Abstract
Purpose
The follow-up of anthropometric percentiles such as triceps and mid-thigh skinfold thickness (TSF, MTSF), mid-upper arm and mid-thigh circumferences (MUAC, MTC), and arm and mid-thigh muscle areas (AMA, MTMA) after a resistance training might allow for detecting nutritional progress of fat and muscular tissue during the treatment of anorexia nervosa restricting (AN-R) type patients.
Methods
A total of 44 AN-R patients were randomized for control (CG 13.0 ± 0.6 years) and intervention (IG 12.7 ± 0.7 years) groups after hospitalization. The intervention group underwent a resistance training program of 8 weeks following the guidelines for healthy adolescents (3 days/week; 70 % of 6 RM). All measurements were obtained prior to starting the program (PRE) and after 8 weeks of training (POST) in both groups. TSF, MTSF, MUAC, and MTC were measured, and AMA and MTMA were calculated. Data were matched with percentile tables for general population. Changes were assessed using statistical tests for categorical data.
Results
The distribution of percentile categories within the groups did not differ statistically after 8 weeks (p > 0.05). After training, 73 % of the patients were at the same percentile interval of MUAC, 18 % higher and 9 % lower, while 30 % of CG was at lower percentile categories. Further, 54 % of the IG patients remained at the same percentile interval of MTC after training, and 36 % higher, while 20 % were at lower categories in the CG. The AMA increased (32 %) after training or remained at the same interval (59 %) in the IG, while the IG showed greater frequency of percentile decreases (45 %).
Conclusions
Anthropometric measurements could be useful for assessing muscle status in AN-R patients during the treatment process. However, exact standard deviation scores should be used instead of percentile categories to increase the sensitivity to changes in TSF, MTSF, MUAC, MTC or AMA.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Severe malnutrition and weight loss are considered as core elements of anorexia nervosa (AN) [1]. Despite the fact that restoration of fat and muscle tissues is critical in the process of cure, weight and body mass index (BMI) are commonly used as criteria to assess the nutritional status [2, 3]. In this sense, weight and BMI percentiles have been used to set up cutoff points of healthy nutritional status [4–6], but neither fat mass nor muscle mass gains are distinguished in the process. The control of fat mass gains seems to be critical in the short and long term owing to its relationship to increased dissatisfaction with self-body image and increased return to restrictive eating behaviors [7]. In a clinical environment, skinfold thicknesses, circumferences, and muscle areas might provide advantage as a predictor of fat tissue and muscle tissue recovery. These anthropometric measurements might allow for recognition of nutritional progress of both fat and muscular tissue during different treatment protocols [1].
Previous research includes studies that analyze the effect of the inclusion of physical activity and specific training programs in psychological and physiological outcomes as a part of the treatment process. The inclusion of AN-R patients in recreational activities and training programs was associated with improvements in self-well-being and self-image in AN patients [8, 9], improvements in aerobic capacity and muscular strength [10], reduction in the emotional stress [11], improvements in the peak force of the upper and lower body [12], and a reduction of disorder symptoms [13]. Overall, exercise programs included mainly flexibility exercises [13, 14], and low-intensity resistance exercises as a core of the sessions [12, 14, 15]. Consistently, training programs studies have focused on their effects in the body weight, BMI, percentage of fat, and fat mass recovery [11–13, 15, 16], reporting improvements in BMI, body fat percentage, or body weight [9, 14]. Although AN-R results in an impaired physical fitness [17], such as reduced muscle mass (15–45 % of the total body weight) [18] and a reduced ability to generate force [17], to our knowledge, there is a lack of studies that analyze the effect of exercise programs or physical activity programs in specific muscle areas, segment-related muscle function, segment circumferences, skinfold thicknesses, and/or their percentiles in AN-R patients. Interestingly, weight restoration studies analyzed the effect of re-feeding programs on anthropometric variables (skinfold thicknesses, circumferences, or muscle areas) to assess nutritional status. Those studies reported gains in BMI associated with gains in skinfold thicknesses or segment circumferences [1, 7, 19–21]. However, none of these studies was aimed to analyze muscle mass-related variables during recovery.
