Abstract
Purpose
This study sought to identify the prefrontal cortex hemodynamic response that is dependent on cognitive performance in patients with bulimic disorders (BD), and investigate its association with personality characteristics.
Methods
Nineteen female patients with BD and 23 healthy women were recruited. Their personality characteristics related to eating disorders were examined using a self-reporting questionnaire, namely the eating disorder inventory-2 (EDI-2). Cerebral blood flow response in the prefrontal cortex during the digit span backward task (DSBT) was measured using near-infrared spectroscopy (NIRS). Change in oxygenated hemoglobin concentration (ΔoxyHb), obtained using NIRS, were used as an index of brain activity. Further, the relationship between prefrontal cortical activity and personality characteristics was investigated in patients with BD.
Results
The cognitive performance of patients with BD was significantly lower in the DSBT compared with healthy subjects. There was no difference between the groups in ΔoxyHb during the task. Task scores of patients with BD correlated with asceticism and perfectionism. Moreover, the asceticism score was negatively correlated with ΔoxyHb of the bilateral prefrontal cortex in patients with BD.
Conclusion
The results suggest that cognitive performance and brain activity induced during DSBT might be affected by asceticism in BD patients.
Level of evidence
III, case-control study.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bulimia nervosa (BN) and binge eating disorder (BED) are both defined in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) by regular and sustained binge eating episodes, with a lack of control over eating during the episode [1]. According to DSM-5, BN is characterized by self-evaluation that is unduly influenced by body shape and weight, focused on controlling weight by inappropriate compensatory behavior [2,3,4]. However, although BED may also occur due to overvaluation of shape and weight, behavior preventing weight gain is uncommon [2]. In both cases, the patients’ body weight may be normal or high. Since the subtype of disorders that a patient is diagnosed with may change over time, and because some patients may have a history of AN or may develop AN in the future [5], a standard approach for the treatment of eating disorders (ED) based on the “transdiagnostic” theory, which focuses on psychopathology beyond diagnosis, is used [5]. According to this theory, individuals may have some personality characteristics that may act as symptom maintenance factors for eating disorder (ED) [6, 7].
Personality characteristics in ED are often evaluated using the Eating Disorder Inventory-2 (EDI-2) which includes 11 characteristics, such as perfectionism and asceticism [8, 9]. For example, asceticism is defined as a tendency to pursue the ideal of self-sacrificing spirit, such as self-discipline, self-denial, self-restriction, and excessive control of physical needs [9, 10]. It has been reported that the high asceticism scores are positively correlated with the frequency of vomiting in the purging type of BD [11]. Personality characteristics influence lifestyle and methods of adaptation; they are individual-specific patterns that are closely related to thought, emotion and behavior. In addition to the personality characteristics, cognitive impairment in BD has attracted attention recently as a reason for the maladaptive behavior and symptom maintenance factors [12,13,14]. The prefrontal cortex (PFC) is responsible for cognitive functions such as working memory and executive function, which ensure appropriate judgments and selection for achieving an intended purpose [15]. Indeed, patients with AN show a higher tendency of perfectionism, resulting in lower scores in tasks that require cognitive flexibility. These patients also show a higher tendency toward social insecurity, with severely low weight, and higher scores in the verbal fluency task [16].
Near-infrared spectroscopy (NIRS) is a non-invasive neuroimaging method using near-infrared light, which allows the detection of slight changes in blood oxygenation induced by cognitive processing [17,18,19,20]. The results of NIRS can be helpful in the assessment of cortical activity related to cognitive function [21,22,23,24,25]. Some neuropsychological examinations using NIRS have been performed for patients with ED [16, 26,27,28,29,30]. For example, it has been reported that patients with AN have lower frontal cortical oxygenated hemoglobin concentration (oxyHb), but without performance deficits during cognitive tasks [29]. Uehara et al. concluded that hemoglobin concentration changes in the prefrontal cortex, occurring during a verbal task, are different in patients with ED (specifically AN and BN), relative to healthy subjects (HS) [30]. Furthermore, Sutoh et al. reported that patients with BD require strong prefrontal cortical activation to perform “difficult-to-lose” tasks with an accuracy equal to that of healthy controls [28]. In addition, they concluded that increased impulsivity and disinhibition (lack of control) are seen more frequently in BN patients compared with AN patients, and are manifested as episodes of eating behaviors [28].
