione, L., Cascone, C. et al. Multicenter study on parathyroidectomy (PTX) in Italy: preliminary results. J Nephrol 31, 767–773 (2018). https://doi.org/10.1007/s40620-018-0527-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-018-0527-x