The aim of this study was to detect changes in body composition in two groups of anorexia nervosa restricting type (AN-R) patients using the percentiles of triceps skinfold (TSF), mid-thigh skinfold (MTSF), mid-upper arm circumference (MUAC), mid-thigh circumference (MTC), and arm muscle area (AMA).
Materials and methods
Ethics and sample
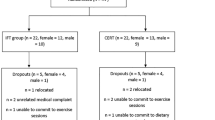
A total of 44 Caucasian female patients in treatment at the Unit of Psychiatry and Psychology of the Pediatric Hospital Niño Jesus (Madrid, Spain) were contacted and accepted to participate in the study. The parental written consent form was obtained and the Hospital Ethics Committee approved the intervention protocol (ID Ethics Committee: R-0034/08; January 2009). Assuming from the literature that medium effect sizes would be expected, a total sample size of 34 patients was calculated with a power of 80 % and a significance level of 0.05. To prevent the possible attrition, the final sample was increased to 22 patients in each group. Forty-four Caucasian female patients were randomly allocated on the basis of a coin toss into intervention and control groups (n = 22 each). Forty-two (IG: n = 22, CG: n = 20) patients completed the study. Two patients from the CG who declined to continue after a period of 4 weeks were not included in the analysis. The intervention was carried out in accordance with the ethical standards on human experimentation of the Declaration of Helsinki of 1975, as revised in 2008. The inclusion and exclusion criteria in the study were as follows: AN-R diagnosed using the International Classification of Diseases 10 (ICD-10) F50.0 [22]; age = 12–14 years; attend to a daily care program; body mass index (BMI) >14.0 kg/m2; <6 h/week of physical activity upon admission [23]; have no contraindications to perform exercise (e.g., cardiovascular or orthopedic) [24]. Participants were randomly allocated to intervention (IG) and control (CG) groups (n = 22 each). Continuance criteria were added to ensure the patients’ safety during the study [24]. Energy intake consisted of 55 % carbohydrate, 30 % protein, and 15 % fat, and the daily calorie intake was 1,800–2,500 kcal/day depending on the patient.
Assessment
The patients completed a familiarization period of three sessions in 1 week. A six repetition maximum (6 RM) test of bench press, leg press, and lateral row was performed following this period [15]. Anthropometric assessments were performed before (PRE) and after (POST) the intervention of 8 weeks following the International Standards for the Advancement of the Kinanthropometry (ISAK) [25], and were carried out by a certified technician (ISAK level 3). BMI was calculated as weight/height2 (kg/m2). TSF (mm), MTSF (mm), MUAC (cm), and MTC (cm) were measured on the dominant side of the body at appropriately marked sites using a Harpenden caliper (British Indicators Ltd., St Albans, Hertfordshire, UK) and Lufkin Executive W606P tape (Cooper Industries, USA), respectively [25]. AMA (cm2) and mid-thigh muscle area (MTMA) (cm2) were calculated using Heymsfield’s equations [26]. The assessor was involved in anthropometric measurements only and was blinded to the group allocation. The trainer was involved with intervention group patients only, just for the application of resistance training program.
Training program
The training program was based on the National Strength and Conditioning Association (NSCA) guidelines for adolescents [27]. Patients from the IG participated in a supervised training program performed over 8 weeks, 3 days per week, of lateral row, bench press, leg press, leg extension, lateral pull-down, abdominal crunch, low back extension, and push-ups, whereas the CG did not. The instructor-to-patient ratio was 1:2 for every training session. Each session was performed before psychological therapy, lapsed 50–60 min, and started and ended with warm-up and cool-down. The patients performed 3 sets of 8–10 repetitions with resting periods of 1–2 min starting at 70 % of 6 RM. We increased the load gradually 5–10 % as participants’ strength increased, up to a 100 % of 6 RM at the end of the program [27].