Thus, these changes in factors such as among cognitive function, cognitive ability-dependent PFC hemodynamics, and personality characteristics, influence the other factors. However, compared to AN, little is known about the relationship among personality characteristics, cognitive function and brain activity in BD.
The aim of this study was to identify the hemodynamic PFC responses that are dependent on cognitive performance in BD, and investigate the association between cognitive performance and personality characteristics.
Methods
Subjects
Patients were recruited through the outpatient unit of Chiba University Hospital. HS were recruited via advertisements placed on a website and the hospital notice board. A psychiatrist assessed 31 HS and 24 patients with BD. Patients with a severe psychiatric comorbidity causing impairment of cognitive function, and neurological disorders, head injury, suicidal ideation, substance abuse, or body mass index (BMI) < 17 kg/m2 were excluded from this study. Among the 55 participants who entered this study, 12 were excluded due to incomplete responses to questionnaires (8 HS and 4 patients with BD). Therefore, the final analysis comprised of 23 HS and 19 patients with BD. All the participants were female. The age range was 18–39 years (HS, 27.8 ± 7.1 years; patients with BD, 29.2 ± 5.9 years; p = 0.50). Mean BMI for HS and patients with BD was 20.6 ± 1.8 kg/m2 and 20.3 ± 2.3 kg/m2, respectively (p = 0.61). Wechsler Adult Intelligence Scale-Revision was used for all subjects to match intelligence levels for the cognitive task. Intelligence quotient was 98.2 ± 14.9 for HS, and 91.9 ± 15.2 for patients with BD (p = 0.19). Years of education were 14.4 ± 1.6 years for HS, and 14.6 ± 2.2 years for patients with BD (p = 0.75). All patients were diagnosed with BD or BED by a psychiatrist familiar with ED based on fulfillment of the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision criteria (American Psychiatric Association, 2000) for BD.
Among the 19 patients with BD, 17 were diagnosed with BN, and 2 with BED. Regarding psychiatric comorbidities, 2 patients exhibited mood disorders, and 2 exhibited mild depression. Out of these 4 patients, 2 were being administered an antidepressant, 1 an anxiolytic and 1 an antiepileptic by their primary care doctors. All but 4 of the subjects were right-handed (2 patients and 2 HS), as confirmed by the Edinburgh Handedness Inventory [31].
Design and procedure
The clinical symptoms of BD and each subjects’ personality characteristics were examined in advance using self-reported questionnaires: namely, the Bulimic Investigatory Test, Edinburgh (BITE) [32] for assessment of the severity of bulimic symptoms, Hospital Anxiety and Depression Scale [33], for evaluation of anxiety and depression, and EDI-2 [8, 9] for assessment of psychopathological characteristics. A psychiatrist familiar with ED assessed the patients, using a structured interview, Eating Disorder Examination version 16.0 [34]. Prefrontal cortical activity was individually evaluated by measuring changes in concentration of oxyHb (ΔoxyHb) using NIRS, during performance of the digit span backward task (DSBT), as described below.
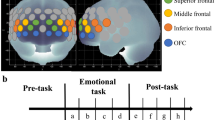
The subjects were instructed to sit in a comfortable chair in the experimental room. After the task instructions were given, an NIRS device was fitted on their forehead. The subjects were instructed to focus their gaze on the monitor screen to perform the task according to the visual cues. On the monitor screen, numbers were presented individually every second. Once the series ended, a blank screen appeared, on which the subjects were required to recite the numbers in the reverse order. The time allotted for answering one question was 10 s. The screen automatically proceeded to the next question regardless of whether the subject gave the correct or incorrect answer (Fig. 1b). The subject continued this procedure through the task period (60 s). Six questions were asked. The task consisted of a 40-s pre-task period, a 60-s DSBT period and a 60-s post-task period. The subjects were required to repeat “1, 2, 3…” during the pre- and post-task period as a baseline (and recovery) values.
a Position of near-infrared spectroscopy (NIRS) probes. Changes in oxygenated hemoglobin (oxyHb) were measured using a 16-channel NIRS machine (Spectratech OEG-16) at two wavelengths of near-infrared light (780 and 830 nm). Absorption of near-infrared light was measured and oxyHb calculated. b Task procedure
Cognitive task and NIRS measurements
The DSBT is associated with executive function and working memory, and is a reliable task used in the Japanese version of the WAIS-R (Wechsler, 1981) [35,36,37]. The DSBT requires transformation to reorder the input digits in a reversed sequence to generate the output. Due to the difficulty of the task, this executive process is considered relatively “heavier” than other tasks such as the digit span forward task (DSFT) [38, 39]. DSBT performance is impaired in the case of a frontal lobe lesion, concordant with evidence from NIRS that DSBT activates bilateral dorsolateral PFC to a greater extent than DSFT in HS [17, 18]. Accordingly, this burdensome task is more suitable for our current study.
A 16-channel optical topography system (OEG-16; Spectratech Inc., Tokyo, Japan) with 6 light emission and 6 light detection probes, was used for NIRS recordings during DSBT. The deoxygenated hemoglobin concentrations were sampled at 1.5 Hz (approximately 0.67 s/sample). Channels 8 and 9 were placed on the top and bottom of Fpz, according to the International 10–20 system used in electroencephalography. Detection and emission probes were placed at 3-cm intervals of each other (Fig. 1a).
Data analysis
Near-infrared spectroscopy (NIRS) data consisted of 160-s measurement durations. The duration of each section was 40 s for the baseline, 60 s for the task, and 60 s for the post-task. Data analysis was performed using custom-made software working on MATLAB R2013a (Mathworks, Natick, MA, USA). A high-pass filter with a 0.13-Hz discrete cosine transform and low-pass filter using a 51-point moving average were applied. After filtering, conversion to z values was performed using the baseline average [19, 20]. Average z values for the ΔoxyHb for each section (baseline, task, and post-task) were calculated for bilateral foreheads of each subject. The ΔoxyHb in bilateral dorsolateral PFC was averaged across channels 2 to 6 (right forehead) and channels 11 to 15 (left forehead). Taking advantage of a higher signal-to-noise ratio, the time axis was divided into 9 sections: 1 section for baseline, and 8 sections for task and post-task, which were divided into 15-s periods.
Statistical analysis
The values obtained from patients with BD and HS were compared using Student’s t test, controlling for demographic parameters. For the NIRS data, two-way repeated-measures analysis of variance (ANOVA), followed by Bonferroni post-hoc test, was used to compare the differences between the two groups. In addition, correlation coefficients between two variables (Spearman’s rs) were used to determine the correlation among peak ΔoxyHb values, task scores, and each psychopathological score. Finally, multiple regression analysis was performed for two groups to identify the best predictors of ΔoxyHb, using ΔoxyHb as the dependent variable and all EDI-2 subscale scores as independent variables. All statistical analyses were performed using SPSS version 21 (IBM Corp., Armonk, NY, USA), and the statistical significance level was set at p < 0.05 (two-tailed tests).
Results
Demographic analyses and clinical characteristics
Mean values (M), standard deviation (SD) and p values (Student’s t test) for all demographic parameters are presented in Table 1. As shown in Table 1, the BITE symptom score and severity scale were significantly higher for patients with BD (symptom scale 21.3 ± 5.5; severity scale 11.2 ± 5.5) compared with HS (symptom scale 5.3 ± 3.7, p < 0.001; severity scale 1.4 ± 1.1, p < 0.001). Anxiety score and depression scores (Table 2) were significantly higher for patients with BD (anxiety 8.8 ± 4.4; depression 9.3 ± 5.2) compared with HS (anxiety 4.9 ± 3.4, p < 0.001; depression 3.4 ± 2.8, p < 0.001). No difference was observed in “maturity fear” on the EDI-2 subscale between HS and BD patients (HS 8.7 ± 3.6; BD patients 10.0 ± 4.9; p = 0.353). However, all other subscale scores were significantly higher for BD patients than for HS.