Statistical analyses
Means and standard deviations of age, days lapsed from admission to enrollment, lowest weight and height at hospitalization, and initial weight, height and BMI, were obtained. Percentiles of TSF for age, MTSF for age, MUAC for age, MTC for age, and AMA for age were estimated prior and after the intervention period using reference tables. Intervals of percentiles were designated using the values provided in the references as upper limit for each interval. Chi squared and Fisher exact tests of association between PRE and POST and percentile categories were performed. The individual change in raw data was also inspected sorting the participants of each group by each variable and comparing the PRE and POST measurements in figures. Mid-thigh muscle area (MTMA) values were also analyzed despite the lack of percentile reference values. Student’s T tests were used to compare CG and IG prior to start the study and to compare the percentage of increase in the mentioned six anthropometric variables. Cohen’s d was used to test standardized effect sizes, taking reference values of small (d = 0.2), medium (d = 0.5) and large (d = 0.8) from Cohen [28]. The significance level was set to p < 0.05.
Results
No statistically significant differences were found between CG and IG in any of the assessments performed prior to the intervention. As shown in Table 1, age, height, weight, BMI and days lapsed from admission to enrollment were similar in both groups.
The distribution of percentile intervals across the groups did not show statistically significant differences after 8 weeks of intervention (p > 0.05). The results of each group are shown in Tables 2 and 3.
Upper body
After the training program, 16 of the IG participants were at the same percentile interval of MUAC for age, 4 were higher and 2 were lower. In contrast, 6 of controls were lower, four were higher and ten remained in the same percentile interval. The AMA for age percentile interval was higher in 7 IG participants after training, 2 were lower and 13 remained in the same interval. Within the CG, 4 participants went to higher intervals, 7 remained at the same category and 9 were lower after the period of study. None of the controls went to a lower category of TSF for age percentile, while 7 IG did. Five participants of the CG resulted in a higher percentile category of TSF and 6 participants did for the IG. In contrast to the 9 participants of IG, 15 of the controls remained within the same interval of TSF for age percentile. AMA was below the PRE measure in 12 participants from CG and in 3 participants from IG.
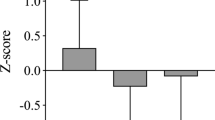
Figure 1a–c shows changes of TSF, MUAC and AMA for each particular score in both groups. These figures show different trends in each variable depending on the group. When comparing groups by the increment in TSF, we found that CG increased on average 14.9 %, 95 % CI (0.9, 28.9 %), t19 = 2.08, p = 0.051, d = 0.35, while IG decreased 0.7 %, 95 % CI (−13.2, 11.8 %), t 21 = −0.72, p = 0.48, d = −0.18. No statistically significant differences were found for the averaged percentage of increase in MUAC, as CG presented almost null increase of 0.3 %, 95 % CI (−2.4, 2.9 %, t 19 = 0.14, p = 0.893, d = 0.02), and IG increased 2.6 %, 95 % CI (−2.3, 7.5 %), t 21 = 0.7, p = 0.494, d = 0.16. However, for the averaged percentage of increase in AMA, we found that CG tended to decrease −3.6 %, 95 % CI (−11.2, 3.9 %), t 19 = −1.5, p = 0.15, d = −0.25, while IG tended to increase 11.6 %, 95 % CI (0.5, 22.8 %), t 21 = 1.74, p = 0.097, d = 0.36, and the difference between both groups was statistically significant (t 40 = 2.25, p = 0.030, d = 0.7) (Fig. 1c).
Lower body
After the training program, 12 of the IG participants were at the same percentile interval of MTC for age, but 10 were at a higher percentile, showing a significant difference (p = 0.002) from PRE to POST. In contrast, four participants in the CG exhibited a lower percentile, seven were higher and nine remained in the same percentile interval. One participant from each group (CG and IG) moved to a lower category of MTSF for age percentile in each group (CG and IG). Five participants of the CG and three participants of the IG resulted in a higher percentile category of MTSF. Changes in MTMA percentiles for age were not calculated since reference tables were not available.