Task performance
The number of correct answers in the DSBT for BD patients (M = 3.2, SD = 1.2) was significantly lower compared with HS (M = 4.1, SD = 1.0, p = 0.009) (Table 2).
The ΔoxyHb in PFC during DSBT
Compared with baseline values, significant increases in oxyHb were observed for both groups during DSBT. However, BD patients exhibited a more gradual increase in oxyHb than HS at the beginning of the task period, which continued to gradually increase until the beginning of the post-task period. The ΔoxyHb during the task showed a tendency to be higher for BD patients than for HS. However, this difference was not statistically significant.
Figure 2 shows the raw NIRS data for ΔoxyHb. The ΔoxyHb of bilateral PFC was notably increased for both groups during the task, in comparison with baseline (pre-task). However, two-way repeated measures ANOVA showed no significant difference in ΔoxyHb between groups at any point during the task (F (1,39) = 0.03, p = 0.864). Peak value of ΔoxyHb and the time to peak also showed no correlation between groups.
Correlation of prefrontal cortical activity with DSBT performance and personality characteristics
The number of correct answers was lower for BD patients than for HS. Further, there was no difference in ΔoxyHb between groups during DSBT (Table 2). The DSBT scores of BD patients correlated with the EDI-2 subscales, asceticism (rs = 0.513, p = 0.025) and perfectionism (rs = 0.514, p = 0.025) (Fig. 3). No other correlation was detected between DSBT performance and EDI-2 personality characteristics. The asceticism score negatively correlated with ΔoxyHb of the bilateral PFC for BD patients (left PFC, rs = − 0.506, p = 0.027; right PFC, rs = − 0.542, p = 0.016) (Fig. 4).
Multiple regression analysis for the bilateral PFC showed no significant differences between HS and BD patients, however, ΔoxyHb in the bilateral PFC of BD patients during DSBT, negatively correlated with asceticism score (left: β = − 0.57, p = 0.01; right: β = − 0.911, p = 0.001) (Table 3). However, asceticism of HS showed no association with ΔoxyHb (asceticism: left PFC, rs = − 0.045, p = 0.839; right PFC, rs = − 0.236, p = 0.279). (DOI for the above data is https://doi.org/10.6084/m9.figshare.5398015).
Discussion
To our knowledge, this is the first study, using NIRS, in BD patients to evaluate the association between personality characteristics and prefrontal cortical activity during a cognitive task. In relation to cognitive performance and personality characteristics of BD patients, we investigated ΔoxyHb in PFC during DSBT and compared with HS. During DSBT, both groups exhibited greater activation of the bilateral PFC. However, no difference in PFC activation was observed between the two groups. BD patients exhibited lower DSBT performance than HS, and performance correlated positively with asceticism that is a subscale of EDI-2. Moreover, asceticism negatively correlated with bilateral PFC activation of BD patients but not with that of HS.
Greater PFC activation during DSBT
In this study, we observed a significant increase from baseline value in oxyHb during DSBT, for each group. While the difference in oxyHb between the two groups did not reach statistical significance, a distinct increase in oxyHb was observed during the first half of the task. In HS, oxyHb reached a peak by the first half of the task, and although the increase in oxyHb of BD patients was initially comparable with HS, it continued to increase beyond the HS peak, and peaked in the latter half of the task. However, the overall magnitude of ΔoxyHb was not significantly different between two groups. Generally, HS exhibit a larger activation than ED patients, and HS exhibit an initial rapid increase followed by a gradual decrease in oxygenated- and total-Hb [30]. Conversely, patients with ED exhibit a smaller activation than HS, and a gradual elevation of oxygenated-Hb [30]. Our results are in accordance with a reported ΔoxyHb pattern in HS showing an initial rapid increase, followed by a gradual decrease [30]. Oxygenation changes in patients with ED are characterized by a lower and more gradual increase in regional cerebral blood volume [29]. This may be explained by attempts by ED patients to reduce metabolism to conserve energy [30]. However, in our study, oxyHb of BD patients was not lower compared with HS, rather it tended to be higher, which is not consistent with previous results [30]. Regarding the subjects of the study of Uehara et al., the number of AN was larger than BN. Therefore, BMI of the participants in their study was lower than participants in our study. This may explain, in part, our differing results.