Figure 2a–c shows changes in MTSF, MTC and MTMA for each particular score for both groups. The trends are similar to those shown for the upper arm assessments. When comparing groups by the increment in MTSF, we found that CG increased 5 %, 95 % CI (−4.5, 14.5 %), t 19 = 1.37, p = 0.187, d = 0.16, and similarly IG increased 3.5 %, 95 % CI (−2.5, 9.6 %), t 21 = 0.77, p = 0.451, d = 0.06. However, the increment in MTC showed non-significant change for the CG, −0.2 %, 95 % CI (−3.1, 2.6 %), t 19 = −0.29, p = 0.779, d = −0.05, while IG significantly increased the same circumference 2.1 %, 95 % CI (0.4, 3.7 %), t 21 = 2.44, p = 0.023, d = 0.21 (see Fig. 2b). We found the same trend for the MTMA, where CG decreased −1.6 %, 95 % CI (−8.3, 5 %), t 19 = −0.84, p = 0.410, d = -0.21, while IG increased the muscle area by 4.4 %, 95 % CI (0.01, 8.8 %), t 21 = 1.98, p = 0.061, d = 0.25 (Fig. 2c).
Discussion
Our results suggest that resistance training had a positive effect in the muscle mass in AN-R patients when performed after hospitalization. This is reflected by the statistical differences in the percentage of change in AMA when IG (11.6 %) and CG (−3.6 %) are compared. A small effect size for the change in MUAC is consistent with the literature, where re-feeding studies found improvements in the body fat or BMI [1, 7, 19, 20, 29] associated with changes in the arm circumference. However, re-feeding studies did not find significant changes in the AMA, which might be caused by the lack of exercise programs to promote muscle mass gains [29]. Since changes in both fat and muscle tissues in this study might contribute to an increase in the MUAC, a difference in the circumference between groups should not be expected. Even though the difference in the percentage of increase in TSF showed a medium effect size (0.5), the difference was not statistically significant due to a substantial intra-group variability in both groups (14.9 % CG and 0.7 % IG). In this sense, previous studies have shown that large increases in skinfold thicknesses were achieved as a result of re-feeding programs, and have been correlated to increases in BMI, fat mass and percentage of body fat [1, 7, 19]. Furthermore, those increases were associated with greater weight preoccupation, body dissatisfaction and an increased tendency to return to restrictive eating behaviors [7, 29]. Contrary to re-feeding studies, MTC changes in the IG (2.1 %) occur simultaneously to the increment of MTMA (4.4 %). Although previous studies associated circumference enlargements to improvements in the body fat or BMI [1, 19, 20], the gains on MTC and marginal gains MTMA for the IG, and the lack of changes on MTST for both CG and IG, show that resistance training elicited changes in muscle mass at the lower extremity.
The responses in TSF, MTSF, MUAC, MTC, AMA, and MTMA to high intensity resistance training obtained in this study shed light on how the inclusion of resistance training programs may elicit more desirable outcomes compared to traditional treatment programs. The use of simple and inexpensive measurements, as shown in this study, might be of clinical importance for the monitoring of the nutritional status beyond the BMI, fat mass, % of body fat, and weight assessment. However, the lack of significant results, when percentile categories of TSF, MTSF, MUAC, MTC and AMA were compared, questions their effectiveness to detect inter-group differences. There are important intra-individual changes that are not detected, therefore, reducing the clinical applicability of the anthropometry as a method to assist the re-feeding programs and monitor the nutritional status in AN-R patients. For example, a patient that has a 2 cm change in MUAC could still remain assigned to the same percentile category despite the clinical importance of such change. Small changes in body composition during the treatment are of great importance for the short- and long-term psychological recovery of the AN-R patient. The use of anthropometric measurements related to both muscle and fat mass for feedback might be critical for the psychological therapy. This is of high importance due to the correlation of muscle mass content with the increased rate of urgent hospitalization among AN patients [30]. Clinicians need a tool to ensure a healthy—both muscle and fat mass—recovery and not only a weight or BMI restoration “at all costs”, which usually results in mainly fat accumulation. Still, more research related to muscle mass recovery, psychological effects, and rates of relapse and hospitalization is needed to understand the long-term psychological and physiological effects of treatment.