Association among DSBT performance, asceticism, and bilateral PFC activation
We found a significantly higher asceticism score in patients with BD, compared with HS, that positively correlated with DSBT patient performance. Moreover, asceticism scores of BD patients were the best predictor of ΔoxyHb in the bilateral PFC, based on multiple regression analysis. Therefore, patients with higher asceticism scores exhibited lower ΔoxyHb during DSBT. Since 1991, asceticism has been one of three new EDI-2 subscales, along with impulse regulation and social insecurity. Schoemaker et al. [40] suggested that the asceticism scale is valuable for distinguishing BD patients from general psychiatric outpatients. The EDI-2 includes specific questions for the assessment of asceticism that represent idealistic desires for self-realization, such as “I do not like being weak”, “It can be good to suffer”, and “I want to control everything”. Asceticism indicates a will “to train” or “to discipline” oneself for achieving certain purposes by suppressing desires and/or feelings. Garner et al. mention that asceticism measures the tendency to pursue spiritual ideals such as self-discipline, self-denial, self-limitation, hypercontrol over bodily needs, and self-sacrifice [8,9,10]. As mentioned earlier, patients with BD have a strong desire to lose weight, which is the pursuit of an ideal; consequently, asceticism may manifest strongly. Moreover, Sassaroli et al. [41] indicate that the patients with ED are more concerned about making mistakes than patients with other psychiatric disorders (such as obsessive–compulsive disorder or major depressive disorder). Consequently, these conditions may lead to maintenance of aroused consciousness.
Limitations
Our study has some limitations that should be noted. The number of subjects examined was small. We targeted only BN in this study and did not include AN patients. Furthermore, we did not control for the presence of other axis I and axis II disorders, such as anxiety, depression, and personality disorders, or for the effects of pharmacological interventions.
Some NIRS studies have raised concerns about skin blood flow [42, 43]. The results of NIRS may be influenced by scalp and skin blood flow, as near-infrared light must pass through these tissues to reach the brain [44]. We have not addressed this issue in our study, and cannot eliminate the possibility that our results might be affected by skin blood flow.
Although our results may be considered less reliable due to the limitations noted above, we consider the identification of personality characteristics of ED from a transdiagnostic view rather than a subscale classification to be clinically useful. Our findings showed that personality characteristics might be associated with cognitive function and ΔoxyHb of the prefrontal cortex.
Conclusions
In conclusion, our results show that ΔoxyHb in PFC shows no difference between groups, although cognitive performance in a relatively challenging task was lower for BD patients than HS. This difference might be mere fluctuation within an error range. However, a higher tendency toward asceticism correlated with a smaller task-induced increment in oxyHb.
Our results suggest that the values of cognitive function and cognitive ability-dependent PFC hemodynamics may be influenced by asceticism values. This study might lead to a better understanding of BD in terms of cognitive function and personality traits, and may also help develop improved therapeutic approaches. Further studies with a larger number of subjects are needed for ensuring the results of this study.