The diagnosis over an individual’s nutritional status can be registered in qualitative (e.g., thick, normal, overweight, obese; categories within percentiles) or quantitative (e.g., SD scores) scales. Further, percentiles have not been published for all the anthropometric-related variables (e.g., mid-thigh muscle area) and only some percentiles are available (e.g., 5, 15, 25, 50….). As shown in this study, the published percentile categories are too broad, thus reducing the ability to detect meaningful clinical changes. We suggest that future research should focus on the development of standards for age, based on SD scores. Moreover, the performance and sensibility of these scores should be tested on samples close to the lower limit of the distribution (−2SD and under), which are frequently required for the assessment of AN-R patients.
References
Konstantynowicz J, Abramowicz P, Jamiolkowski J, Kadziela-Olech H, Bialokoz-Kalinowska I, Kierus-Jankowska K, Piotrowska-Jastrzebska J, Kaczmarski M (2011) Thigh circumference as a useful predictor of body fat in adolescent girls with anorexia nervosa. Ann Nutr Metab 58:181–187. doi:10.1159/000329437
Haas VK, Kohn MR, Clarke SD, Allen JR, Madden S, Muller MJ, Gaskin KJ (2009) Body composition changes in female adolescents with anorexia nervosa. Am J Clin Nutr 89:1005–1010. doi:10.3945/ajcn.2008.26958
Probst M, Goris M, Vandereycken W, Van Coppenolle H (1996) Body composition in female anorexia nervosa patients. Br J Nutr 76:639–647. pii: S0007114596001754
de Onis M, Onyango AW, Borghi E, Siyam A, Nishida C, Siekmann J (2007) Development of a WHO growth reference for school-aged children and adolescents. Bull World Health Organ 85:660–667
Cole T, Lobstein T (2012) Extended international (IOTF) body mass index cut-offs for thinness, overweight and obesity. Pediatric obesity 7:284–294. doi:10.1111/j.2047-6310.2012.00064.x
Ogden CL, Kuczmarski RJ, Flegal KM, Mei Z, Guo S, Wei R, Grummer-Strawn LM, Curtin LR, Roche AF, Johnson CL (2002) Centers for Disease Control and Prevention 2000 growth charts for the United States: improvements to the 1977 National Center for Health Statistics version. Pediatrics 109:45–60
Nova E, Varela P, Lopez-Vidriero I, Toro O, Ceñal M, Casas J, Marcos A (2001) A one-year follow-up study in anorexia nervosa. Dietary pattern and anthropometrical evolution. Eur J Clin Nutr 55:547–554
Vandereycken W (1983) Outpatient management of anorexia nervosa. Pediatrician 12:118–125
Calogero RM, Pedrotly KN (2004) The practice and process of healthy exercise: an investigation of the treatment of exercise abuse in women with eating disorders. Eat Disord 12:273–291
Bratland-Sanda S, Sundgot-Borgen J, Ro O, Rosenvinge JH, Hoffart A, Martinsen EW (2010) Physical activity and exercise dependence during inpatient treatment of longstanding eating disorders: an exploratory study of excessive and non-excessive exercisers. Int J Eat Disord 43:266–273. doi:10.1002/eat.20769
Tokumura M, Yoshiba S, Tanaka T, Nanri S, Watanabe H (2003) Prescribed exercise training improves exercise capacity of convalescent children and adolescents with anorexia nervosa. Eur J Pediatr 162:430–431. doi:10.1007/s00431-003-1203-1
Chantler I, Szabo CP, Green K (2006) Muscular strength changes in hospitalized anorexic patients after an eight week resistance training program. Int J Sports Med 27:660–665. doi:10.1055/s-2005-865812
Carei TR, Fyfe-Johnson AL, Breuner CC, Brown MA (2010) Randomized controlled clinical trial of yoga in the treatment of eating disorders. J Adolesc Health 46:346–351. doi:10.1016/j.jadohealth.2009.08.007
Thien V, Thomas A, Markin D, Birmingham CL (2000) Pilot study of a graded exercise program for the treatment of anorexia nervosa. Int J Eat Disord 28:101–106