References
American Psychiatric Association (2013) Diagnostic and statistical manual of mental disorders, DSM-5. American Psychiatric Publishing, Arlington, VA
Hay P, Chinn D, Forbes D, Madden S, Newton R, Sugenor L, Touyz S, Ward W, Royal Australian and New Zealand CoP (2014) Royal Australian and New Zealand College of Psychiatrists clinical practice guidelines for the treatment of eating disorders. Aust N Z J Psychiatry 48(11):977–1008. https://doi.org/10.1177/0004867414555814
Smink FR, Hoeken D, Oldehinkel AJ, Hoek HW (2014) Prevalence and severity of DSM-5 eating disorders in a community cohort of adolescents. Int J Eat Disord 47(6):610–619. https://doi.org/10.1002/eat.22316
Thomas JJ, Eddy KT, Murray HB, Tromp MD, Hartmann AS, Stone MT, Levendusky PG, Becker AE (2015) The impact of revised DSM-5 criteria on the relative distribution and inter-rater reliability of eating disorder diagnoses in a residential treatment setting. Psychiatry Res 229(1–2):517–523. https://doi.org/10.1016/j.psychres.2015.06.017
Fairburn CG, Harrison PJ (2003) Eating disorders. Lancet 361(9355):407–416. https://doi.org/10.1016/s0140-6736(03)12378-1
Treasure J, Schmidt U (2013) The cognitive-interpersonal maintenance model of anorexia nervosa revisited: a summary of the evidence for cognitive, socio-emotional and interpersonal predisposing and perpetuating factors. J Eat Disord. https://doi.org/10.1186/2050-2974-1-13
Fairburn CG, Cooper Z, Shafran R (2003) Cognitive behaviour therapy for eating disorders: a “transdiagnostic” theory and treatment. Behaviour research therapy 41(5):509–528. https://doi.org/10.1016/s0005-7967(02)00088-8
Garner DM, Olmstead MP, Polivy J (1983) Development and validation of a multidimensional eating disorder inventory for anorexia nervosa and bulimia. Int J Eat Disord 2(2):15–34. https://doi.org/10.1002/1098-108X(198321)2:23.0.CO;2-6
Garner DM (1991) Eating disorder inventory-2. Psychological Assessment Resources, Odessa, FI, USA
Fassino S, Piero A, Gramaglia C, Daga GA, Gandione M, Rovera GG, Bartocci G (2006) Clinical, psychological, and personality correlates of asceticism in anorexia nervosa: from saint anorexia to pathologic perfectionism. Transcult Psychiatry 43(4):600–614. https://doi.org/10.1177/1363461506070785
Abbate-Daga G, Piero A, Gramaglia C, Fassino S (2005) Factors related to severity of vomiting behaviors in bulimia nervosa. Psychiatry Res 134(1):75–84. https://doi.org/10.1016/j.psychres.2004.01.013
Kaye W (2008) Neurobiology of anorexia and bulimia nervosa. Physiol Behav 94(1):121–135. https://doi.org/10.1016/j.physbeh.2007.11.037
Kaye WH, Fudge JL, Paulus M (2009) New insights into symptoms and neurocircuit function of anorexia nervosa. Nat Rev Neurosci 10(8):573–584. https://doi.org/10.1038/nrn2682
Iglesias MobbsO.Golay. K, Van der Linden A. M (2011) Cognitive deficits in obese persons with and without binge eating disorder. Investigation using a mental flexibility task. Appetite 57(1):263–271. https://doi.org/10.1016/j.appet.2011.04.023
Satomura Y, Takizawa R, Koike S, Kawasaki S, Kinoshita A, Sakakibara E, Nishimura Y, Kasai K (2014) Potential biomarker of subjective quality of life: prefrontal activation measurement by near-infrared spectroscopy. Soc Neurosci 9(1):63–73. https://doi.org/10.1080/17470919.2013.861359
Katayama H, Kohmura K, Tanaka S, Imaeda M, Kawano N, Noda Y, Nishioka K, Ando M, Aleksic B, Iidaka T, Ozaki N (2014) Social insecurity in relation to orbitofrontal activity in patients with eating disorders: a near-infrared spectroscopy study. BMC Psychiatry. https://doi.org/10.1186/1471-244X-14-173
Hoshi Y, Oda I, Wada Y, Ito Y, Yamashita Y, Oda M, Ohta K, Yamada Y, Tamura M (2000) Visuospatial imagery is a fruitful strategy for the digit span backward task: a study with near-infrared optical tomography. Cogn Brain Res 9(3):339–342. https://doi.org/10.1016/S0926-6410(00)00006-9
Gerton BK, Brown TT, Meyer-Lindenberg A, Kohn P, Holt JL, Olsen RK, Berman KF (2004) Shared and distinct neurophysiological components of the digits forward and backward tasks as revealed by functional neuroimaging. Neuropsychologia 42(13):1781–1787. https://doi.org/10.1016/j.neuropsychologia.2004.04.023
Takeda K, Gomi Y, Imai I, Shimoda N, Hiwatari M, Kato H (2007) Shift of motor activation areas during recovery from hemiparesis after cerebral infarction: a longitudinal study with near-infrared spectroscopy. Neurosci Res 59(2):136–144. https://doi.org/10.1016/j.neures.2007.06.1466
Takeda K, Gomi Y, Kato H (2014) Near-infrared spectroscopy and motor lateralization after stroke: a case series study. Int J Phys Med Rehabil 2(3):192. https://doi.org/10.4172/2329-9096.1000192
Doi H, Nishitani S, Shinohara K (2013) NIRS as a tool for assaying emotional function in the prefrontal cortex. Front Hum Neurosci 7:770. https://doi.org/10.3389/fnhum.2013.00770
Herrmann MJ, Ehlis AC, Fallgatter AJ (2003) Frontal activation during a verbal-fluency task as measured by near-infrared spectroscopy. Brain Res Bull 61(1):51–56. https://doi.org/10.1016/s0361-9230(03)00066-2
Herrmann MJ, Ehlis AC, Fallgatter AJ (2003) Prefrontal activation through task requirements of emotional induction measured with NIRS. Biol Psychol 64(3):255–263. https://doi.org/10.1016/s0301-0511(03)00095-4
Fallgatter AJ, Strik WK (1998) Frontal brain activation during the Wisconsin Card Sorting Test assessed with two-channel near-infrared spectroscopy. Eur Arch Psychiatry Clin Neurosci 248(5):245–249. https://doi.org/10.1007/s004060050045
Usami M, Iwadare Y, Kodaira M, Watanabe K, Saito K (2014) Near infrared spectroscopy study of the frontopolar hemodynamic response and depressive mood in children with major depressive disorder: a pilot study. PLoS One 9(1):e86290. https://doi.org/10.1371/journal.pone.0086290
Matsumoto R, Kitabayashi Y, Narumoto J, Wada Y, Okamoto A, Ushijima Y, Yokoyama C, Yamashita T, Takahashi H, Yasuno F, Suhara T, Fukui K (2006) Regional cerebral blood flow changes associated with interoceptive awareness in the recovery process of anorexia nervosa. Prog Neuropsychopharmacol Biol Psychiatry 30(7):1265–1270. https://doi.org/10.1016/j.pnpbp.2006.03.042
Sutoh C, Koga Y, Kimura H, Kanahara N, Numata N, Hirano Y, Matsuzawa D, Iyo M, Nakazato M, Shimizu E (2015) Repetitive transcranial magnetic stimulation changes cerebral oxygenation on the left dorsolateral prefrontal cortex in bulimia nervosa: a near-infrared spectroscopy pilot study. Eur Eat Disord Rev 24(1):83–88. https://doi.org/10.1002/erv.2413
Sutoh C, Nakazato M, Matsuzawa D, Tsuru K, Niitsu T, Iyo M, Shimizu E (2013) Changes in self-regulation-related prefrontal activities in eating disorders: a near infrared spectroscopy study. PLoS One 8(3):e59324. https://doi.org/10.1371/journal.pone.0059324
Suda M, Uehara T, Fukuda M, Sato T, Kameyama M, Mikuni M (2010) Dieting tendency and eating behavior problems in eating disorder correlate with right frontotemporal and left orbitofrontal cortex: a near-infrared spectroscopy study. J Psychiatr Res 44(8):547–555. https://doi.org/10.1016/j.jpsychires.2009.11.005
Uehara T, Fukuda M, Suda M, Ito M, Suto T, Kameyama M, Yamagishi Y, Mikuni M (2007) Cerebral blood volume changes in patients with eating disorders during word fluency: a preliminary study using multi-channel near infrared spectroscopy. Eat Weight Disord 12(4):183–190. https://doi.org/10.1007/BF03327596
Oldfiel RC (1971) The assessment and analysis of handedness: the edinburgh inventory. Neuropsychologia 9(1):97–113. https://doi.org/10.1016/0028-3932(71)90067-4
Henderson M, Freeman CP (1987) A self-rating scale for bulimia. The ‘BITE’. Br J Psychiatry 150(1):18–24. https://doi.org/10.1192/bjp.150.1.18