del Valle MF, Perez M, Santana-Sosa E, Fiuza-Luces C, Bustamante-Ara N, Gallardo C, Villasenor A, Graell M, Morande G, Romo GR, Lopez-Mojares LM, Ruiz JR, Lucia A (2010) Does resistance training improve the functional capacity and well being of very young anorexic patients? A randomized controlled trial. J Adolesc Health 46:352–358. doi:10.1016/j.jadohealth.2009.09.001
Touyz SW, Lennerts W, Arthur B, Beumont PJ (1993) Anaerobic exercise as an adjunct to refeeding patients with anorexia nervosa: does it compromise weight gain? Eur Eat Disord Rev 1:177–182
Alberti M, Galvani C, Capelli C, Lanza M, El Ghoch M, Calugi S, Dalle Grave R (2013) Physical fitness before and after weight restoration in anorexia nervosa. J Sports Med Phys Fit 53:396–402
Melchiorri G, Rainoldi A (2008) Mechanical and myoelectric manifestations of fatigue in subjects with anorexia nervosa. J Electromyogr Kinesiol 18:291–297
Kerruish KP, O’Connor J, Humphries IR, Kohn MR, Clarke SD, Briody JN, Thomson EJ, Wright KA, Gaskin KJ, Baur LA (2002) Body composition in adolescents with anorexia nervosa. Am J Clin Nutr 75:31–37
Martin AC, Pascoe EM, Forbes DA (2009) Monitoring nutritional status accurately and reliably in adolescents with anorexia nervosa. J Paediatr Child Health 45:53–57. doi:10.1111/j.1440-1754.2008.01427.x
Mattar L, Godart N, Melchior JC, Pichard C (2011) Anorexia nervosa and nutritional assessment: contribution of body composition measurements. Nutr Res Rev 24:39–45. doi:10.1017/S0954422410000284
APA: American Psychiatric Association (2000) Diagnostic and statistical manual of mental disorders: DSM-IV-TR. American Psychiatric Publishing, Inc., Washington, DC
Davis C, Katzman DK, Kaptein S, Kirsh C, Brewer H (1997) The prevalence of high-level exercise in the eating disorders: etiological implications. Compr Psychiatry 38:321–326
Beumont PJ, Arthur B, Russell JD, Touyz SW (1994) Excessive physical activity in dieting disorder patients: proposals for a supervised exercise program. Int J Eat Disord 15:21–36
Marfell-Jones MJ, Stewart AD, de Ridder JH (2012) International Standards for anthropometric assessment. International Society for the Advancement of Kinanthropometry (ISAK), Lower Hutt, New Zealand
Heymsfield SB, McManus C, Smith J, Stevens V, Nixon DW (1982) Anthropometric measurement of muscle mass: revised equations for calculating bone-free arm muscle area. Am J Clin Nutr 36:680–690
Faigenbaum AD, Kraemer WJ, Blimkie CJ, Jeffreys I, Micheli LJ, Nitka M, Rowland TW (2009) Youth resistance training: updated position statement paper from the national strength and conditioning association. J Strength Cond Res 23:S60–S79. doi:10.1519/JSC.0b013e31819df407
Cohen J (1988) Statistical power analysis for the behavioral sciences. Hillsdale, NJ, Erlbaum
Scalfi L, Polito A, Bianchi L, Marra M, Caldara A, Nicolai E, Contaldo F (2002) Body composition changes in patients with anorexia nervosa after complete weight recovery. Eur J Clin Nutr 56:15–20
Kawai K, Yamashita S, Yamanaka T, Gondo M, Morita C, Nozaki T, Takakura S, Hata T, Yamada Y, Matsubayashi S (2011) The longitudinal BMI pattern and body composition of patients with anorexia nervosa who require urgent hospitalization: a case control study. Biopsychosoc Med 5:14. doi:10.1186/1751-0759-5-14
Acknowledgments
The authors thank the participants and the personnel of the Unit of Psychiatry and Psychology at the hospital for their cooperation. We thank also Texas Tech Writing Center for the contribution in editing this manuscript.
Conflict of interest
The authors have no conflict of interests to disclosure.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fernandez-del-Valle, M., Larumbe-Zabala, E., Graell-Berna, M. et al. Anthropometric changes in adolescents with anorexia nervosa in response to resistance training. Eat Weight Disord 20, 311–317 (2015). https://doi.org/10.1007/s40519-015-0181-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40519-015-0181-4