Zigmond AS, Snaith RP (1986) The hospital anxiety and depression scale. Acta Psychiatr Scand 67(6):361–370. https://doi.org/10.1111/j.1600-0447.1983.tb09716.x
Fairburn CG (2008) Eating disorder examination (16.0 D). Guilford Press, New York
Kessels R, van den Berg E, Ruis C, Brands A (2008) The backward span of the corsi block-tapping task and its association with the WAIS-III digit span. Assessment 15(4):426–434. https://doi.org/10.1177/1073191108315611
Saggino A, Balsamo M, Grieco A, Cerbone MR, Raviele NN (2004) Corsi’s block-tapping task: standardization and location in factor space with the WAIS-R for two normal samples of older adults. Percept Mot Skills 98(3):840–848. https://doi.org/10.2466/pms.98.3.840-848
Wilde N, Strauss E (2002) Functional equivalence of WAIS-III/WMS-III digit and spatial span under forward and backward recall conditions. Clin Neuropsychol 16(3):322–330. https://doi.org/10.1076/clin.16.3.322.13858
Jones G, Macken B (2015) Questioning short-term memory and its measurement: why digit span measures long-term associative learning. Cognition 144:1–13. https://doi.org/10.1016/j.cognition.2015.07.009
Kaneko H, Yoshikawa T, Nomura K, Ito H, Yamauchi H, Ogura M, Honjo S (2011) Hemodynamic changes in the prefrontal cortex during digit span task: a near-infrared spectroscopy study. Neuropsychobiology 63(2):59–65. https://doi.org/10.1159/000323446
Schoemaker C, Verbraak M, Breteler R, Van der Staak C (1997) The discriminant validity of the eating disorder inventory-2. Br J Clin Psychol 36:627–629. https://doi.org/10.1111/j.2044-8260.1997.tb01268.x
Sassaroli S, Lauro LJ, Ruggiero GM, Mauri MC, Vinai P, Frost R (2008) Perfectionism in depression, obsessive–compulsive disorder and eating disorders. Behav Res Ther 46(6):757–765. https://doi.org/10.1016/j.brat.2008.02.007
Takahashi T, Takikawa Y, Kawagoe R, Shibuya S, Iwano T, Kitazawa S (2011) Influence of skin blood flow on near-infrared spectroscopy signals measured on the forehead during a verbal fluency task. NeuroImage 57(3):991–1002. https://doi.org/10.1016/j.neuroimage.2011.05.012
Kohno S, Miyai I, Seiyama A, Oda I, Ishikawa A, Tsuneishi S, Amita T, Shimizu K (2007) Removal of the skin blood flow artifact in functional near-infrared spectroscopic imaging data through independent component analysis. J Biomed Opt 12(6):062111. https://doi.org/10.1117/1.2814249
Jobsis FF (1977) Noninvasive, infrared monitoring of cerebral and myocardial oxygen sufficiency and circulatory parameters. Science 198(4323):1264–1267. https://doi.org/10.1126/science.929199
Acknowledgements
The study was supported by grants from JSPS KAKENHI (Grant numbers JP25461750 and JP15H05359). This work was also supported by the Academic Contributions from Pfizer Japan Inc.
Author information
Authors and Affiliations
Contributions
NN, SC, and YH designed the project and DM and KT prepared and provided the NIRS data. NN performed the experiment and collected the data. YH and NN analyzed the data. NN and YH drafted the manuscript. MN, ES, and RS supervised the study.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Research involving human participants
All procedures performed for experiments involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The Institutional Ethics Committee of the Chiba University Graduate School of Medicine approved this study.
Informed consent
Informed consent was obtained from all participants of this study.
Rights and permissions
About this article
Cite this article
Numata, N., Hirano, Y., Sutoh, C. et al. Hemodynamic responses in prefrontal cortex and personality characteristics in patients with bulimic disorders: a near-infrared spectroscopy study. Eat Weight Disord 25, 59–67 (2020). https://doi.org/10.1007/s40519-018-0500-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40519-018-0500-7